Abstract
Introduction
Rising healthcare costs, driven by demographic changes, advancements in medical technology, and growing pharmaceutical development expenses, emphasize the need for affordable medications, particularly in emerging economies like Iran. Generic and branded generic alternatives aim to improve accessibility and affordability due to their lower prices compared to original brands. Policymakers, producers, and service providers argue over the benefits and constraints of each, but little is known about consumers’ perspectives, which are ultimately the decision-makers.
Aim
This study aimed to compare customers’ perspectives towards branded-generic medicines and to identify major variables determining their acceptability.
Method
In 2023, a descriptive-analytical cross-sectional survey was conducted on 390 consumers in Tehran, Iran. Online data collection was performed by a valid questionnaire, evaluating knowledge, perception, and practices. Averages of indices in these areas were analyzed and correlated with socio-demographic and background variables.
Results
Findings indicated that respondents’ knowledge about branded generic drugs was average. However, 70% of them had average to high acceptance of branded generics over generics and original brands, meaning that their drug choice processes were high. Chronic illness, working in the healthcare industry, exposure to adverse drug reactions, negative substitute experiences, and insurance coverage were notable variables which induced positive attitudes and behaviors. Interestingly, acceptance was inversely associated with greater awareness of branded generics.
Conclusion
Customers gave more importance to branded generics than original brands and generics even though they were less aware and perceived. This preference indicates that branded generics are more appealing and trustworthy, especially for elderly patients who form a vast majority of the user base. Branded generic marketing will bring about greater patient preference from a marketing point of view, but making patients more aware of generics can result in using cheaper alternatives instead of brand-name generics, and this will be to the advantage of the healthcare system.
• A significant portion of respondents incorrectly view branded generics as more effective and of higher quality than their generic equivalents, highlighting the need for improving knowledge in generic concept. • Despite knowledge gaps and negative perceptions, 59% of respondents trust service providers and are willing to choose branded generic medications. • Knowledge, attitude, and practice of patients regarding branded generic can be vary according to socio-demographic characterizations and personal backgrounds.
Impacts statements
Introduction
The rising healthcare usage and expenses can be attributed to factors like increased life expectancy, demographic shifts towards older populations, advancements in medical technologies and emergence of new illnesses. 1 Additionally, escalated pharmaceutical expenses driven by greater medication usage and extravagant costs of pharmaceutical research and development, contributes to the overall increase in healthcare expenditures. As the pharmaceutical industry advances and new medicine emerge, ensuring medicine affordability without compromising quality becomes imperative. This need is particularly important for populations in middle-income countries such as Iran, where significant inequalities in healthcare remain unresolved. 2
For years, branded medicines were the prevalent form of medicines until the introduction of generic medicines aimed at improving accessibility and affordability. Generic Medicine, which are bioequivalent to brand-name medicines priced 20%–90% lower than branded counterparts, are therapeutically equivalent alternatives produced after patent expiration. Branded generics, which are bioequivalent to branded medicine but priced around 33% cheaper, offer a middle ground between original branded and generic medicine. 3 The pharmaceutical market in Iran is segmented into branded, generic, and branded generic medicines. The generic brand policy in Iran was introduced in 2000 and implemented between 2002 and 2004 to distinguish pharmaceutical products from several companies. 2 Despite the substantial amount of research focusing on the perspective of patients on branded medicine and generic equivalents, there is limited investigation into consumers’ views on branded generics.
In addition, despite the differing opinions of policy makers, producers, and service providers on advantages and disadvantages of generic versus branded generic choices, a significant gap remains in understanding consumer perspectives—the end users who play a crucial role in purchasing decisions. This gap calls for further research to better address patients’ attitudes and preferences.
The study begins with a comprehensive review of factors shaping patients’ perspective, followed by a field survey, and an analysis which explore the average measures and correlation of data across knowledge, attitude, and practice of patients, further exploring their associations with demographic variables to address research inquiries.
Theoretical background
Although patients' willingness to uptake branded generics may increase health system expenditures, it is crucial for fostering strong relationships with medical providers, ensuring adherence to treatment regimens, and enhancing overall health outcomes. If branded generic naming structure were not confusing for patients and service providers, branded generic medicines may influence both patients’ well-being and providers' success. The pharmaceutical industry, driven by scientific advancements and market demands, continually innovates to improve quality of life. Although generic medicines offer cost-effective alternatives, patients’ perspectives often favour branded medicines. Enhancing the image and confidence in generic medicines could significantly impact acceptability, affordability, and accessibility of medicines. Branded generics as a middle solution between original brands and generic medicines, warrant the perspective of patients on cheaper alternatives.
Consumers often have mixed views about generic equivalents. Some studies suggest that consumer confidence in generic medicines is increasing, particularly in developed countries. 4 However, other research indicates that patients may harbor negative perspective concerning the quality, efficacy, and safety of generic medicines,5–9 even when they are familiar with the concept of generic substitution. For instance, some individuals correlate generic medicines with poorer quality in comparison to their brand-name equivalents.10,11 Some studies perceived a lack of understanding regarding the approval processes for generic medicines as to contribute to this perception. 12 In some cases, patients have had negative experiences after switching to generics, leading to decreased willingness to use them again.10,13 Concerns about safety, efficacy, and a perceived difference in effectiveness compared to branded counterparts remain significant barriers to generic medicine acceptance.
