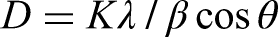Abstract
Hydroxyapatite (HAp) is an essential material in the biomedical field because of its chemical composition, similar to the apatites found in bones. The lack of bactericidal properties of HAp-based materials cannot prevent the adhesion and growth of bacteria. However, it can develop composite biomaterials using nanoparticles (NPs) such as silver (Ag) and zinc (Zn), which exhibit biocidal behaviours to control resistant microorganisms. This study aimed to prepareHAp–AgNPs and HAp–Ag@ZnNPs nanocomposite and evaluate their antimicrobial properties against Gram-positive and Gram-negative bacteria. HAp powders were produced from sheep bone. An aqueous extract of Marrubium astracanicum was used as a reducing and stabilising agent for the synthesis of NPs. The composites were characterized by XRD, SEM–EDX, SEM and FTIR. In vitro biocompatibility assessment was determined by agar well diffusion tests. The HAp–Ag@ZnNPs nanocomposites showed stronger antimicrobial activity than HAp–AgNPs against bacteria and fungi, while pure HAp did not show any effect.
This is a visual representation of the abstract.
Introduction
Biomaterials are natural or synthetic materials used to provide structural support to organs or tissues in the human body. In the last few decades, hydroxyapatite (HAp), which is among the bioactive ceramic materials, has been widely studied because of its chemical similarity with the mineral component of bones Hap, which is a biocompatible, bioactive, and thermodynamically stable structure in the body. For this reason, its applications have begun to attract the attention of biomedicine. 1 HAp is obtained in two ways, synthetically and naturally. Synthetic HAp biomaterials are highly reliable, but their production is complex and expensive. Natural HAp, which is usually extracted from biological sources such as sheep and beef bone, eggshell, and seaweeds, is easy and economical.2,3 Despite its numerous advantages, the lack of antibacterial properties of pure HAp can increase the risk of developing the disease due to its use as the bone implant material. 4 In recent years, infections caused by drug-resistant viruses, fungi, and bacteria have emerged as serious health problems worldwide. 5 Over the last decade, metal nanoparticles (NPs) have become important for medical, pharmaceutical, and antimicrobial applications. According to the World Health Organization, metal-based NPs that are promising solutions against the resistance to traditional antibiotics have also proven to be effective against primarily listed pathogens. 6 Silver NPs (AgNPs), one of these nanometals, have shown high toxicity against microorganisms due to their antibacterial properties. 7 Zinc and zinc oxide NPs (Zn/ZnO NPs) also exhibited activity against different microorganisms. It prevents microbes from adhering to biomedical surfaces and can reduce their viability. In addition, Zn transforms into nanocomposites with metals such as silver (Ag) or copper and increases their antimicrobial activity. 8 Therefore, Ag and ZnNP-doped HAp filling material can be used to prevent implant-associated infection. Metal NPs can be synthesised using physical, chemical, green, and biological techniques. 9 Physical and chemical methods have been used for a long time. Nevertheless, these methods are quite expensive, toxic, and can have undesirable adverse effects on human health and the environment. 10 Thus, green chemistry has become increasingly attractive in producing metallic NPs. Plant extracts are more suitable candidates for green synthesis because they do not require toxic reducing and capping agents, radiation, or high temperatures. 11 Many plant extracts are rich in natural antioxidants and antimicrobial compounds. These properties can be imparted to the NPs during synthesis, making them useful in various applications related to antimicrobial coatings. This study aims to impart antibacterial properties to HAp produced from sheep bone for use in the biomedical field. Therefore, Ag and Ag@ZnNPs synthesised using a green method that is non-toxic, environmentally friendly and biocompatible, were doped into the HAP powder Marrubium astracanicum, which is used in the synthesis of NP, is a plant species in Turkey. This plant, called mountain tea, treats cough, phlegm, sore throat, and respiratory system diseases due to its rich antioxidant content. 12 The physicochemical properties of the prepared composite were characterised by spectrometric and microscopic methods. Moreover, ion release amounts were determined using inductively coupled plasma mass spectrometry (ICP-MS). Finally, antibacterial activity was evaluated against Pseudomonas aeruginosa, Escherichia coli, Staphylococcus aureus and Candida albicans.
Materials and methods
Materials
Silver nitrate (AgNO3, 99.8%), ethanol (C2H6O), sodium hydroxide (NaOH), zinc nitrate hexahydrate (Zn(NO3)26H2O) used in AgNPs and ZnNPs syntheses were obtained from Sigma-Aldrich. The plant used for synthesis was purchased from the local market. Furthermore, sheep bones were purchased from veterinary-controlled local butchers in Turkey.
