Abstract
Background
Hippocampus (HIP) was an important limbic structure, and concurrent emotion disorders may occur in medication-overuse headache patients. The aim of this study is to investigate altered HIP and HIP subfields volume in relation with the anxiety in medication-overuse headache patients using a state-of-the-art hippocampal segment method.
Results
The current study presented that a significant lower HIP and HIP subfields volume were identified in medication-overuse headache compared with that in normal controls except right HIP tail, bilateral parasubiculums, and HIP fissure. The left HIP and right subiculum presented negative correlation with headache variables, and the right subiculum, Cornu Ammonis 4, granule cell layer of dentate gyrus, bilateral Cornu Ammonis 1, molecular layer, and whole HIP presented negative correlation with Hamilton Anxiety Scale score, which were further confirmed by the linear regression analysis with the exclusion of psychological variables and headache variables, respectively.
Conclusions
The lower HIP and HIP subfields volume were identified in medication-overuse headache patients, and negatively related with anxiety condition. The potential mechanism for the comorbidity medication-overuse headache and anxiety might be interpreted as the reciprocal causation relationship and co-occurrence relationship.
Keywords
Introduction
Hippocampus (HIP) was an important medial temporal lobe structure and mainly included dentate gyrus (DG), subiculum, presubiculum, parasubiculum, and Cornu Ammonis (CA), which played key roles in the memory 1 and healthy aging. 2 Besides, HIP was also an important one of limbic structure as like cingulate cortex, amygdala, and olfactory cortex, which could be associated with the mood disorders.3,4 Some previous studies demonstrated that different hippocampal subfields would participate in the different mood disorders, e.g., volume reductions of CA One (CA1) and the DG in schizophrenia, 5 volume loss of subiculum and CA Two–CA Three (CA2–3) in medication-naïve female patients with major depressive disorder (MDD), 6 and multiple hippocampal subfields volume reduction in bipolar disorder and MDD. 3 Therefore, these findings suggested that HIP was not a uniform structure, and the precise structural changes of hippocampal subfields may provide more information on the neuromechanism of the brain disorder.
Medication-overuse headache (MOH) was one of chronic headache characterized by repetitive and excessive overconsumption of analgesics or acute anti-migraine drugs. 7 In a previous study, functional connectivity increased between precuneus and HIP, and voxel-based morphometry did not detect any significant brain volume change in this MOH patient entity. 8 The other structure studies identified that volume gain of periaqueductal gray matter (PAG) presented in MOH patients,9,10 and one study confirmed that there was no significant brain volume changes in MOH patients. 11 However, the elaborate hippocampal subfields measurement were not performed in MOH patients up to now.
Headache perception may include physiological and psychological factors, and the increased functional connectivity of HIP indicated that HIP might participate in the emotion control, 8 but the link between the hippocampal subfield volume and the MOH had not been investigated.
The previous studies recognized that MOH was associated with mood disorders in 27%–85% and anxiety disorders in 61%–83%.12,13 The voxel-based morphometry study demonstrated that the increased volume in PAG was positively correlated with anxiety scores, and anxiety might be associated with the dysfunction of antinociceptive system in MOH. 10 A recent review study suggested that the affective and anxiety disorders had an increased risk in migraine patients compared with non-migraine patients, and prevailed in patients with chronic forms or transform of headache and substance use than in patients with migraine alone. 14 However, the neuromechanism of migraine concomitant with anxiety was not totally understood.
The current study is aimed to investigate the relationship between MOH and anxiety. We hypothesize that HIP volume change may be associated with MOH. To address this hypothesis, we prospectively obtained high-resolution structural images from 31 MOH patients and 31 normal controls (NCs) to calculate and analyze total HIP and hippocampal subfield volume change using a state-of-the-art hippocampal segmentation approach. 15 The generalized linear model was used to compute the linear regression equation to determine the effect of the headache variables, anxiety, depression, and cognitive variables on the HIP volume change in MOH patients.