Consumer attitudes towards generic medicines, including those with a brand name attached, are influenced by multiple factors, including past experiences, personal beliefs, and social influences. 4 Positive experiences with generic medications can give rise to higher acceptance rates. A study by Hassali et al. 14 found that patients who had previously used and were satisfied with generic medicines were more inclined to select them again in the future. Conversely, negative experiences, such as perceived side effects or perceived lack of efficacy, can foster negative attitudes and resistance to using generic medicines.4,12 This underscores the importance of addressing these concerns and providing personalized counselling to patients, particularly those who have had negative experiences. Branded generics give patients who had a bad experience with use of generic medications a second chance to choose another alternative with affordable prices. This is especially important when dealing with chronic conditions like epilepsy, where patients may be more hesitant to switch medications due to concerns about seizure control. 15
Healthcare professionals, particularly physicians and pharmacists have a pivotal role in shaping consumer perceptions and acceptance of generic medicines. They can educate patients about generic medicines, address their concerns, and promote informed decision-making through clear and concise communication about bio-equivalency, safety, and cost-effectiveness. Additionally, government policies and regulations have a significant impact in influencing the utilization of generic medications. 16 Policies promoting generic substitution, such as mandatory substitution laws or financial incentives, can encourage generic medicine adoption while balancing the need for patient choice. For example, in Germany, pharmacists are obligated to replace brand-name prescriptions with generics when available. However, in other countries, like Spain, pharmacists have more flexibility in substitution practices. 17 In addition to government, and service provider, pharmaceutical companies may play a key role by producing generic medicines as high quality as original branded ones and by improving their quality image of their branded generic. In summary, the underlying personal factors influencing patients’ knowledge, perception, and exercise regarding branded generic medicine determined, as age, 18 educational level,19,20 Positive past experiences with branded generics, experience of switching between medicine, trust in pharmacists’ recommendations,13,21 trust in physicians’ prescribed medication choices,3,9,22,23 efficacy concerns, 5 Patient familiarity with branded generics, price consideration,24,25 disease severity, 7 packaging and appearance, 26 Manufacturer’s origin,27,28 and previous experience of unexpected side effects.15,17,19,29
Aim
The aims of this study were to compare customers’ perspectives towards branded-generic medicines and to identify major variables determining their acceptability. Specifically, the research aimed to examine patients’ knowledge, attitude, and practice (KAP) toward branded generics, exploring factors that influence their acceptance within the context of the Iranian pharmaceutical market.
Ethics approval
This study did not require formal ethical approval due to its nature. Nevertheless, ethical principles were upheld throughout the process. Participants were provided with detailed information about the study on the initial page of the questionnaire. They were given the option to decline participation at the outset or withdraw at any point during the study. Respondents were assured that all responses would remain confidential and that no personal identifiers would be disclosed either during the research or in the final report.
Method
Representative sample
The data were gathered from a descriptive, cross-sectional KAP survey conducted in Tehran in 2023. Respondents were randomly selected from consumers visiting private and public pharmacies in Tehran. The questionnaire was delivered to consumers through a domestic online platform, Porsline. 30 Out of 851 shared questionnaires, 390 were found to have complete answers, yielding a response rate of 45.8%, achieving the sufficient sample size of 385 calculated by Cochrane’s formula. The sample size for this study was calculated using the Cochrane Equation, a widely recognized formula for determining sample sizes to ensure sufficient statistical power to detect meaningful differences in assessing perspectives of consumers toward branded generics. To this end, by using the latest population size of Tehran (8.7 million 31 ) sample size was determined through Cochrane Equation, considering a significance level of 0.05. The data gathering process lasted from April 26 to June 18, 2023.
Questionnaire development
The initial questionnaire draft, comprising 38 items derived through deductive approach (literature review), was reviewed by 15 specialists to develop the content validity. The specialists had over 15 years of experience in clinical practice. Using Content Validity Index (CVI) and Content Validity Ratio (CVR), the instrument was examined for its relevance and comprehension, as well as for lack of bias and appropriateness of the items, 32 which reduced the number of questions from 38 to 28. Additionally, applying Cronbach’s alpha coefficient to 15 completed surveys confirmed the validity and reliability of the updated questionnaire.
This created a valid, self-administered survey, which consists of 28 questions, focused on consumers’ perspective of branded generic medications. Seven concerning patients’ knowledge about branded generic medicine; 10 questions covering patients’ perceptions and attitude; and 11 for evaluating the practice of patients toward branded generic medicines. In addition to the core 28 items, the questionnaire incorporated nine questions designed to gather socio-demographic data (i.e. gender, age, educational level, occupation, type of insurance coverage) and other relevant background information (i.e. employment in the healthcare sector, having chronic diseases, familiarity with prescription substitutions with branded generics, and experiences of medical side effects reported by respondents.