Preparation of HAp powder
HAp powder was obtained from sheep bone by thermal calcination with modifications in the previously reported literature.13,14 First, the bones were cleared of all soft and cartilaginous tissue with a knife. Next, it was boiled in a pressure cooker using water for about an hour to separate the organic materials (meat and marrow). Then 4% NaOH solution was prepared. Cleaned bones were boiled in a 4% (w/w) NaOH solution for half an hour in a pressure cooker (at about 125 °C) to prevent uncontrolled burning during the process of calcination. This process was repeated five to six times. After that, the bones were washed under tap water to remove chemical residue and calcined at 600 °C for 4 h, at a ramp rate of 5 °C min−1, using a muffle furnace. The calcinated bone samples were ground using a mechanical grinder to attain fine powder.
Biosynthesis of HAp–AgNPs and HAp–Ag@ZnNPs nanocomposites using plant extract
Preparation of the plant extracts
The plant obtained from the Tunceli region was washed in tap water and dried in the shade. The plant, which was ground into a blender, was weighed 20 g and added to the 1:2 ethanol/water mixture. It was boiled for 10 min at 60 °C in a 250 ml beaker. The mixture was kept at room temperature. It was centrifuged at 4000 rpm and filtered through filter paper. The filtrate was stored at 4 °C. 15
Synthesis of HAp–AgNPs and HAp–Ag@ZnNPs nanocomposites
The synthesis of AgNPs and Ag@ZnNPs on the HAp surface was based on the biological reduction of Ag+ and Zn2+ ions using plant extract as the reducing/stabiliser agent. Initially, 0.01 M 100 ml AgNO3 solution was prepared. Next, 2 g of HAp powder was added to the AgNO3 solution and mixed for 30 min on a magnetic stirrer. Then, 10 ml of plant extract was added to the HAp–AgNO3 mixture and stirred at room temperature for 24 h at 400 rpm. The HAp–AgNPs were collected by filtration and the residue was washed with ethanol. Finally, it was dried in an oven at 40 °C for 24 h. To synthesise HAp–Ag@Zn nanocomposite, firstly, 100 ml of Zn(NO3)26H2O (0.05 M) and 50 ml of AgNO3 (0.01 M) solution were prepared and mixed in a 250 beaker. Then, the same procedure was used for the HAp–AgNP synthesis. 16
Characterisation
The crystallinity and phase of all samples were characterised by X-ray diffraction (XRD: D/max 2550 V, Rigaku Miniflex, Japan) with monochromatic CuKa radiation. Data were collected in the 2θ range of 20° to 80° with a scanning step of 0.02°. Fourier-transform infrared spectroscopy (FT-IR) analysis was performed using ATR-FTIR 6700 Jasco (Japan). Spectral data were acquired in transmittance mode with 16 scans at a resolution of 4 cm−1 in the 4000–400 cm−1 range. Surface morphology was examined with a scanning electron microscope (SEM; 400 F Field Emission, UANTA) equipped with EDX. Before SEM analysis, samples were sputter coated with gold. TEM analysis was performed on a JJOEL Brand, JEM-1011 electron microscope at an accelerating voltage of 100 kV. Samples were prepared by ultrasonically dispersing the powders in ethanol before being collected on carbon-coated copper grids.
Ag and Zn ion release behaviour
A 0.3 g of HAp–AgNPs and a 0.3 g HAp–Ag@ZnNPs were separately introduced into glass bottles containing 25 ml of phosphate-buffered saline (PBS) and incubated at 37 °C in a CO2 incubator for 1, 3, 7, 14, 21, and 28 days to determine the release of Ag and Zn ions. At the end of each period, a 2 ml sample was taken with a sterile syringe and filtered through an MILTEX-HP filter unit with a pore size of 0.45 μm. The amount of Ag+ and Zn2+ ion concentration in the collected samples was determined by ICP-MS. 17
Antimicrobial activity
The agar well diffusion method was used to examine the antimicrobial activity of HAp, HAp–AgNPs, and HAp–Ag@ZnNPs nanocomposite against Gram-negative E. coli (ATCC 25922), Pseudomonas aeroginosa (P. aeroginosa, ATCC PAO1), Gram-positive S. aureus (ATCC 25923) and C. albicans (ATCC 90028) fungus. Gram-positive and Gram-negative microorganisms were incubated overnight on Tryptic soy agar at 37 °C, while C. albicans fungus was set on potato dextrose agar at 28 °C. After incubation, suspensions of microorganisms at 0.5 McFarland turbidity were prepared in physiological saline, and standardised suspensions of each bacterium were placed on Müller–Hinton agar plates. Next, 8 mm diameter wells were drilled in the Petri using a sterile cork borer. Each sample was prepared at three different concentrations (5, 10, and 20 mg/ml), and 100 µl was loaded into the wells. Gentamicin and fluconazole solutions were used as control antibiotics for bacteria and fungi. Antimicrobial activity was measured as a zone of inhibition (mm) around the sample discs using a standard steel ruler after 24 h of incubation. The experiment was carried out in triplicate. 18
Results and discussion
Observing the colour change
The milky white slurry of HAp turned dark brown when AgNP was loaded and grey-brown when ZnNP was loaded at the end of the reaction (Figure 1). The solution colour changes from yellow to dark brown to greyish brown during the formation of AgNPs and ZnNPs by bioreduction. The discernible colour change was observed due to the surface plasmon resonance excitation, one of the most important indicators of NP formation. 19

Photo image of (a) pristine HAp, (b) HAp–AgNPs, and (c) HAp–Ag@ZnNPs solution and powder.