Materials and methods
Subjects
Thirty-one MOH patients were recruited from the International Headache Center, Department of Neurology, Chinese PLA General Hospital. The diagnostic criteria of MOH was based on the International Classification of Headache Disorders, third Edition (beta version) (ICHD-III beta), 16 including the diagnosis of 8.2 MOH, 1.1, and 1.2 migraine. The inclusion criteria should be included as follows: (1) no migraine preventive medication used in the past three months; and (2) patient’s willingness to engage in the study. The patients with the following criteria should be excluded out: (1) with any chronic disorders, including hypertension, diabetes mellitus, cardiovascular diseases, etc.; and (2) with cranium trauma, illness interfering with central nervous system function, psychotic disorder, and regular use of a psychoactive or hormone medication. Thirty-one NCs were recruited, who had the same inclusion and exclusion criteria with MOH patients. NCs should never have any primary headache disorders or other types of headache in the past year. General demographic and headache information were registered and evaluated in our headache database. Additionally, we evaluated anxiety, depression, and cognitive function of all the participants by using the Hamilton Anxiety Scale (HAMA), 17 the Hamilton Depression Scale (HAMD), 18 and the Montreal Cognitive Assessment (MoCA) Beijing Version (www.mocatest.org). The study protocol was approved by the Ethical Committee of Chinese PLA General Hospital and complied with the Declaration of Helsinki. Informed consents were obtained from all participants before the study. Magnetic resonance imaging (MRI) scans were taken at least three days after a migraine attack for MOH patients. All the patients were assessed using the Visual Analogue Scale (VAS) and Migraine Disability Assessment (MIDAS). All the subjects were right-handed and underwent conventional MRI examination to exclude the subjects with cerebral infarction, malacia, or occupying lesions. Alcohol, nicotine, caffeine, and other substances were avoided for at least 12 h before MRI examination.
MRI acquisition
The brain structure images and conventional MR images were obtained from a GE three-tesla MR system (DISCOVERY MR750, GE Healthcare, Milwaukee, WI, USA), and a conventional eight-channel quadrature head coil was used. All subjects were instructed to lie in a supine position, and formed padding was used to limit head movement. An axial three-dimensional T1-weighted fast spoiled gradient recalled echo (3D T1-FSPGR) sequence was performed with the following parameters: TR (repetition time) = 6.3 ms, TE (echo time) = 2.8 ms, flip angle = 15°, FOV (field of view) = 25.6 cm × 25.6 cm, Matrix = 256 × 256, NEX (number of acquisition) = 1. Conventional T2-weighted imaging (T2WI), T1 fluid-attenuated inversion recovery (T1-FLAIR) weighted imaging, and diffusion weighted imaging (DWI) were also acquired. All imaging protocols were identical for all subjects. No obvious structural damage and T2-visible lesion were observed based on the conventional MR images.
MR image processing
All MR structural image data were processed using the hippocampal subfield segmentation package in Freesurfer image analysis software (http://surfer.nmr.mgh.harvard.edu/) (stable v6.0.0)15,19–20 running under Ubuntu 12.04.2. The image processing included following steps: (1) The structural images were performed with volumetric and surface processing using Freesurfer recon-all—all pipeline; 21 (2) The segmentation of individual hippocampal subfields was performed using Freesurfer hippocampal-subfields-T1 pipeline, which included the following steps: (1) Whole HIP segment were performed from the automated pipeline for volumetric segmentation of subcortical structures implemented in Freesurfer; (2) HIP subfields segment was performed using a Bayesian inference approach according to a novel atlas of the HIP built in Freesurfer; (3) HIP was segmented into 12 subfields: hippocampal tail, subiculum, presubiculum, parasubiculum, Cornu Ammonis 1 (CA1), CA2/3, CA4, granule cell layer of dentate gyrus (GC-DG), molecular layer, fimbria, HIP-amygdala-transition-area, and hippocampal fissure (Figure 1).15,19 (4) The final hippocampal subfields volume and brain parenchymal volume were measured for the statistical analysis.