Questions aimed to assess respondents’ knowledge regarding branded generics categorized into sub-sections including level of familiarity with branded generic medicines, having information about their quality, safety, efficacy, and price, we examine the knowledge of patients using a two-point response system with “Yes” and “No”. The respondents’ attitude regarding branded generic medicines examined through 10 questions about their perception on efficacy, quality and safety. The respondent’s practice level was also assessed using 11 practical questions using a 5-point Likert scale.
Then, respondents’ socio-demographic variables were gathered in nominal and ordinal classifications, while background data were assessed using a 2-point response format (“Yes” or “No”) response format.
Data analysis
After sending the questionnaire to 851 pharmacies’ customers in different part of Tehran through online platform, 390 full responses were obtained, and SPSS software was used to perform descriptive and inferential statistics on the data. Descriptive statistics (such as frequency, mean, and standard deviation) offered the main perspective on the data, and the Kolmogorov-Smirnov test was used to determine if the data were normal. The Mann-Whitney U and Kruskal-Wallis tests were used when necessary to assess the effects of respondents’ demographic and background characteristics on their knowledge, perception, and attitude. A relationship was deemed statistically significant when the p-value was below or equal to 0.05.
Results
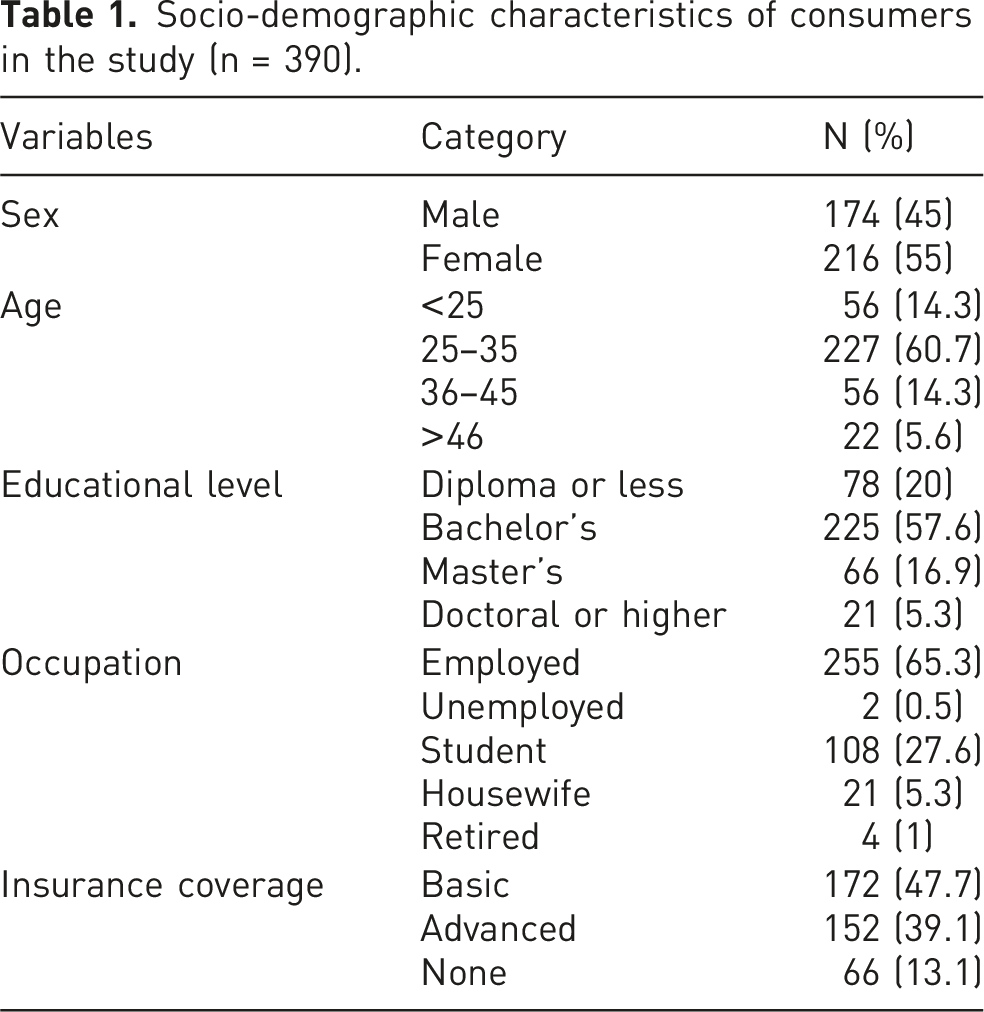
Socio-demographic characteristics of consumers in the study (n = 390).
Background factors of participants in the study (n = 390).

Consumers’ knowledge of generic medicine substitution (n = 390).