SEM–EDX results
SEM and EDX are used to analyse the morphological and chemical composition of the sample. Figure 2 shows the surface morphology of HAp, HAp–AgNPs, and HAp–Ag@ZnNPs nanocomposite samples. HAp has a porous structure according to the SEM image, HAp particles appear irregular in shape and size. 20 EDX analysis results of HAp–AgNPs and HAp–Ag@ZnNPs nanocomposite powders show that Ag and Ag@ZnNPs are successfully doped into the HAp structure. On the other hand, it was observed that the Ca/P ratio values based on the EDX spectrum were 2.39 for the pure HAp sample. This value was 2.10 for Ag–HAp and 2.20 for Ag@Zn–Hap. The theoretical composition of pure HAp consists of 39.68% calcium and 18.45% phosphorus by weight. The molar stoichiometric HAp value is 1.67, and the atomic is 2.15. 21 The high Ca/P ratio in the calcined bone sample may be due to phosphate ions substituted with carbonate ions in the HAp lattice structure. 4 It was observed that the Ca/P ratio decreased in AgNPs and Ag@ZnNPs loaded HAp. This may be due to calcium deficiency in HAp due to the cationic exchange of Ag+ and Zn2+ ions with Ca2+ ions.
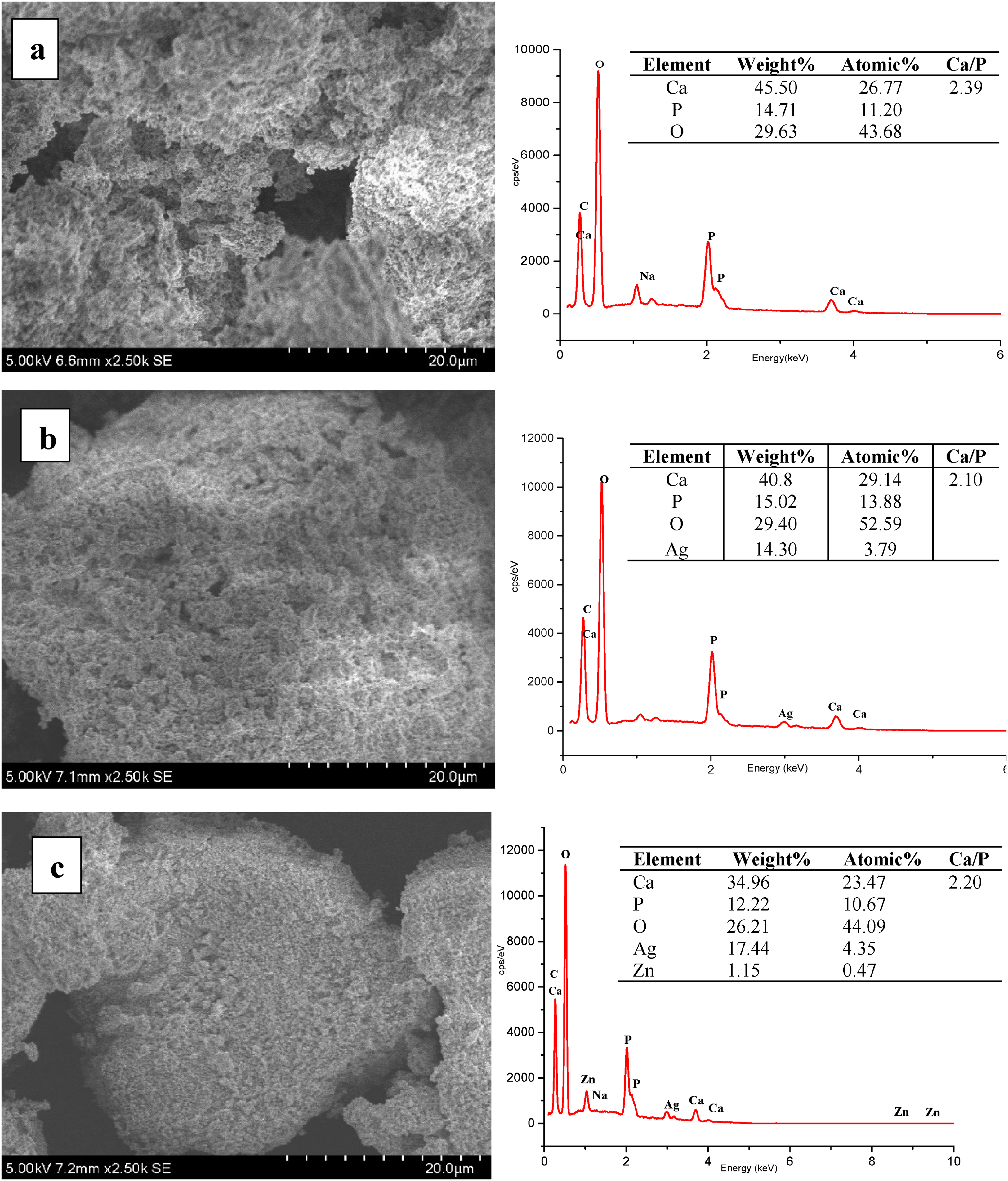
SEM image and EDX analysis of (a) pristine HAp, (b) HAp–AgNPs and (c) HAp–Ag@ZnNPs composites.
