Abstract
Neurofibromatosis type 1 is a genetic syndrome characterized by a wide variety of tumor and non-tumor manifestations. Bone-related issues, such as scoliosis, tibial dysplasia, and low bone mineral density, are a significant source of morbidity for this population with limited treatment options. Some of the challenges to developing such treatments include the lack of consensus regarding the optimal methods to assess bone health in neurofibromatosis type 1 and limited data regarding the natural history of these manifestations. In this review, the Functional Committee of the Response Evaluation in Neurofibromatosis and Schwannomatosis International Collaboration: (1) presents the available techniques for measuring overall bone health and metabolism in persons with neurofibromatosis type 1, (2) reviews data for use of each of these measures in the neurofibromatosis type 1 population, and (3) describes the strengths and limitations for each method as they might be used in clinical trials targeting neurofibromatosis type 1 bone manifestations. The Response Evaluation in Neurofibromatosis and Schwannomatosis International Collaboration supports the development of a prospective, longitudinal natural history study focusing on the bone-related manifestations and relevant biomarkers of neurofibromatosis type 1. In addition, we suggest that the neurofibromatosis type 1 research community consider adding the less burdensome measurements of bone health as exploratory endpoints in ongoing or planned clinical trials for other neurofibromatosis type 1 manifestations to expand knowledge in the field.
Introduction
Neurofibromatosis type 1 (NF1), a disorder caused by loss of function of the tumor suppressor protein neurofibromin,1,2 impacts multiple organ systems. 3 The bone-related phenotypes of NF1 have been well described and include skeletal malformations such as long bone and sphenoid wing dysplasias,4,5 abnormalities that evolve over time such as dystrophic scoliosis, 6 and a decrease in bone mineral density (BMD) and overall bone quality compared with the general population.7–10 These skeletal manifestations cause significant morbidity for people with NF1; however, monitoring recommendations and treatment options remain limited.
With the advent of effective therapies for certain tumor-related manifestations of NF1 (e.g. mitogen-activating protein kinase kinase inhibitors), there is optimism that similar targeted approaches may lead to improved bone health in people with NF1. 11 However, there is no consensus regarding the optimal assessment of bone health in NF1, either in clinical practice or in the context of a clinical trial. While BMD measurement by dual-energy X-ray absorptiometry (DXA) is readily available and widely studied, BMD does not assess other properties which are important to bone health, including microarchitecture, degree of mineralization, extent of microdamage, and composition of the skeletal matrix and mineral that are important to bone strength and health, and may not ideally reflect the ongoing bone changes seen in NF1. 12
In this review, the Functional Committee of the Response Evaluation in Neurofibromatosis and Schwannomatosis (REiNS) International Collaboration: (1) presents the available techniques for measuring overall bone health and metabolism in persons with NF1, (2) reviews data for use of each of these measures in the NF1 population, and (3) describes the strengths and limitations for each method as they might be used in clinical trials targeting NF1 bone manifestations. Measurement techniques targeting other major bone-related manifestations of NF1, such as scoliosis and long bone dysplasia, as well as patient-reported outcome measures of the impact of these issues, are outside the scope of this review.
Bone health in NF1
Abnormal bone mineralization and metabolism have been demonstrated in people with NF1 compared with unaffected controls. 13 Lower BMD in the whole body has been demonstrated in both children and adults with NF1 and may occur in as many as 50% of individuals.14,15 In a small study of people with NF1-related scoliosis, the severity of curvature correlated with the decrease in lumbar spine density, 16 indicating a potential overlap. Similarly, plexiform neurofibromas, benign peripheral nerve sheath tumors in NF1, may have a direct negative impact on the mineralization of adjacent bones. 17 In addition, increased plexiform neurofibroma burden has been correlated with lower BMD in patients with NF1. 9 While this local effect may have a different etiology than systemic bone-related issues, both are important when considering potential clinical trial endpoints and ultimately clinical screening and treatment options. In addition, with ongoing use of mitogen-activating protein kinase kinase inhibitors and other targeted therapies for other NF1 manifestations, it will be important to monitor for potential impacts of these treatments on bone health.