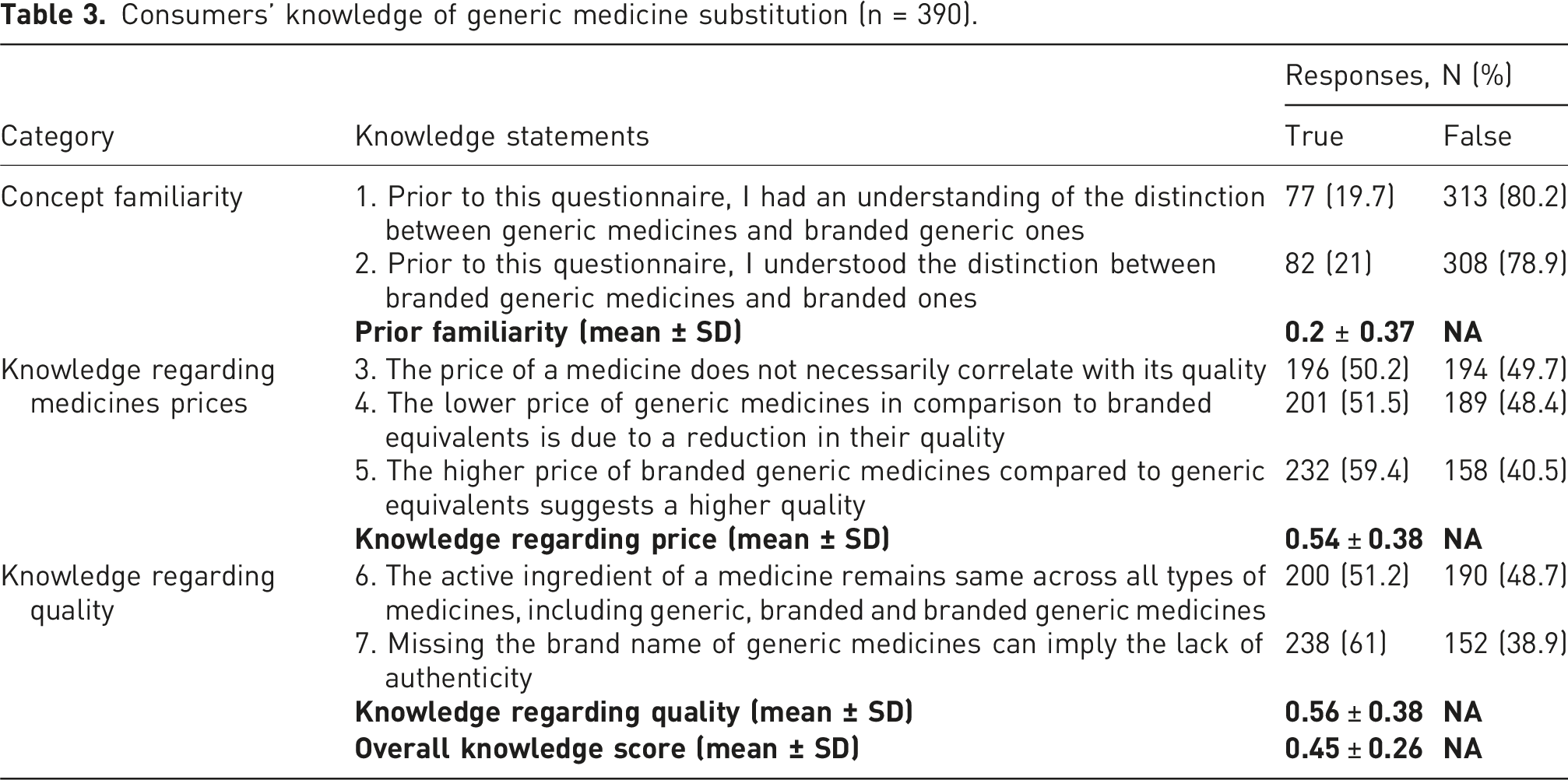
According to descriptive statistics derived from the frequency data in Table 3, approximately 80% of participants did not possess knowledge concerning the distinction between the three types of medicines, generic, branded generic, and original brands.
Although majority of the participants correctly do not attribute the absence of a brand name to a drug’s lack of authenticity, many of them (approximately half) mistakenly perceive that the medicine price are directly correlated with its quality, then identified branded generic medicines as of higher quality compared to the generic alternatives (∼40%) due to their higher prices. Moreover, approximately half of them (∼49%) wrongly do not acknowledge the uniformity of active ingredients across all generic, branded generic and branded medicines.
For categorizeing consumers’ knowledge regarding branded generic medicines to high, medium, and low knowledge, we assume six to seven correct answers out of 7 item as high knowledge, four to five correct responses as medium knowledge, and low knowledge included participants with fewer than three correct answers. The findings indicated that approximately 9.5% of participants possess high knowledge, around 31% displayed medium level of knowledge, and roughly 60% demonstrated low knowledge (not knowledgeable) regarding generic medicines. The mean score calculated by assigning 0 and 1 to each false and true questions.
We grouped respondents based on their answers into positive, average, and negative perception regarding branded generic medicine. Approximately 5% showed a positive perception (8-10 positive answers), 28% had an average perception (5-7 positive answers), and 68% exhibited a negative perception (4 positive answers or fewer) regarding branded generic medicine.
Consumers’ attitude of generic medicines substitution (n = 390).