TEM results
Figure 3 shows the TEM image of the prepared HAp–AgNPs and HAp–Ag@ZnNPs nanocomposite. The presence of Ag and Ag@ZnNPs in different shapes and sizes is observed on the surface of the HAp crystal. These outcomes confirm that the Ag and Zn ions had been reduced from AgNO3 and ZnNO3 salts using plant extract. 22 The TEM images show that the HAp powder sizes ranged from 0.1 to 0.5 µm and particles formed agglomerated. It can also be seen that HAp powder has a spherical shape. All NPs were essentially spherical and the size distribution ranges from 5 to 30 nm.

TEM image of (a) HAp–AgNPs and (b) HAp–Ag@ZnNPs composites.
XRD results
XRD analyses are presented in Figure 4. The Miller indices of the HAp, AgNPs, and Ag@ZnNPs are labelled in black, red, and blue, respectively. The crystal structure of HAp revealed peaks at 2θ values of 25.7°, 28.8°, 31.7°, 33.9°, 32.1°, 32.9°, 39.8°, 46.6°, 48.0°, and 49.4° corresponding to (0 0 2), (2 1 0), (2 1 1), (1 1 2), (3 0 0), (2 0 2), (3 1 0), (2 2 2), (3 1 2) and (2 1 3) planes of HAp (JCPDS No. 9-0342).2,23 The corresponding Miller indices of (111), (200), (220), and (311) confirm the presence of AgNPs (JCPDS No. 04-0783).
24
Furthermore, the XRD peaks of the ZnNPs were observed at 002, 101, and 110 (JPCDS No. 36-1451)
25
and Ag@ZnNPs peaks (111), (200), (220) and (311), confirming the doping of Ag on the Zn.
16
HAP has a hexagonal crystal structure, but when the metal is added to the system, causes an asymmetry. Therefore, the crystallinity of the material decreases and the density peaks decrease.
26
