Abstract
Ventilation plays a crucial role in influencing airflow and particle dispersion, highlighting the importance of effective systems to mitigate the spread of viruses in indoor spaces. While most research has concentrated on non-residential buildings, less attention has been given to optimizing ventilation in residential settings. This study examines a typical ventilation system in a detached house in British Columbia, Canada, using Computational Fluid Dynamics simulations to evaluate its effectiveness in reducing airborne infection risks. The study investigates eight different scenarios, varying diffuser locations (near the ceiling and floor), two types of ventilation rates based on standards, and the positioning of the infected individual while considering flow, heat, and particle dynamics. The concentration of injected and distributed particles is used to assess infection probabilities as an additional risk indicator. In addition, the Wells-Riley model is applied to quantify the infection probability. The results indicate that the location of the infected person and diffuser placement significantly impact particle dispersion and infection risk, with near-ceiling outlets generally reducing concentrations more effectively than near-floor outlets. Higher ventilation rates decrease particle concentration when diffusers are near the ceiling but can increase concentrations with near-floor diffusers due to turbulence. Optimizing diffuser placement and ventilation rates is crucial for minimizing airborne transmission in residential settings.
Keywords
Introduction
The airborne transmission of COVID-19 significantly impacts indoor air quality, especially in poorly ventilated spaces where infected aerosols can linger or travel beyond typical distances. The World Health Organization emphasizes the importance of enhancing indoor ventilation to mitigate the virus’s spread in enclosed areas. In response, governments have adapted Heating, Ventilation, and Cooling (HVAC) system regulations to address the pandemic’s challenges (Zheng et al., 2021). Epidemiological studies highlight that direct interactions, such as face-to-face conversations, coughing, and sneezing, are primary modes of infection transmission (Wiersinga et al., 2020). Shao et al. (Shao et al., 2021) demonstrated that normal breathing emits approximately 11 particles per second while coughing and sneezing release far more, with up to 1 × 104–1.5 × 105 particles per second (Rudnick and Milton, 2003; Lindsley et al., 2012) and 3 × 106 particles per second (Duguid, 1946), respectively. These particles, especially smaller aerosols, can remain airborne longer, increasing infection risk in indoor spaces (Busco et al., 2020; Kohanski et al., 2020). The behavior of these particles, influenced by their size and emission velocity, determines whether they quickly settle or are carried further by indoor airflow (Chao et al., 2009; Tang et al., 2013).
The relationship between ventilation rates in indoor environments and the concentration of infectious particles forms the foundation to assess airborne transmission risks, as demonstrated by the development and adaptation of models such as the Wells–Riley equation, which correlates indoor ventilation with infection probability (Riley et al., 1978; Sun and Zhai, 2020). Computational Fluid Dynamics combined with Discrete Particle Modeling (CFD-DPM) effectively tracks aerosols and is another crucial approach for infection risk assessment. These simulations are essential for determining minimum ventilation rates that balance low contagion risk with energy efficiency (Ferrari et al., 2022). CFD has also been used to model droplet dynamics from coughing and sneezing, which is vital for mitigating respiratory disease transmission in spaces like aircraft cabins and outdoor environments (Kotb and Khalil, 2020; Li et al., 2020). Vuorinen et al. (2020) utilized CFD and Large Eddy Simulation (LES) to model aerosol exposure from speaking, introducing Monte Carlo Modeling for probabilistic risk assessment in public spaces. Table 1 provides a structured summary of relevant CFD studies, focusing on ventilation effectiveness for infection control.
A summary of previous works confirming the ventilation effectiveness.
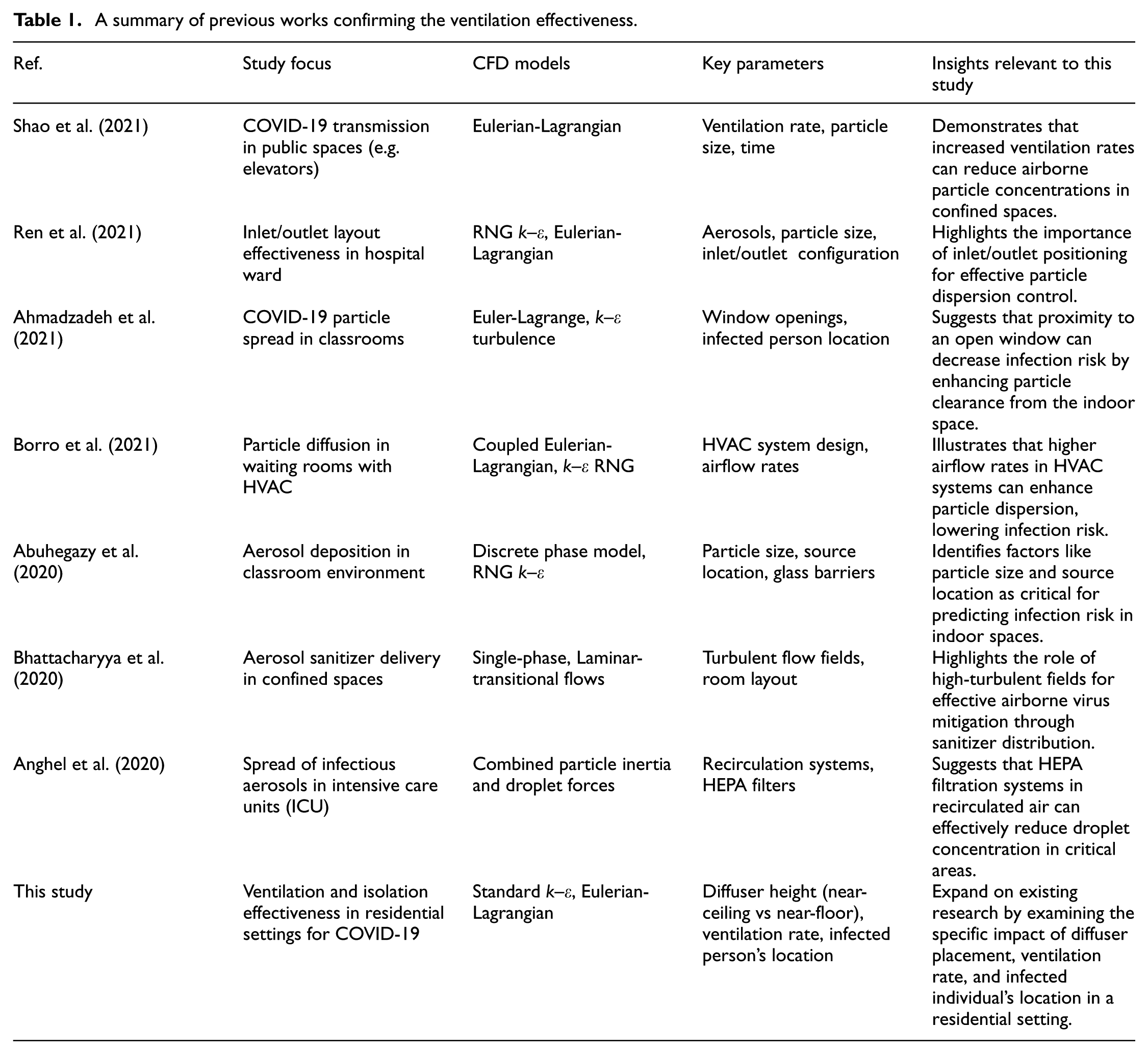
Table 1 establishes a foundation by identifying the CFD models, turbulence methods, and parameters utilized across various studies. Summarizing these aspects shows how different ventilation strategies impact infection risk, offering context for the specific CFD approaches adopted in this study. This study builds upon these previous methodologies by applying similar CFD models in a residential setting, filling a gap in the literature, as most existing research has focused on non-residential spaces. Recent research (Quang et al., 2024) highlights the use of Deep Neural Networks (DNNs) alongside CFD to efficiently predict airflow patterns in residential buildings, aiding ventilation design.
Current CFD studies on infection analysis predominantly focus on commercial or institutional settings, such as hospitals and classrooms, where airflow and particle distribution dynamics vary significantly from those in residential spaces. Moreover, these studies often overlook key variables relevant to smaller, enclosed environments, such as the positioning of diffusers and the spatial arrangement of rooms in a typical house. Additionally, many of these studies focus on high-efficiency ventilation systems, which may not be feasible for residential applications. By addressing these limitations, this study aims to offer practical insights specific to home environments, considering factors like diffuser placement and the location of an infected individual to assess their influence on airborne particle dispersion.
The objectives of this study are to evaluate the effectiveness of four ventilation strategies in a residential setting, specifically a typical detached house in British Columbia, Canada. The objectives can be divided into three sections:
Quantitatively assessing how different diffuser placements (near-ceiling vs near-floor) impact infection risk.
Analyzing the effect of two ventilation rates to understand the role of airflow intensity.
Investigating the influence of the infected individual’s location (living room vs bedroom) on particle spread and infection probability.
These contributions provide critical insights for optimizing residential ventilation system design to mitigate airborne virus transmission, filling a gap in infection control research within home environments.
Problem definition and boundary conditions
The simulation aims to analyze COVID-19 transmission within a detached residential building in Columbia BC, using layouts derived from the BC Housing annual publication (Housing, 2019). The environment is detailed, featuring three occupants, one infected. According to Figure 1, the house includes two bedrooms, a living room, a kitchen, two washrooms, a laundry room, and a storage room. The living room is furnished with sofas and a coffee table. At the same time, the bedrooms have two beds, and the kitchen and washrooms are equipped with essential fixtures such as sinks, toilets, and cabinets, creating a realistic setting for the simulation. Interior boundaries are defined at the entrances to each space. The occupants are modeled as cylinders, a simplification used to analyze the dynamics of respiratory particles, focusing on particles expelled at different velocities during speech, coughing, and sneezing. The mouth diameter is assumed to be 5 cm.
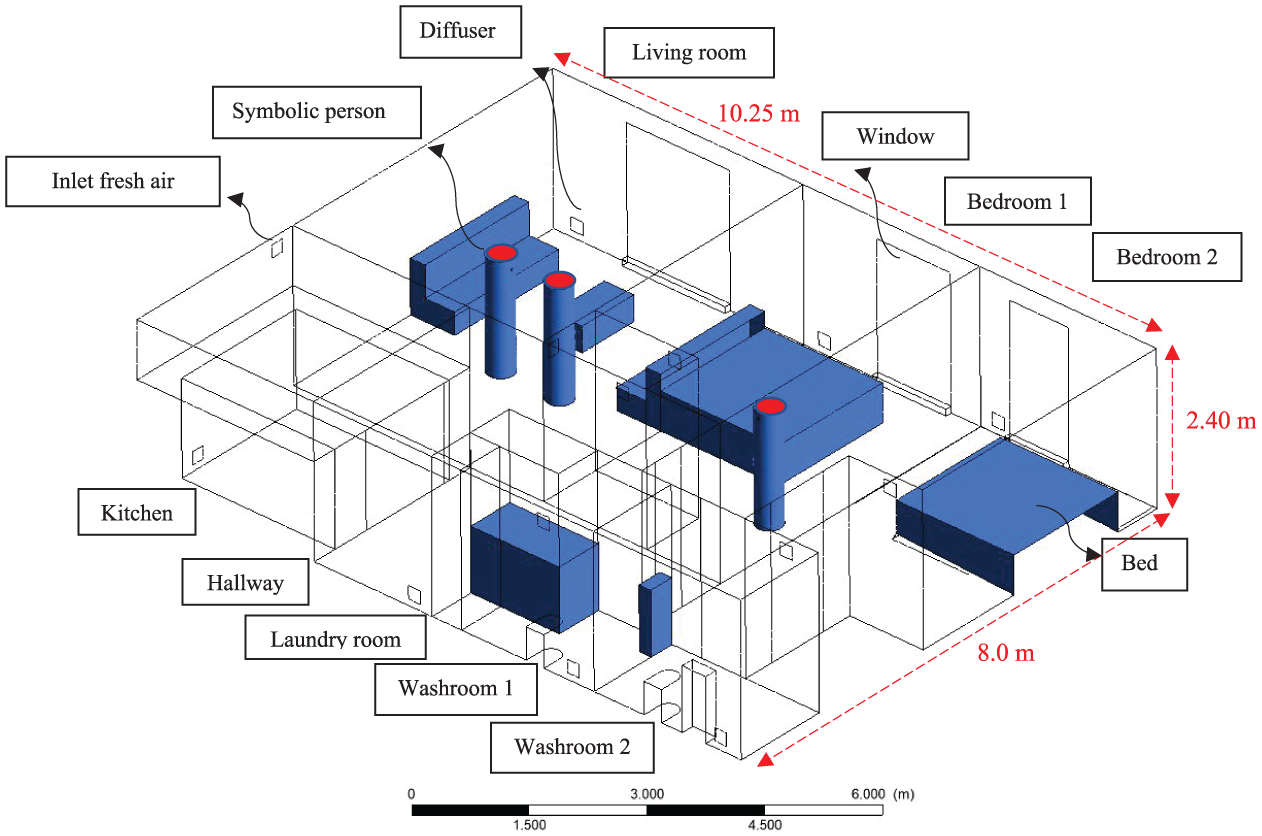
Layout of a residential building with various zones, boundaries, and symbolic individuals.