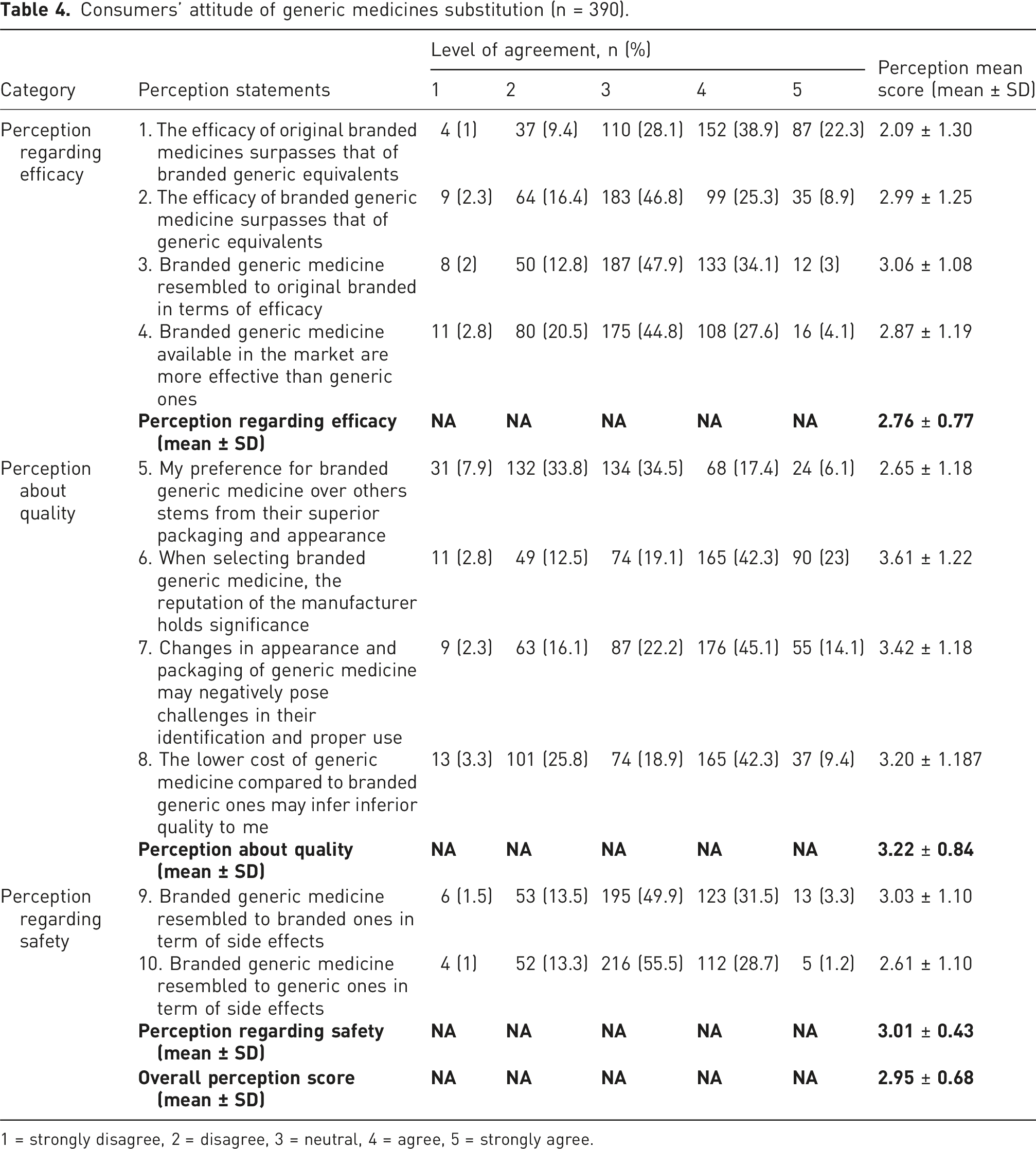
1 = strongly disagree, 2 = disagree, 3 = neutral, 4 = agree, 5 = strongly agree.
As majority of respondents declare the changes in appearance and packaging of generic drugs, as well as manufacturer reputation influence their favor to a branded medicine, more than half of participants attribute their choice to higher price of branded generics as believe it indicates higher quality compared to generic equivalents.
Moreover, regarding safety, particularly in relation to side effects, while most of answers inclined to “no idea”, respondents viewed branded generic medicine as more akin to original branded medicine than to generic ones. We grouped respondents based on their answers into positive, average, and negative perception regarding branded generic medicine. Approximately 5% showed a positive perception (8-10 positive answers), 28% had an average perception (5-7 positive answers), and 68% exhibited a negative perception (4 positive answers or fewer) regarding branded generic medicine.
Consumers’ attitude of generic medicines substitution (n = 390).