The planes of the crystal structure of HAp in HAp–AgNP and HAp–Ag@ZnNP do not lead to significant effects on the crystal form of the NPs. However, HAp showed sharper (narrower) XRD peaks, while HAp–AgNP and HAp–Ag@ZnNP showed broader XRD peaks. It revealed that the metals synthesised and deposited on the surface of HAp by green synthesis changed the crystallinity to a more amorphous structure. The crystallite size of the mean HAp was calculated from the XRD peak broadening using the Debye–Scherrer equation for a characteristic peak:
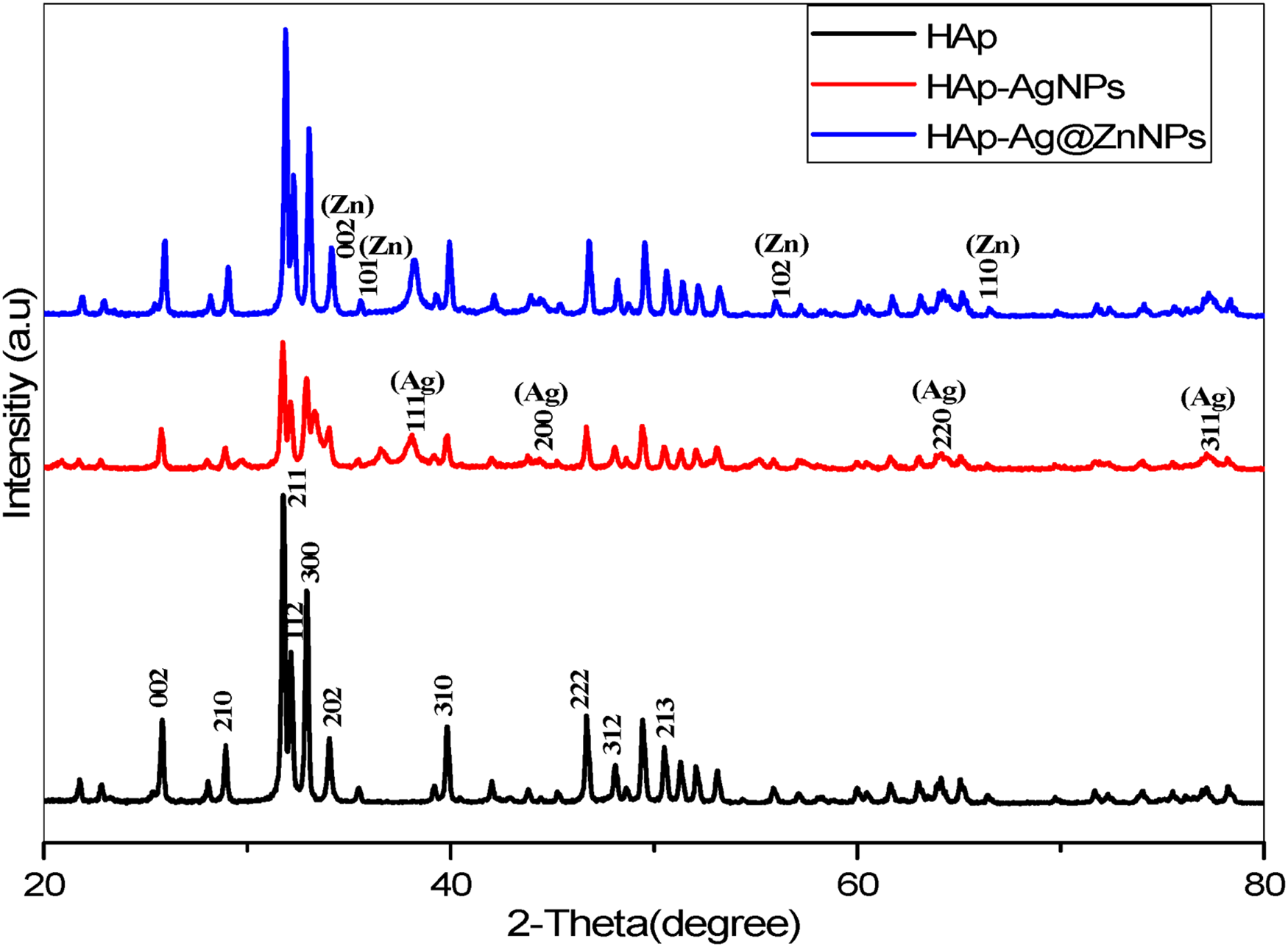
XRD patterns of pristine HAp, HAp–AgNPs and HAp–Ag@ZnNPs composites, respectively.
FT-IR Results
FT-IR analysis was used to determine the functional-component groups of AgNPs and Ag@ZnNPs-doped HAp microparticles, as shown in Figure 5. FT-IR spectra are shown as HAp (red line), HAp–AgNPs (blue line), and HAp–Ag@ZnNPs (black line). In these spectra, bands placed at 1455, 1412, and 873.6 cm−1, corresponding to the C–O symmetrical vibrational bending or stretching groups, can be identified. The broad and characteristic bands at 1017 and 561 cm−1 are assigned to the PO43− ions. Three peaks at 1093, 1023 and 960 cm−1 should be attributed to asymmetric and symmetric stretch, and two peaks at 601 and 562 cm−1 are attributed to the symmetric bend of the phosphate group. Antisymmetric stretching vibration of CO32− in the 1500–1400 cm−1 region and asymmetric bend in 873 cm−1 are observed. 28 On the other hand, HAp–AgNPs and HAp–Ag@ZnNPs nanocomposites indicated a band of properties similar to natural HAp.
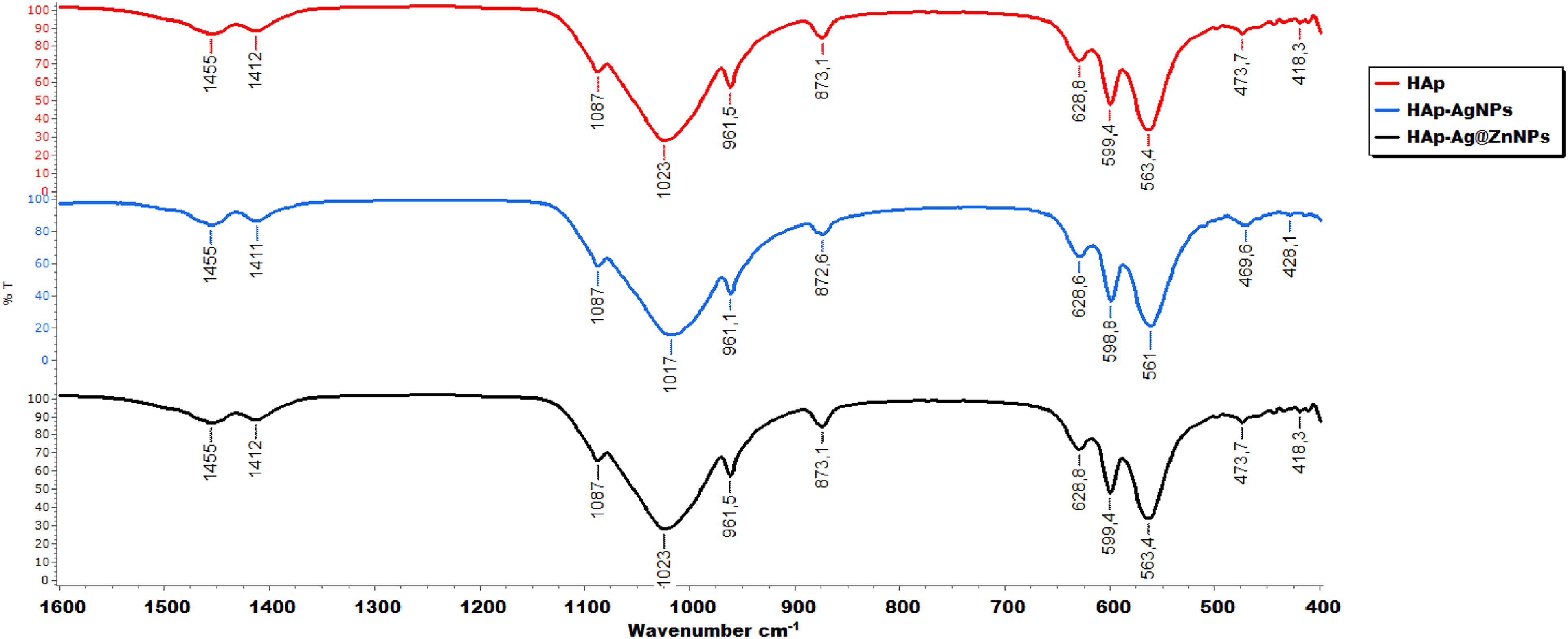
FT-IR spectrum of pristine HAp (black), HAp–AgNPs (red) and HAp–Ag@ZnNPs (blue) composites, respectively.
