Abstract
Background
After spinal cord injury, central neuropathic pain develops in the majority of spinal cord injury patients. Spinal hemisection in rats, which has been developed as an animal model of spinal cord injury in humans, results in hyperexcitation of spinal dorsal horn neurons soon after the hemisection and thereafter. The hyperexcitation is likely caused by permanent elimination of the descending pain systems. We examined the change in synaptic transmission of substantia gelatinosa neurons following acute spinal hemisection by using an in vivo whole-cell patch-clamp technique.
Results
An increased spontaneous action potential firings of substantia gelatinosa neurons was detected in hemisected rats compared with that in control animals. The frequencies and amplitudes of spontaneous excitatory postsynaptic currents and of evoked excitatory postsynaptic currentss in response to non-noxious and noxious stimuli were not different between hemisected and control animals. On the contrary, the amplitude and frequency of spontaneous inhibitory postsynaptic currents of substantia gelatinosa neurons in hemisected animals were significantly smaller and lower, respectively, than those in control animals (P < 0.01). Large amplitude and high-frequency spontaneous inhibitory postsynaptic currents, which could not be elicited by mechanical stimuli, were seen in 44% of substantia gelatinosa neurons in control animals but only in 17% of substantia gelatinosa neurons in hemisected animals. In control animals, such large amplitude spontaneous inhibitory postsynaptic currents were suppressed by spinal application of tetrodotoxin (1 µM). Cervical application of lidocaine (2%, 10 µl) also inhibited such large amplitude of inhibitory postsynaptic currents. The proportion of multi-receptive substantia gelatinosa neurons, which exhibit action potential firing in response to non-noxious and noxious stimuli, was much larger in hemisected animals than in control animals.
Conclusions
These suggest that substantia gelatinosa neurons receive tonic inhibition by spinal inhibitory interneurons which generate persistent action potentials. Spinal hemisection results in hyperexcitation of substantia gelatinosa neurons at least in part by eliminating the tonic descending control of spinal inhibitory interneurons from supraspinal levels.
Keywords
Introduction
After spinal cord injury (SCI), central neuropathic pain syndrome, which is refractory to currently available modes of treatment, develops in the majority of SCI patients. 1 Spinal hemisection in rats has been developed as an animal model of incomplete SCI in humans.2–4 Increases in spontaneous activity and responsiveness to non-noxious and noxious stimuli can be seen in spinal laminae V-VI neurons located caudally to and on both sides of the hemisection within 1 h after the SCI and thereafter for several weeks.3,5–8
Hyperexcitation of spinal dorsal horn (SDH) neurons is most likely caused by permanent elimination of descending inhibitory inputs from rostral segments of the spinal cord and from supraspinal sites, including the raphe magnus and locus ceruleus in the brain stem,6,9,10 which is thought to be a common mechanism of the hyperexcitation over periods of acute (several hrs) and chronic phases (several weeks) after hemisection of the spinal cord in the rat.2,3,7,8,10 In addition to the inhibitory systems, descending influences from supraspinal sites can also facilitate activity of spinal nociceptive neurons. 11 However, functional alterations of SDH neurons after interruption of the descending inhibitory and facilitatory inputs to SDH neurons following SCI have not been fully elucidated.
Noxious information is transmitted through fine myelinated Aδ and unmyelinated C afferents from the periphery to the superficial dorsal horn, especially to the substantia gelatinosa (SG, lamina II of Rexed). This sensory information is modified and integrated in the SG and consequently regulates the outputs of projection neurons located in lamina I and laminae V-VI. 12 In addition, the descending inhibitory influences from rostral segments of the spinal cord and supraspinal structures on SG neurons are known to be modified under certain pathological conditions.13,14 A new technique for patch-clamp recordings in vivo from SG neurons has been established.15–18 The in vivo recording, but not in vitro recording by spinal slice preparations, is suitable for analyzing the descending influences on SG neurons since the synaptic connectivity between the spinal cord and the brain remains intact.
The aim of the present study was to determine the characteristics of inhibitory and excitatory synaptic transmission in SG neurons located caudally to the site of spinal hemisection compared with the characteristics in control animals by using the in vivo whole-cell patch-clamp technique.
Materials and methods
Animal preparation and spinal cord hemisection
All procedures were performed according to the Ethical Guidelines of the International Association for the Study of Pain and approved by the Animal Care and Use Committee of Sapporo Medical University School of Medicine, Sapporo, Japan (Approval No. 05-049) and Shinshu University School of Medicine, Matsumoto, Japan (Approval No. 09-015). Efforts were made to minimize the number of animals used.
The methods used for the current experiment were modifications of those used in previous studies.19–22 Adult male Sprague-Dawley rats (six–seven weeks of age, 180–220 g) were anesthetized with urethane (1.2–1.5 g kg−1, intraperitoneal) and artificially ventilated through a tracheotomy tube. In most animals, a PE-10 polyethylene catheter was intraperitoneally implanted after the first dose of urethane (0.7–0.9 g kg−1) had been administered, and additional doses (0.2–0.4 g kg−1) of urethane were administered through the catheter until withdrawal reflex were eliminated. The rectal temperature was kept at 37.0 ± 0.5℃ with a heating pad placed beneath the animal. A lumbar laminectomy was performed at the level of the T12 to L2 vertebrae, and the animal was then placed in a stereotaxic apparatus (Model ST-7, Narishige, Tokyo, Japan). After removing the dura, the pia-arachnoid membrane of the right L3 or L4 dorsal root entry zone was cut to make a window large enough for insertion of a patch electrode (Figure 1(a)). The surface of the spinal cord was irrigated with 95% O2-5% CO2-equilibrated Krebs solution (in mM: 117 NaCl, 3.6 KCl, 2.5 CaCl2, 1.2 MgCl2, 1.2 NaH2PO4, 11 glucose, and 25 NaHCO3) at 37.0 ± 0.5℃.
Schematic diagrams of in vivo rat preparation and schematic diagrams of the extent of spinal hemisection. (a) While animals were mechanically ventilated with tracheotomy, the lumbar spinal cord at the level T13 to L5 was exposed by laminectomy. The superficial dorsal gray matter lateral to the dorsal root entry zone was discernible as a relatively translucent band under Lissauer’s tract (gray zone). The pia-arachnoid membrane was cut to make a window for penetration of the patch electrode into the spinal cord. The spinal cord was hemisected along a bar between T13 and L1 spinal segments. Non-noxious air puff stimuli and noxious pinch stimuli were applied to the most sensitive site of the receptive field. (b) Extent of the surgical lesion to the spinal cord was determined in histological sections of the animals used in this study. The minimum lesion (left) and the maximum lesion (right) observed in this study are indicated by horizontal lines. (c) Schematic drawing depicting an electrode in the transverse plane. The electrode was advanced into the SG at an angle of 30°–45°. SG: substantia gelatinosa.