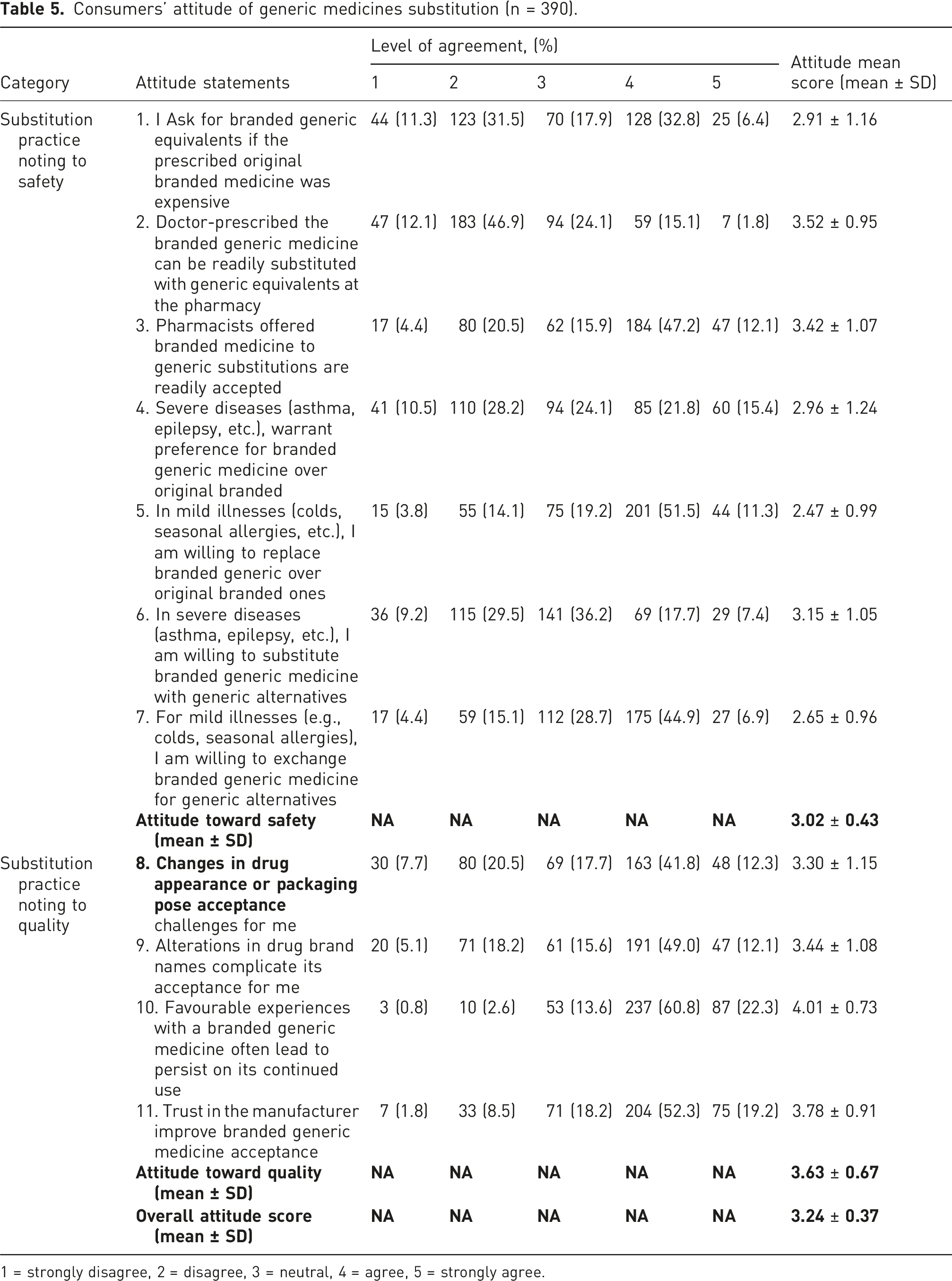
1 = strongly disagree, 2 = disagree, 3 = neutral, 4 = agree, 5 = strongly agree.
Regarding medication quality, the findings indicated a notable level of sensitivity among participants towards a range of factors. In Practice, over half (∼54%) of customers displayed hesitation towards changes in packaging appearance, while a substantial majority exhibited confidence in previous positive experiences (∼83%) and trust in the country and manufacturing reputation (71.5%) when selecting branded generic medication.
To explore participants’ habits, participants were grouped into three categories—positive, average, and negative habits regarding branded generic substitution—depending on their number of positive answers. Those with 8-11 positive responses were deemed positive practice, 5-7 were considered average, and 4 or fewer were categorized as negative routines. Results indicated that approximately 18% demonstrated positive practice, 51% showed average, and 31% exhibited low practice regarding branded generic medicine substitution.
Finally, we try to test the influence of socio‐demographic and background factors of participants on their knowledge, attitude, and practice towards branded generic substitution.
The impact of professional and demographic characteristics of participants’ KAP regarding branded generic medicines.
