Abstract
Numerous successful gene-targeted therapies are arising for the treatment of a variety of rare diseases. At the same time, current treatment options for neurofibromatosis 1 and schwannomatosis are limited and do not directly address loss of gene/protein function. In addition, treatments have mostly focused on symptomatic tumors, but have failed to address multisystem involvement in these conditions. Gene-targeted therapies hold promise to address these limitations. However, despite intense interest over decades, multiple preclinical and clinical issues need to be resolved before they become a reality. The optimal approaches to gene-, mRNA-, or protein restoration and to delivery to the appropriate cell types remain elusive. Preclinical models that recapitulate manifestations of neurofibromatosis 1 and schwannomatosis need to be refined. The development of validated assays for measuring neurofibromin and merlin activity in animal and human tissues will be critical for early-stage trials, as will the selection of appropriate patients, based on their individual genotypes and risk/benefit balance. Once the safety of gene-targeted therapy for symptomatic tumors has been established, the possibility of addressing a wide range of symptoms, including non-tumor manifestations, should be explored. As preclinical efforts are underway, it will be essential to educate both clinicians and those affected by neurofibromatosis 1/schwannomatosis about the risks and benefits of gene-targeted therapy for these conditions.
Introduction
Neurofibromatosis 1 (NF1) and schwannomatosis (SWN), including
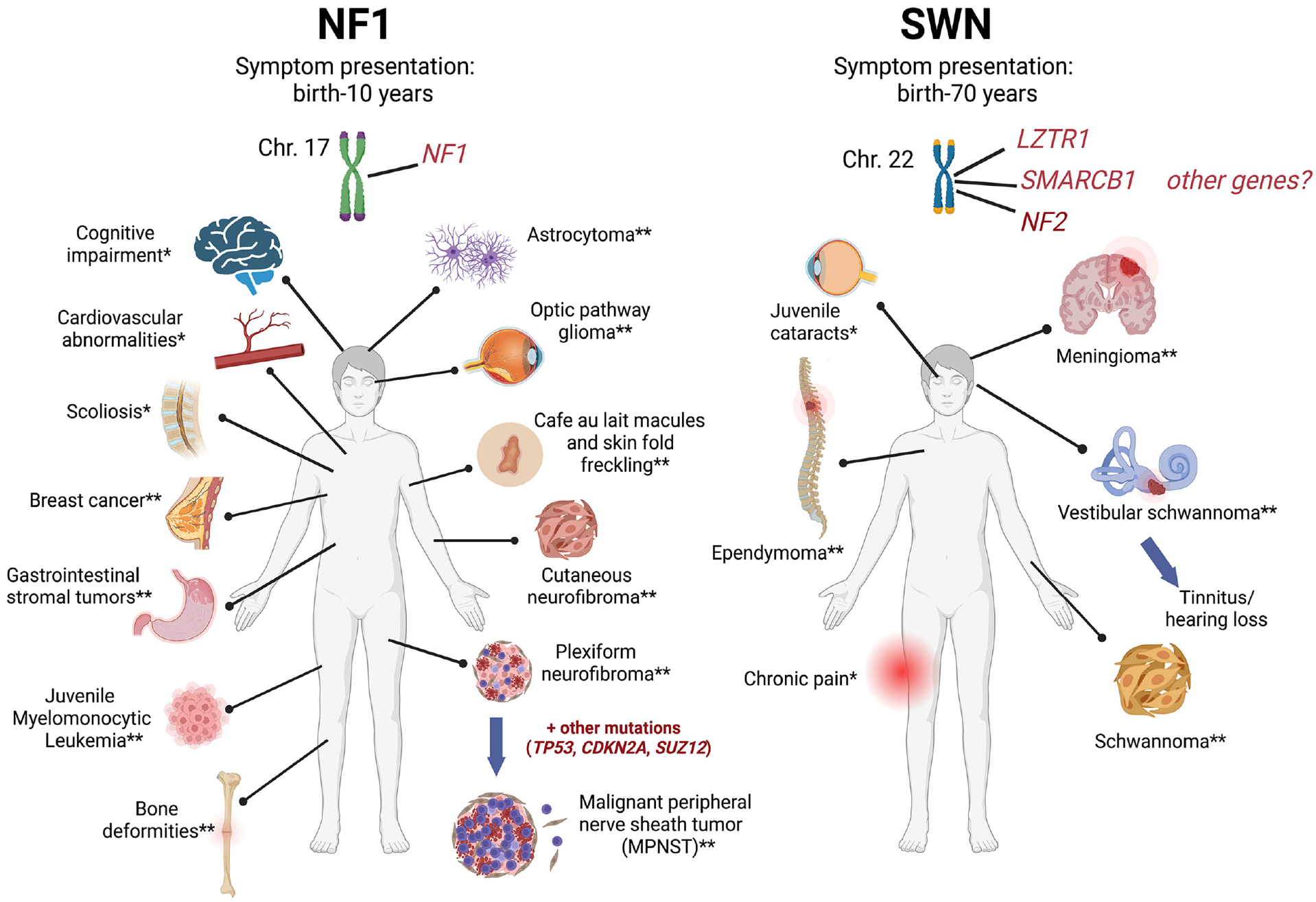
NF1/SWN symptom presentation.
Current success of approved gene-targeted therapy