The mechanisms by which loss of neurofibromin function leads to lowered BMD and abnormal bone metabolism in NF1 are incompletely understood. In people with NF1, histomorphometric analyses of bone biopsies have shown an overall decreased mineral content compared with age and sex-matched controls, in addition to reduced trabecular bone volume, increased osteoid volume, and increased osteoblast and osteoclast numbers. 18 Preclinical studies suggest that functional neurofibromin protein is necessary for normal osteoblast differentiation and mineralization19,20 and that loss of functional neurofibromin (e.g. protein haploinsufficiency) leads to increased bone resorption by osteoclasts.21–23 In addition, the hyperactivation of the Ras-Erk pathway also results in lower levels of tissue nonspecific alkaline phosphatase. 24 When present, alkaline phosphatase hydrolyzes inorganic pyrophosphate, which is a potent inhibitor of bone mineralization, to inorganic phosphate. Therefore, lower levels of alkaline phosphatase lead to elevated inorganic pyrophosphate levels which may contribute to the NF1-associated bone phenotype. 24
While the clinical impact of lower BMD in individuals with NF1 is not fully understood, several retrospective studies have documented an increase in the rate of fracture, particularly in the leg and foot, in both children 25 and adults8,26 with NF1. The largest of these retrospective studies compared 460 individuals with NF1 to nearly 4000 age-matched control subjects and found adults with NF1 ≥ 41 years had a risk ratio of 5.2-fold for fractures, and children with NF1 had a 3.4-fold increased risk for fractures. 26 However, none of these studies demonstrated an association between increased fracture risk and low BMD in individual patients. One retrospective review of 69 children with NF1 found no statistically significant correlation between those with reduced BMD (Z-score ≤ −2) and history of fracture; 9 however, the study was not sufficiently powered to answer this question. There does appear to be an association between the presence and severity of skeletal manifestations of NF1, such as scoliosis or tibial dysplasia, and low BMD. Yilmaz et al. 27 found an increased risk of lumbar Z-score ≤ −2 in children with NF1 and at least one skeletal manifestation, and Illés et al. 16 found that the severity of scoliosis in a small cohort of people with NF1 was inversely correlated with lumbar spine Z-score, with more severe scoliosis being associated with lower BMD. The fracture risk assessment tool (FRAX) calculator is one of the several tools to determine fracture risk, but has not been applied to a population of patients with neurofibromatosis and has not been calibrated for people below the age of 40. 28
To date, there have been few prospective clinical trials targeting bone health in NF1, and those which have been undertaken have struggled to be successful. For instance, a phase 2 trial of vitamin D supplementation for adults with NF1 (NCT01968590) was terminated due to lack of accrual, despite promising retrospective clinical data. 29 The feasibility of clinical trials has been limited due to an incomplete understanding of the natural history of abnormal bone health across the lifespan and a lack of consensus as to the most clinically meaningful measures to study.
Measures of bone health
Dual Energy X-Ray Absorptiometry (DXA)
DXA is the most commonly used technique for measurement of BMD. It is generally considered the gold-standard, and DXA test results are incorporated into accepted definitions of both osteoporosis and osteopenia. 30 The effective dose of ionizing radiation from DXA varies based on an individual’s size, but is relatively low (0.01–0.03 mSv). 31 Therefore, DXA is considered relatively safe for those with NF1 despite the potential for ionizing radiation to increase risk of malignant transformation of tumors in people with NF1. 32
DXA measures bone mineral content, which is the amount of mineral in a specific bone site scanned. Areal BMD (aBMD, g/cm2) is calculated by dividing the bone mineral content by the total area scanned. Volumetric BMD (vBMD, g/cm3) can be measured utilizing quantitative computed tomography (QCT) or calculated from DXA measurements (bone mineral apparent density, g/cm3). 33 BMD is measured either in a particular body region (e.g. femoral neck) or as a measure of the entire skeleton (e.g. whole body).
For postmenopausal women and men more than 50 years, BMD is reported as a T-score, which compares the person’s value versus the young adult normal mean and reports a standard score. In children and younger adults, BMD is reported as a Z-score, which compares the patient’s values with controls for age, sex, and ethnicity. 34 In children, particularly those with short stature, which is common in NF1, using comparisons that do not account for body size can lead to an increase in the proportion of patients incorrectly diagnosed with low BMD. 35 To address this issue, using height-adjusted Z-scores is recommended. 36
aBMD and bone mineral apparent density are the measures most widely studied in NF1 (Table 1). Both children and adults with NF1 have been shown to have lower BMD when compared with healthy controls.7,9,10,14,16,18,27,29,37–41 While lower BMD values compared with the general population have been reported in all measured body regions, one study found that the lowest aBMD in those with NF1 tends to be in load-bearing regions of the body, that is, the legs and trunk. 41
Techniques for measuring bone health in NF1.