The spinal cord was hemisected between T13 and L1 spinal segments using scissors for microsurgery without damage to the posterior vessel or its branches according to a modification of our previously reported method (Figure 1(a) and 1(b)). 8 Sham surgery, in which the surgical procedure was identical to that described above but without hemisectioning of the spinal cord, was performed in animals as controls. Animals were stabilized at least for 1 h before electrophysiological recordings were obtained. The extent of the hemisection lesion was assessed from histological sections fixed with 4% paraformaldehyde in 0.1 M phosphate buffer and was confined to one side and included the dorsal column, Lissauer’s tract, lateral and ventral column systems, and the gray matter ipsilaterally (Figure 1(b)).2,8 At the end of the experiments, the rats were given an overdose of urethane and were then killed by exsanguination.
Electrophysiological recordings
The patch electrodes were pulled from thin-walled borosilicate glass capillaries (outer diameter of 1.5 mm, TW150F-4, World Precision Instruments, Sarasota, FL) using a puller (p-97, Sutter Instruments, Novato, CA) and were filled with a patch-pipette solution having either of the following compositions (in mM): 135 K-gluconate, 5 KCl, 0.5 CaCl2, 2 MgCl2, 5 EGTA, 5 ATP-Mg, 5 HEPES, KOH (pH 7.2) for excitatory postsynaptic currents (EPSCs) recordings and current clamp recordings and 110 Cs2SO4, 0.5 CaCl2, 2 MgCl2, 5 EGTA, 5 HEPES, 5 ATP-Mg, 5 tetraethylammonium, CsOH (pH 7.2) for inhibitory postsynaptic currents (IPSCs) recordings. An electrode with a resistance of 5–10 MΩ was advanced at an angle of 30°–45° into the SG at the right L3 or L4 dorsal root entry zone through the window using a micromanipulator (Model MHW-4, Narishige, Tokyo, Japan) (Figure 1(c)). The distance from the recording site to the site of hemisectioning of the spinal cord was more than 10 mm. After making a gigaohm seal (resistance of at least 10 GΩ), the membrane patch was ruptured by a brief period of more negative pressure, resulting in whole-cell configuration. Recordings were made using a patch-clamp amplifier (Axopatch 200B, Axon Instruments, Union City, CA), and the data were digitized with an A/D converter (Digidata 1322A, Axon Instruments), stored in a personal computer using a data acquisition program (pCLAMP8, Axon Instruments), and analyzed with Mini Analysis Program version 6.03 (Synaptosoft, Fort Lee, NJ). In voltage clamp mode, the holding potential (VH) were −70 mV and 0 mV, at which glycine- and GABA-mediated IPSCs and glutamate-mediated EPSCs, respectively, were negligible. 15 The recorded neurons were identified as being in the SG based either on the depth (50–150 µm) of the neurons from the surface of the spinal cord. Some neurons identified with an intracellular injection of biocytin were located in the SG (data not shown), as was found in previous studies.19–22 An animal preparation could be maintained in a stable condition for over 8 h and patch-clamp recordings could be maintained for up to 3 h, comparable to those in previous experiments.19,21,22
Experimental protocol
The experimental protocol both in animals with spinal hemisection (hemisected animals) and animals without spinal hemisection (control animals) was as follows. After obtaining the whole-cell recording configuration using patch electrodes filled with a patch-pipette solution for IPSC recordings, a receptive field was roughly determined by applying non-noxious stimuli with untoothed forceps on the shaved skin of the right hindquarter (lumbar and gluteal regions of the rat), and the most reactive site was defined as the stimulating point (Figure 1(a)) in the voltage clamp mode in which VH was kept at −70 mV. The response to non-noxious stimulation was assessed with puffing air (150 p.s.i.) through a pipette (outer diameter of 200 µm) applied repetitively (duration of an injection, 100 ms; frequency, 10 Hz; injection time, 10 s) using a pico-injector (PLI-100, Harvard Apparatus, Holliston, MA) according to a previously described method.15,16 The air puff stimulation did not evoke pain or discomfort sensation in examiners in a preliminary study.
The noxious mechanical stimulation was applied with toothed forceps fixed on a rod, and a weight (40 g) was placed on the forceps for 10 s, by which examiners experienced pinching pain. In a preliminary study, the frequency of EPSCs of SG neurons increased in proportion to the increase in weight and no significant accommodation was observed, indicating that the responses were mediated by the activation of nociceptors. Stimuli (air puff stimuli and pinch stimuli) were applied several times at 1-min intervals. In most SG neurons, both non-noxious “air puff” stimuli and noxious “pinch” stimuli applied to the ipsilateral hindquarter elicited a barrage of EPSCs (see mechanical responses in Figure 2(b)).
15
However, in some neurons, a barrage of EPSCs was maximally and constantly elicited during application of non-noxious air puff stimuli, but occurrence of evoked EPSCs in response to noxious pinch stimuli was restricted to the beginning and end of the stimuli (Figure 2(b)). These responses of EPSCs have been interpreted as responses to the initial “touch” contact of the forceps during pinching and were qualitatively similar to an action potential (AP) firing pattern of a subset of SG neurons classified as light-touch neurons in previous studies.20,23 These neurons in which EPSCs were maximally elicited by air puff stimuli were not used in the present study since the neurons were commonly located deeply (120 µm ∼ from the dorsal surface of the spinal cord), that is, possibly located in lamina III and since we could not completely distinguish these neurons from lamina III neurons, which are involved in non-noxious but not noxious sensory information, with respect to their responses to mechanical stimuli.