Gene-targeted therapy, the process of manipulating a pathogenic gene variant or its product through a variety of different modalities, has provided novel treatment options for several inherited and acquired conditions, including hematologic disorders, immunodeficiencies, cancer, infectious diseases, ophthalmologic and dermatologic conditions, and neurological diseases. 12 As of December 2022, more than 27 gene therapy products are approved by the US Food and Drug Administration (FDA), with 5 therapies targeting monogenic disorders. 13 While these therapies may be life-changing for affected individuals, 12 they also fundamentally change how life-threatening disorders can be treated.14,15 Gene-targeted therapy is under intense investigation in oncology; however, its application for genetic conditions linked to tumor predisposition such as NF1 and SWN is still in its infancy.
Current treatment approach to NF1 and SWN
Most treatments for NF1 and SWN focus on symptomatic tumors. While surgery is the primary treatment, several chemotherapy trials for NF1 and SWN have been completed. Currently, the only drug approved specifically for people with NF1 is the Mitogen-activated protein kinase kinase (MEK) inhibitor selumetinib.6,16,17 Bevacizumab, a neutralizing antibody for vascular endothelial growth factor, is recommended for treatment of hearing loss related to progressive vestibular schwannomas in
Rationale and challenges for gene-targeted therapy for NF1 and SWN
For NF1 and SWN, gene-targeted therapies are designed to increase levels of functional proteins in cells that have either one functional gene copy, known as haploinsufficient cells, or no functional copies, defined as diploinsufficient cells, of a tumor suppressor gene. While diploinsufficiency is required for tumor formation, haploinsufficiency results in abnormal cell function that can also cause disease manifestations or can promote tumor progression through changes in the microenvironment. Thus, increasing functional protein levels holds promise for treating established tumors, for preventing tumor formation, and for treating non-tumor manifestations of these conditions.