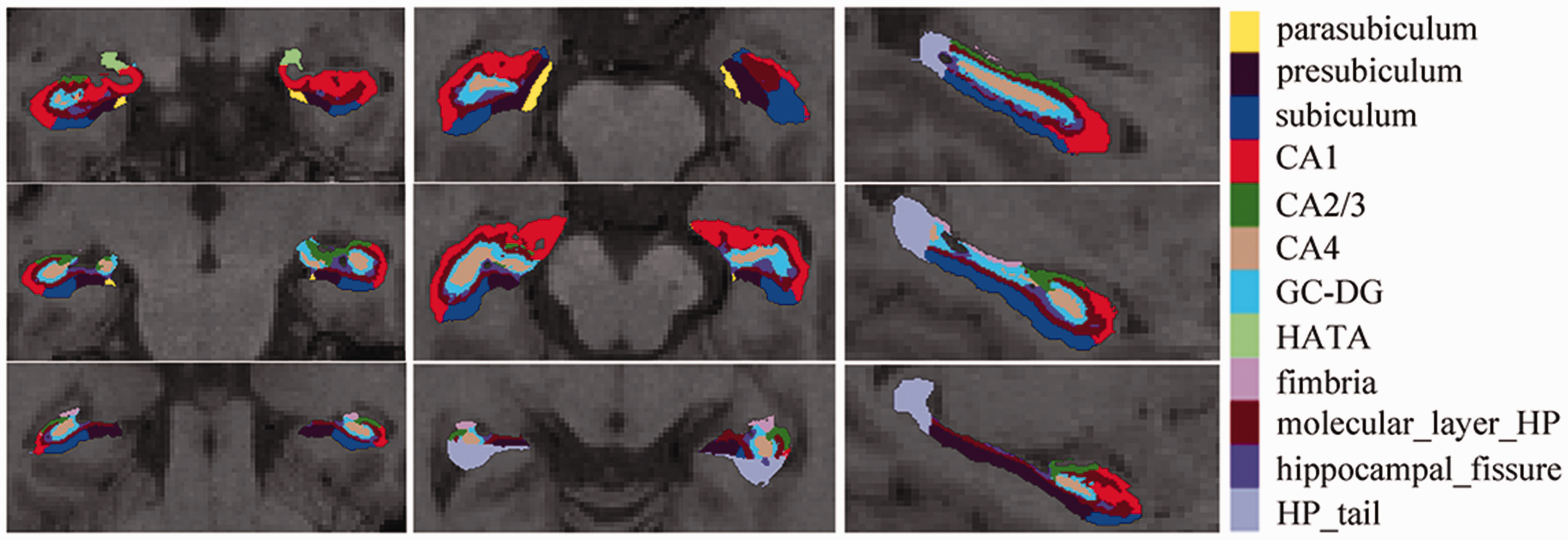
The 12 hippocampal subfields were segmented and labeled with different colors. CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area; HP: hippocampus.
Statistical analysis
The statistical analysis was performed by using PASW Statistics 18.0. The significant differences of hippocampal volume between MOH group and NC group were compared with general linear model (independent univariate t-test with age, sex, and total brain parenchymal volume as covariates). The Pearson's correlation analysis was applied between hippocampal volume and the clinical variables in MOH. The age, HAMD, and MoCA were performed with independent samples t-test, HAMA was performed with Welch test because of variance nonhomogeneity, and sex was performed with Chi-Square test. Linear regression (Backward method) was performed between the volume of HIP and HIP subfields and the clinical variables including disease duration (DD), headache days per month (HDPM), VAS, MIDAS, drug pills taken (DP), HAMA, HAMD, and MoCA, which would be used to identify the clinical parameters participating in the hippocampal volume changes. The linear regression analysis over the whole brain was performed by QDEC tool of Freesurfer 6.0 software (with Monte Carlo simulations for multiple comparisons correction). Significant difference was set at a P value of < 0.05.
Results
The clinical characteristics of MOH and NCs
There was no significant difference for age between MOH (42.55 ± 10.31 years old) and NC (45.09 ± 10.50 years old), and there was a significant difference for sex between two groups. There were significant high scores of HAMA and HAMD and lower MoCA in MOH than that in NC (P < 0.05). The disease duration was 18.32 ± 9.45 years, and the scores of VAS and MIDAS were 8.23 ± 1.61 and 136.10 ± 72.10, respectively. Table 1 shows that HDPM was 27.58 ± 4.57 days and the number of drug pills taken per day was 5.76 ± 4.43.
The clinical characteristics of MOH and normal controls.