This study considers four types of ventilation strategies regarding the diffuser’s height and ventilation rate. According to the ASHRAE Standard 62.2-2022 (ASHRAE, 2022b), the required minimum whole-building ventilation rate
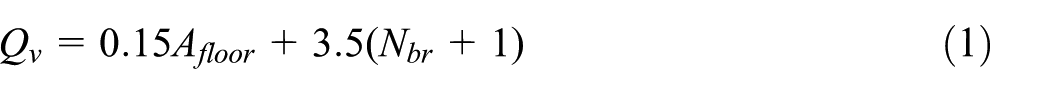
Q v is the total ventilation flow rate (L/s), A floor is the conditioned floor area of the building (m2), and Nbr is the number of bedrooms (N br = 2). For this calculation, the conditioned floor area was 82 m2. Plugging in these values, the total ventilation flow rate was 22.8 L/s (82.08 m3/hour). To ensure that each zone within the building receives an appropriate share of the total ventilation rate, the ventilation rate was distributed proportionally based on the area of each zone relative to the total area of the building. The resulting airflow for each zone was then used to determine the corresponding inlet boundary conditions, expressed in terms of velocity (m/s) based on the calculated flow rates and zone areas. For instance, the velocity for the living room inlet was determined by dividing the flow rate allocated to the living room by the cross-sectional area of the inlet (0.0129 m2), resulting in a velocity of 0.457 m/s. Similar calculations were performed for other zones, such as bedrooms, the kitchen, and washrooms, ensuring the ventilation rates were correctly distributed and translated into appropriate inlet boundary conditions. These calculated velocities are then applied as boundary conditions at the inlets in the CFD model to simulate the ventilation system accurately.
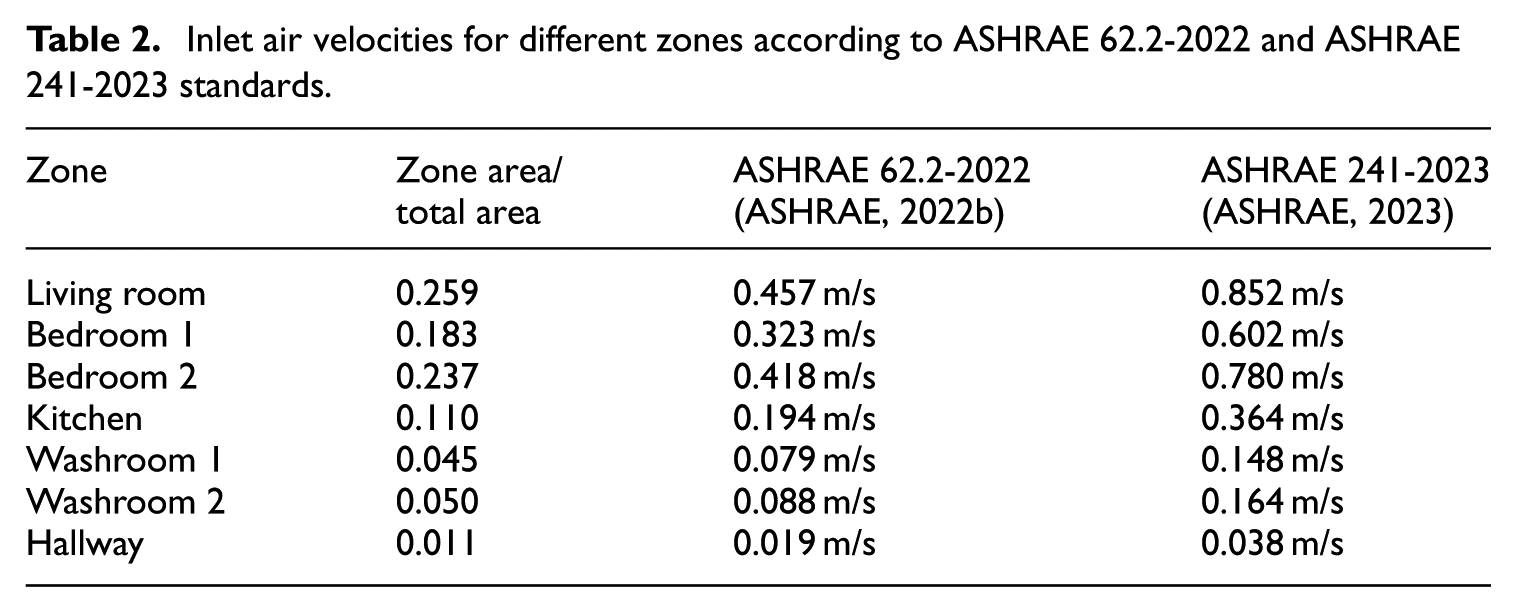
Another approach for finding the minimum ventilation rate per person was applied to various rooms within the residential building based on ASHRAE 241-2023 Control of Infectious Aerosols (ASHRAE, 2023). Utilizing a baseline rate of 50.97 m2/hour per person, and with three occupants, the total ventilation rate was set at 152.91 m2/hour. This rate was then distributed among the rooms based on specific fractional allocations, similar to previous calculations. Tables 2 and 3 present the calculated inlet air velocities in different zones according to the two referenced standards and a summary of other boundary conditions.
Inlet air velocities for different zones according to ASHRAE 62.2-2022 and ASHRAE 241-2023 standards.
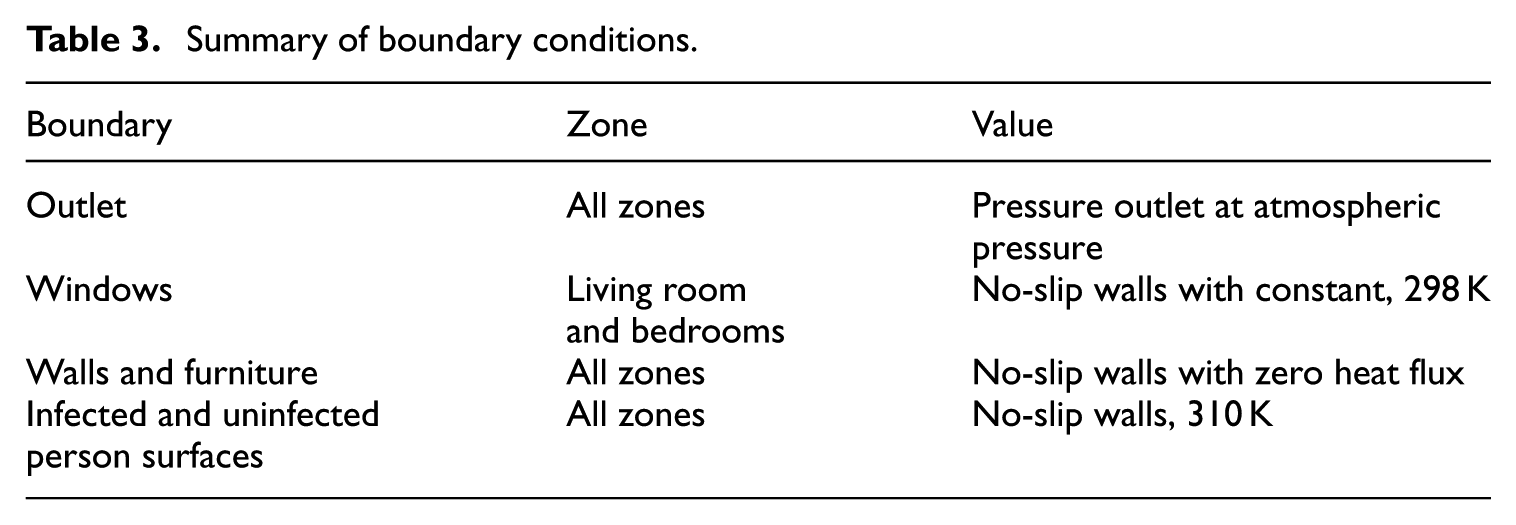
Summary of boundary conditions.
To identify the most conservative scenario regarding the transmission of airborne particles, a comparative analysis was conducted across three distinct activities (speaking, coughing, and sneezing) performed by an infected individual in the living room. By simulating these activities, the study aimed to identify which poses the highest risk regarding particle distribution and potential exposure to other occupants to ensure that subsequent simulations focus on the most critical scenario. Table 4 outlines the different ventilation scenarios considered in the study, specifying the ventilation rates, diffuser locations, and the positions of the infected person within the residential building. It compares scenarios based on the ASHRAE 62.2-2022 and ASHRAE 241-2023 standards, highlighting variations in diffuser placement, either on the ceiling or the floor, and the location of the infected person, either in the living room or Bedroom 1.
Overview of ventilation scenarios, diffuser locations, and infected person’s position.