Examples of spontaneous action potentials (APs) elicited in SG neurons of control animals (Control) and spinally hemisected animals (Hemisection) and representative excitatory synaptic responses evoked in SG neurons. (a) The majority of SG neurons (89%, 16/18) in control animals did not show any spontaneous AP firing. Forty-two percent of SG neurons (9/21) in spinally hemisected animals showed spontaneous AP firing. The mean frequency of spontaneous AP firing of SG neurons was significantly higher in hemisected animals (n = 21) than in control animals (n = 18). *P < 0.05 versus control. Data are shown as means ± SEM. (b) Spontaneous excitatory postsynaptic currents (EPSCs) and mechanical stimuli-evoked EPSCs recorded from control animals in the voltage clamp mode (VH = −70 mV). EPSCs were elicited both by air puff stimuli and pinch stimuli in most SG neurons that were more sensitive to pinch than air puff stimuli (upper traces). Bars above the traces show the duration of the stimulation. These mechanical stimuli-evoked EPSCs did not differ from those in hemisected animals (see text and
Table 2
). In some SG neurons commonly located deeply (120 µm∼ from the dorsal surface of the spinal cord), EPSCs were maximally elicited by application of non-noxious air puff stimuli, but pinch stimuli-evoked EPSCs were restricted to the beginning and end of the stimuli (lower traces). These responses of EPSCs have been interpreted as responses to the initial and final “touch” contact of the forceps during pinching. These SG neurons were not used in this study.
Data of stable spontaneous IPSCs (sIPSCs) were obtained over a period of 5 min. At 0 mV of VH. Air puff stimuli and pinch stimuli were then applied to the same site in the same manner as that described above for EPSC recordings. In SG neurons that exerted large amplitude (>50 pA) and high-frequency (>20–30 Hz) sIPSCs, in addition to the stimulating site, many portions of the skin of the body such as the face, ipsilateral and contralateral forepaws, rostral regions of the back, ipsilateral and contralateral hindpaws, and tail were mechanically stimulated by air puff and pinch stimuli. With respect to sIPSCs and evoked IPSCs, SG neurons were classified as described below in the Results section.
In a separate study, after obtaining of the whole-cell recording configuration using patch electrodes filled with the patch-pipette solution for EPSC recordings described in the Electrophysiological recordings section, the SG neurons were confirmed to be responsive to both non-noxious and noxious stimuli in the voltage clamp mode at VH of −70 mV. Then, spontaneous excitatory postsynaptic potentials (EPSPs) and evoked EPSPs in response to non-noxious and noxious stimuli were recorded in the current clamp mode. Non-noxious air puff stimuli and noxious pinch stimuli were applied to the site at which a barrage of EPSCs was most greatly evoked by the pinch stimuli in the voltage clamp mode.
In order to evaluate effects of anesthetics on occurrence of IPSCs, anesthesia was induced and maintained with pentobarbital (60 mgkg−1 i.p.) or halothane (1.1%) in oxygen in some experiments, and electrophysiological recordings were made as described above. In some other experiments, anesthesia was induced with urethane (0.8–1.0 g kg−1) so that withdrawal reflexes to pinch stimuli were slightly seen during the data collection period. Then, additional doses (0.2–0.4 g kg−1) of urethane were given through the intraperitoneally implanted catheter. Electrophysiological recordings were made in the same neurons before and after administration of the additional doses of urethane.
The drugs used in the present study were tetrodotoxin (TTX) (Sigma, St. Louis, MO), bicuculline methiodide (bicuculline) (Sigma), strychnine hydrochloride (strychnine) (Sigma), and lidocaine hydrochloride monohydrate (lidocaine) (Astra-Zeneca, Osaka, Japan). TTX, bicuculline, and strychnine were dissolved in Krebs solution and were perfused over the surface of the spinal cord according to a previously described method.15,17,18 In some animals, 10 µl of lidocaine (2%) was applied to the cervical spinal cord through a 3-cm-long piece of PE-10 polyethylene tube inserted intrathecally at the cisterna magna and advanced to the caudal aspect of the cervical spinal cord according to a previously described method. 24 Each injection was flushed with 10 µl of intrathecal saline. At the end of the experiment, the location of the distal end of the intrathecal catheter was verified by independent post-mortem examination of the rats. Each animal received an intrathecal injection of 10 µl of brilliant green ink and each injection was flushed with 10 µl of intrathecal saline. Each rat was examined for the existence of dye only in the cervical spinal cord. All catheters were found to be placed correctly.
Data analysis
Numerical data are expressed as means ± SEM. Unpaired Student’s t test and one-way analysis of variance with the Fisher’s PLSD post-hoc test were used for parametric statistical analysis. The Mann–Whitney U test was used for nonparametric statistical analysis. The frequencies of proportions of cell types with respect to response properties to non-noxious and noxious stimuli were statistically analyzed by the chi-square test. P < 0.05 was considered significant. In all cases, n refers to the number of neurons studied.
Results
Passive membrane properties in control and hemisected animals and change in spontaneous firing following hemisection
Electrophysiological properties of SG neurons obtained from control animals and hemisected animal.

Note. SG: substantia gelatinosa; RMP: resting membrane potential; IR: input resistance. Cell location was determined as the distance of the electrode tip from the dorsal surface of the spinal cord; RMPs and IRs were determined using a K-gluconate patch-pipette solution. There was no significant difference in cell location, RMP, or IR between SG neurons obtained from control animals and hemisected animals (hemisection) in which the spinal cord was hemisected between the T13 and L1 segments of the spinal cord.
Excitatory synaptic transmission in control and hemisected animals
Amplitudes and frequencies of spontaneous and evoked EPSCs.