Note: HAMA: Hamilton Anxiety Scale; HAMD: Hamilton Depression Scale; MoCA: Montreal Cognitive Assessment; DD: disease duration; HDPM: headache days per month; VAS: Visual Analogue Scale; MIDAS: Migraine Disability Assessment; DP: the number of drug pills taken per day; MOH: medication-overuse headache; NC: normal control; MMSE: Mini-mental State Examination.
aPearson Chi-Square test with Chi-Square value 5.391.
bWelch test because of variance nonhomogeneity.
Comparison of hippocampal subfields volume between MOH and NC
There was a significant lower hippocampal subfields volume in MOH than that in NC (P < 0.05) except right HIP tail, bilateral parasubiculums, and HIP fissure (P > 0.05) (Table 2). Figure 2 shows that bilateral whole HIPs had lower volume in MOH than that in NC (P < 0.05).
Comparison of the volume of hippocampal subfields between MOH and NC.

Note: HIP: hippocampus; CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area; MOH: medication-overuse headache; NC: normal control.
aWelch test because of variance nonhomogeneity.

The hippocampal subfields and whole hippocampus volume in MOH and NC. MOH: Medication-overuse headache; NC: normal controls; CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area; HIP: hippocampus.
Correlation analysis between hippocampal subfields volume and the clinical variables
Table 3 shows that left HIP tail showed a negative correlation with VAS score (r = −0.361, P = 0.046), and right subiculum and presubiculum had a negative correlation with HDPM (r = −0.384 and P = 0.033, r = −0.363 and P = 0.045, respectively). Besides these, right CA4 identified a positive correlation with disease duration (r = 0.375, P = 0.038) (Figure 3).