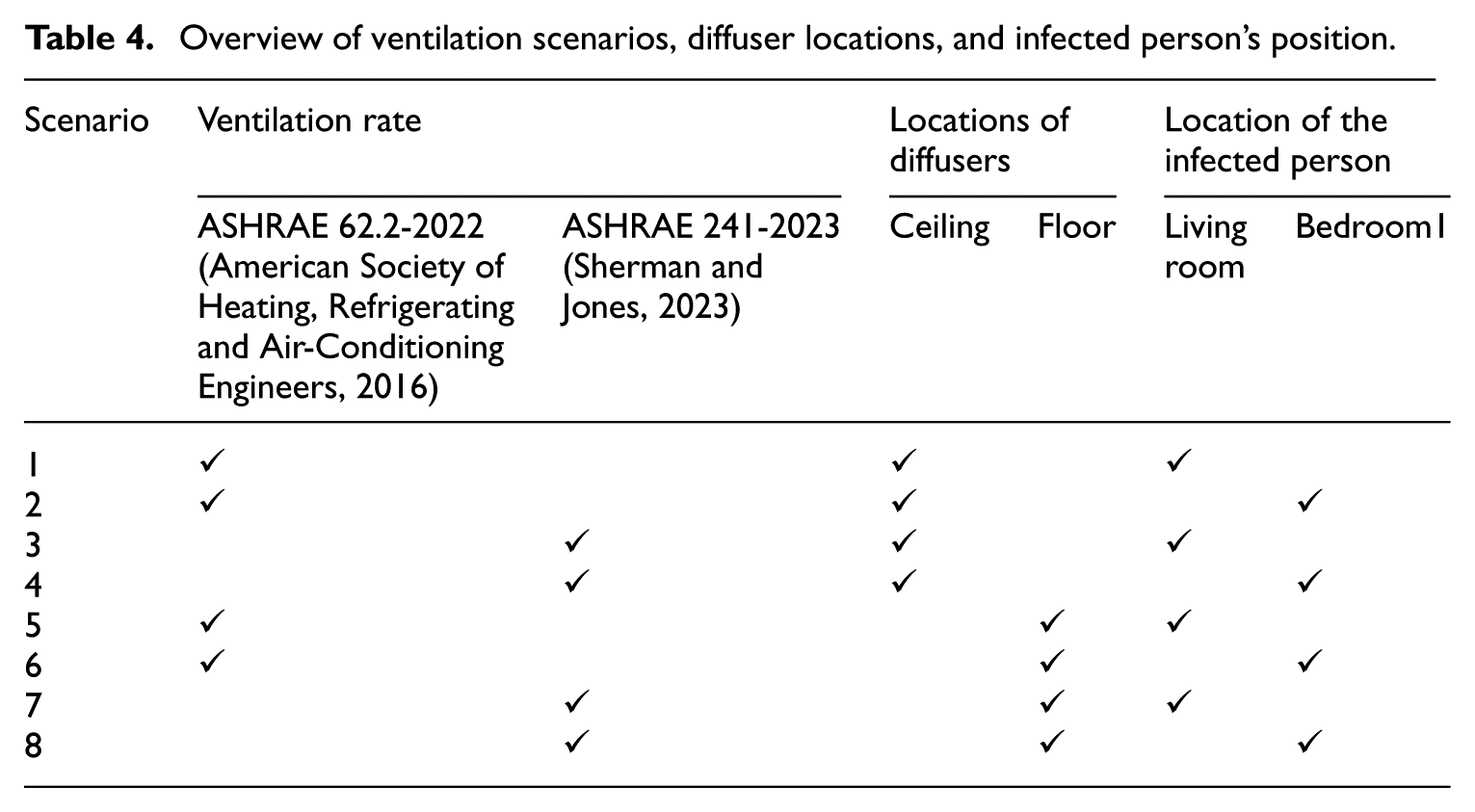
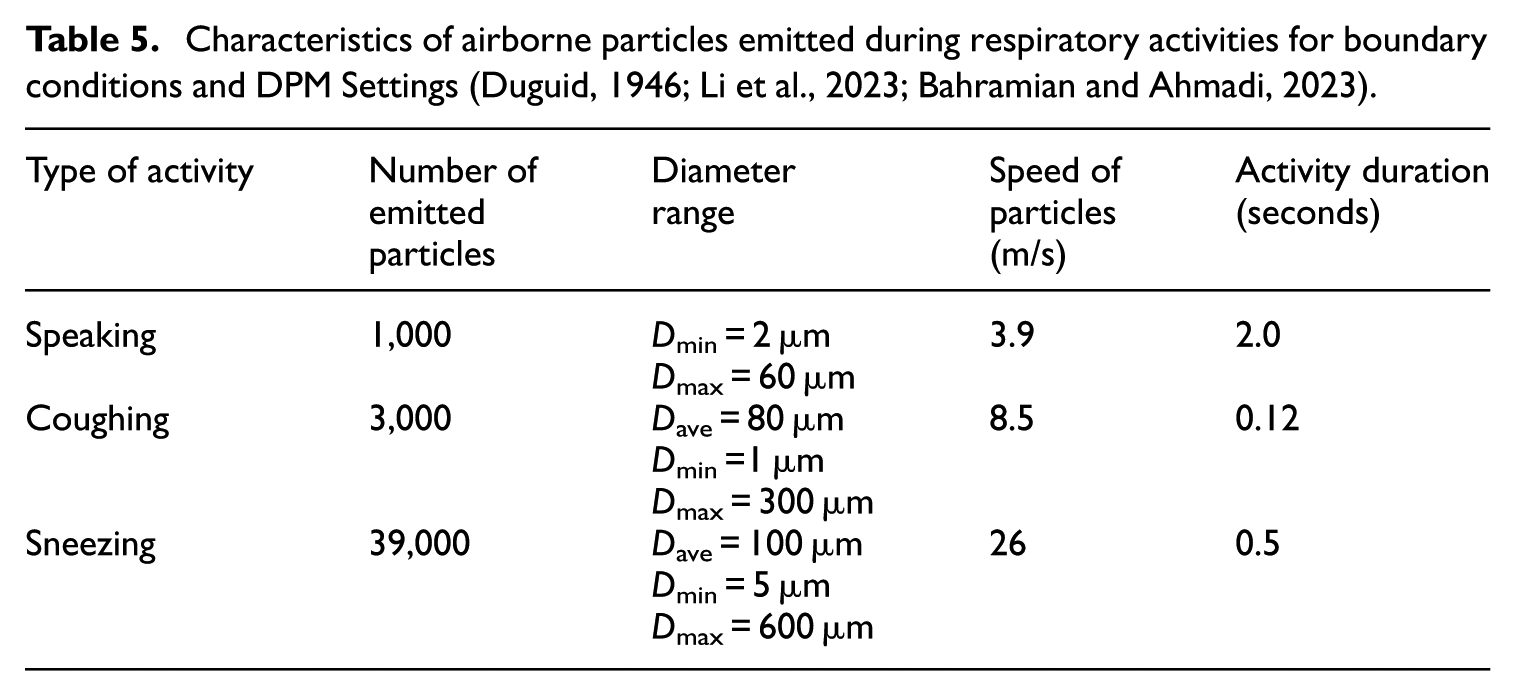
Table 5 details the number of emitted particles, their diameter range, speed, and the duration of each activity. Speaking involves a steady-state emission with particle sizes ranging from 2 to 60 µm at a speed of 3.9 m/s. Coughing releases 3000 droplets, ranging from 1 to 300 µm in diameter, at 8.5 m/s over 0.12 seconds. Sneezing emits 39,000 droplets with sizes between 5 and 600 µm at 26 m/s, lasting 0.5 seconds. These parameters were directly applied to define particle injection, velocity boundaries, and time-based emission settings in the DPM section.
Characteristics of airborne particles emitted during respiratory activities for boundary conditions and DPM Settings (Duguid, 1946; Li et al., 2023; Bahramian and Ahmadi, 2023).
The outlet was modeled as a pressure outlet at atmospheric pressure, allowing particles to escape. Walls in different zones and furniture surfaces were treated as no-slip walls with zero heat flux, with particles set to reflect upon contact. The surfaces of the person’s body were also considered no-slip walls, maintained at a temperature of 310 K, with particles similarly set to reflect.
Methodology and numerical approach
The CFD simulation consists of three main stages: preprocessing, processing, and post-processing. During preprocessing, the geometry of the system is created and meshed, and boundary conditions are defined, ensuring an accurate representation of the physical environment. Processing involves activating energy, turbulent, or laminar flow models to simulate fluid dynamics, accounting for factors like air conditioning flow rates, droplet evaporation, and dispersion patterns (Mui et al., 2022). This stage generates detailed data on droplet behavior, including size, velocity, and trajectory over time (Christakis et al., 2024; Yi et al., 2023). In post-processing, the simulation results are analyzed to provide data and contours. It’s crucial to validate the data against experimental studies or numerical assessments, with mesh independence tests ensuring reliable results (Yi et al., 2023).
All simulations are conducted using ANSYS Fluent 22.0 R2 software with a double-precision solver. The governing equations for continuity, momentum, energy, the turbulent k–ε model, and the DPM are solved to predict the behavior of flow, heat, and particle concentration within the system. The particles are injected perpendicular to the patient’s mouth. It is important to note that the particle decay rate is considered negligible in this study, as evaporation is not taken into account. To account for the various droplet size distributions expelled during speaking, coughing, and sneezing, the Rosin-Rammler distribution is applied (Li et al., 2023). The SIMPLE algorithm is employed to couple the momentum and mass conservation equations. The second-order upwind discretization method is used to achieve higher convergence and accuracy of results. The residual criteria for solving all governing equations are set to 10−6. Additionally, to track the particles after the infected person closes their mouth, the boundary condition for the injection in the DPM is changed from a velocity inlet to a wall. This adjustment allows for the continued simulation of particle behavior in the environment after the cessation of particle emission. Table 6 summarizes the key numerical settings and modeling parameters employed in the CFD simulations using ANSYS Fluent 22.0 R2, including solver type, turbulence model, discretization scheme, and particle tracking approach.
Summary of CFD setup parameters.

Governing equations
This section presents the governing equations, based on the Navier-Stokes equations for mass conservation, momentum, and energy. The airflow field is modeled using the RANS equations for incompressible turbulent flow, with turbulence captured by the Standard k–ε model due to its stability, efficiency, and reliability in indoor airflow simulations. This model choice aligns with the validation study by Zhang and Chen (2006), which demonstrated close agreement with experimental data.
Continuity equation

where ρ (kg/m3) is fluid density, and
Conservation of momentum
Conservation of momentum equation incorporating the Boussinesq approximation to account for density variation effects (Fluent, 2016):
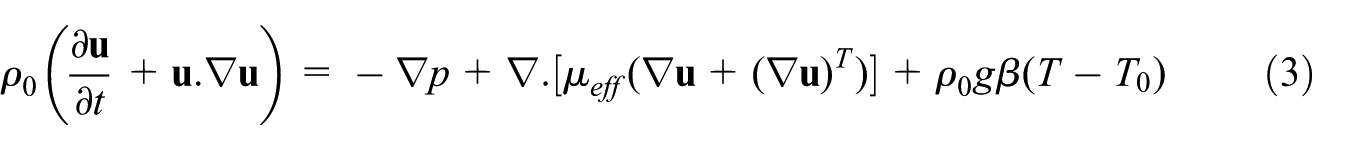
The Boussinesq approximation is used to account for buoyancy effects due to small temperature-induced density variations. Where
Turbulent k–ε model
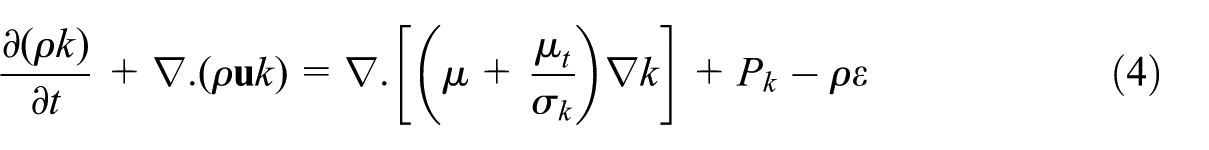

Equation (4) describes the transport of turbulent kinetic energy. The source term Pk represents energy transferred from the mean flow into turbulent fluctuations. This formulation is essential for predicting velocity fluctuations within mechanically ventilated and buoyant indoor flows (Fluent, 2016).
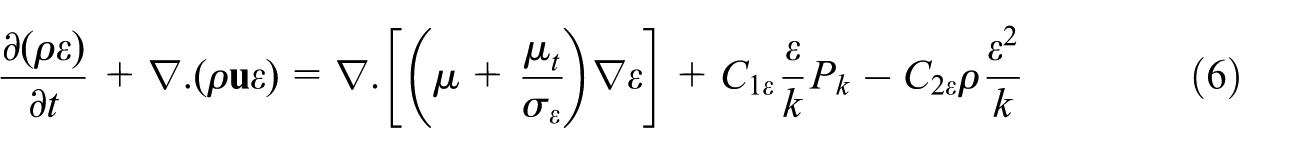

This relationship defines the turbulent eddy viscosity used to approximate the Reynolds stresses via the Boussinesq hypothesis. It enables the momentum and energy equations to account for unresolved turbulent fluctuations.
k is the turbulent kinetic energy (J/kg); ε is the rate of dissipation of turbulent kinetic energy (m2/s3); S k , S ε are the source terms; v is the fluid velocity vector; C μ , σ e , σ k , C 1e , and C 2e are the empirical constants (Fluent, 2016).
Energy balance equation:
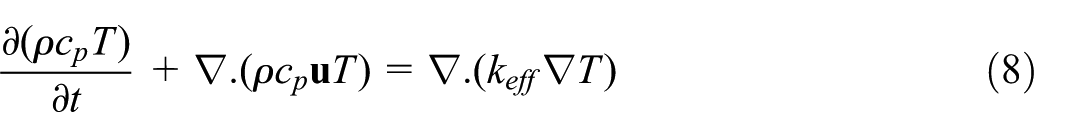

This equation governs heat transfer through both convection and conduction. The term k eff represents the turbulent-enhanced thermal conductivity, where μ t is the turbulent viscosity from the standard k–ε model (Fluent, 2016).
Discrete phase model (DPM)
The particle motion equation is also used to calculate each particle trajectory by solving the momentum equation (Fluent, 2016).
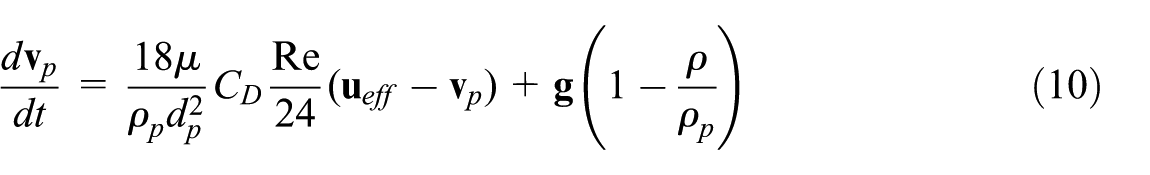
This Lagrangian equation governs the motion of particles expelled from the mouth during respiratory activities. The random walk model modifies the fluid velocity using ueff to account for turbulent fluctuations, improving the realism of particle dispersion patterns in turbulent indoor airflows.
The Wells-Riley model is designed for prolonged, steady-state exposure conditions where infectious aerosols are gradually emitted over time, such as talking or breathing. By using sneezing for contour visualization and talking for infection risk modeling, the dual dimensions of the problem is captured spatial dimension. Sneezing highlights critical zones of high particle dispersion that need targeted mitigation. Temporal dimension: Talking captures the steady-state exposure that dominates cumulative infection risk. This combined approach ensures a comprehensive evaluation of ventilation effectiveness under both transient and steady-state conditions. The modified Wells-Riley (W-R) model used in this study calculates the probability P (Sun and Zhai, 2020) based on factors such as the number of infectors I, exposure time t, infected airborne rate q, pulmonary ventilation rate p, room ventilation rate Q, and a distance index P d . The final equation, equation (10), incorporates these parameters to model airborne disease transmission, capturing the effects of social distancing and ventilation effectiveness.

t:Exposure time (hour) = 1 hour
E z :Ventilation effectiveness for both ventilation configurations, E z = 1 (ASHRAE-62.1, 2022) (ASHRAE, 2022a)
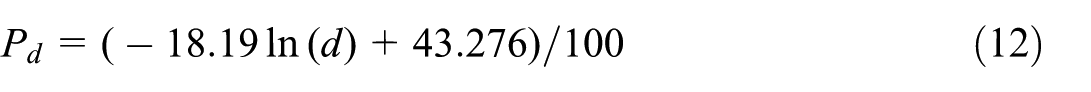
The distance index P d in equation (8) was calculated through theoretical analysis of how droplets are distributed and transmitted when speaking.