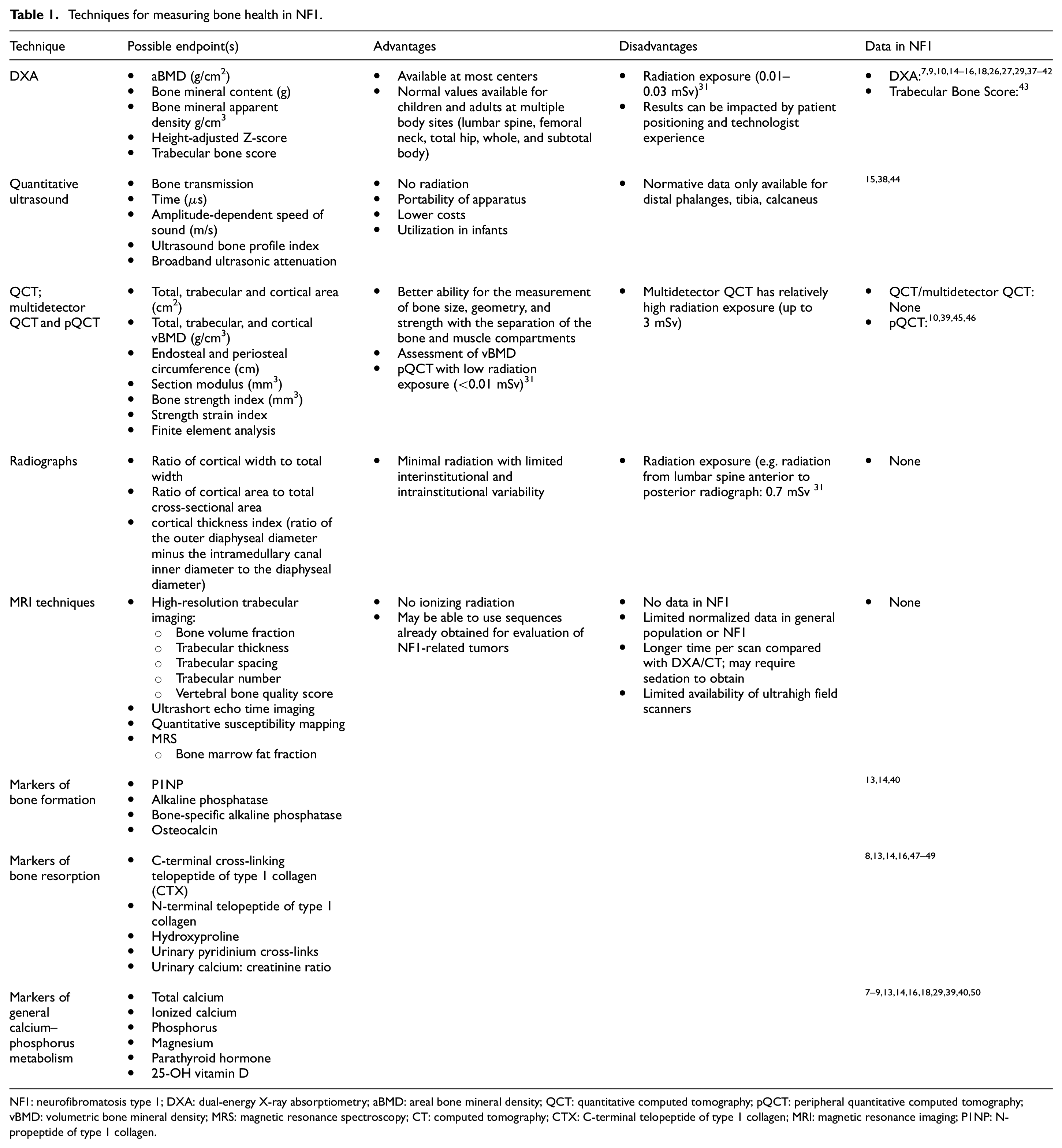
NF1: neurofibromatosis type 1; DXA: dual-energy X-ray absorptiometry; aBMD: areal bone mineral density; QCT: quantitative computed tomography; pQCT: peripheral quantitative computed tomography; vBMD: volumetric bone mineral density; MRS: magnetic resonance spectroscopy; CT: computed tomography; CTX: C-terminal telopeptide of type 1 collagen; MRI: magnetic resonance imaging; P1NP: N-propeptide of type 1 collagen.