NF1 is caused by pathogenic variants (PVs) in the
The main challenge in treating
Similar to NF1, diverse cell types give rise to schwannomas, meningiomas, and ependymomas; this necessitates defining the optimal target cells for treatment to avoid unnecessary exposure of off-target tissues and to optimize treatment window. The cellular responses to merlin overexpression in normal (merlin-expressing) tissues are not known. Moreover, people with
Currently investigated gene-targeted treatment modalities in NF1 and SWN
There are numerous potential gene-targeted therapy approaches and strategies that have recently been thoroughly reviewed and discussed in detail.37,38 Hence, we provide a brief overview. Understanding how each approach works informs selection of the most appropriate therapy for specific PVs, tissues, and manifestations. Genome editing can be used to alter the nucleotide sequence or the chemistry of targeted DNA bases. 39 These changes occur on a base-by-base basis, and are highly personalized, with new reagents designed for nearly each type of PV. Alternatively, additional copies of DNA (encoding all or a significant portion of a gene) can be introduced in gene replacement therapy, which either restores gene function (for most loss-of-function variants), or as suicide gene therapy to kill the target cell independent of the PV. Alternatively, small molecules and antisense oligonucleotides can target transcription and translation enhancers to increase protein expression from a remaining wild-type allele, or modulate protein degradation, each ultimately resulting in more functional protein. 