Therefore, the final version of the modified W-R model for this study is obtained as follows:
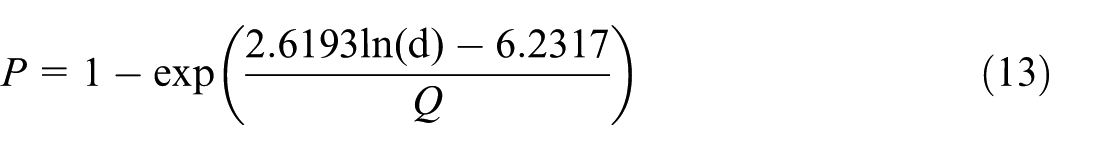
It should be noted that employing standard wall functions introduces known limitations in modeling convective heat transfer at solid surfaces. The wall-function approach (used with the high-Reynolds-number k–ε model) assumes fully turbulent near-wall flow and uses a fixed wall Prandtl number; as a result, it may not accurately resolve low-velocity, buoyancy-driven thermal boundary layers along the walls with constant temperatures. In indoor simulations with significant heat exchange at boundaries, this simplification can lead to underestimation of wall convective heat transfer and can affect the temperature distributions (Zhang et al., 2013). While the use of wall functions drastically reduces computational cost by avoiding extremely fine near-wall meshes, the trade-off is some loss of fidelity in capturing near-wall thermal gradients. Therefore, using standard wall functions provides a computationally efficient solution while preserving the accuracy of large-scale airflow features and particle dynamics, especially in turbulent flow fields. Instead, the mesh near surfaces was refined to maintain Y+ values within the standard wall function validity range (30–300), ensuring acceptable prediction of mean flow and thermal fields, consistent with approaches used in validated indoor airflow studies (e.g. Zhang and Chen, 2006).
Validation
To ensure the accuracy and reliability of the computational fluid dynamics (CFD) model employed in this study, simulations were conducted under conditions identical to those in Zhang and Chen’s study (Zhang and Chen, 2006). Their work utilized a full-scale environmental chamber (4.91 m × 2.44 m × 4.31 m) equipped with an underfloor air distribution system featuring floor-level inlets and ceiling-mounted exhausts, operating at a supply flow rate of 0.0944 m2/s. Human simulators and heat sources were used to mimic realistic indoor conditions.
A structured mesh with 389,512 control volumes was used in the present study, closely matching the 389,338 cells used by Zhang and Chen (2006) to ensure consistency.
Zhang and Chen (2006) applied standard k–ε turbulence model and the SIMPLE algorithm for pressure–velocity coupling. Second-order upwind and PRESTO! schemes were adopted for numerical accuracy. Particle transport was simulated using the Lagrangian approach, with turbulent dispersion accounted for via the discrete random walk (DRW) model. They assumed uniform inflow, no-slip walls, negligible deposition, and tracked 10,000 to 50,000 particle trajectories to ensure statistical convergence. Both studies applied the standard k–ε turbulence model with standard wall functions. No enhanced wall treatment or low-Reynolds-number models were employed, aligning with the original setup.
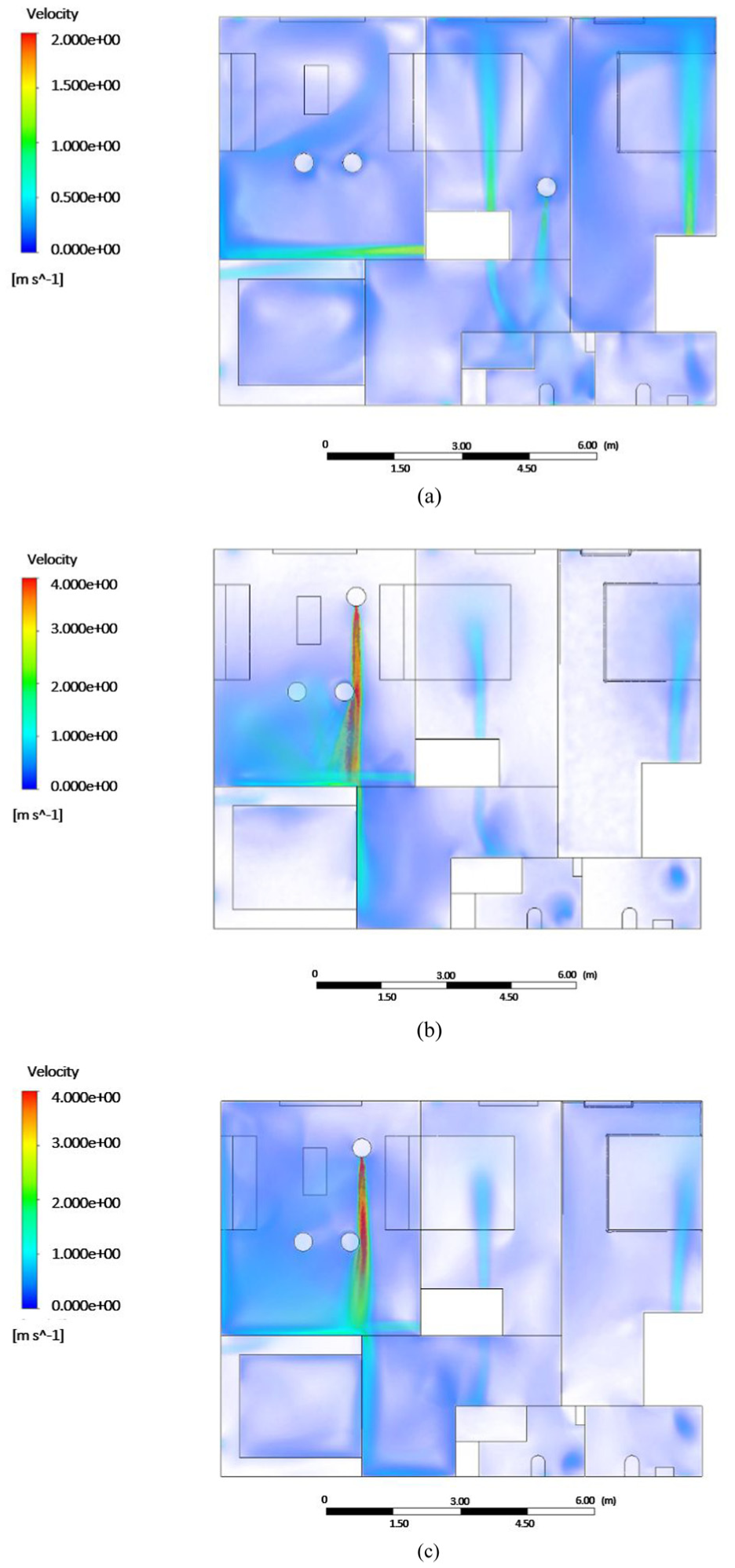
Figure 2 presents a schematic of the test domain highlighting the locations where velocity, temperature, and particle concentration data were extracted for validation. Figure 2(a) shows vertical line positions for cases (a) to (f). Figure 2(b) marks the position of symbolic individuals and sampling points from the top view used for comparison with the study by Zhang and Chen (2006).
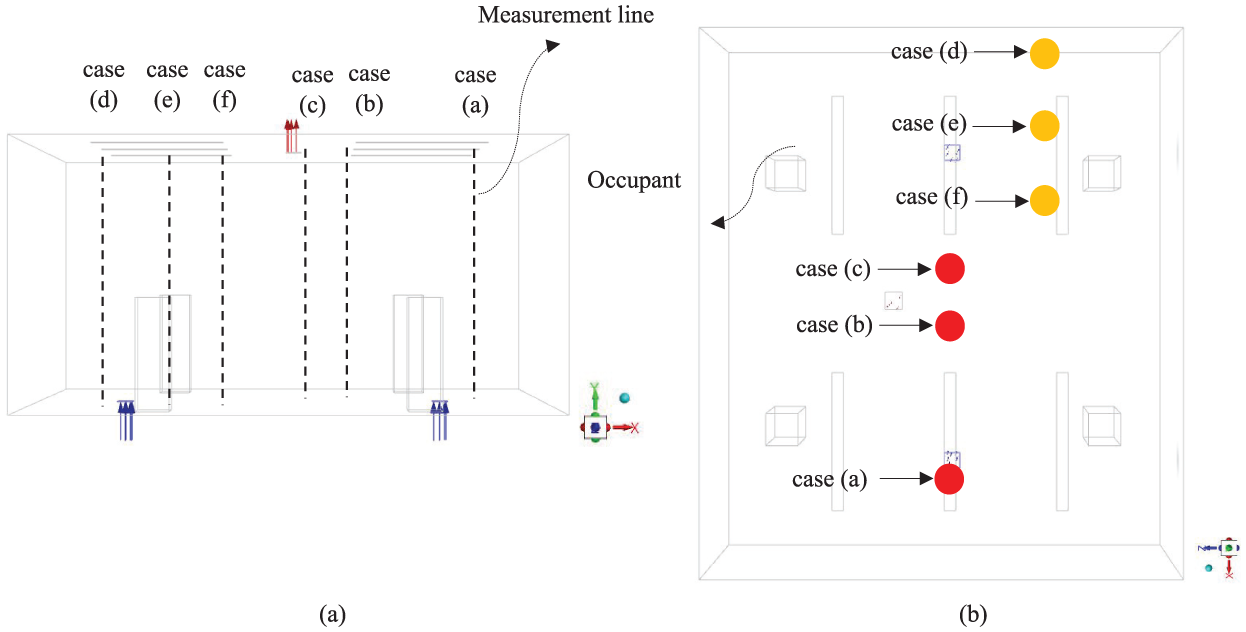
Sampling locations for validation: (a) side view showing vertical lines for the velocity and particle concentration profiles (cases a–f) and (b) top view indicating occupant positions and sampling lines.
The validation focused on three aspects,
Airflow characteristics, comparison of temperature and velocity profiles at multiple heights for cases (a), (b), and (c), presented in Figure 3(a) to (c).
Particle concentration distribution, spatial comparison of concentration profiles, for cases (d), (e), and (f), presented in Figure 3(d) to (f).
To further support the validation of the present model, contour plots of velocity magnitude and static temperature are shown in Figure 3(g) and (h), respectively. These plots correspond to the same environmental chamber setup and flow conditions described by Zhang and Chen (2006). The results closely resemble the airflow patterns reported in the reference study, reinforcing the accuracy of the CFD model in replicating indoor airflow behavior.
The current CFD model reproduced the test conditions and followed the same modeling assumptions.
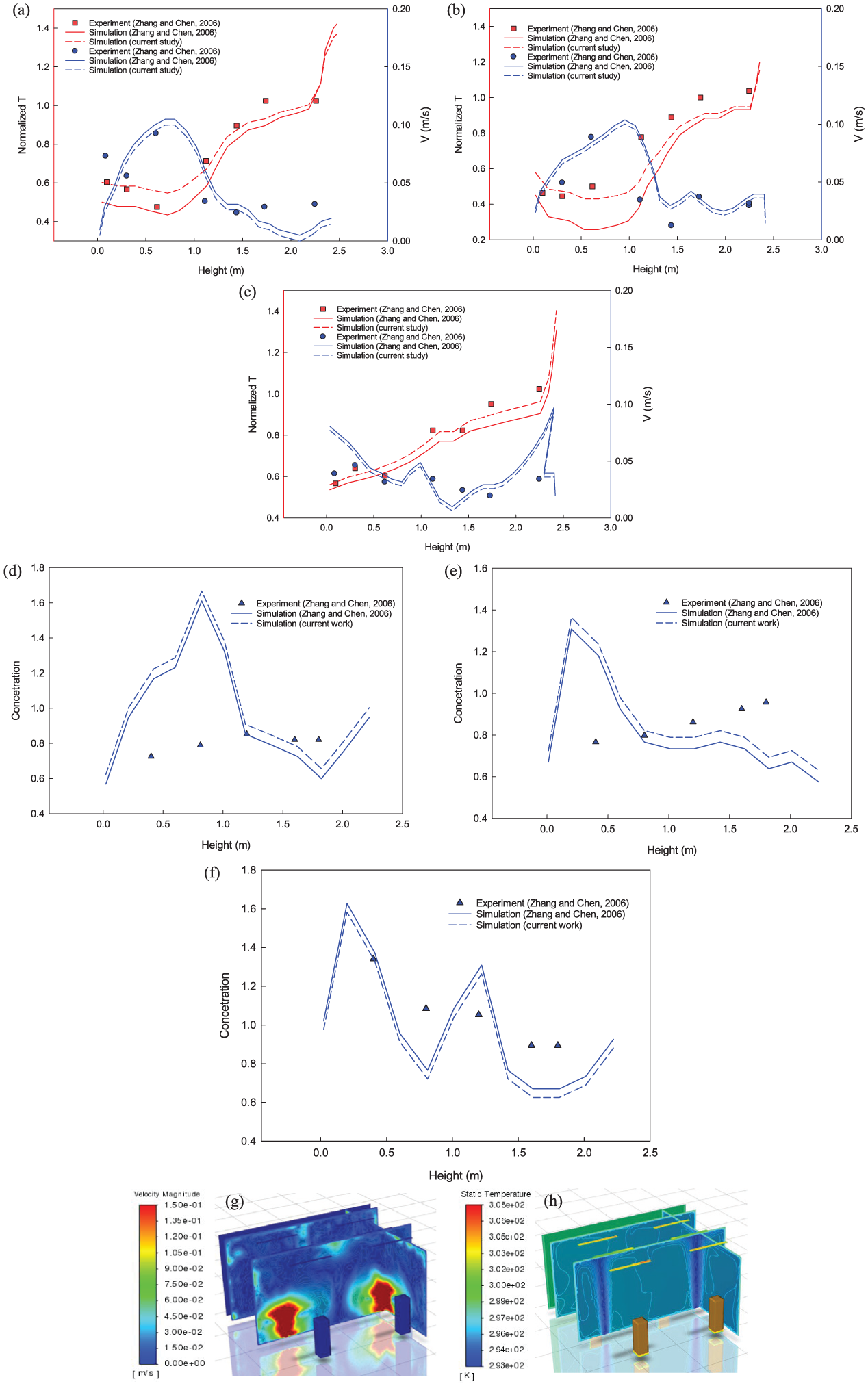
Validation of the CFD model against (Zhang and Chen, 2006) : (a–c) Comparison of normalized temperature and velocity profiles at multiple heights; (d–f) Comparison of particle concentration profiles at different sampling positions; (g) Contour plot of velocity magnitude; and (h) Contour plot of static temperature.