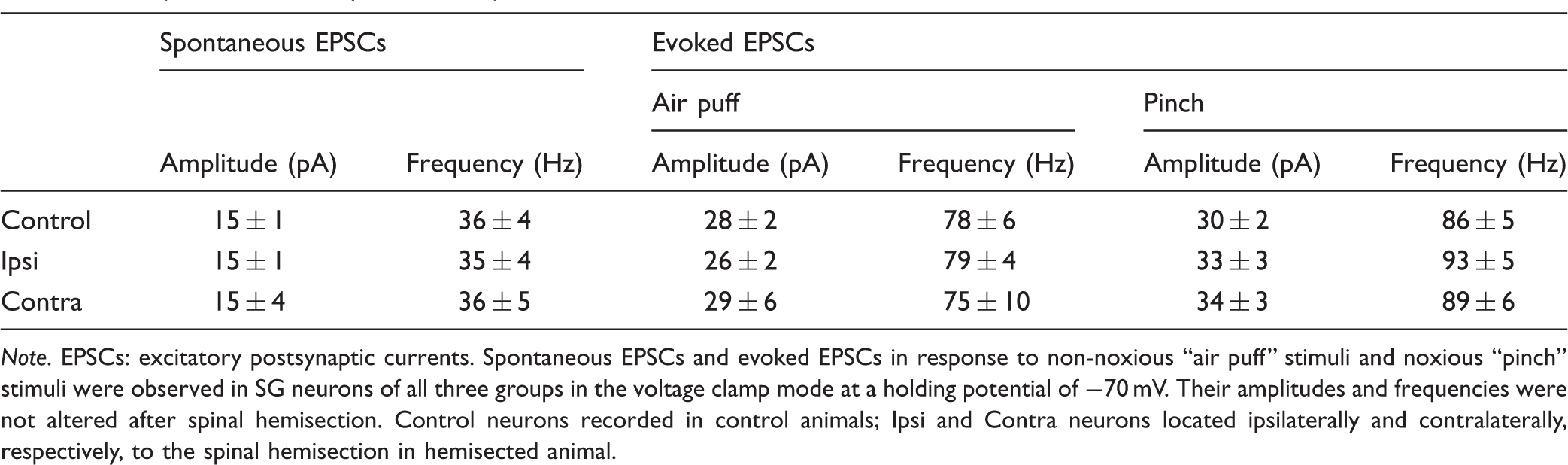
Note. EPSCs: excitatory postsynaptic currents. Spontaneous EPSCs and evoked EPSCs in response to non-noxious “air puff” stimuli and noxious “pinch” stimuli were observed in SG neurons of all three groups in the voltage clamp mode at a holding potential of −70 mV. Their amplitudes and frequencies were not altered after spinal hemisection. Control neurons recorded in control animals; Ipsi and Contra neurons located ipsilaterally and contralaterally, respectively, to the spinal hemisection in hemisected animal.
Changes in sIPSCs and evoked IPSCs following hemisection
One of the other possibilities for the increased spontaneous firing rate in SG neurons is a change in inhibitory synaptic transmission. We therefore tested inhibitory synaptic responses evoked in SG neurons under the voltage clamp mode (VH = 0 mV). The majority of SG neurons (44%) in control rats had large amplitude and high-frequency sIPSCs, which were blocked by spinal application of bicuculline (20 µM) and strychnine (4 µM) by perfusion of Krebs solution (data, not shown). In contrast, the majority of SG neurons (66%) of hemisected animals had relatively small-amplitude and low-frequency sIPSCs (traces in Figure 3(a)). The mean amplitude and frequency of the sIPSCs recorded from SG neurons in control animals (n = 36) were 48 ± 4 pA and 76 ± 7 Hz, respectively. Those obtained from ipsilateral SG neurons in hemisected animals (amplitude, 33 ± 2 pA; frequency, 43 ± 4 Hz; n = 23) were significantly smaller and lower, respectively, than those in control animals (P < 0.01) (left graph in Figure 3(a)). In SG neurons contralateral to the hemisection site, the mean amplitude and frequency of the sIPSCs (38 ± 3 pA and 55 ± 6 Hz, n = 16) were also smaller and lower, respectively, than those in control animals. However, there was no statistically significant difference between the two groups (P = 0.12 and 0.059, respectively). The proportions of sIPSCs with large amplitudes (>50 pA) were less in ipsilateral SG neurons in hemisected animals than in control animals (P < 0.01, right graph in Figure 3(a)). When TTX (1 µM) was applied to the surface of the spinal cord in type 1 neurons (n = 6) with large amplitude and high-frequency sIPSCs (as described below) obtained from control animals, IPSCs were suppressed in a reversible manner (upper trace and graph in Figure 3(b)). When cervical application of lidocaine (2%, 10 µl) was performed in type 1 neurons (n = 4) with large amplitude and high-frequency sIPSCs obtained from other control animals, sIPSCs were also greatly suppressed in a reversible manner (lower trace and graph in Figure 3(b)). The amplitude but not frequency was significantly decreased by spinal application of TTX (P < 0.01 vs. before application) and cervical application of lidocaine (P < 0.05 vs. before application) (Figure 3(c)).
Examples of two patterns of spontaneous inhibitory postsynaptic currents (sIPSCs) seen in SG neurons under voltage clamp conditions at a holding potential of 0 mV, and drug actions on sIPSCs. (a) Large amplitude and high-frequency sIPSCs, which could not be elicited by non-noxious or noxious mechanical stimuli, were observed in the majority of SG neurons (44%) in control animals (upper traces). Relatively small-amplitude and low-frequency sIPSCs werre seen in the majority of SG neurons (66%) in hemisected animals (lower traces). The frequency of sIPSCs in SG neurons in control animals was larger than those in SG neurons located in the ipsilateral side to the lesion in hemisected animals (left graph). Proportions of SG neurons exerting sIPSCs with amplitudes above 50 pA was also higher in control than that in hemisected animals (right graph). **P < 0.01 vs. control. Data are shown as means ± S.E.M. (b) Spinal application of tetrodotoxin (TTX, 1 µM) to the recording site by perfusion of Krebs solution reversibly abolished large amplitude spontaneous IPSCs (sIPSCs) in SG neurons in control animals (upper trace). Application of lidocaine (2%, 10 µl) onto the dorsal surface of the cervical spinal by a PE-10 catheter (see text) reversibly reduced amplitudes of sIPSCs with large amplitude and high-frequency in SG neurons in control animals (lower trace). Cumulative probability histograms show deceases in amplitude (upper panel) of sIPSCs after application of TTX to the recording site and those in amplitude of sIPSC after cervical application of lidocaine (right two graphs). (c) Relative amplitude (upper) and frequency (lower) of sIPSCs after application of TTX to the recording site and cervical application of lidocaine were calculated as the percentages of those before the applications. Application of TTX and cervical application of lidocaine significantly reduced the amplitude of sIPSCs (**P < 0.01 and *P < 0.05, respectively, vs. before drug application). TTX, application of TTX to the recording site; LID, cervical application of lidocaine (2%, 10 µl).