27 At the RNA level, target mRNAs may be edited using trans-splicing ribozymes40–44 or SMaRT (Switching Mechanism at the 5′ end of RNA Template) technology,43,45,46 a strategy that does not permanently change the genome. mRNA splicing, as well as miRNA function, can also be modulated with antisense oligonucleotides, to control gene expression in a targeted manner.37,47 While mRNA editing and splicing are also highly personalized, they are well tolerated in pediatric patients with Duchenne muscular dystrophy or spinal muscular atrophy. 48 Nonsense variants, which result in negligible full-length protein expression, can be targeted by nonsense suppression therapy, which uses small molecules that target the translational machinery to restore translation of full-length, functional protein.49,50 Finally, proteins can be replaced, and modifier compounds can be utilized to translocate, stabilize, or otherwise help restore defective function. We consider each modality in detail and elaborate on advantages and disadvantages, their status for NF1/SWN, and examples of clinical success in Table 1.
Approaches for gene-targeted therapies for rare disease and their advantages, disadvantages, status for NF1/SWN, and examples of success.
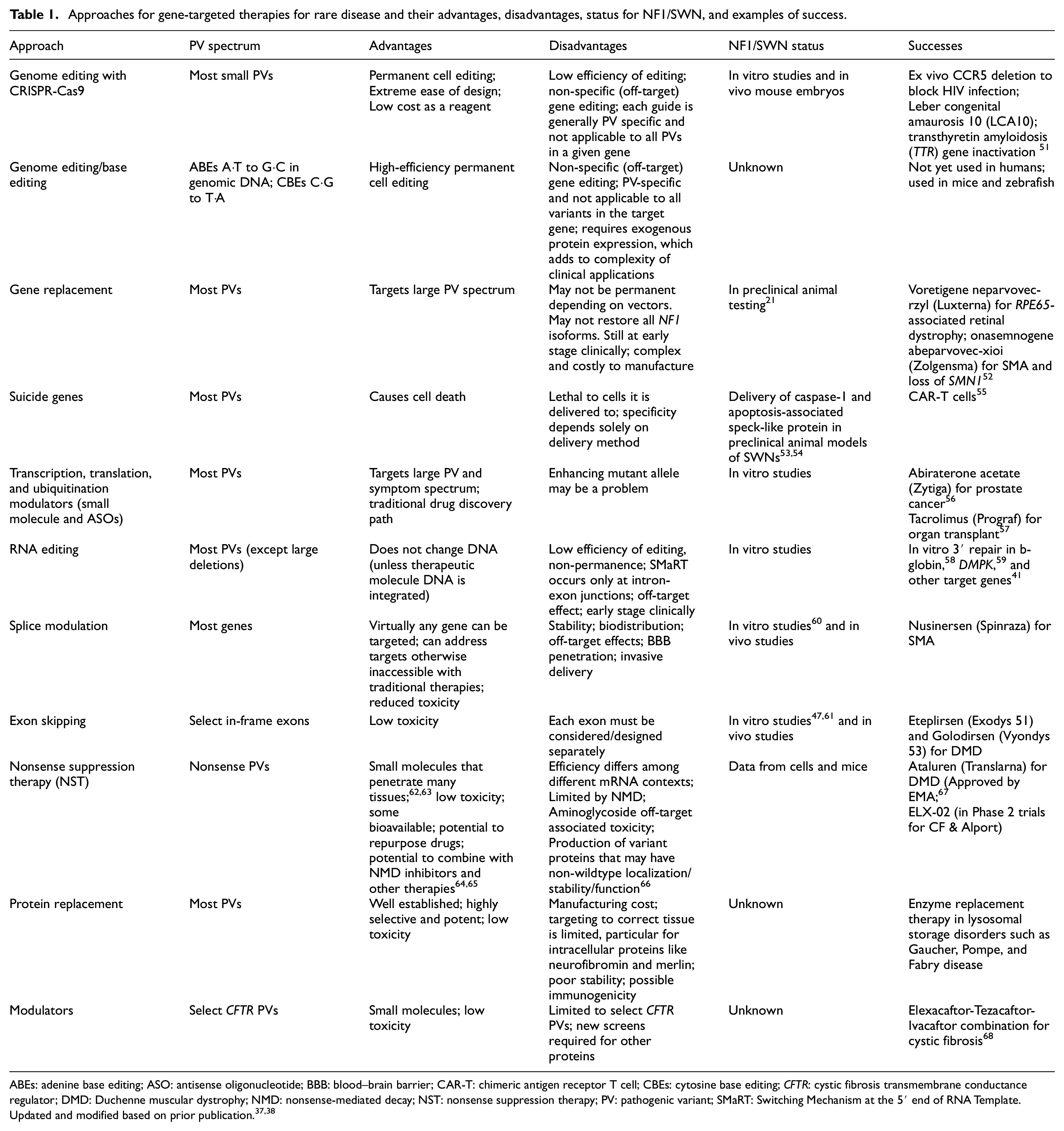
ABEs: adenine base editing; ASO: antisense oligonucleotide; BBB: blood–brain barrier; CAR-T: chimeric antigen receptor T cell; CBEs: cytosine base editing;
Delivery vector platforms
Intracellular delivery of gene-targeted therapy remains a major hurdle in the clinical translation of these treatments, and the safety and efficacy of therapies are largely dependent on the vehicle used to deliver the genetic material or cargo to the target cells. Outside of traditional small-molecule compounds (which travel across the cell membrane due to small size or lipophilicity), other delivery strategies include both viral and non-viral methods. Common viral vectors include adeno-associated virus, adenovirus, lentivirus/retrovirus, and herpes simplex virus 1. 