Normalized temperature profiles at various heights demonstrated strong agreement with both the experimental and simulated results from Zhang and Chen (2006). Minor deviations observed near the ceiling may result from differences in near-wall treatment or slight variations in boundary condition specifications. Velocity profiles captured the overall flow patterns and vertical stratification observed in the benchmark study, affirming the capability of the standard k–ε model. Slight mismatches are attributed to potential measurement uncertainties and turbulence intensity variations.
Simulated concentration profiles at different sampling heights aligned well with those reported by Zhang and Chen (2006). The current model successfully captured both spatial distribution and trends, reinforcing its suitability for predicting particle behavior in indoor environments.
Mesh independence analysis
Figure 4 illustrates the full view of the computational mesh, zoomed-in view of the living room with three individual, zoomed-in view hallway and bedrooms.
(a) Full view of the computational mesh, (b) zoomed-in view of the living room with three individuals, and (c) zoomed-in view hallway and bedrooms.
A tetrahedral volume mesh is utilized throughout all domains, with a boundary layer mesh specifically applied to the mouths of the symbolic individuals.
In this study, the near-wall region was modeled using standard wall functions as part of the Standard k–ε turbulence model. This approach requires that the first computational cell adjacent to the wall be positioned within the logarithmic layer of the boundary layer. To ensure this, the mesh was designed so that the first cell center is approximately 1.9 cm or more from the wall. These spacing yields non-dimensional wall distances Y+ greater than 30 in most near-wall regions, satisfying the requirements for using standard wall functions (Canonsburg, 2017; Versteeg, 2007). This approach enables efficient and accurate simulation of indoor airflow while avoiding the high computational cost of resolving the viscous sublayer. The selected near-wall treatment is also consistent with the approach used in the validation study by Zhang and Chen (2006) which successfully applied the same model to indoor ventilation cases with accepted experimental agreement.
The minimum orthogonal quality was maintained above 0.35, and maximum aspect ratio remained below 8, with smooth cell size transitions. Mesh skewness was controlled to remain below 0.4 across the domain (Fluent, 2011).
As shown in Figure 5(a), mesh independence analysis reveals that the minimum number of mesh elements required to ensure mesh independence is 14,150,101. The area-weighted average outlet velocity of the diffuser in the living room was selected as a reference point to identify the optimal number of mesh elements for the simulation. Also, as indicated in Figure 5(b), a mesh independence study was conducted using five different mesh densities ranging from 6.0 × 106 to 1.6 × 107 cells. The velocity profiles along a selected diagonal line within a representative plane were compared across all mesh levels. The diagonal line spans the plane dimensions of 10.25 m × 8 m, resulting in a total length of approximately 13.00 m. In this graph, the x-axis labeled as “Position” represents the cumulative parametric distance along this diagonal path. The results from the finer meshes (1.4 × 107 and 1.6 × 107 cells) exhibit negligible differences. Based on this analysis, the mesh with 1.4 × 107 cells was selected for subsequent simulations.
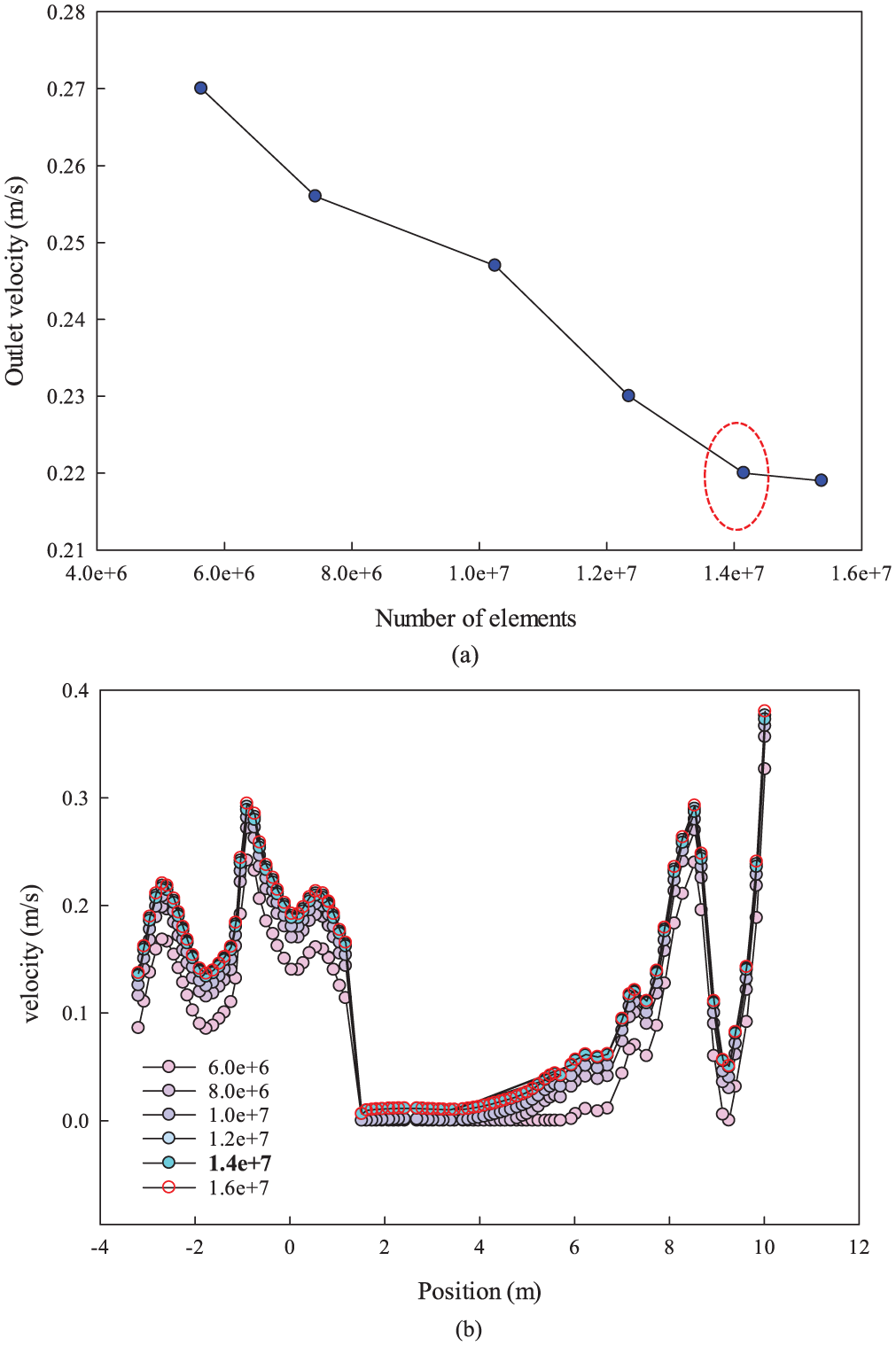
Mesh independence analysis based on: (a) outlet velocity for the first scenario and (b) velocity profiles along a selected diagonal line.
Comparison of airflow patterns for different respiratory activities
Figure 6 illustrates airflow patterns for speaking, coughing, and sneezing under the minimum ventilation rate as per ASHRAE 62.2-2022. In the first case, speaking generates a low-velocity airflow, which causes particles to disperse gently around the mouth. Coughing, shown in the second image, produces a more forceful jet, leading to a broader and faster distribution of particles. The third figure, representing sneezing, exhibits the highest velocity, causing a powerful and extensive spread of particles across the room. This progression from speaking to sneezing underscores the increasing risk of particle dispersion with higher exhalation velocities, concluding that sneezing represents the most conservative respiratory activity for evaluating ventilation strategies. As a result, sneezing was chosen as the critical scenario for further analysis to ensure the effectiveness of ventilation in mitigating airborne particle spread in residential settings. As detailed earlier in Table 5, the duration of each respiratory activity differs, 2.0 seconds for speaking, 0.12 seconds for coughing, and 0.5 seconds for sneezing. The snapshots shown in Figure 6 were extracted at the end of these specific durations, corresponding to the transient injection phase of each activity. The simulation remained fully transient during this phase, ensuring accurate representation of particle release and airflow dynamics for each case.
Airflow patterns for different respiratory activities of an infected person:(a) speaking, (b) coughing, and (c) sneezing.
In Figure 6, the color scale for the speaking case was refined to appropriately capture the lower velocity magnitudes compared to coughing and sneezing. Variations in the infected individual’s location are acceptable, as this phase of analysis aimed to characterize the hydrodynamic features and dispersion profiles of exhaled airflow under different respiratory activities, rather than to model location-specific transmission pathways.
The sneeze event was simulated over the first 0.5 seconds using a fine time step of 0.01 seconds to resolve the high-velocity jet and particle emission. After the injection phase, the simulation continued for an additional 390 seconds using a larger time step of 0.1 seconds to capture the long-term particle dispersion driven by mechanical ventilation. This total simulation time of 390.5 seconds was selected based on recommendations from prior studies on airborne transmission indoors. For example, Vuorinen et al. (2020) analyzed particle dynamics and infection risk over durations of 300 seconds, confirming the importance of modeling several minutes of post-emission airflow in enclosed spaces.
Results and discussion
Section 4 is organized to align with the three primary objectives outlined in the introduction: (1) evaluating the impact of diffuser placement, (2) assessing the effect of ventilation rates, and (3) investigating the influence of the infected person’s location on airborne particle dispersion and infection risk.
Comparison of streamlines with ceiling and floor diffusers
According to Figure 7, in cases 3 and 7, where the ventilation rate is higher, the streamlines indeed appear more turbulent. This is expected because increasing the ventilation rate introduces more energy into the airflow, which can lead to more complex and chaotic movement patterns, resulting in increased turbulence. Depending on the diffuser placement, this turbulence can help disperse particles more thoroughly but may also keep them suspended in the air longer. Comparing cases 1 and 3 (near-ceiling diffusers) with cases 5 and 7 (near-floor diffusers), it is evident that the streamlines in cases 5 and 7 are denser and more concentrated near the floor. This suggests that near-floor diffusers create a more confined airflow pattern, leading to higher particle concentrations in certain areas, as particles may not be dispersed as widely or effectively as near-ceiling diffusers. The denser streamlines in cases 5 and 7 indicate that the airflow is more localized and less efficient at moving air throughout the entire space, leading to potential zones of higher particle concentration, especially with the increased turbulence from higher ventilation rates.