Lower BMD measured by DXA has been directly correlated with increased fracture risk in postmenopausal women across multiple prospective studies. 51 However, it has not yet been shown to directly correlate to fracture risk in people with NF1. This lack of association is a potential limitation when considering the use of BMD as the primary endpoint of a clinical trial for a targeted therapy in people with NF1. BMD as a trial endpoint is also limited by the potential contribution of other factors that may impact BMD, such as sun exposure and physical activity. 40 These contributors should be considered and controlled for in any clinical trial utilizing BMD as an endpoint.
Trabecular bone score is a measure of vertebral bone texture that is derived from DXA data and is an indirect measure of trabecular microarchitecture.52,53 To assess fracture risk, trabecular bone score can be used alone or in combination with BMD. 54 Filopanti et al. 43 found that trabecular bone score was lower in people of both sexes with NF1 (n = 74) compared with the general population; this was independent of other factors such as 25-OH vitamin D levels and low exercise levels. It is notable, however, that trabecular bone score can be calculated from previously obtained DXA studies and, therefore, might present an opportunity for additional retrospective analyses.
While DXA-measured BMD is widely used for guiding clinical decisions, primarily in older adults, there are several ways in which the results of these studies can be easily skewed or misinterpreted, resulting in potentially erroneous conclusions. First, the technologist must be well trained to ensure proper patient positioning; this can be a challenge (and sometimes impossible) for patients with scoliosis, significant tumor burden, or chronic pain. Regions of interest automatically placed by the DXA software are not always correct, so manual adjustments may be necessary. In addition, incorrect patient demographic information can result in widely discrepant T- or Z-scores. 55 For any potential clinical trial, centralized analysis of scans, as well as measures to ensure adequate quality control and training of technologists, is essential. 34 Another limitation with the use of DXA in the NF1 population is that some patients with the most severe bone-related manifestations, such as dystrophic scoliosis or tibial dysplasia, may have metal implants that make calculations of bone mineral content in that region impossible.
QCT and pQCT
Computed tomography (CT) is an X-ray-based imaging technique commonly used in clinical practice. Quantitative tomography using body CT is a three-dimensional (3D) measurement technique that converts the values obtained by CT (Hounsfield units) to a bone-equivalent value to calculate a vBMD (g/cm3). 31 The BMD calculated by quantitative CT (QCT) is a volumetric measurement since (unlike DXA) the slice thickness is always known. 56 Multidetector QCT is a 3D technique which allows for assessment of both BMD and bone geometry. The International Society of Clinical Densitometry official position statement in 2007 stated that “trabecular BMD of the lumbar spine measured by QCT can be used to monitor age-, disease- and treatment-related BMD changes.” 56 However, obtaining high-resolution images capable of differentiating individual trabeculae requires a relatively ionizing radiation exposure (up to 3 mSv), and therefore this technique should be used judiciously in people with NF1 who have an increased risk of secondary malignancy.
Peripheral QCT (pQCT) is a 3D nonprojection technique used to quantify BMD specifically in the proximal femur, forearm, and tibia. pQCT has been utilized in several studies of individuals with NF1 (Table 1). High-resolution pQCT is performed using a small scanner that is specifically intended to measure bone characteristics. Some advantages of pQCT over DXA include the ability to separate out cortical and trabecular bone, and to calculate 3D geometric parameters of bone health, such as the stress–strain index. 57 Bone strength can also be estimated from pQCT using a numerical simulation modeling method known as finite element analysis. Finite element analysis has been validated in several clinical trials and estimates bone fragility from various factors including the distribution of the bone mass measured on pQCT, the properties of the materials in the extracellular matrix, and general principles of mechanics. 58 In addition, pQCT can directly account for bone size and shape, and can separate bone and muscle compartments, which DXA cannot. 59 pQCT uses less radiation than multidetector QCT, usually delivering doses <0.003 mSv, which is an even lower dose than DXA.31,57 One of the limitations of pQCT for assessment of young children, however, is the need for patients to cooperate with placement of the leg or arm in the scanner’s gantry, as pQCT is more sensitive to movement than DXA and hence prone to motion artifacts. 57 A second limitation is that pQCT assesses only a small region of the body, and hence, values obtained may not be representative of overall bone health. Although there are normative data available for the pediatric population, and some data exist in the NF1 population, there is overall less published data for pQCT in comparison with DXA.