Ag and Zn ion release result
The release of HAp–AgNPs and HAp–Ag@ZnNPs nanocomposites, Ag ions, and Zn ions in 25 ml of PBS at 37 °C for 1, 3, 7, 14, 21, and 28 days was investigated (Figure 6). As seen in the TEM images, AgNPs and Ag@ZnNPs were generally synthesised on the outer surface and a small amount on the inner structure. As anticipated, HAp–AgNPs released higher amounts of Ag than HAp–Ag@ZnNPs nanocomposite in the first seven days. The decrease in Ag ion release in HAp–Ag@ZnNPs nanocomposites is due to the presence of Zn. Also, a slow and sustained release was obtained after an initial burst release in the composites. After the initial release of Ag at the surface, the release has become limited diffusion. The Zn ion released from the ZnNP burst on the first day and reached equilibrium.29,30
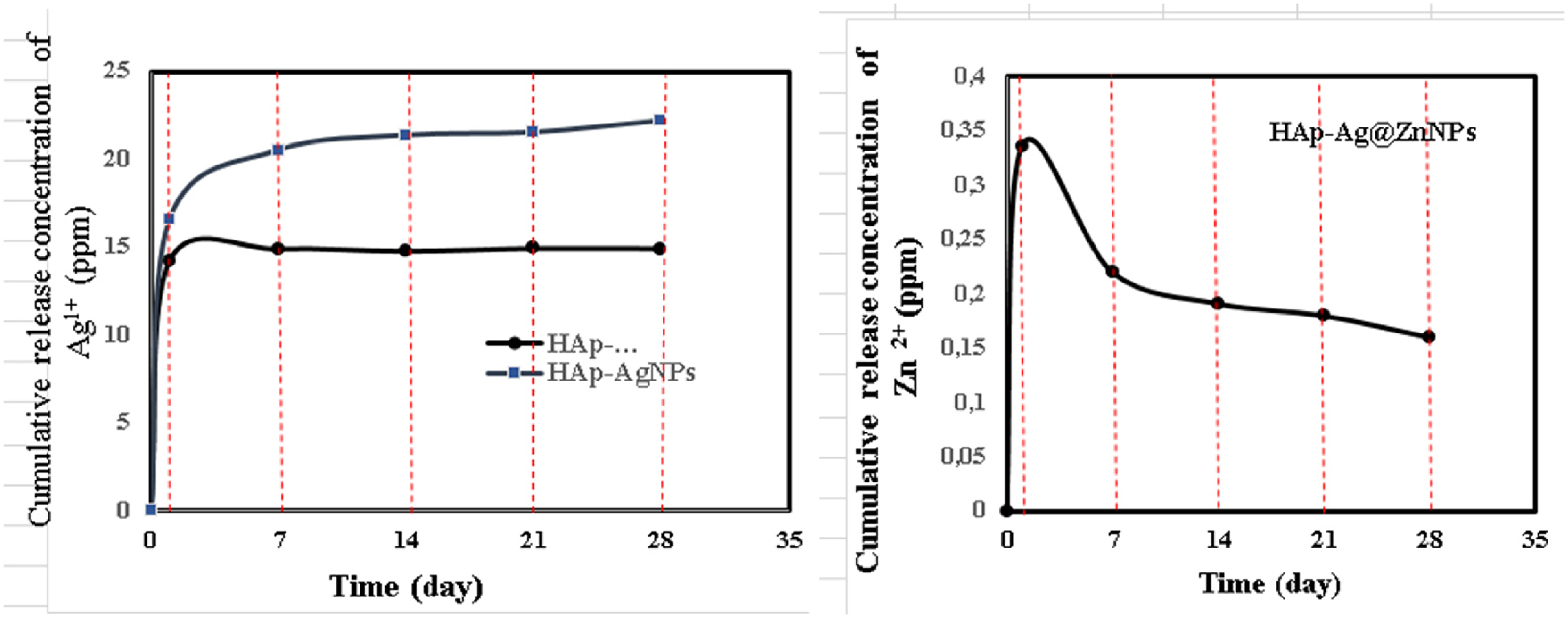
The cumulative release concentration (ppm) of Ag+ and Zn2+ after soaking in PBS for a different time, respectively.