14 Adeno-associated viruses are the most used vectors because they can infect multiple vertebrate species, including humans and non-human primates, can be engineered to increase tissue tropism, and are naturally replication deficient, a distinctive feature that is highly advantageous for their use as delivery vectors. 14 Non-viral methods include cell-penetrating peptides, nanoparticles, and bacteria. We consider each method in detail and elaborate on advantages and disadvantages of each, their status for NF1/SWN, and examples of success in Table 2.
Delivery methods for therapeutics, their applications, advantages, disadvantages, and success.
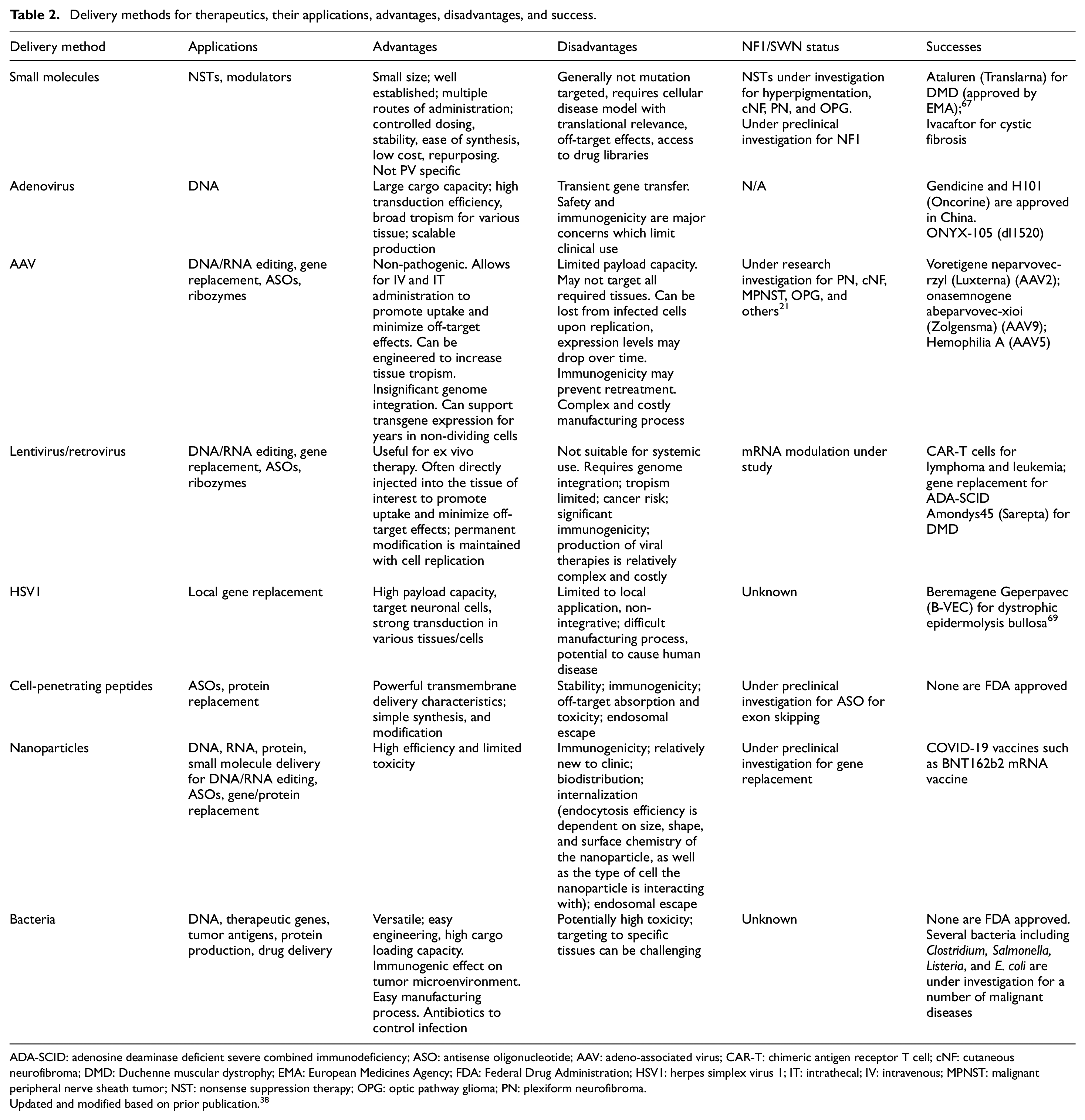
ADA-SCID: adenosine deaminase deficient severe combined immunodeficiency; ASO: antisense oligonucleotide; AAV: adeno-associated virus; CAR-T: chimeric antigen receptor T cell; cNF: cutaneous neurofibroma; DMD: Duchenne muscular dystrophy; EMA: European Medicines Agency; FDA: Federal Drug Administration; HSV1: herpes simplex virus 1; IT: intrathecal; IV: intravenous; MPNST: malignant peripheral nerve sheath tumor; NST: nonsense suppression therapy; OPG: optic pathway glioma; PN: plexiform neurofibroma.
Updated and modified based on prior publication. 38
Some primary delivery concerns include transgene capacity, tissue tropism, and transduction efficiency (the rate or level of transfer of genetic material into the cell of interest). Frequently, the cargo size is a determining factor in choosing the optimal delivery method. For example, the size of the
Preclinical testing in NF1 and SWN models
Ideally, gene-targeted therapy approaches should be tested in animal models that are based on human PVs and that recapitulate tumor natural history as far as growth rate, location, and therapeutic responses (model validation). However, due to the diverse spectrum of
Cre-drivers and variants for mammalian NF1 and SWN models.