Of note, opportunistic CTs obtained for other medical indications can be used to measure Hounsfield units as a proxy for BMD. These measurements are currently used to assess response to medical therapy and by surgeons to determine whether bone can support spinal instrumentation.60,61 Improvements in CT technology, sometimes referred to as low-dose CT, may result in significantly lower radiation exposure (e.g. 0.8 mSv per scan), though this is not routinely available at all institutions. 62
Quantitative ultrasound
Quantitative ultrasound is another method used to assess bone health in specific peripheral body regions (e.g. forearm, phalanges, tibia, and calcaneus).63,64 Quantitative ultrasound is based on the premise that the speed of sound and broadband attenuation correlate with material density, and has been used in the general population to assess fracture risk. 64 The probe is a handheld device with transmitting and receiving transducers which simultaneously measure both the speed of sound through the bone and the broadband attenuation.
Quantitative ultrasound has potential benefits for the NF1 population. First, compared with other modalities, it can be used in young infants/children which is typically when long bone dysplasias present in NF1. Second, there is existing control data for the typically affected skeletal region for tibial bowing (diaphysis of the tibia). Other potential advantages include lack of radiation, portability, and comparably lower costs of imaging/equipment.
Stevenson et al. 44 utilized quantitative ultrasound to assess both the affected and unaffected lower limbs of individuals with NF1 and tibial bowing and found that quantitative ultrasound could differentiate between the affected and unaffected tibia, providing rationale for use in clinical trials for tibial dysplasia. A unique advantage of quantitative ultrasound is that the unaffected leg can be used as an internal control when developing clinical trials for tibial dysplasia in NF1.
Like pQCT, one limitation is that quantitative ultrasound focuses on the peripheral skeleton, and results may not represent the more central regions of the skeleton. One technology that may address this limitation, radiofrequency echographic multispectrometry, uses ultrasound to provide an estimated BMD (BMDUS) in the femur and lumbar spine which has been shown to correlate with aBMD DXA results. 65 However, radiofrequency echographic multispectrometry is primarily available in research settings, and there are no data in people with NF1. Another challenge for quantitative ultrasound is that there are several devices in use, each with its own normative database, which complicates standardization for use in clinical trials.
Radiography (X-ray)
Another assessment of bone health is the measurement of cortical thickness of bone on plain radiograph. Specifically, a cortical thickness index for a bone can be calculated as the ratio of the outer diaphyseal diameter minus the intramedullary canal inner diameter to the diaphyseal diameter. Recently, in a retrospective study of more than 500 healthy volunteers, the cortical thickness index of the proximal femur was predictive of the femoral neck BMD. 66 While cortical thickness index has yet to be specifically evaluated in people with NF1, the relative ease of obtaining a radiograph compared with other modalities makes this an appealing consideration. Also, radiographs may be available from earlier studies or clinical care which could be analyzed retrospectively for bone health in NF1. If a similar correlation between low cortical thickness and low BMD can be established in people with NF1, this could be a potential endpoint for future clinical trials.
A newer technology with potential utility is biplanar, low-dose radiography (digital slot scanning). This technology obtains simultaneous anterior/posterior and lateral radiographs from a standing or sitting individual, which can be paired to create a secondary 3D image of the skeleton at lower doses of ionizing radiation than conventional radiographs or CT imaging.67,68 Utilized primarily for presurgical scoliosis assessments, biplanar, low-dose radiography has the potential to capture the whole skeleton in a short time and may also allow for calculation of trabecular bone score and BMD.69,70 This technology is relatively new, and information on people with NF1 has not been published.