Antimicrobial activity
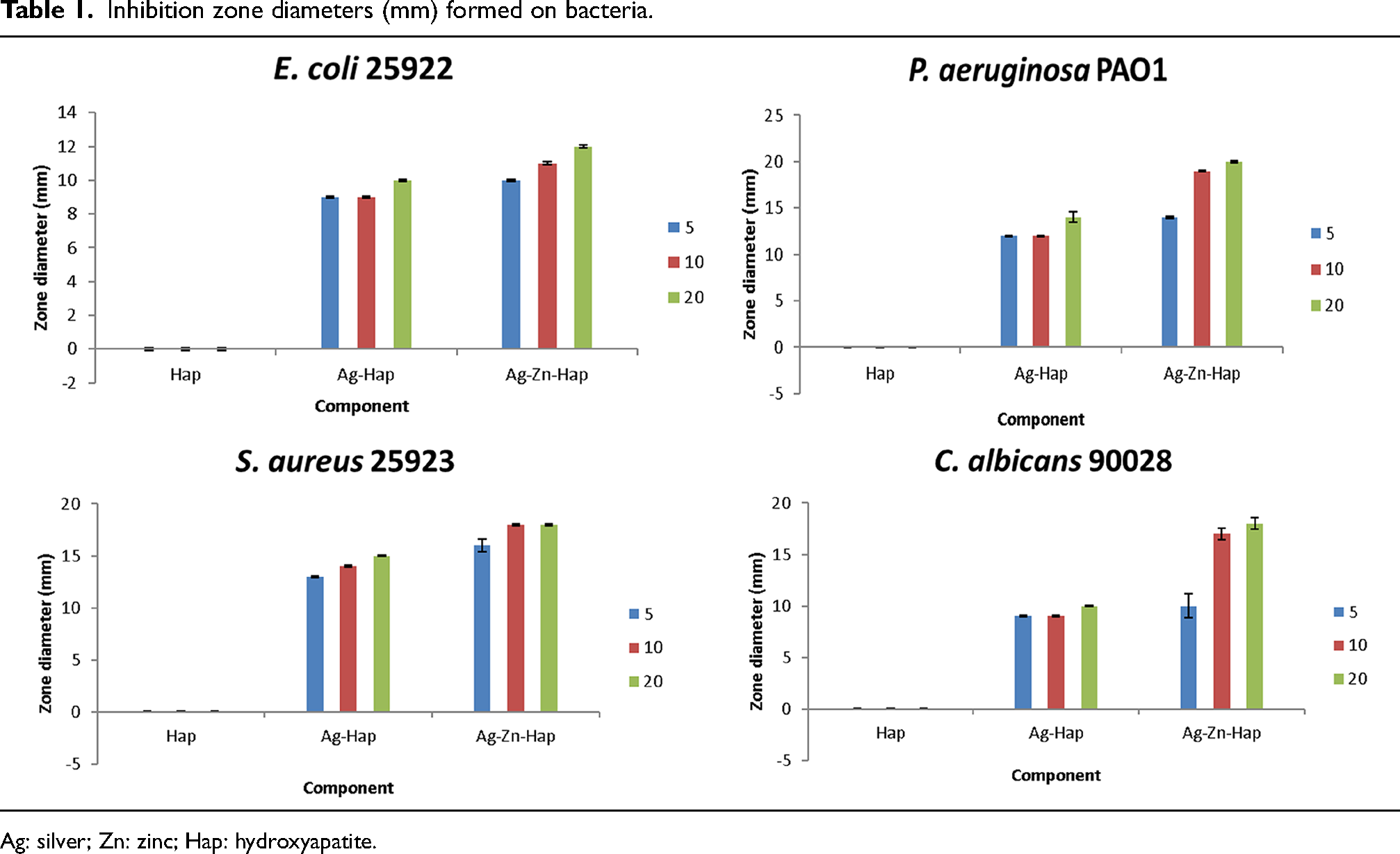
The results revealed that a clear antimicrobial activity is observed for all the prepared HAp–AgNPs and HAp–Ag@ZnNPs composites against P. aeruginosa, E. coli, S. aureus, and C. albicans (Figure 7). Pristine HAp did not lead to any traceable antimicrobial activity against the strains. When compared the inhibition zone shows that the higher the concentration of HAp–AgNPs and HAp–Ag@ZnNPs nanocomposite, the greater the inhibition zone. The antimicrobial effect increased as the concentration increased. Moreover, the antibacterial and antifungal activities of HAp–Ag@ZnNPs nanocomposite were higher than HAp–AgNPs (Table 1). HAp–AgNPs displayed a large inhibition zone against S. aureus bacteria, while HAp–Ag@ZnNPs showed against P. aeruginosa bacteria at 20 mg ml−1 concentration. In previous studies, it was observed that HAp-doped Ag and ZnO NPs increased antimicrobial activity. 5 However, AgNPs and ZnNPs induce toxicity through increased reactive oxygen species production. In addition, ions released from NPs disrupt cell stability and lead to death. 31