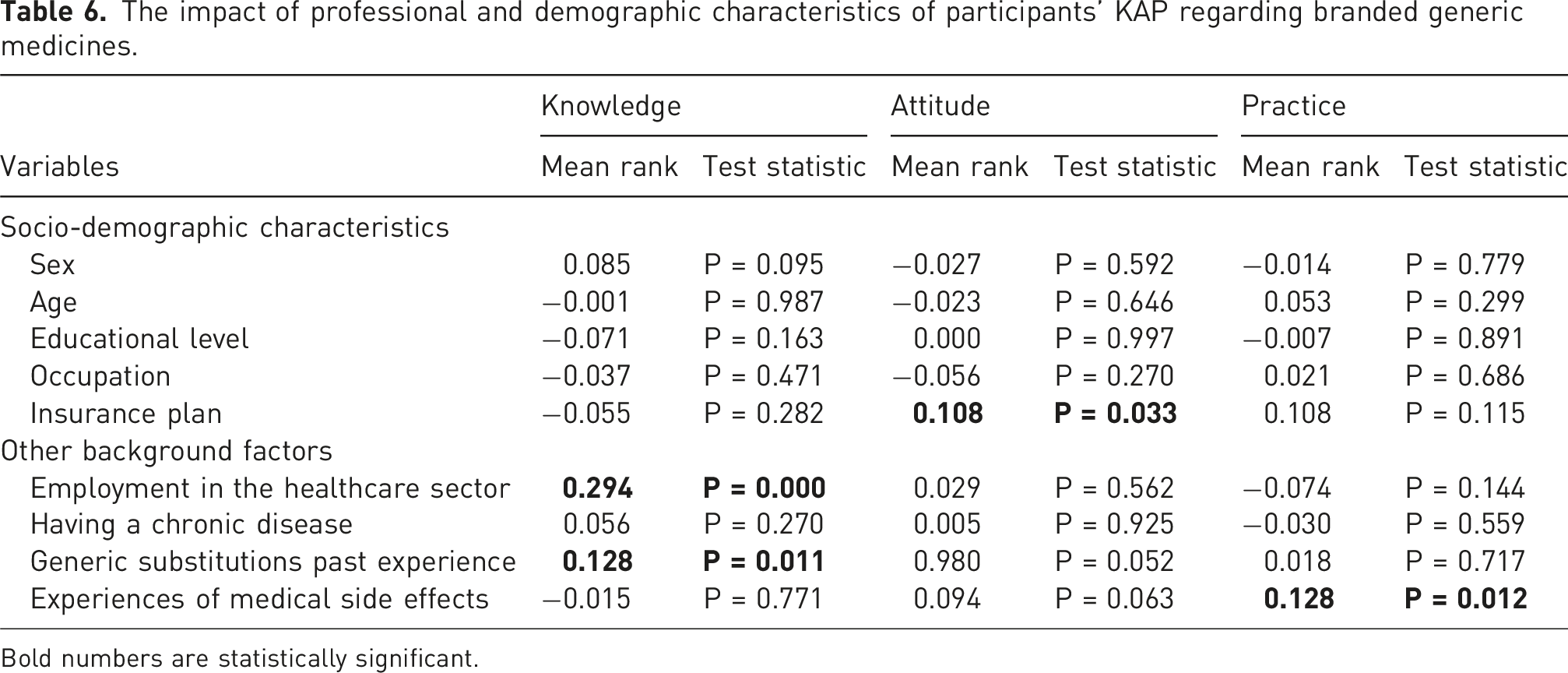
Bold numbers are statistically significant.
Although the knowledge, attitude, and practice of customers were different in our sample based on their gender, age, educational level, and etc, these differences were not statistically significant and only 4 of them (experience of medical side effect, generic substitution experience, being health system member, and insurance plan) can be generalized to the population.
Discussion
The choice and use of therapeutic agents may be influenced by consumers’ attitudes, perceptions, and knowledge about branded generic medications.
Regarding respondents’ knowledge of branded generic medicines in our study, approximately 80% of the participants did not have prior familiarity or knowledge for distinguishing between original brands, generics and branded generic medicines prior to this study. The fact that only 20% of respondents had familiarity with branded generics medicines is far lower than similar studies conducted in Auckland, New Zealand (51.6%), 11 Kuala Lumpur, Malaysia (67.5%), 26 Germany (63%), 13 India (65%), 33 France (80%), Canada (72%), Spain (58%), and the United Kingdom (32%). 17 Moreover, from the total participants, approximately 51% correctly cited that all three types of above-mentioned medications contain the same quantity of same active pharmaceutical ingredients. And this is also lower than the similar study carried out in Ethiopia (81.1%) 6 and Japan (71.1%). 8 Nevertheless, this finding is almost identical to the study conducted in Hong Kong (41.2%). 34
Additionally, approximately 40% of respondents view pricing as an indicator of quality, perceiving branded medications to be of higher quality than branded generics and generics, primarily due to the price differences. This finding is consistent with the results of a study conducted in Germany (36.7%), 13 but is this finding seems far more sceptic regarding branded generics compared to the studies conducted in Ireland (29%), 10 but it is lower than the similar study done in Ethiopia (56.8%). 35
The important point is while people have shown a lack of knowledge regarding the branded medicine, branded generics, and generics, they simultaneously maintain a positive practice toward original brand and branded generic medicines, which is consistent with studies carried out in Auckland,11,36 Ireland, 10 and United Kingdom. 17
The current study indicates that although the overall respondents’ perception showed a negative view about branded generic medicines, approximately 61% believe that original branded medicines are more effective than their branded generic and generic equivalents.