Magnetic resonance imaging
Magnetic resonance imaging (MRI) is a standard imaging technique used to follow the tumor-related manifestations of NF1 including plexiform neurofibroma and low-grade gliomas. To date, there have not been any studies evaluating MRI to assess bone health in NF1; however, this technology has been explored in other conditions. Specific MRI sequences—the bone marrow fat fraction—can be used to evaluate not only BMD and trabecular microarchitecture, but also aspects of bone that are not evaluable by other methods, such as the proportion of nonmineralized trabecular bone composed of fat cells (adipocytes) versus hematopoietic cells. 71 The most common measures reported are bone volume fraction, trabecular thickness, spacing, and number. 72
The main advantage of MRI when compared with radiographs, DXA, or QCT is the absence of ionizing radiation. However, there remain several limitations, including the fact that scans are generally much longer, and therefore, children may require sedation or general anesthesia to successfully obtain imaging. Another limitation is that a generally accepted measurement technique for assessing bone with MRI does not yet exist. In recent years, the advent of high-field (3 Tesla) and ultrahigh field (7 Tesla) scanners have improved the signal-to-noise ratio and allowed for better quantification of bone. 72 Chang et al. 73 demonstrated that by using this type of ultrahigh field scanner they were able to differentiate between postmenopausal women with a history of fragility fractures and those without. Notably, there were no differences in DXA-derived BMD between the two groups, indicating that the level of detail in ultrahigh field MRI imaging may provide additional clinical information beyond what can be detected by DXA. However, these ultrahigh field scanners are only available at specialized centers. Other MRI techniques including ultrashort time echo for imaging of cortical bone and quantitative susceptibility mapping for trabecular bone have also been evaluated in the setting of osteoporosis, but it is not clear what additional clinical utility these may provide beyond more standard measurements of BMD.71,74
More recently, a measure known as the vertebral bone quality score has emerged as a potential endpoint which has the advantage of being calculated based on routinely obtained noncontrast, T1-weighted MRI images of the lumbar spine. Like the ultrahigh field MRI technique described above, in a population of patients with known low BMD as assessed by DXA, the vertebral bone quality score was able to independently differentiate between those with and without fragility fractures. 75 A significant difference between the vertebral bone quality scores of healthy adolescents and those with idiopathic scoliosis has also been demonstrated. 76
Additional prospective studies of these MRI bone quality endpoints are needed, both in the general population and in people with NF1, to better understand how they may be incorporated into future care or clinical trials.
Bone turnover markers and other measures of bone metabolism
Bone turnover markers are protein, or protein-derivative, biomarkers which are indicators of bone homeostasis. They are typically divided into markers of bone formation and bone resorption. The most popular marker of bone formation is serum N-propeptide of type 1 collagen (P1NP); others include osteocalcin and bone-specific alkaline phosphatase. For bone resorption, the most commonly used is serum C-terminal telopeptide of type 1 collagen (CTX); other markers include N-terminal telopeptide of type 1 collagen in serum or urine, urine deoxypyridinoline and serum tartrate-resistant acid phosphatase, 5b. 77 Changes in bone turnover markers have been directly correlated with changes in BMD on imaging and have the advantage of being detectable more quickly than changes based on imaging findings.
P1NP (bone formation) and CTX (bone resorption) are the most widely studied bone turnover markers in the general population, and robust normative data sets exist for both healthy adult 78 and pediatric 79 populations. CTX is a breakdown product of type 1 collagen that is released during bone breakdown and resorption and has been used to effectively monitor antiresorptive therapy.78,80 Similarly, P1NP is cleaved from procollagen and released into circulation during the process of bone formation 78 and is also used clinically for monitoring of response to anabolic treatments. 77
There are relatively few studies investigating bone turnover markers in NF1. One study demonstrated that individuals with NF1 have a higher level of CTX 40 when compared with healthy controls, consistent with elevated levels of bone turnover and resorption. Urinary pyridinium cross-links (pyridinoline and deoxypyridinoline), which are collagen breakdown products that also measure bone resorption, 81 were also shown to be higher in people with NF1 compared with the general population. 47 In terms of bone formation markers, both osteocalcin and bone-specific alkaline phosphatase have been shown to be increased in adults with NF1. 14 There is early evidence that persons with NF1 and known skeletal abnormalities have a significantly lower level of osteocalcin compared with persons with no skeletal changes. 13 To date, there are no prospective studies to establish the natural history of bone turnover markers in people with NF1 and very limited data in childhood. One potential limitation of bone turnover markers is that they increase with fracture healing and may remain increased for months after a fracture, possibly confounding results in the setting of a traumatic fracture.