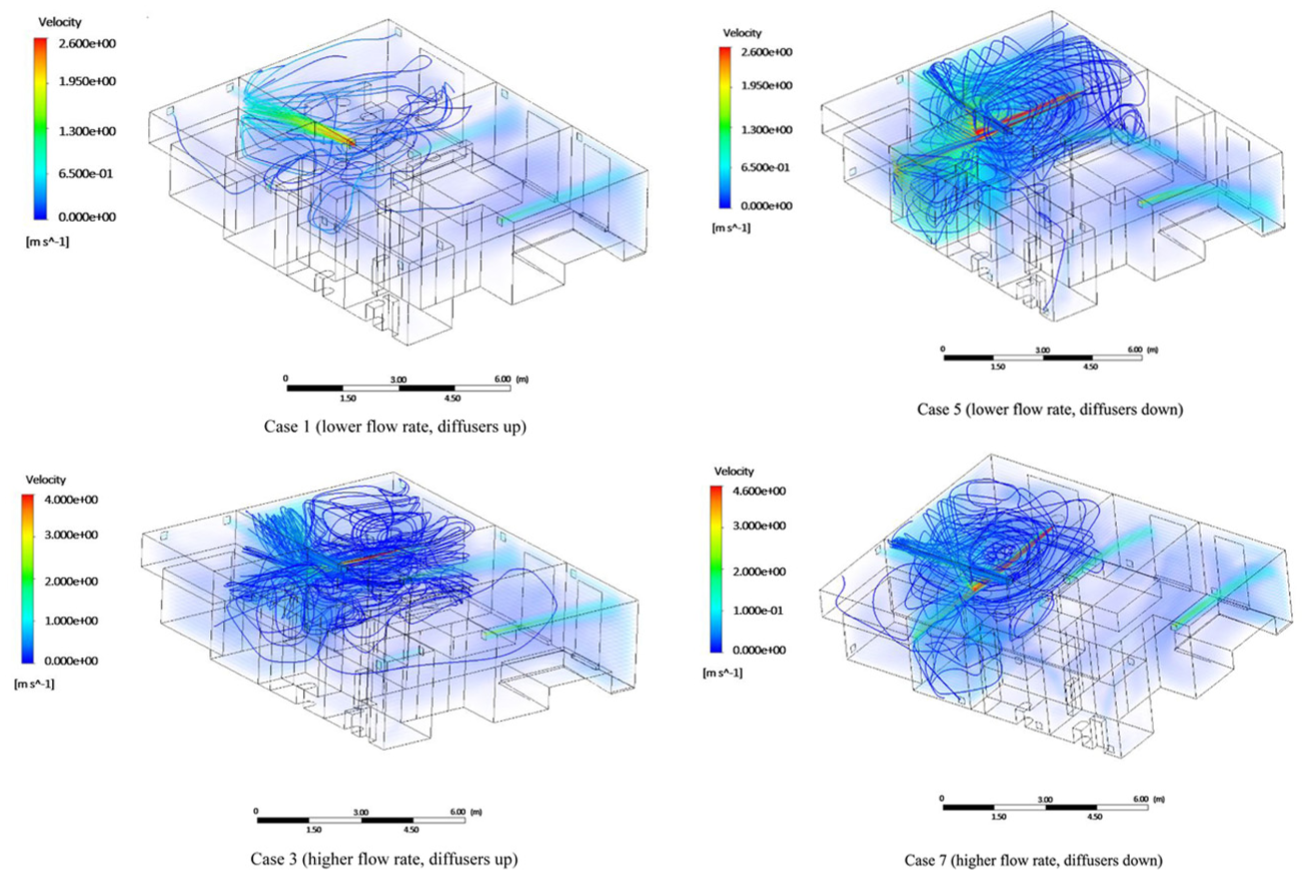
Comparison of airflow streamlines across different ventilation rates and diffuser placements (infected person in the living room). Case 1 (lower flow rate, diffusers up). Case 3 (higher flow rate, diffusers up). Case 5 (lower flow rate, diffusers down). Case 7 (higher flow rate, diffusers down).
Temporal analysis of particle mass concentration following a sneeze
Figure 8 shows the distribution of particle mass concentration over time (at 0.1, 0.3, and 0.5 seconds) after an infected person sneezes in the living room. At t = 0.1 seconds, the particles are initially concentrated near the source of the sneeze and in the hallway, with a high concentration in the immediate vicinity. The particles begin to spread outward, but the distribution remains relatively localized. By t = 0.3 seconds, the particles have dispersed more widely across the living room, covering a larger area, which indicates that the particles are being carried by the airflow and are starting to occupy the surrounding space more uniformly. Some particles are also beginning to move toward the kitchen, bedroom 1, and washroom 2. By t = 0.5 seconds, the particles have spread significantly, covering almost the entire living room and beginning to enter other areas of the house. The concentration is more evenly distributed, and particles are observed moving toward the right side of the house.
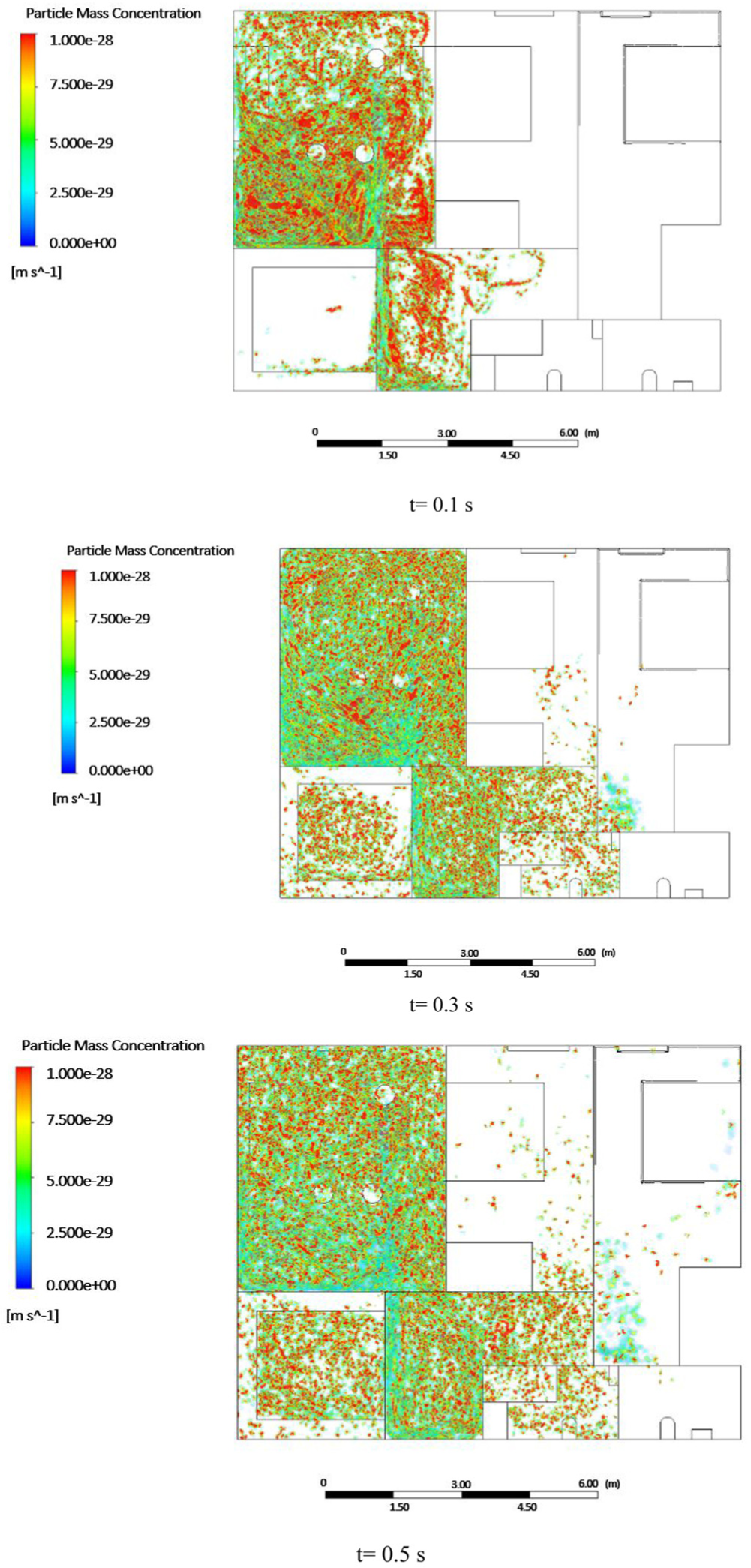
Temporal distribution of particle mass concentration following a sneeze in a living room at 0.1, 0.3, and 0.5 seconds from the top view.
Effect of infected person’s location
This subsection addresses the third objective by examining how the infected person’s location affects airborne particle dispersion.
The position of the infected individual influences how particles interact with the airflow generated by the ventilation system and how they spread throughout the indoor environment. Based on Figure 9, regarding Cases 1, 3, 5, and 7, when the infected person is in the living room, which is a central and often larger space in the house, the particles tend to disperse widely. The living room’s central location allows the particles to interact with multiple airflows. Because the living room is typically connected to multiple other rooms (e.g. hallways and bedrooms), there is a higher potential for particles to spread to adjacent areas, especially if the ventilation system is not optimized.
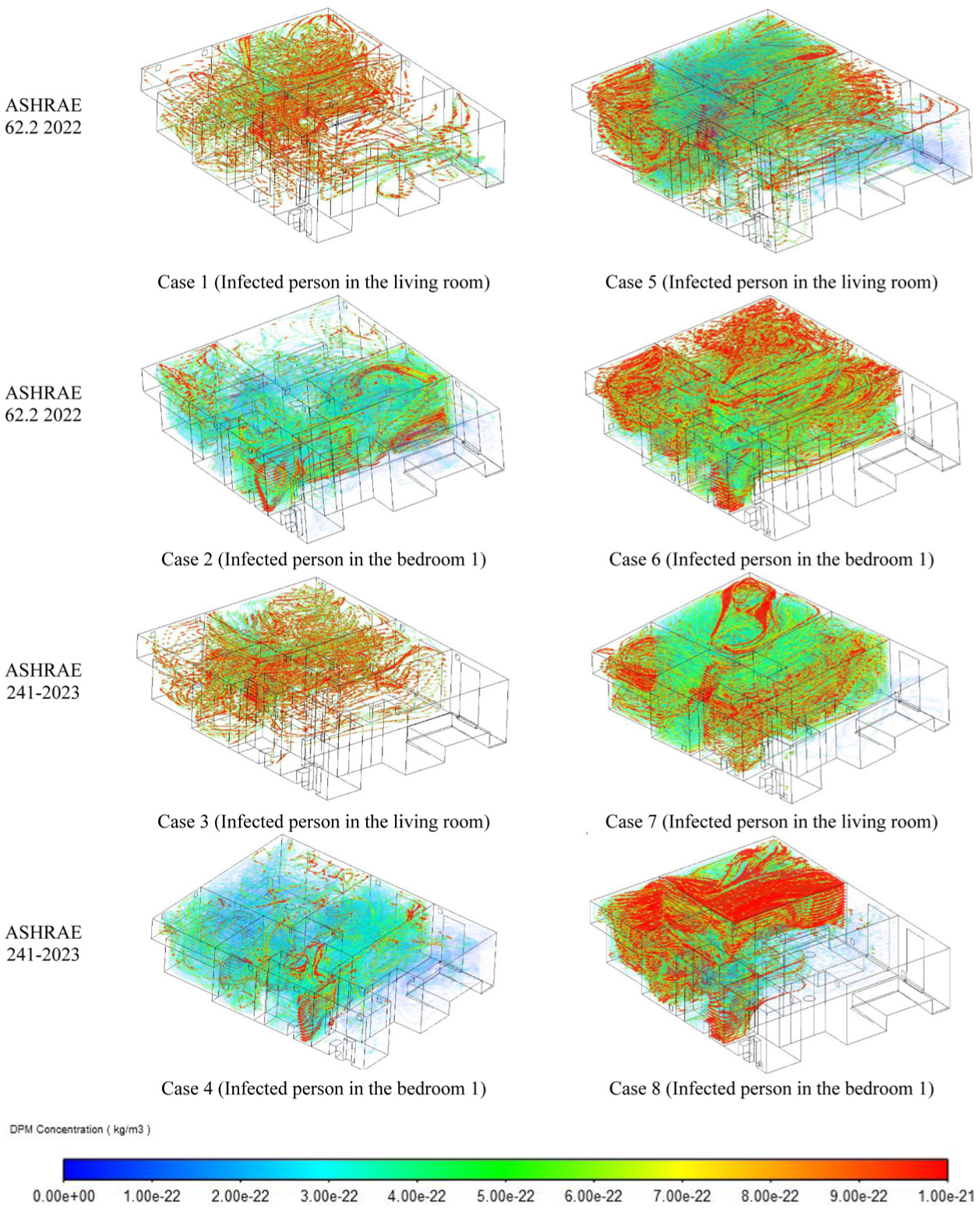
Comparison of particle dispersion across eight ventilation scenarios with varying ventilation rates, diffuser’s heights, and infected person’s locations.
In cases with a higher ventilation rate (e.g. Case 3 and Case 7), there is a noticeable reduction in particle concentration, particularly when the diffuser is near the ceiling. However, when the diffuser is near the floor, the increased ventilation rate can actually increase particle concentration due to enhanced turbulence.