The study also showed that about 37% of respondents thinks branded generic medicine has an analogous efficacy to original branded ones. This finding is lower than similar study conducted in India (75%). 37 In addition, about 59% of respondents correctly tended to believe changes in appearance and packaging of generic drugs influence their decision for utilization, this measure is analogous to another finding. However, 65% of respondents attribute manufacturer’s reputation as an influencing factor on their acceptance of medicine which is much lower than India (78.6%). 38 On the other hand, regarding safety, particularly in relation to side effects, about 35% of respondents believed generic alternatives brings more side effects than branded ones. This finding is also similar to other studies conducted in the United States (33.7%), 19 and Norway (24%). 29
Concerning respondents’ practice, the current study indicated 59% of participants in the study had shown a preference to switch from their prescribed branded medicine to branded generic alternatives if suggested by a pharmacist. But this finding is much different from other studies took in Netherland (73%) 39 and India (63.9%), 37 where a substantial number of respondents did not support pharmacists substituting generics. This finding can be perceived as a high level of trust and confidence on pharmacists’ suggestion in Iran’s community pharmacies. Also, about 42.7% of respondents did not favour branded generics over their prescribed branded medicine if there were a potential cost savings. This finding is similar to another study on antiepileptic drugs in the United States (43%). 15 This can be a demonstration of patients’ high level of trust in their practitioner and their faith in what is prescribed for them.
A serious disease scenario also lowered switching from branded medicine to generic alternatives under these conditions, as about 40% of respondents did not agree to substitute their branded medicine in case of contracting to a serious disease. This is quite different from another study in New Zealand, 11 where 58.7% of participants experience levels of anxiety and such as asthma, diabetes and heart problems, 58.7% were found to be willing to switch to a generic alternative. But the statistics were comparatively different in mild disease, where 50 to 60% of respondents were in favor of switching into generic and branded generic equivalents. This is similar to another study carried out in New Zealand (78%). 11
Regarding quality, attitudes of respondents showed more sensitivity towards various factors in practice; as 54% of respondents stated that even a change in appearance and packaging of medicine would pose a change in their buying decision. This finding is similar to another study held in Netherland (50%), 39 but it is much more than other studies in New Zealand (36%), 11 and Finland (1%). 24 Also, about 83% counted on their previous experience of medicine as indicator of medicine quality and choose a medicine that they have a positive experience with it. This is analogous but more than the other studies held in Malaysia (40%). 26 Moreover, approximately 70% of participants exhibited confidence in manufacturing reputation and trust to the medicine origin country when selecting their medicines. This result is extremely higher than similar study conducted in Finland (10%). 40
We found no significant association between the socio-demographic characteristics and the respondents’ knowledge, perception, and attitude towards branded generic medicine. But some background factors conceived to have an impact on the perspective of consumers.
Our finding in this study not only help pharmaceutical manufacturers for expanding the market share of their branded generic medicines by attention to patients attitudes and preferences, but also shed a light for health policy makers on patients’ perceptions about branded generics to improve health outcome by substitution original brand with branded generics, and branded generic with generic medicines or vice versa based on the priority of costs or acceptability in defined situation.
The present study, akin to prior research endeavours, is not immune to scientific and operational constraints that warrant careful interpretation and generalization of findings. These limitations include the restricted pharmaceuticals market in Iran. The economic downturn in Iran has resulted in a scarcity of original brands in Iran, leading to a dominance of branded generic and generic drugs in the pharmaceutical market. Educational disparities may had effect on data collection due to online method of data gathering particularly among individuals have less access to online services.
Conclusion
This research contributes to our understanding of the knowledge and attitude of consumers when choosing their medicines. The lack of knowledge regarding the differences and distinction between groups original brands, branded generic, and generic medicines may lead to expense for choices which do not have added any added value for patients’ health outcomes.
This research also showed the perception of consumers regarding the efficacy, appearance and manufacturer reputation, and their view of side effects in each group of medicine when making purchases. More than 68% of respondents recognized to have a negative perception regarding branded generic, and perceived original brands more efficacious. They also have an uncertain perception regarding the side effects of branded generic medicines as they were not sure they could be similar to original brands.
This research also adds to that body of knowledge, via a deeper understanding of practice of consumers’ perceiving branded generics over branded and generic alternative when making purchases as a safer option in regard to generics and comparable to branded alternatives, while attributing the quality of medicine to reputation of manufacturer, packaging, and physical characteristics of branded generic medicines.
The research also shows the effects of these variables could be independent of each other and level of knowledge or negative perception of branded medicine would not necessarily result in a poor practice in branded generic selection by patients.
Footnotes
Acknowledgements
We are thankful to all study participants who kindly shared their thoughts and feelings with us.
Ethical consideration
The study was approved by the Ethics Committee of the Medical University of Shahid Beheshti (ref. no. IR. SBMU.PHARMACY.REC.1400.264, date 14 December 2021).
Author contributions
Conceptualization, N.Y.; methodology, N.Y. and S.H.; software, S.H.; validation, N.Y. and S.H.; analysis, S.H.; writing—original draft preparation, S.H. ; writing—review and editing, N.Y and N.M.; supervision, N.Y. and N.M. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