Classification using peripheral cutaneous stimuli proceeded for neurons after assessment of their sIPSCs. With respect to evoked IPSCs, SG neurons could be divided into the following four types by response to non-noxious (air puff) and noxious (pinch) mechanical stimuli applied to the skin in the present study (Figure 4(a)). SG neurons were classified as type 1 if the neurons had large amplitude (>50 pA) and high-frequency (>20–30 Hz) sIPSCs and were not elicited IPSCs in response to non-noxious “air puff” stimulation or noxious “pinch” stimulation in any portions of the body. SG neurons were classified as type 2 if the neurons had large amplitude and high-frequency sIPSCs and responded poorly to both air puff and pinch stimuli. In those neurons, IPSCs evoked by mechanical stimuli seemed be masked because of their sIPSCs. SG neurons were classified as type 3 if the neurons had small-amplitude and low-frequency sIPSCs and responded maximally to non-noxious stimuli. The type 3 neurons also exerted large amplitude and high-frequency IPSCs evoked by noxious stimuli; however, these responses were mostly restricted to the beginning and end of pinch stimuli and were qualitatively similar to the corresponding brush responses.
18
SG neurons were classified as type 4 if the neurons had small-amplitude and low-frequency sIPSCs and responded to non-noxious stimuli and noxious stimuli. In contrast to type 3 neurons, type 4 neurons exerted persistent IPSCs during noxious pinch stimuli.
Typical response profiles of mechanical stimuli-evoked inhibitory postsynaptic currents (IPSCs) seen in SG neurons. (a) Responses of IPSCs from four different neurons during application of air puff stimuli (left traces) and pinch stimuli (right traces) in the voltage clamp mode (VH = 0 mV). Bars above the traces show the duration of the stimulation. Responses to non-noxious and noxious stimuli were not observed in type 1 neurons because of their high-frequency and large amplitude sIPSCs. Although type 2 neurons exert relatively high-frequency and large amplitude sIPSCs, responses were consistently elicited during non-noxious and noxious stimuli. In type 3 neurons, non-noxious stimuli produced a barrage of IPSCs that persisted during the stimuli, while pinch stimuli evoked IPSCs only at the beginning and end of stimuli. In type 4 neurons, both non-noxious stimuli and noxious stimuli produced a barrage of IPSCs that persisted during the stimuli. Bars above the traces show the duration of the stimuli. (b) Proportions of types of SG neurons in animals in which the spinal cord was hemisected (Hemisection) and left intact (Control). The proportion of type 1 neurons was smaller in SG neurons recorded in hemisected animals than in control animals, although there was no statistical significance (P = 0.067). The proportion of type 4 neurons was significantly larger in SG neurons recorded in hemisected animals than in control animals (P < 0.01).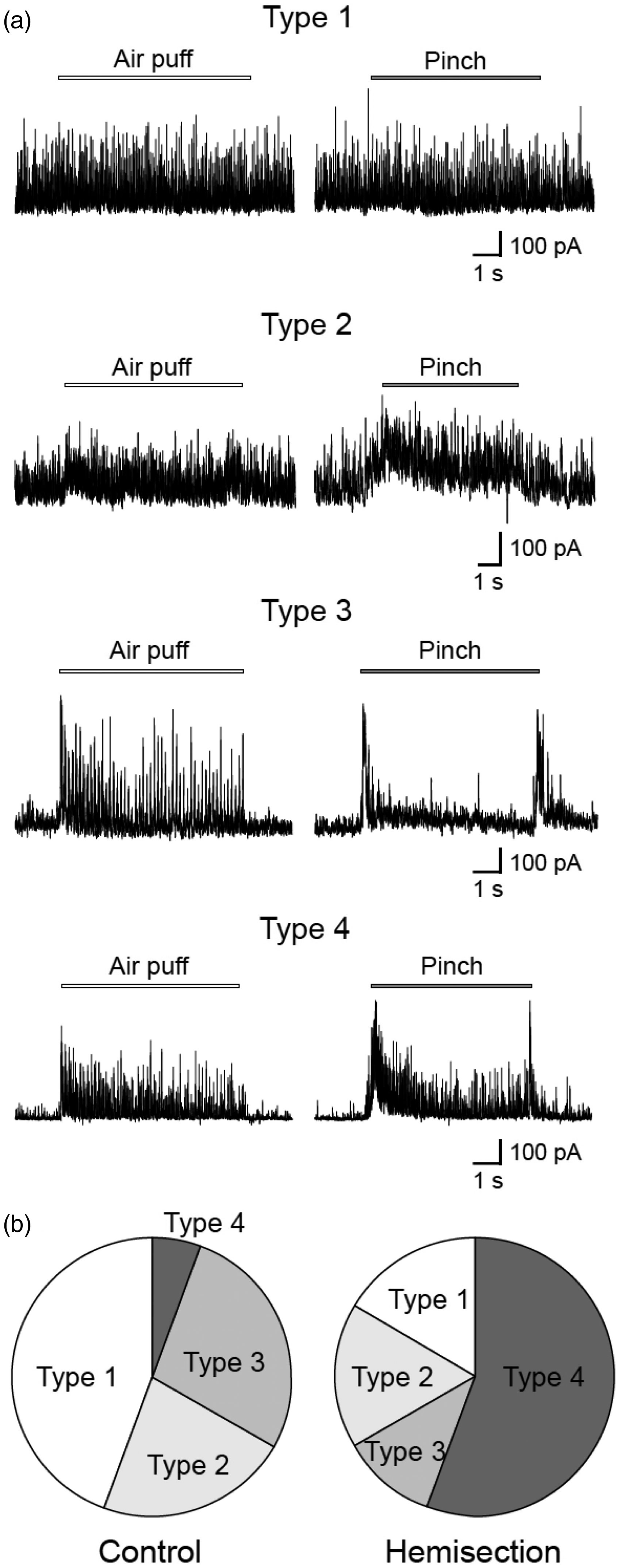
The proportions of type 1, type 2, type3, and type 4 neurons in SG neurons (n = 36) in control animals were 44%, 22%, 28%, and 6%, respectively (Figure 4(b)). The proportions of type 1, type 2, type3, and type 4 neurons in SG neurons located ipsilaterally to the spinal hemisection (n = 28) were 17%, 17%, 11%, and 55%, respectively. The proportions of type 1, type 2, type 3, and type 4 neurons in SG neurons located contralaterally to the spinal hemisection (n = 8) were 0%, 38%, 13%, and 50%, respectively. The proportion of type 4 neurons was significantly larger in SG neurons recorded in hemisected animals than that in SG neurons recorded in control animals (P < 0.01). The total proportions of type 3 and type 4 neurons, in which a barrage of IPSCs were elicited by non-noxious stimulation and/or noxious stimulation, were significantly larger in hemisected animals (66%) than in control animals (34%) (P < 0.05).