When the infected person is located in a bedroom, the particles are more contained within that smaller, relatively isolated space. The risk of particles spreading to other parts of the house is reduced, but the concentration within the bedroom itself can be higher. In cases where the ventilation rate is lower (e.g. Case 2 and Case 6), the particles tend to remain concentrated within the bedroom, with a limited spread to other areas.
As with the living room, increasing the ventilation rate (e.g. Case 4 and Case 8) generally helps reduce particle concentration when the diffuser is near the ceiling. However, with a near floor diffuser, the increased ventilation rate can lead to higher concentrations within the bedroom due to increased airflow turbulence.
These results indicate that the location of the infected individual significantly affects the spread and containment of airborne particles. When located in open and interconnected spaces like living rooms, the potential for widespread contamination increases due to multiple airflow paths. Conversely, being located in a more enclosed area like a bedroom limits the spread to other spaces but can lead to higher localized particle concentrations. Moreover, while increasing the ventilation rate helps in cases with near-ceiling diffusers by promoting particle removal, it can have adverse effects when combined with floor-level diffusers, where enhanced turbulence may elevate exposure risk.
In addition, the contours at 1.70 m height in Figure 10 demonstrate that the location of the infected person plays a crucial role in the extent of particle spread. When the infected individual is positioned in the living room (Cases 1, 3, 5, and 7), which is a central and open area, particles disperse more widely, resulting in elevated concentration levels across multiple zones. Conversely, when the infected person is located in Bedroom 1 (Cases 2, 4, 6, and 8), the particles largely remain confined within the bedroom, with significantly reduced spread to adjacent spaces. This observation confirms that open, interconnected spaces facilitate wider particle dispersion, whereas isolated rooms help contain airborne contaminants, consistent with previous streamline analyses.
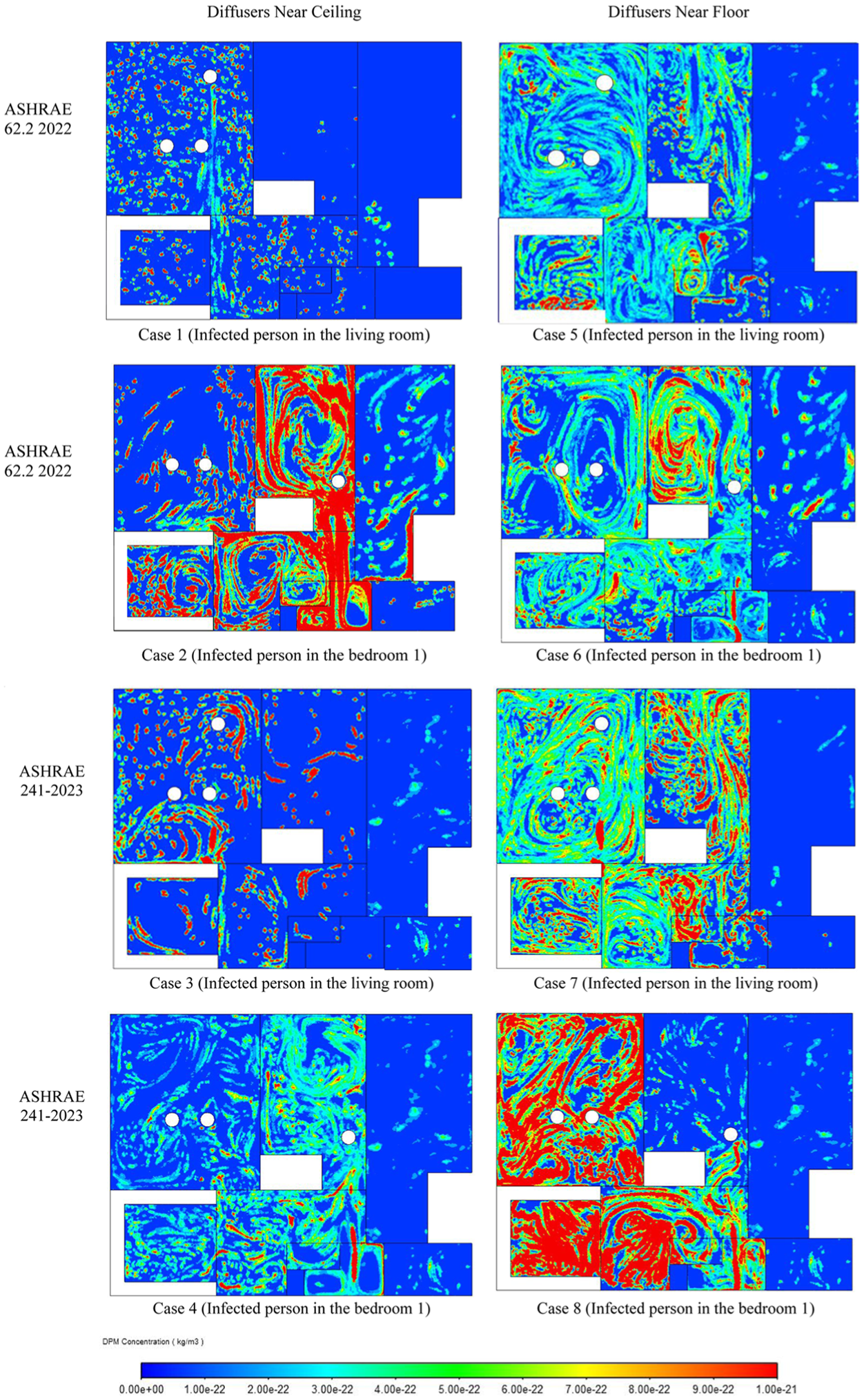
Particle mass concentration at 1.70 m height across eight ventilation scenarios with varying ventilation rates, diffuser heights, and infected person’s locations.
Effect of the diffuser’s height
This subsection focuses on the first objective, investigating the impact of diffuser placement on airflow and particle distribution.
Diffuser placement plays a critical role in controlling particle dispersion within indoor environments. Based on Figure 9, diffusers near the ceiling appear to be more effective at reducing overall particle concentration, due to their ability to leverage natural convection currents and promote better mixing of air.
In contrast, diffusers near the floor may result in denser particle concentrations and less effective dispersion, which could increase the risk of airborne transmission. The outlets near the ceiling typically enhance the removal of warm, particle-laden air that rises naturally due to buoyancy.
This helps create a more efficient air exchange, allowing for better removal of contaminated air. When the outlet is near the floor, the airflow path may not align as effectively with the natural rise of warm air, leading to less efficient removal of airborne particles.
With a floor-level outlet, the airflow might not efficiently carry particles away from the breathing zone. Instead, particles could circulate and settle more easily, resulting in denser concentrations throughout the space. This scenario could lead to higher overall particle density in the room, particularly near the source of the contamination.
These observations suggest that diffuser placement significantly affects ventilation effectiveness and indoor air quality. Ceiling-mounted diffusers, by aligning with natural buoyancy-driven air movement, promote more effective removal of contaminated air and thus reduce airborne particle concentration.
In contrast, floor-mounted diffusers can hinder efficient air circulation, increasing the risk of particle accumulation and potentially elevating exposure risks, especially near the source of contamination. Therefore, near-ceiling diffuser placement is preferred for minimizing infection risk through enhanced particle clearance.
In addition, based on Figure 10 ceiling-mounted diffusers result in lower and more uniformly distributed particle concentrations at 1.70 m height, indicating more efficient mixing and removal of airborne contaminants. In contrast, floor-mounted diffusers produce localized zones of higher particle concentration, particularly around the source, suggesting less effective particle removal. These findings reinforce the earlier conclusion that ceiling-mounted diffusers enhance natural convection patterns, improving particle clearance compared to floor-mounted systems.
Effect of overall ventilation rates using two ASHRAE standards
This subsection relates to the second objective, analyzing the effect of different ventilation rates on infection risk and particle spread.
Referring to Figure 9, in Case 1, the diffuser is positioned near the ceiling, but the ventilation rate follows the lower standard set by ASHRAE 62.2-2022. The airflow dynamics in this scenario are less robust, leading to a higher concentration of particles within the living room. Due to the lower ventilation rate, the particles have more time to spread within the living room and can more easily enter adjacent areas, such as Bedroom 2, increasing the risk of contamination in those spaces.
In Case 3, the diffuser is also near the ceiling, but the ventilation rate is approximately doubled, as per ASHRAE 241-2023. The increased ventilation rate creates stronger airflow patterns, which enhances the dispersion of particles and reduces their overall concentration in the living room. Notably, the higher ventilation rate in Case 3 is more effective at containing the particles within the living room, preventing them from entering Bedroom 2. This containment reduces the risk of cross-contamination between the living room and other areas of the house.
In Case 5, the diffuser is located near the floor, and the ventilation rate is set according to the lower standard of ASHRAE 62.2-2022. The airflow generated is relatively mild, which means particles may not be dispersed as effectively. However, the lower ventilation rate keeps the airflow less forceful, resulting in a somewhat limited spread of particles. In this scenario, the particle concentration in the living room remains moderate, as the weaker airflow does not significantly disturb the particles or carry them extensively throughout the room.
In Case 7, the diffuser remains near the floor, but the ventilation rate is increased according to ASHRAE 241-2023, which nearly doubles the airflow. Interestingly, the increased ventilation rate in this configuration leads to a higher concentration of particles within the room. This is contrary to the expected outcome observed in cases with near-ceiling diffusers. The stronger airflow generated by the higher ventilation rate seems to cause more turbulence near the floor, which stirs up particles and keeps them suspended in the air for longer periods. This results in a higher overall particle concentration within the living room and potentially increases the risk of exposure.
The comparison between Case 5 and Case 7 reveals that, with the diffuser near the floor, increasing the ventilation rate can inadvertently increase particle concentration. The stronger, more turbulent airflow in Case 7 appears to keep particles suspended and circulating within the room, which contrasts with the results observed with near ceiling diffusers.
The comparison between Case 6 and Case 8 demonstrates that increasing the ventilation rate with a near floor diffuser can lead to higher particle concentrations, similar to the observation in the living room cases (5 and 7). The increased airflow rate appears to generate more turbulence near the floor, preventing particles from settling and leading to a higher risk of exposure.
Bedroom 2 consistently shows low particle concentration, which aligns with the building’s ventilation design that directs airflow away from Bedroom 2. Airflow is primarily directed toward other areas, such as exhaust or return vents in nearby spaces, effectively isolating Bedroom 2 from the living room airflow patterns, which is previously confirmed by the streamlines presented in Figure 7.
In other words, in Case 8, similar to Cases 5 and 7, the streamlines are denser and more concentrated close to the floor, indicating that near-floor diffusers produce a more restricted airflow pattern. This results in certain areas exhibiting higher particle concentrations, as particles may not disperse as widely or effectively as they would with near-ceiling diffusers. The concentrated streamlines in these cases suggest that the airflow is more localized, reducing its efficiency in circulating air throughout the entire space. This effect potentially creates zones of increased particle buildup, especially when combined with the increased turbulence associated with higher ventilation rates.
These findings indicate that diffuser placement plays a crucial role in how ventilation rates influence particle dispersion and concentration. While higher ventilation rates are generally beneficial when diffusers are located near the ceiling, promoting stronger airflow, better particle dilution, and containment within specific zones, the same approach can have adverse effects when the diffusers are placed near the floor.
Increased ventilation rates with floor-level diffusers enhance turbulence, suspending particles longer in the breathing zone, leading to higher local concentrations and elevated exposure risks. Therefore, optimizing ventilation effectiveness requires careful consideration of both the ventilation rate and diffuser placement strategy.
The influence of ventilation rates is also evident in the contour plots presented in Figure 10.
Cases following the higher ventilation standard (ASHRAE 241-2023; Cases 3, 4, 7, and 8) exhibit overall lower particle concentrations compared to those with the lower ventilation rate (ASHRAE 62.2-2022; Cases 1, 2, 5, and 6), particularly when ceiling-mounted diffusers are used.
However, in scenarios with floor-mounted diffusers, higher ventilation rates lead to intensified airflow turbulence near the floor, causing the formation of localized high-concentration regions.
Thus, while increased ventilation generally benefits airborne contaminant removal, its effectiveness strongly depends on the alignment of airflow patterns with diffuser placement.
Infection risk quantification
As mentioned, sneezing represents the most extreme respiratory activity in terms of particle velocity, volume, and dispersion. Visualizing sneezing helps to identify the critical areas of concern to ensure the most conservative assessment of ventilation performance under a challenging scenario. The transition from a brief sneezing event to steady talking reflects real-life scenarios where a person may sneeze and then continue talking or breathing.