There are several reports assessing calcium–phosphorus metabolism and the role of vitamin D in NF1. In general, a higher proportion of people with NF1 had 25-hydroxy vitamin D in the insufficient/deficient range compared with healthy controls, but there was not a direct association between low vitamin D and abnormal BMD. 50
Recommendations for development of clinical trial endpoints in NF1
REiNS is charged with evaluating and recommending consensus endpoints for use in future clinical trials targeting specific conditions, such as bone health in NF1. The goal of this review is to provide a starting point for these discussions and guidance about which measures could be included in future studies to allow for more specific consensus recommendations in the future.
Nearly all information on bone-related natural history and associated measurement techniques in NF1 is based on either cross-sectional cohort studies or retrospective reviews. The lack of prospective, longitudinal data regarding bone health in patients with NF1, including changes in the measurements over time, makes designing a clinical trial to improve bone health much less feasible. In NF1, as with other rare diseases, implementing large prospective randomized clinical trials is not practical, particularly given the variability in phenotype; therefore, natural history data will be crucial for studying any future targeted intervention. There is significant need for a prospective, longitudinal natural history study focusing on the bone-related manifestations and relevant biomarkers of NF1.
The REiNS International Collaboration recommends that such a study focus on both children and adults with NF1 and incorporate a variety of the measurement techniques and biomarkers herein outlined. Most existing data in NF1 focus on BMD as measured by DXA, and DXA is readily available at most centers with low radiation exposure; therefore, this should be considered in any future trial. However, a future study should also aim to establish whether there is any correlation in measurements of bone health obtained from different modalities in people with NF1. For example, such a study could address: (1) whether changes in serum bone turnover markers can be used to predict changes in DXA in people with NF1, (2) whether imaging modalities looking at localized bone changes, such as quantitative ultrasound, pQCT, or radiography, may be reflective of the BMD as measured by DXA, (3) how these measurements change over time, and (4) the reliability and cost of these measurements in those with NF1.
REiNS also recommends that the NF1 research community consider adding some of the less burdensome measurements of bone health, such as DXA scans or serum bone turnover markers and measures of bone metabolism, as exploratory endpoints in ongoing or planned clinical trials for other NF1 manifestations. Further exploration could provide additional ways to assess bone health through opportunistic imaging obtained for other indications. Collecting serial measurements prospectively for these bone-related issues in clinical trials targeting the tumor or nontumor manifestations of NF1 is a low-cost opportunity, which would add to the knowledge in this field.
Conclusion
Overall, bone-related issues contribute to clinical morbidity in people with NF1, and therefore, it is critical to target these issues in future clinical trials. We have summarized here the currently available methods for measuring overall bone health and described what is known about each of them in the setting of NF1. Most data in people with NF1 are derived from small case series or retrospective reviews which, while valuable, do not provide comprehensive data that a prospective natural history study would be able to obtain. In addition to recommendations for future investigations, REiNS recommends that the less burdensome measurements of bone health be used as exploratory endpoints in NF1 clinical trials going forward to better inform future studies.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745231201338 – Supplemental material for Potential endpoints for assessment of bone health in persons with neurofibromatosis type 1
Supplemental material, sj-docx-1-ctj-10.1177_17407745231201338 for Potential endpoints for assessment of bone health in persons with neurofibromatosis type 1 by Andrea M Gross, Scott R Plotkin, Nelson B Watts, Michael J Fisher, Laura J Klesse, Andrés J Lessing, Miranda L McManus, A Noelle Larson, Beverly Oberlander, Jonathan J Rios, Herb Sarnoff, Brittany N Simpson, Nicole J Ullrich and David A Stevenson in Clinical Trials
Footnotes
Acknowledgements
Members of the Response Evaluation in Neurofibromatosis and Schwannomatosis (REiNS) Functional Outcomes Bone Subcommittee including Kelly Carpenter, Gareth Evans, Renie Moss, Elizabeth Schorry, Heather Thompson, David Viskochil, and Brigitte C Widemann. The authors acknowledge the support of the Children’s Tumor Foundation for publication of this article. This article is submitted on behalf of the REiNS International Collaboration, the full membership of which is listed in the supplemental material.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AMG and LJK have served as unpaid advisors for AstraZeneca/Alexion; AMG has been an unpaid advisor for SpringWorks Therapeutics. DAS has been a paid consultant for Alexion. HS is CEO of Infixion Bioscience, Inc., a pharmaceutical R&D company targeting NF1.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