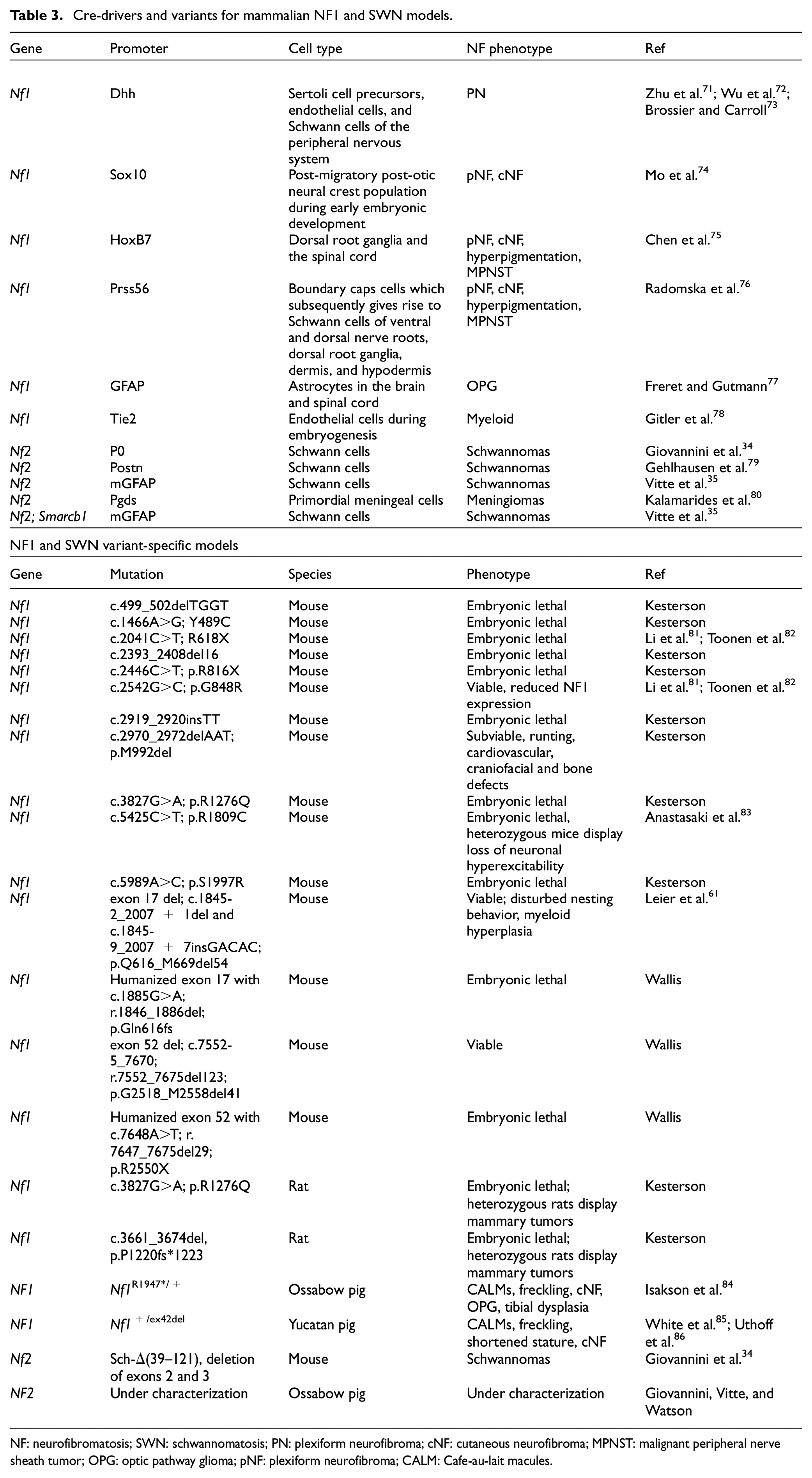
NF: neurofibromatosis; SWN: schwannomatosis; PN: plexiform neurofibroma; cNF: cutaneous neurofibroma; MPNST: malignant peripheral nerve sheath tumor; OPG: optic pathway glioma; pNF: plexiform neurofibroma; CALM: Cafe-au-lait macules.