As shown in Figure 11, the distance between the infected person and healthy individuals 1 and 2 is demonstrated in two different scenarios based on the infected person’s location in the house. The distances between the infected individual (marked in red) and persons 1 and 2 were measured using indirect paths rather than direct line-of-sight measurements. This approach was chosen due to the presence of walls and structural divisions, which place the individuals in separate zones. Direct measurements would not accurately represent the separation effect created by these barriers. However, for cases where all occupants are within the same zone (living room), direct measurement is applied.
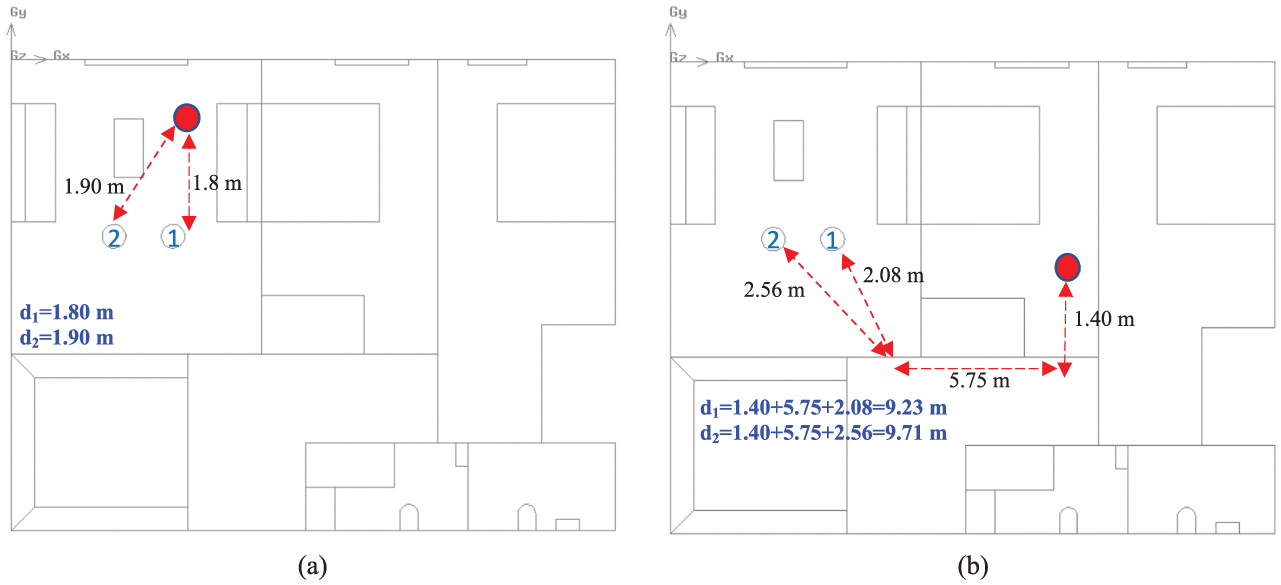
Social distance measurement approach between the infected person and other occupants: (a) occupants at the same zone and (b) occupants at the different zones
For example, in the first case, the distance between the infected person and person 2 is 1.90 m as they are within the same area. However, in the second scenario, where the infected person is in the bedroom, the distance to person 2, measured along the navigable path, increases to 9.71 m due to the wall barriers.
Table 7 provides a comparison of infection risks for different ventilation scenarios, demonstrating how the ventilation rate and the proximity to the infected person affect the transmission risk.
Infection risk results for different scenarios based on Equation (10)
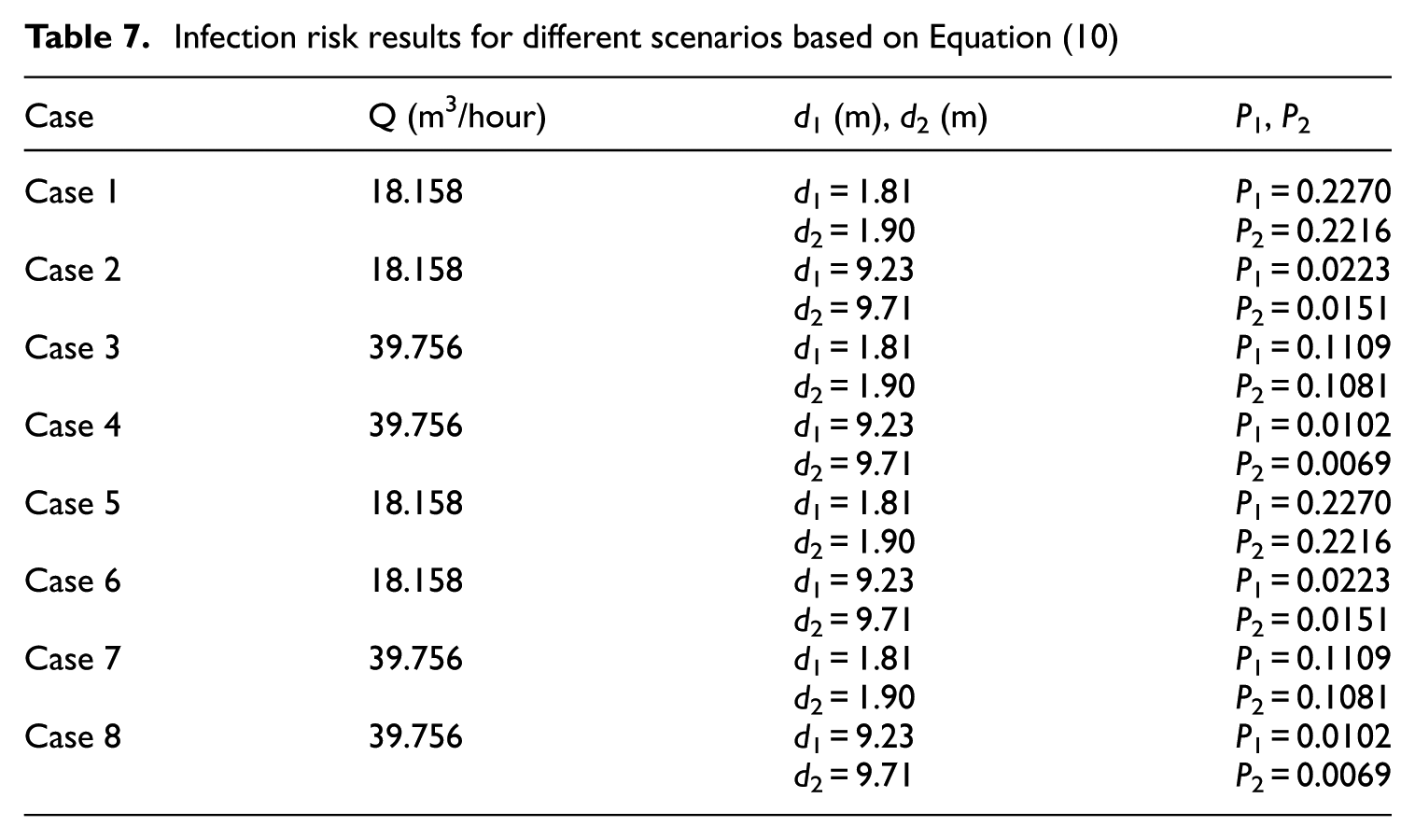
In Cases 1 and 3, all individuals are situated in a living room, with smaller distances (d1 = 1.81 m, d2 = 1.90 m) between the infected person and others. The infection risk is notably higher in these cases due to the closer proximity, with P1 reaching 0.2270 and P2 reaching 0.2216 in Case 1 (Q = 18.158 m2/hour). When the ventilation rate increases to 39.756 m2/hour in Case 3, P1 and P2 reduce to 0.1109 and 0.1081, respectively, indicating that increased ventilation can mitigate the infection risk around 50%.
In Cases 2 and 4, the infected individual is positioned in a bedroom, resulting in larger distances (d1 = 9.23 m and d2 = 9.71 m) between individuals. This greater separation lowers infection risk significantly. For example, at Q = 18.158 m2/hour in Case 2, P1 and P2 are reduced to 0.0223 and 0.0151, respectively. When ventilation is increased to 39.756 m2/hour in Case 4, P1 decreases slightly to 0.0102, and P2 drops to 0.0069. These comparisons show that both increased distance and ventilation rate contribute to lowering infection risks, with ventilation reducing P1 by approximately 54% from Case 2 to Case 4.
In Cases 5–8, the results are consistent with Cases 1–4, as they feature identical infection risk values to their counterparts due to the equal values for E z based on Sun and Zhai (2020).
These trends indicate that infection risk strongly depends on the proximity between individuals and the applied ventilation rate. When individuals are in close proximity, even under enhanced ventilation conditions, infection risks remain relatively high compared to scenarios where physical distancing is maintained.
Greater separation between the infected and susceptible individuals significantly reduces the infection probability, showing that spatial configuration plays a critical role in mitigating airborne transmission.
Additionally, the results emphasize that while increasing the ventilation rate effectively reduces infection risk, its effectiveness is substantially enhanced when combined with larger distances between occupants. This finding highlights the importance of not relying solely on ventilation strategies but also incorporating spatial layout considerations for effective infection control in residential settings.
Effective ventilation and isolation strategies
Placement of diffusers near the ceiling is recommended to enhance particle dispersion and reduce particle concentration through natural convection. In rooms where floor-level diffusers are necessary, it is recommended to apply moderate ventilation rates to prevent excessive turbulence, which could prolong particle suspension and increase transmission risk.
For shared or high-traffic areas, such as living rooms, a higher ventilation rate in accordance with ASHRAE 241-2023 guidelines is recommended to facilitate particle dilution and containment within the primary area. In more isolated rooms, such as bedrooms occupied by symptomatic individuals, maintaining a moderate ventilation rate can help contain particles within that room, thereby reducing the risk of transmission to adjacent spaces.
To minimize cross-contamination, establishing high-ventilation zones in central areas (e.g. living rooms) with ceiling-based diffusers is recommended. This configuration effectively contains particles within these areas, preventing their movement into adjacent spaces, such as bedrooms, which can maintain lower particle concentrations due to limited airflow interaction.
In shared environments, maximizing physical distance from infected individuals is strongly advised to reduce infection risk substantially. Layout adjustments that increase separation, in combination with optimal ventilation, provide additional protection against airborne transmission, further enhancing isolation strategies in multi-room settings.
Limitations and future recommendations
This study provides insights into the effectiveness of residential ventilation strategies for airborne infection control using CFD simulations; however, certain limitations remain. The simplified geometry and boundary conditions, such as the representation of occupants and furnishings, may not fully capture real-world complexities. The assumptions regarding negligible particle decay and the exclusion of environmental factors, such as humidity and temperature variations, limit the applicability of the results to dynamic conditions. Additionally, the focus on two ventilation rates prescribed by ASHRAE standards and short-term particle dispersion overlooks the potential effects of broader ventilation scenarios and long-term exposure. Furthermore, the validation was limited to a single study, necessitating further experimental or field-based verification to ensure broader applicability. Moreover, the wall-block risk estimation neglected the particle path resistance introduced by doors, leading to an overestimation of risk probabilities for wall-involved scenarios. Future research should aim to incorporate more detailed environmental modeling, evaluate diverse ventilation configurations, explore the integration of advanced filtration technologies, and assess the long-term impact of particle dispersion under varying conditions. These advancements would provide a more comprehensive framework for optimizing residential ventilation systems to mitigate airborne infection risks effectively.
Conclusions
This study uses CFD simulations to evaluate a typical ventilation system in a detached house in British Columbia, focusing on eight scenarios with varying diffuser locations, ventilation rates, and infected person positioning. The analysis assesses particle concentrations and infection risks, providing insights into the effectiveness of different ventilation strategies.
The location of the infected person significantly influences particle dispersion and infection risk within the building, with central locations like the living room leading to a higher potential for widespread particle distribution.
In more isolated spaces, such as bedrooms, particles tend to be contained within the room, but the risk of high particle concentration increases if the ventilation system is not effectively designed.
The placement of diffusers (pressure outlets) plays a crucial role in ventilation effectiveness; near-ceiling outlets better align with natural convection currents, reducing particle concentration and transmission risk.
Near-floor diffusers may lead to higher particle concentrations due to less effective air removal, particularly at higher ventilation rates, potentially increasing the risk of airborne transmission.
Increasing the ventilation rate generally reduces particle concentrations when the diffuser is near the ceiling, but it can have the opposite effect with near-floor diffusers due to increased turbulence keeping particles suspended.
These findings underscore the complexity of ventilation system design, emphasizing the need to carefully optimize both diffuser placement and ventilation rates to effectively manage airborne particle distribution and minimize infection risks in residential environments.
Increased physical separation, such as placing the infected individual in a separate room, significantly reduces infection risk, with larger distances yielding notably lower transmission probabilities.
Enhanced ventilation rates can reduce infection risk depending on proximity, highlighting the combined importance of spatial separation and ventilation in mitigating airborne transmission risks.
Footnotes
Appendix
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by BC Housing through the Building Excellence Research & Education Grant.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and analyzed during the study are available from the corresponding author upon reasonable request.
