Abstract
Background/Aims
Determining whether clinical trial findings are applicable to diverse, real-world patient populations can be challenging when the full demographic characteristics of enrolled patients are not consistently reported. Here, we present the results of a descriptive analysis of racial and ethnic demographic information for patients in Bristol Myers Squibb (BMS)–sponsored oncology trials in the United States (US) and describe factors associated with increased patient diversity.
Methods
BMS–sponsored oncology trials conducted at US sites with study enrollment dates between 1 January 2013 and 31 May 2021 were analyzed. Patient race/ethnicity information was self-reported in case report forms. As principal investigators (PIs) did not report their own race/ethnicity, a deep-learning algorithm (ethnicolr) was used to predict PI race/ethnicity. Trial sites were linked to counties to understand the role of county-level demographics. The impact of working with patient advocacy and community-based organizations to increase diversity in prostate cancer trials was analyzed. The magnitude of associations between patient diversity and PI diversity, US county demographics, and recruitment interventions in prostate cancer trials were assessed by bootstrapping.
Results
A total of 108 trials for solid tumors were analyzed, including 15,763 patients with race/ethnicity information and 834 unique PIs. Of the 15,763 patients, 13,968 (89%) self-reported as White, 956 (6%) Black, 466 (3%) Asian, and 373 (2%) Hispanic. Among 834 PIs, 607 (73%) were predicted to be White, 17 (2%) Black, 161 (19%) Asian, and 49 (6%) Hispanic. A positive concordance was observed between Hispanic patients and PIs (mean = 5.9%; 95% confidence interval (CI) = 2.4, 8.9), a less positive concordance between Black patients and PIs (mean = 1.0%; 95% CI = −2.7, 5.5), and no concordance between Asian patients and PIs. Geographic analyses showed that more non-White patients enrolled in study sites in counties with higher proportions of non-White residents (e.g. a county population that was 5%–30% Black had 7%–14% more Black patients enrolled in study sites). Following purposeful recruitment efforts in prostate cancer trials, 11% (95% CI = 7.7, 15.3) more Black men enrolled in prostate cancer trials.
Conclusion
Most patients in these clinical trials were White. PI diversity, geographic diversity, and recruitment efforts were related to greater patient diversity. This report constitutes an essential step in benchmarking patient diversity in BMS US oncology trials and enables BMS to understand which initiatives may increase patient diversity. While complete reporting of patient characteristics such as race/ethnicity is critical, identifying diversity improvement tactics with the highest impact is essential. Strategies with the greatest concordance to clinical trial patient diversity should be implemented to make meaningful improvements to the diversity of clinical trial populations.
Keywords
Introduction
Patients, caregivers, health authorities, and the medical community have long recognized the importance of representativeness in clinical trials,1–3 but effective, sustainable strategies to increase diversity have been historically elusive. 4 Translation of clinical trial findings to applications in diverse, real-world patient populations is essential to delivering optimal care in a medically responsible and socially equitable manner and to addressing an epidemiological imperative for healthcare delivery. In the United States (US), certain cancers are higher in underserved racial and ethnic groups, 5 but certain groups are disproportionately represented in clinical trials.6,7 The reasons for inadequate diversity and representation for patients are multifactorial and complex; 4 however, eligibility parameters, study site selection, and recruitment efforts are significant modifiable factors that can impact patient diversity in clinical trial participation.
The US Food and Drug Administration (FDA) has published guidance for study sponsors to enhance the diversity of clinical trial patient populations in terms of demographic and non-demographic characteristics. This draft guidance, which addresses race/ethnicity, recommends integration of inclusive recruitment plans early in the design and development of human trials. 8 The FDA has also established Project Equity (www.fda.gov/about-fda/oncology-center-excellence/project-equity) within the agency’s Oncology Center of Excellence to ensure that data submitted for approval of oncology medical products adequately reflect the demographic representation of patients for whom these products are intended.
Industry stakeholders are making substantial commitments to tangible action and progress, 9 such as integrating diversity strategies into clinical trial protocols, engaging study sites that serve under-represented populations, increasing patient education about clinical trials, and pursuing multistakeholder partnerships to improve awareness, trust, and participation. 9 Industry is also conducting analyses to better understand the benchmarks and opportunities to address patient under-representation in clinical trials. 10
Here, we describe an analysis of racial and ethnic demographic information for patients participating in Bristol Myers Squibb (BMS)–sponsored US oncology trials to answer the following questions: (1) What is the concordance between patient and principal investigator (PI) race/ethnicity?; (2) What is the relationship between patient diversity in trials with sites located in more ethnically diverse regions?; (3) What is the effect of collaborating with advocacy and community-based organizations on patient enrollment?; and (4) How do results of these analysis compare with those of the general population? These baseline data are essential to understanding the current status of clinical trial diversity within the BMS clinical program in oncology and to inform future plans to improve diversity.
Methods
Identification of BMS-sponsored US oncology clinical trials
Oncology clinical trials with active enrollment between January 1, 2013 and May 31, 2021, inclusive, were identified. Eligible trials had to be conducted at US study sites for the treatment of solid tumors and be accessible to the BMS Clinical Data Reporting and Analytics team on the BMS Statistical Computing Environment (the organizational computing platform for BMS clinical trials). PIs had to be identifiable in the accompanying validated system that enables traceable documentation of study site interactions. Trials could have a status of “active,”“recruiting,”“not recruiting,”“terminated,”“completed,”“unknown status,” or “no longer available” (as indicated on www.ClinicalTrials.gov). Trials sponsored by entities acquired by BMS during the analysis period, trials of hematologic cancers, and those sponsored by the investigators were excluded.