Murine models have been used in several gene therapy studies as the initial preclinical evaluation platform, including those for cancer, hematological, and neurological conditions.
87
Several mouse models have been designed to mimic loss of heterozygosity at the
Several genetically engineered mouse models of
The use of large animal models (pig) with physiology and metabolism closer to humans could improve the predictivity of preclinical testing.
87
Porcine models are well situated to assess different delivery approaches and toxicity studies, as was shown in spinal muscular atrophy and Duchenne muscular dystrophy,
87
whereas lower species (mouse) can be used to address biologic/mechanistic questions. In NF1, two fairly well-characterized porcine minipig models exist, manifesting clinical features of cutaneous neurofibromas, neurocognitive symptoms, and optic pathway gliomas;84,85 in
Finally, non-human primates have played an important role in the evaluation of gene therapies because they are genetically closest to humans. 87 Their nervous systems and anatomy are similar to humans, making them a crucial model for evaluating biodistribution and safety of gene therapies targeting NF1 and SWN; however, non-human primates modeling NF1 and SWN currently do not exist. Hence, not only would utilization of these models to evaluate efficacy require precision model development, but also extensive phenotypic characterization including natural history studies. Notably, non-human primate models were not required for approval of selumetinib and may not be essential for NF-related gene therapy efforts.
Efficacy considerations related to gene-targeted therapy for NF1 and SWN
The therapeutic goals of gene-targeted therapy include stabilization, shrinkage, or prevention of tumors, and treatment of non-tumor manifestations. Given the rarity of these genetic conditions, randomized trials are not practical. Thus, appropriate controls are likely to be derived from Natural History Studies in NF1 and NF2 that have prospectively monitored disease manifestations over time. Selection of participants for clinical trials of gene-targeted therapy will require genotyping to confirm and categorize PVs. Application of the UK NF2 Genetic Severity Score or the revised Functional Genetic Severity Score may help define inclusion criteria and response criteria.
NF1 and SWN are simultaneously monogenic disorders and tumor suppressor syndromes. Effective gene-targeted therapies will need to address several key questions related to the germline and somatic genetic events that cause disease manifestations. For
For NF1, targets should initially focus on tumors with the highest morbidity and mortality, such as plexiform neurofibromas, MPNSTs, high-grade glioma, and optic pathway glioma.6–8 Cutaneous neurofibromas, which affect nearly all patients and cause significant disfigurement, may be another attractive target because, unlike plexiform neurofibromas, these tumors are not congenital and typically arise in adolescents and adults rather than young children. These lesions are accessible on the skin, allowing for easy tissue access and monitoring, and may provide opportunities to explore non-systemic administration, as demonstrated by repeated topical application of herpes simplex virus 1 for dystrophic epidermolysis bullosa.69,91 Schwann cells harboring biallelic inactivation of
Due to the significant mortality in
While symptomatic tumors remain the primary focus for NF1/SWN, a wide range of non-tumor manifestations also significantly impact quality of life and could be explored via gene-targeted therapies. Neurocognitive features of NF1, including learning, attention, and social deficits, affect most individuals to some degree, leading to challenges with academic achievement and social relationships. Brain autopsies and mouse studies demonstrate that neurofibromin loss can alter embryonal migration and differentiation of neuroglial progenitor cells,97–99 and causes pervasive myelin dysfunction,
100
which may limit the efficacy of gene therapies targeting neurocognitive manifestations. On the other hand, downregulation of RAS/ERK signaling in
Safety considerations for early-phase clinical trials of gene-targeted therapies
The promise of gene-targeted therapy has been tempered by the historical reality of safety concerns.105–108 Early-phase clinical trials for NF1/SWN must address safety concerns related to specific transgenes and the method of delivery to cells. For gene replacement therapy, it is possible that overexpression of
The potential short- and long-term consequences of overexpression of neurofibromin or merlin are unknown for both
Additional safety concerns for gene replacement are related to viral vectors that integrate into the host genome such as lentiviral vectors. These vectors can activate or inactive nearby genes, giving rise to neoplastic clones. In addition, they can persist in tissues for years after administration, and their effects may be permanent. This property is especially significant for children with NF1/SWN who are severely affected or who are likely to develop severe disease later in life. These children have the most to benefit from gene-targeted therapy but are at highest risk for long-term complications given their expected lifespan. The FDA requires long-term monitoring plans to understand the true safety profile of these products.