Correlation analysis between the volume of hippocampal subfields and the clinical variables about headache information for MOH patients.a
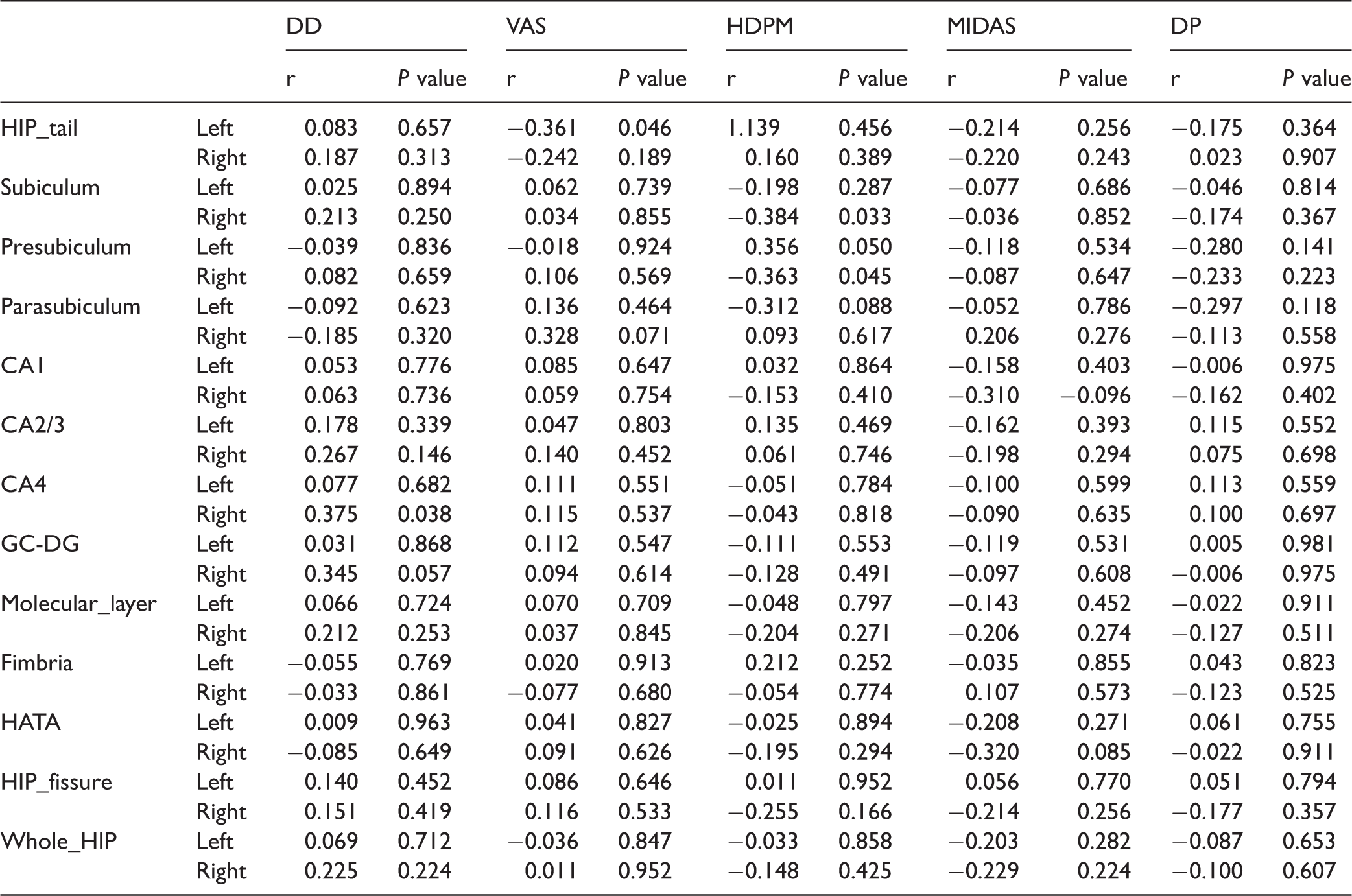
Note: DD: disease duration; VAS: visual analogue scale; HDPM: headache days per month; HIP: hippocampus; CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area; MIDAS: Migraine Disability Assessment; DP: the number of drug pills taken per day.
aThe significance was set as P value > 0.05 with r > 0.3.

The scatter plot between the clinical variables about headache information and hippocampal subfields. HIP: hippocampus; CA: Cornu Ammonis; DD: disease duration; VAS: Visual Analogue Scale; HDPM: headache days per month.
The negative correlation with HAMA score was confirmed in the right subiculum, CA4, GC-DG, bilateral CA1, molecular layer, and whole HIP (Table 4 and Figure 4). The other neuropsychological tests showed no significant correlation (HAMD and MoCA) with hippocampal subfields (P > 0.05).
Correlation analysis between the volume of hippocampal subfields and the clinical variables about psychological evaluation for MOH patients.a

Note: HIP: hippocampus; CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area; HAMA: Hamilton Anxiety Scale; HAMD: Hamilton Depression Scale; MoCA: Montreal Cognitive Assessment.
aThe significance was set as P value > 0.05 with r > 0.3.
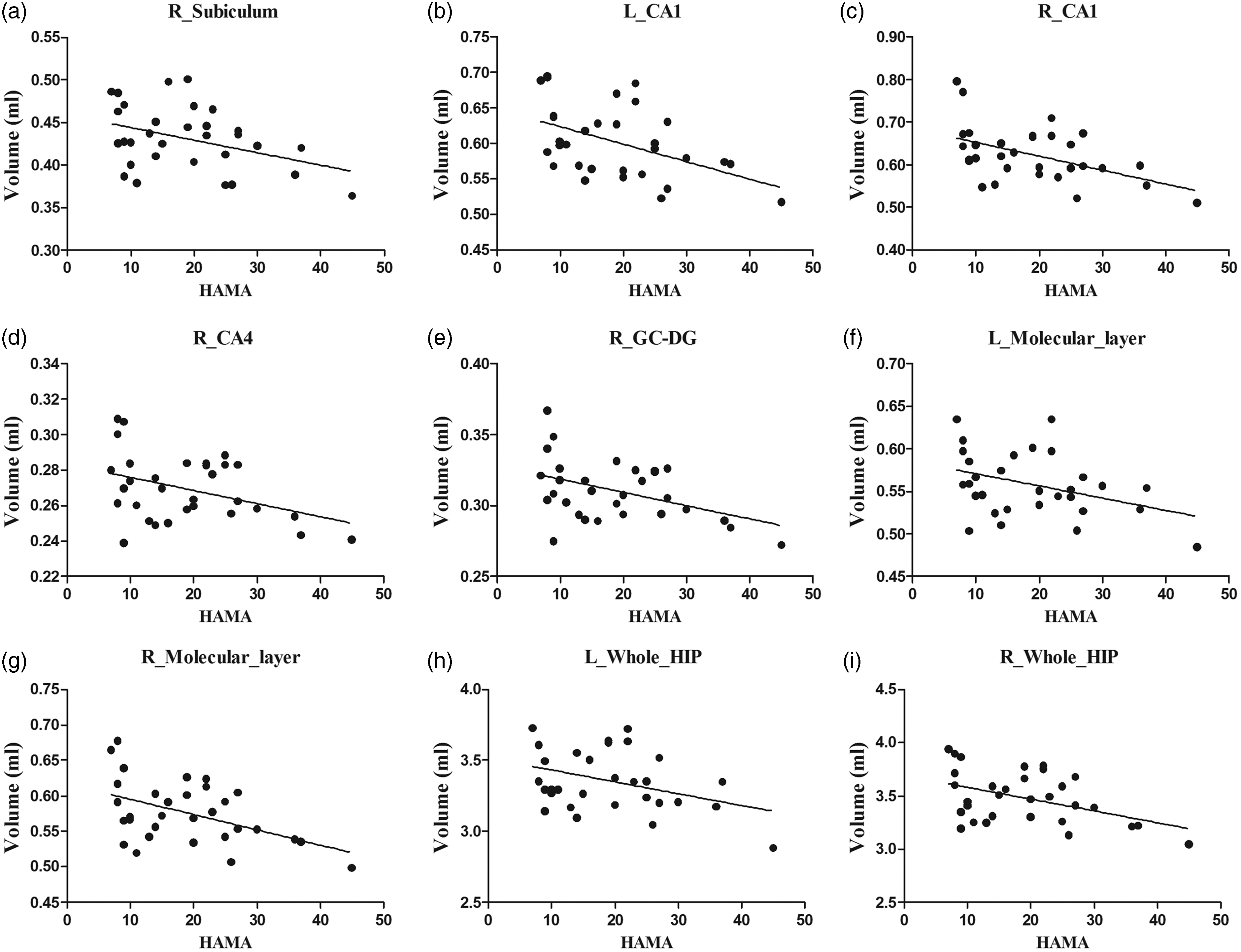
The scatter plot between the psychological scale scores and the hippocampal subfields. a, R_Subiculum vs HAMA; b, L_CA1 vs HAMA; c, R_CA1 vs HAMA; d, R_CA4 vs HAMA; e, R_GC-DG vs HAMA; f, L_Molecular_layer vs HAMA; g, R_Molecular_layer vs HAMA; h, L_Whole_HIP vs HAMA; i, R_Whole_HIP vs HAMA CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HIP: hippocampus; HAMA: Hamilton Anxiety Scale.