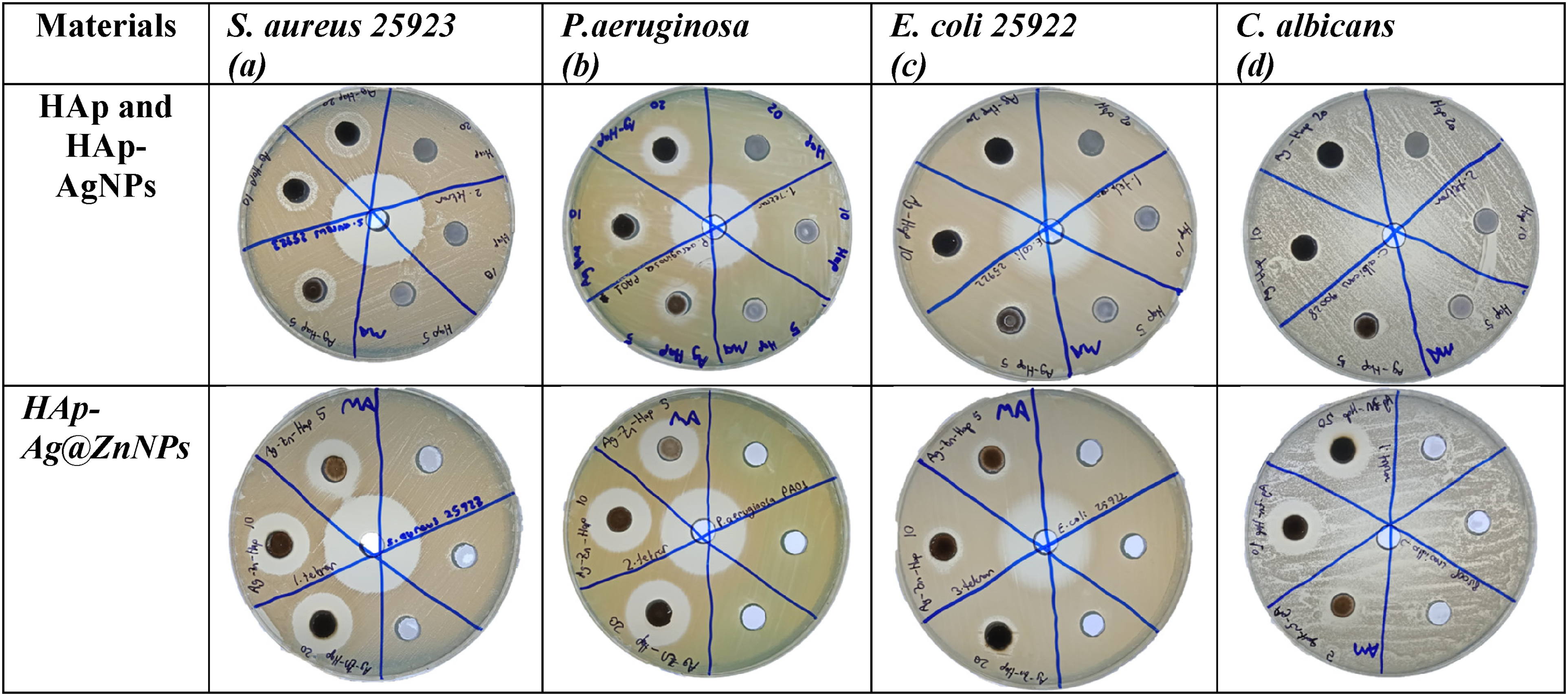
Inhibition zones of pristine HAp, HAp–AgNPs, and HAp–Ag@ZnNPs for: (a) Escherichia coli, (b) Pseudomonas aeroginosa, (c) Staphylococcus aureus, (e) Candida albicans (5, 10, and 20 mg ml−1).
Conclusion
In summary, biosynthesis of AgNPs and Ag@ZnNPs-doped HAp composite was synthesised in a simple, inexpensive, and environmentally friendly method. TEM results showed that NPs were synthesised in different shapes and sizes. Ag+ and Zn2+ releases provided effective bacterial resistance against Gram-negative, Gram-positive, and fungal strains increased proportionally with the increasing concentration. Furthermore, adding ZnNPs increased the antibacterial activity through the synergistic effects of Ag+ and Zn2+. It is important to underline that the synthesised composites as antimicrobial materials have a high potential for medical treatments focused on controlling antibiotic-resistant bacterial diseases.
Footnotes
Author contributions
All authors contributed equally to this work.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.