In addition, most viral vectors have the potential to elicit an immune response when administered repeatedly. In some situations, this immune response may be desirable (e.g. to stimulate an immune response in a tumor). In other situations, it could limit the efficacy, generate adverse events (i.e. autoimmune disease), or limit the ability to deliver subsequent treatments using the same vector. Given the lifelong risk for developing tumors in NF1 and SWN, retreatment with gene-targeted therapy may be desirable. The use of bacterial or viral vectors also raises the possibility that these can shed in bodily fluids, thereby placing others at infection risk.
Finally, the choice of delivery route is a critical issue and is dependent on the target symptom, vector, location, and indication (prevention vs symptomatic). Intravenous administration may be appropriate for non-tumor manifestations, diffuse disease (e.g. multiple tumors or metastatic MPNST), or when considering symptom prevention. However, intravenous administration necessitates large vector doses to achieve target tissue transduction. High doses may increase toxicity and strain manufacturing capacity, thus slowing clinical translation. 109 Direct administration to the central nervous system via intrathecal injections is more efficient, achieving effective transduction throughout the brain and spinal cord at relatively low vector doses. 110 This approach may be suitable for neurocognitive symptoms and tumors of the brain, spinal cord, and nerve roots. Finally, localized or topical gene transfer may be suitable for solitary and accessible tumors. This approach reduces the risk for systemic toxicity and supports repeated administration of viral vectors owing to reduced immunogenicity.
Conclusion
Gene-targeted therapies hold great promise for NF1/SWN. A key preclinical barrier to success involves defining approaches that can sufficiently target tissues of interest and restore functional protein in correct quantity and at the right time; treatment approaches might differ based on genotype, target symptom, and indication. A key clinical barrier involves identifying a cohort of patients with the appropriate genotype that are at sufficient risk to receive proportionate benefit, while avoiding exposure to individuals that are unlikely going to benefit. As preclinical studies of gene-targeted therapy progress, the wider NF1/SWN community needs to continue discussions surrounding the types of gene-targeted therapies and the potential risks associated with each in order to prepare for early-stage clinical trials.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: V.S. serves as a scientific advisor for the Gilbert Family Foundation Gene Therapy Initiative. H.S. is Founder/CEO of Infixion Bioscience, an early-stage drug discovery company targeting NF1. R.K. serves as a Scientific Advisor for Infixion Bioscience Inc. (3210 Merryfield Row San Diego, CA 92121). Y.K. serves as scientific officer at the Gilbert Family Foundation and as a member scientific advisory panel of NTAP’s Gene Therapy program. S.R.P. is co-founder of NFlection Therapeutics and NF2 Therapeutics and consults for AstraZeneca, SonalaSense, and Akouos. B.K. is a consultant for GenomeMedical, Recursion, Healx, and Springworks.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: V.S. is supported by NTAP’s Gene Therapy Initiative, the National Cancer Institute (NCI) (5K08CA230179 and 1U01CA247576), and the Sontag Distinguished Scientist Award. D.B., J.A.W., A.L., and D.W. are supported by the Gilbert Family Foundation Gene Therapy Initiative. J.A.W. is funded by the Children’s Tumor Foundation’s Gene Therapy Initiative. M.G. and J.V. are supported by NF2 Therapeutics. M.G. receives support from the Department of Defense CDMRP NFRP (W81XWH2110448). J.V. is funded by the Drug Discovery Initiative from the Children’s Tumor Foundation (2020-05-004). B.K. is funded by the NIH and DoD. D.W. and R.K. are supported by the NCI (R01CA26593).