Linear regression analysis for the hippocampal subfields with significant correlation with the clinical variables
The left HIP_tail presented a negative linear relationship with VAS scores and the linear regression equation was Volumeleft_HIP_tail = −0.015 × VAS + 0.648 (PVAS = 0.049) (Table 5). Right presubiculum showed a negative linear relationship with HDPM (Volumeright_presubiculum = −0.002 × HDPM + 0.357, PHDPM = 0.024). The other clinical variables were excluded from these two equations (P > 0.05).
A significant negative linear relationship with HAMA score was identified in the bilateral CA1 and right GC_DG, molecular layer, and whole HIP. The linear regression equations were VolumeL_CA1 = −0.002 × HAMA + 0.651 (PHAMA = 0.011), VolumeR_CA1 = −0.003 × HAMA + 0.674 (PHAMA = 0.016), VolumeR_GC_DG = −0.001 × HAMA + 0.331 (PHAMA = 0.020), VolumeR_Molecular_layer = −0.002 × HAMA + 0.610 (PHAMA = 0.025), and VolumeR_Whole_HIP = −0.02 × HAMA + 3.656 (PHAMA = 0.010). Only HAMA scores entered these five linear equations, and the other clinical variable including DD, VAS, HDPM, MIDAS, DP, HAMD, and MoCA did not show any contribution for these equations (P > 0.05).
The right subiculum showed a negative linear equation with HDPM and HAMA with linear equation VolumeR_Subiculum = −0.003 × HDPM−0.002 × HAMA + 0.540 (PHDPM = 0.013, PHAMA = 0.015). The right CA4, left molecular layer, and whole HIP did not presented a linear regression equation (P > 0.05).