Anesthetic effects on passive membrane properties and IPSCs in intact animals
We examined passive membrane properties and IPSCs under anesthetics with halothane and pentobarbital, and compared with those recoded under the urethane anesthesia. The animals were anesthetized with halothane (1.1%) in oxygen (n = 21) or i.p. pentobarbital (60 mgkg−1, n = 18). The mean depths of location, RMP, and IR of SG neurons recorded in the halothane group and pentobarbital group were 101 ± 21 and 93 ± 25 µm, −62.3 ± 3.1 and −61.1 ± 1.9 mV, and 402 ± 75 and 361 ± 47 MΩ, respectively. Compared to those in the animals anesthetized with urethane (see control group in Table 1), there was no significant difference between the depth, RMP, or IR in the animals anesthetized with a different anesthetic (P > 0.2). In the voltage clamp mode (VH = 0 mV), large amplitude and high-frequency sIPSCs, which could not be elicited by mechanical stimuli, were also seen in 53% and 49% of SG neurons in the animals anesthetized with halothane and pentobarbital, respectively, and there were no significant difference in the proportions of IPSC types in the animals anesthetized with urethane, halothane, and pentobarbital. The mean amplitudes of sIPSCs recorded from SG neurons in the animals anesthetized with urethane, halothane, and pentobarbital were 48 ± 4, 49 ± 3, and 41 ± 4 pA, respectively; and the mean frequencies of sIPSCs recorded from SG neurons in the three groups were 76 ± 7, 74 ± 9, and 72 ± 9 Hz, respectively. There was also no significant difference between the three groups in amplitudes or frequencies of sIPSCs (P > 0.1). We further examined effect of supplemental dose of urethane on sIPSCs. In a longtime period of recording under urethane anesthesia from single type 1 SG neuron, withdrawal movements were seen in the animals. We then administered supplemental dose of urethane (0.2–0.4 g kg−1) until withdrawal movements disappeared. sIPSCs evoked in the same neurons were not changed after the administration (Figure 5(a)). The proportions of type 1, type 2, type3, and type 4 neurons in SG neurons (see above) recorded under anesthetics with halothane and pentobarbital were also different from those recorded under anesthetic with urethane (Figure 5(b)).
Effect of additional injection of urethane on sIPSCs and response profiles of mechanical stimuli-evoked IPSCs recorded under anesthetics with halothane and pentobarbital. (a) An example of sIPSCs seen in an SG neuron in a rat under urethane anesthesia. An arrow indicates additional injection of intraperitoneal urethane (0.3 g kg−1) which abolished withdrawal reflexes to pinch. Lower traces are shown in an expanded time course. Cumulative probability histograms did not show significant changes in amplitude or frequency of sIPSCs after additional injection of urethane. (b) Proportions of types of SG neurons in animals anesthetized with halothane (1.1%) and pentobarbital (60 mg). There were no differences among proportions of types of SG neurons in animals anesthetized with urethane, halothane, and pentobarbital.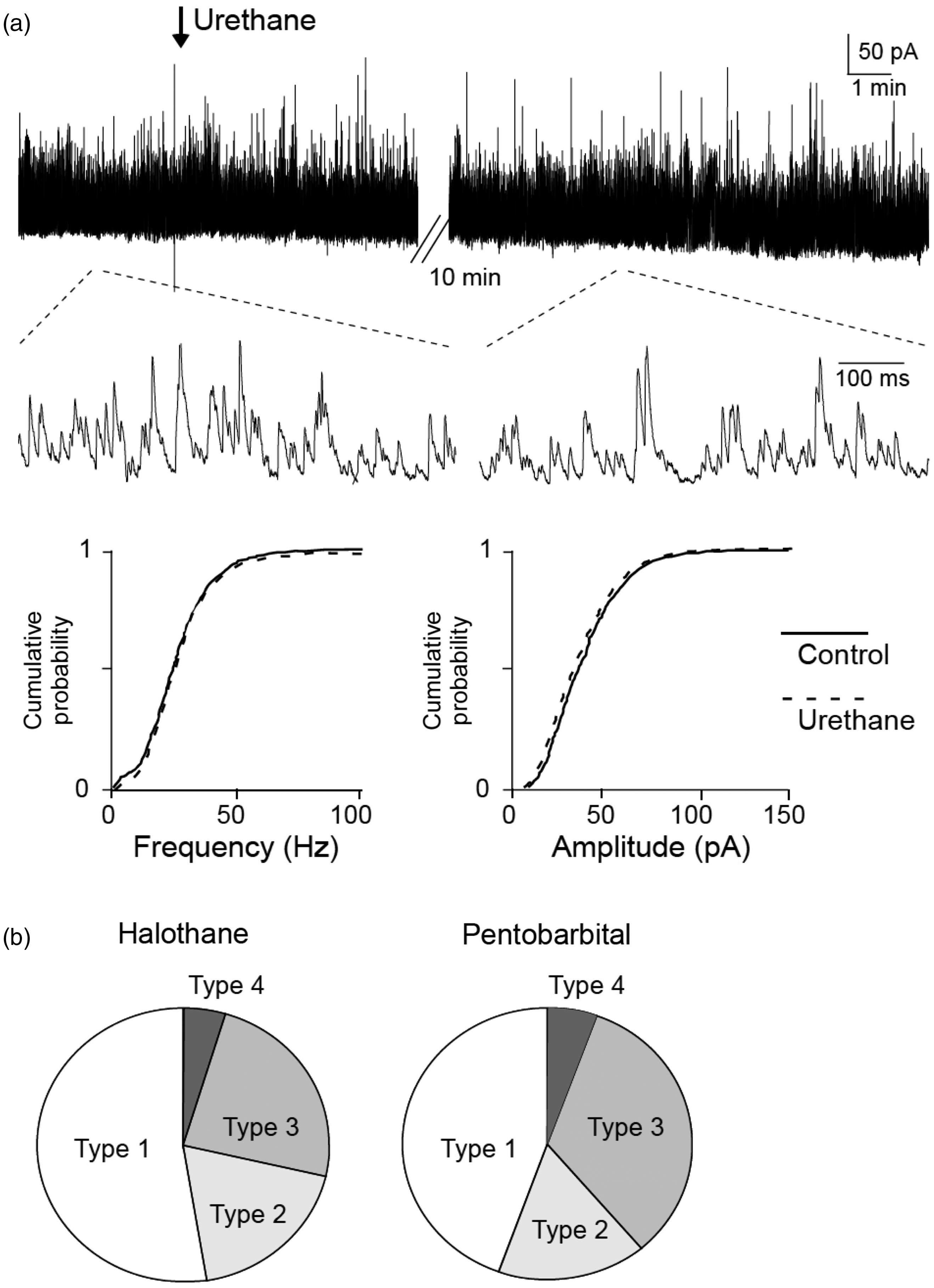
Increased responses to non-noxious and noxious stimuli after spinal hemisection
With respect to evoked responses to both non-noxious and noxious stimuli, it has been reported that SG neurons can be classified as several types according to their response properties of EPSPs and APs to mechanical stimuli on the receptive field.