Linear regression analysis for the hippocampal subfields with significant correlation with the clinical variables.
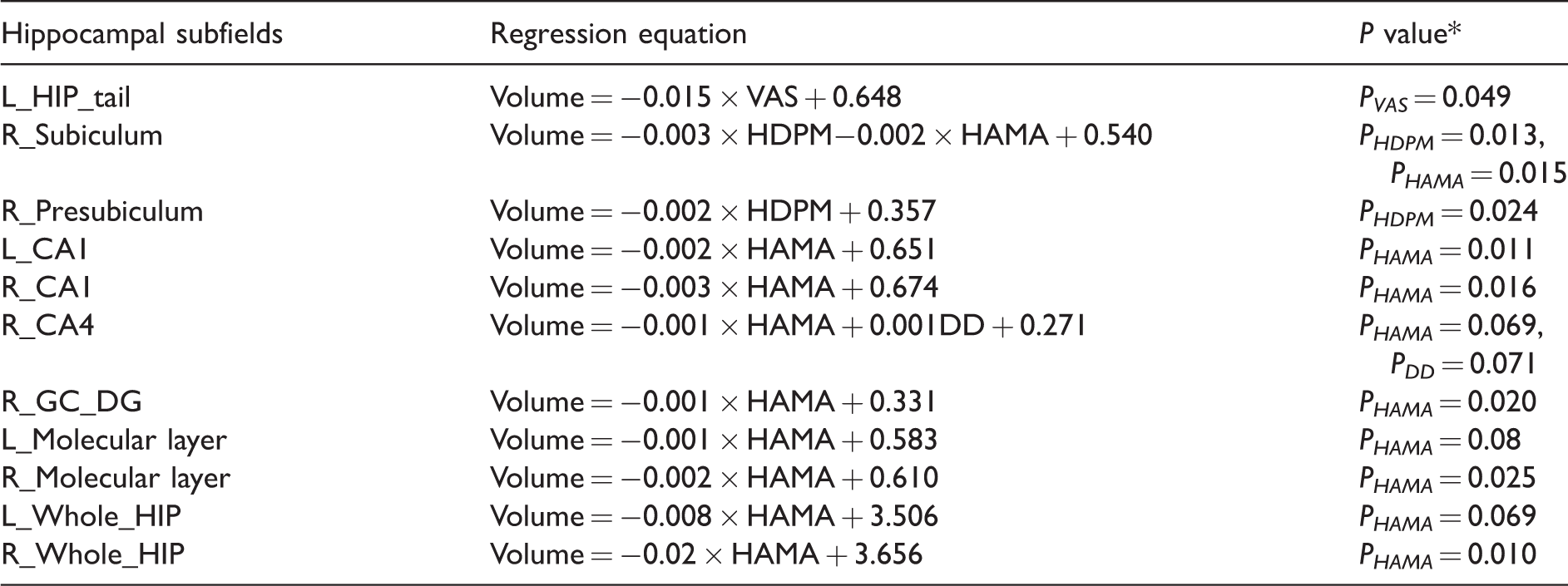
Note: HAMA: Hamilton Anxiety Scale; HDPM: headache days per month; VAS: Visual Analogue Scale; HIP: hippocampus; CA: Cornu Ammonis; GC-DG: granule cell layer of dentate gyrus; HATA: hippocampus-amygdala-transition-area.
*The P value for all the constants was 0.000.
Discussion
In this study, the total HIP and multiple hippocampal subfields detected a decreased volume in MOH compared with NC, which suggested that the lower HIP volume may be associated with MOH itself or the concomitant mood changes. Further correlation analysis demonstrated that some parts of HIP (left HIP_tail, right subiculum, and presubiculum) presented a negative correlation with headache variables, right CA4 showed a positive correlation with headache variables, and multiple HIP subfields (right subiculum, bilateral CA1, molecular layer and whole HIP, right CA4, and GC-DG) presented a negative relation with anxiety scores. The correlation analysis indicated that anxiety may be the main reason of the lower HIP volume.
In the previous studies, the brain morphometric changes were mainly located in the PAG9,22 and left prefrontal cortex, 23 and the HIP and HIP subfield volume changes were not reported up to now. However, a one-year follow-up evaluation study detected the hippocampal volume reduction in episodic migraine. 24 The current study first confirmed the HIP subfield volume decreased in MOH patients, which may present the key value of HIP in understanding the neuromechanism of MOH. A previous study 8 demonstrated that the decreased functional connectivity was detected between the right HIP and the left precuneus in MOH patients, which was positively related with the number of pills per month. Therefore, the lower HIP subfields volume may be associated with default mode networks, which would be sensitive to the cognitive states, 25 and altered in the pain population, such as chronic pain26,27 and diabetic neuropathic pain. 28
The correlation analysis indicated that the headache variables and mood variables participated in the decreased HIP subfields volume. The previous study 8 identified that the right HIP volume was negatively correlated with the migraine duration; however, only the right CA4 region showed positive correlation with migraine duration in MOH patients in the current study. HIP was a complex and heterogeneous structure, and different subfield present different organization and interconnections between the subfields. 29 Regional gray matter volume changes reflected the local extracellular changes in water or the concentration of proteins in neurons and glia. 24 The volume alteration of CA4 regions with migraine duration prolongation may be associated with gliosis. 30 The left HIP_tail, right subiculum, and presubiculum also presented negative correlation with VAS and HDPM, which might be associated with repetitive and frequent exposure to medication. 8
Table 4 shows that multiple HIP subfields was negatively related with HAMA scores, which suggested that anxiety might be an important comorbid psychiatric disorder. The presence of the psychiatric disorder was a risk factor for the transformation of migraine into a chronic form, 31 and anxiety was commonly seen in migraine patients and their relatives. 32 The previous study indicated that MDD had an increased prevalence in migraine patients, 33 and multiple HIP subfield presented decreased volume in MDD.3,6 However, only HAMA scores showed a negative relation with the lower HIP and HIP subfield volume in MOH in the current study. The potential mechanism for the comorbidity between migraine and anxiety may be associated with reciprocal causation relationship or co-occurrence. 14
Therefore, this was a “the chicken or the egg?” question, i.e., the anxiety was generated from MOH itself or the anxiety would lead to the MOH genesis. The further linear regression analysis demonstrated that bilateral CA1, right GC-DG, molecular layer, and right whole HIP had significant linear relationship with HAMA (P < 0.05), and the headache variables, depression, and cognitive variables were excluded from the linear equations (P > 0.05). The linear relationship existence identified that the anxiety was the main reason of the multiple lower HIP volume changes, which indicated that anxiety might play an important role in the development of migraine. 14 Besides these, left HIP_tail and right presubiculum presented the significant linear relation with headache variables (VAS and HDPM), which suggested that MOH could lead to these HIP regions volume decrease and indicated that MOH might be featured on the development of anxiety, i.e., the pain episode might facilitate the development of anticipatory anxiety. 14 The common HIP subfield with decreased volume was the right subiculum resulted from the headache itself and anxiety, which indicated that co-occurrence of MOH and anxiety might share the same etiological factors and determinants. 14
Limitations of this study include the following: First, this study was a cross-sectional study, and the longitudinal observation should be performed to validate the main effect on the HIP for the concurrent anxiety in MOH patients; Second, the anti-anxiety therapy should be investigated to observe the HIP volume changes with the improvement of the anxiety condition, which would be helpful to understand the neuromechanism of lower HIP subfield volume in MOH concurrent with anxiety. Last, the anxiety and depressed patients without migraine should be compared with MOH patients with anxiety in the future study.
In conclusion, the lower HIP and HIP subfields volume were observed in MOH patients and negatively related with anxiety condition. The potential mechanism for the comorbidity MOH and anxiety might be interpreted as the reciprocal causation relationship and co-occurrence relationship.
Footnotes
Authors' contributions
LM and SY contributed to the conception and design; ZC, ML, and XC contributed to the acquisition of data; ZC contributed to the analysis and interpretation of data and drafting the article; and LM and SY revised it for intellectual content. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Special Financial Grant from the China Postdoctoral Science Foundation (2014T70960) and the Foundation for Medical and Health Sci & Tech Innovation Project of Sanya (2016YW37).