19
Neurons are classified as multi-receptive if they exhibit APs in response to non-noxious “air puff” stimuli and maximally in response to noxious “pinch” stimuli. Neurons are classified as nociceptive if they respond only to noxious “pinch” stimuli. Neurons are classified as subthreshold neurons if they respond to air puff stimuli and pinch stimuli with small depolarizations that fail to reach the AP threshold (Figure 6(a)). These types of SG neurons have previously been reported in the SG.16,19 There was a trend for subthreshold neurons to decrease and multi-receptive neurons to increase after hemisection (Figure 6(b)). The proportions of subthreshold, nociceptive, and multi-receptive neurons in control animals were 36% (5/14), 43% (6/14), and 21% (3/14), respectively. The proportions of those neurons in hemisected animals were 21% (4/19), 37% (7/19), and 42% (8/19), respectively.
SG neurons exhibited several response profiles during non-noxious and noxious mechanical stimuli in the current clamp mode. (a) Voltage responses from three different neurons during application of air-puff stimuli (left trace) and pinch stimuli (right trace). Bars above the traces show the duration of the stimulation. Upper traces, multi-receptive neurons exhibited action potential (AP) firing in response to non-noxious air puff stimuli and noxious pinch stimuli. Multi-receptive SG neurons were commonly more sensitive to pinch stimuli than to air puff stimuli. Middle traces, nociceptive neurons only exhibited AP firing to pinch stimuli with or without subthreshold depolarizations during air puff stimuli. Lower traces, subthreshold neurons responded to air puff stimuli and pinch stimuli with small depolarizations that failed to reach the AP threshold. (b) The total proportion of subthreshold and nociceptive neurons was smaller in hemisected animals (58%, 11/19) than in control animals (79%, 11/14). In contrast, the proportion of multi-receptive neurons was larger in hemisected animals (42%, 8/19) than in control animals (21%, 3/14).
Discussion
Tonic descending inhibition on SG neurons
The average amplitude and frequency of sIPSCs of SG neurons in previous studies using in vitro slice preparations were 5–30 pA and 4–13 Hz, respectively,20–22 whereas those of SG neurons in control animals in this study were 48 ± 4 pA and 76 ± 7 Hz, respectively. This was because there was a relatively large proportion of SG neurons (44%) exhibiting large amplitude (>50 pA) and high-frequency sIPSCs (>20–30 Hz). Such sIPSCs of SG neurons were also observed in a previous in vivo study by other workers but have not been analyzed in detail. 23 The large amplitude of the sIPSCs was significantly reduced or abolished by superfusing with TTX, application of lidocaine onto the dorsal surface of the cervical spinal cord, and superfusing with strychnine and bicuculline. The proportion of SG neurons exerting such sIPSCs was relatively small (17%) in hemisected animals. The results suggest that the large amplitude and high-frequency sIPSCs seen in SG neurons are a result of tonic activation of descending pathways and subsequent occurrence of AP firings in GABAergic and glycinergic neurons innervating SG neurons.
Descending noraderenergic and serotonergic (5-hydroxytryptamine: 5-HT) fibers originating from A5, A6, and A7 in the pons and rostral ventromedial medulla including the nucleus raphe magnus, respectively, terminate in superficial dorsal horn neurons including SG neurons25–28 and can reduce nociceptive transmission in the SG by excitatory actions of 5-HT and noradrenaline at GABAergic and glycinergic inhibitory interneurons in the SG.29–31 It is also known that the noradrenergic and serotonergic pathways project both ipsilaterally and contralaterally at the segmental level of termination to the SDH.32,33 In addition, SG neurons directly receive GABAergic fibers and possibly glycinergic fibers from supraspinal sites including the rostral ventromedial medulla.34–36 Propriospinal GABAergic and/or glycinergic neurons also contribute to synaptic inhibition within the spinal cord including the SG.37–40 It appears that GABAergic/glycinergic sIPSCs with large amplitudes and high frequencies seen in SG neurons in the present study reflect a total of tonic inhibition by descending pathways originating from supraspinal sites through dorsolateral portions of the spinal cord. Since application of TTX into the lumbar spinal cord (the recording site of SG neurons) reduced the amplitude of sIPSCs more greatly than cervical application of lidocaine did in this study, in addition to the brain, rostral spinal cord may play an important role in tonic inhibition by descending pathways to the lumbar spinal cord.
Since several anesthetics may exert their actions via activation of descending inhibitory systems41,42 and since surgical exposure of the spinal cord may tonically activate the descending inhibition from the nucleus gigantocelluaris pars alpha, 9 it is possible that the presence of large amplitude and high-frequency sIPSCs was caused by urethane anesthesia and/or surgical injuries in this study. However, the proportions of SG neurons exhibiting large amplitude and high-frequency sIPSCs were not significantly different among animals anesthetized with urethane, halothane, or pentobarbital. Furthermore, although additional injection of urethane abolished withdrawal reflexes in lightly anesthetizing rats, additional administration of urethane did not change the amplitudes or frequencies of sIPSCs in the present study, despite the fact that urethane has anesthetic and analgesic effects. 43 Thus, the large amplitude and high-frequency sIPSCs seen in our study seem to have resulted not from anesthesia or surgical preparations but from the nature of the descending inhibitory systems.
Inhibitory synaptic transmission evoked by non-noxious and noxious stimuli
With respect to response properties of IPSCs evoked by non-noxious and noxious stimuli, type 1 SG neurons have large amplitude and high-frequency sIPSCs and are not responsive to mechanical stimuli. Type 4 SG neurons have relatively small-amplitude and low-frequency sIPSCs and respond to both non-noxious and noxious stimuli, while type 3 SG neurons have relatively small-amplitude and low-frequency sIPSCs and respond only to non-noxious stimuli. There may thus be subpopulations of SG neurons for the modes of receiving inputs from the periphery and from the supraspinal structures. In the present study, the proportions of type 2 and type 3 SG neurons were not significantly different in control animals and hemisected animals, whereas the proportions of type 1 SG neurons and type 4 SG neurons were much smaller and larger, respectively, in the hemisected animals than in the control animals. It is thus possible that some type 1 SG neurons may functionally change to type 4 SG neurons after disruption of the descending inhibitory influence.
Results of previous studies have suggested that primary-afferent Aδ-fibers and thick C-fibers, both of which seem to transmit non-noxious sensory information, innervate glycinergic and/or GABAergic interneurons, the activation of which results in the production of IPSCs in SG neurons.18,20,44 However, it has also been shown that activation of NK1 receptors enhances inhibitory synaptic transmission of SG neurons by increasing their amplitudes and frequencies of sIPSCs, 45 although substance P is released from nociceptive C fiber terminals and evokes excitatory responses in a subset of SG neurons.46,47 Thus, nociceptive primary afferents may also terminate on glycinergic and/or GABAergic inhibitory interneurons in the spinal cord. In the present study, type 4 SG neurons, in which IPSCs were elicited by non-noxious and noxious stimuli, were commonly seen in hemisected animals but rarely seen in control animals. Thus, it is likely that a subset of SG neurons receives inhibitory inputs from GABAergic/glycinergic interneurons to which both non-noxious and noxious primary afferents innervate. Tonic descending influences from supraspinal structures may mask such nociception-evoked IPSCs in a subpopulation of SG neurons in a normal state.
Excitatory synaptic transmission and AP generation in SG neurons after spinal hemisection
It has been postulated that descending serotonergic pathways may not only strengthen inhibitory postsynaptic transmission in the SG by excitatory actions of 5-HT at GABAergic and glycinergic inhibitory interneurons in the SG but also potentiate GABAergic inhibition of primary afferent terminals.48,49 Elimination of the descending pathways by spinal hemisection may thus exaggerate spontaneous and/or evoked EPSCs by presynaptic mechanisms. However, this was not the case in the present study because there were no significant differences between the animal groups in the frequencies and amplitudes of spontaneous EPSCs and evoked EPSCs in response to non-noxious and noxious stimuli. It has also been suggested that descending noradrenergic and/or serotonergic pathways act partly as facilitatory pathways to SG neurons. 50 Thus, with regard to excitatory synaptic transmission in SG neurons, disruption of descending fibers by spinal hemisection may induce loss of descending inhibitory and facilitatory effects on SG neurons. As a result, in contrast to IPSCs, spontaneous EPSCs and evoked EPSCs may not have been changed after spinal hemisection.
In the present study, there was a relatively large proportion (36%) of subthreshold neurons in control animals. These functionally “silent” neurons, which have also been described in the SG,20,23 may be a result of the tonic descending inhibition, since non-noxious and noxious stimuli could induce a barrage of EPSCs even in these subthreshold neurons in the present study (Figure 2(a)). These subthreshold SG neurons may become functional when the descending inhibition is disrupted. On the other hand, in the present study, spontaneous AP firing rate of SG neurons after spinal hemisection was significantly higher in hemisected animals than in control animals. An increase in spontaneous AP firing rate in laminae V-VI neurons is observed immediately after hemisection of the spinal cord and remains high for several weeks7,8 and then gradually decreases to the basal level within the next several weeks.7,10
The proportions of subthreshold neurons and multi-receptive neurons were smaller and larger, respectively, in hemisected animals than in control animals, suggesting that some subthreshold neurons may have functionally changed to multi-receptive neurons after spinal hemisection. Furthermore, 25% of multi-receptive and nociceptive SG neurons exerted after-discharge AP firing in hemisected animals, while such after-discharge AP firing was not seen in control animals. Taken together with the fact that hemisection did not induce strengthening of excitatory synaptic transmission in SG neurons in this study, it seems that the main influence of acute spinal hemisection on SG neurons is elimination of inhibitory transmission, not potentiation of excitatory transmission. Spontaneous activity and responsiveness of SG neurons to mechanical stimuli are thus increased by elimination of the descending inhibitory influence.
In the present study, recordings from SG neurons were performed at an early phase (several hrs) but not at a late phase (several weeks) after spinal hemisection. This is because adhesion to the connective tissue and bleeding due to richness in vascularity in the spinal cord made stable recordings difficult at such a chronic phase in our pilot study. In addition to some common mechanisms that contribute to hyperexcitation of SG neurons both at the early and late phases after SCI, different mechanisms may be involved in development of chronic central pain after SCI. One example might be that is associated with loss of presynaptic inhibition. Although loss of presynaptic inhibition was not seen at the early phase after spinal hemisection in this study, SCI may subsequently induce a devastating loss of descending inhibitory fibers such as serotonergic fibers on primary afferent terminals at the late phase, possibly resulting in loss of presynaptic inhibition by 5-HT. 51 Thus, further study in which recordings are performed in chronically prepared animals after spinal hemisection and/or denervation of serotonergic and adrenergic descending inhibitory fibers by neurotoxins may be required to determine the mechanisms of development of SCI-induced central pain.
Conclusions
SG neurons would receive tonic inhibition by spinal inhibitory interneurons which generate persistent APs. Spinal hemisection results in hyperexcitation of SG neurons at least in part by eliminating the tonic descending control of spinal inhibitory interneurons from supuraspinal levels, which might be a common mechanism of SCI-induced pain.
Footnotes
Authors’ contributions
YK carried out the electrophysiological recordings of the “spinal hemisection” section, analyzed data, and drafted the paper. MK carried out the electrophysiological recordings, conceived and designed the research, and finished the final version of the manuscript. HF assisted in the electrophysiological recordings and helped to finish the final version of the manuscript. TI and ST carried out the electrophysiological recordings of the “effect of anesthetics” section. AN and MY conceived the study, participated in its coordination, and drafted the final version of the manuscript. All authors have read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant-in-aid for scientific research from Japan Society for the Promotion of Science (Tokyo, Japan) to M. K. (grant No. 17390431, 21390432, and 24390365).