Determination of patient and PI race/ethnicity
Patient race/ethnicity information was self-reported and collected from case report forms as provided in the regular conduct of the trial. There was no mechanism for PIs to report their own race/ethnicity. A deep-learning algorithm (ethnicolr; github.com/appeler/ethnicolr) was used to predict the race/ethnicity of PIs using their name strings. It uses a long short-term memory model (Keras, keras.io; TensorFlow, www.tensorflow.org) trained on 1 million randomly sampled names from the Florida voter registration database (total 13 million names) and names from a data set compiled from Wikipedia (130,000 names). 11 The prediction algorithm, as used, generated probabilities of a name string belonging to one of only four categories: non-Hispanic White (White), non-Hispanic Black (Black), Asian, or Hispanic. The highest prediction probability for each name string was used as the race/ethnicity classification for that PI. We performed a validation exercise of the predicted PI race/ethnicity in order to inform the interpretation of our findings (please see Supplemental Tables E1 and E2).
In order to contextualize patient and PI race/ethnicity findings, we informally assessed our findings alongside publicly available race/ethnicity information in the United States. Findings from patients were reported descriptively alongside general population data from the American Community Survey. 12 To account for differences between cancer epidemiology and the general population, we further analyzed patient information alongside cancer incidence rates from the Surveillance, Epidemiology, and End Results (SEER) Program. 13 Incidence rates were averaged for the study period (2013–2018) for melanoma, lung and bronchus cancer, gastrointestinal cancers, and prostate cancer. The SEER-observed age-adjusted incidence rates among White, Black, Asian, and Hispanic people were applied to the average number of people in each category according to the Centers for Disease Control and Prevention (CDC) 14 US population estimates. For BMS US oncology PIs, a descriptive assessment was made alongside an analysis of American Medical Association data on US hematologists/oncologists reported by the Association of American Medical Colleges.
Demographics by study site location
PIs were linked to patient identification codes via site and location identifiers and clinical trial registration identifiers on ClinicalTrials.gov. Study site addresses from eligible BMS-sponsored US oncology trials were standardized using a geocoding application (https://geopy.readthedocs.io/). Zip codes and county information were derived for the standardized addresses (https://uszipcode.readthedocs.io/index.html), and the corresponding county-level demographic information was obtained from the US Census Bureau. 15 Data from the 2019 American Community Survey (www.census.gov/programs-surveys/acs) were used to generate estimates of race and ethnic groups in the general US population.
Analysis of magnitude of association
Bootstrapping was used to estimate the magnitude of association related to each of the objectives: concordance of race/ethnicity between patients and the PIs within the study sites (i.e. having a shared race/ethnicity between a physician and a patient); the relationship between county-level and study-site demographic diversity; and the impact of community outreach initiatives for BMS-sponsored US prostate cancer trials on the recruitment of Black patients.
For all analyses and differences calculated, 10,000 bootstrapped replicate data sets were generated; in each bootstrapped replicate data set, the difference of interest related to the above objectives was calculated and a distribution of proportional differences from the replicate data sets was derived. From this distribution of proportion differences, the mean of the distribution was reported as the point estimate for the proportion difference, and the percentile method was applied to derive the confidence interval (CI) for the difference.
Patients were sampled with replacement from the data set (10,000 samplings with replacement, using all identified patients per sampling). To determine the magnitude of the association for patient–PI concordance, the difference between the proportion of patients with PIs of the same race/ethnicity category and the proportion of patients with PIs of a different race/ethnicity category were compared (e.g. proportion of Asian patients with Asian PIs vs Asian patients with non-Asian PIs). The published precision for ethnicolr was incorporated to account for the uncertainty in the prediction of PI race/ethnicity 16 by randomly unassigning (per bootstrap sampling) a percentage of those PIs identified as a given race/ethnicity, with that percentage being equal to one minus the published precision for the given race/ethnicity.
To determine the magnitude of associations related to county and trial diversity, we measured the percent of (a) Asian patients in trials with study sites in counties with increasing percentages of Asian population and of (b) Asian patients in trials with sites in all other counties, and subtracted (b) from (a) to establish a difference. Analogous measurements and differences for Black and Hispanic patients were also made.
Assessment of proactive partnerships to increase diversity in prostate cancer trial patients
Understanding that the probabilities of developing and dying from prostate cancer are the highest for Black men in the United States, 12 the BMS clinical research teams undertook intentional engagement strategies to increase diversity in three prostate cancer clinical trials: CheckMate 9KD, NCT03338790; CheckMate 650, NCT02985957; and CheckMate 7DX, NCT04100018. These strategies leveraged culturally appropriate materials and content through already trusted community partners, including patient advocacy groups focusing on prostate cancer, for example, ZERO—The End of Prostate Cancer and The Prostate Health Education Network, with targeted efforts specifically to Black patients and community outreach organizations such as Black Health Matters (https://blackhealthmatters.com) to connect with the Black community in a multipronged approach, including live events and digital content focusing on prostate cancer education.
BMS also partnered with research sites, members of the Clinical Trial Ambassadors Program from the Yale Center for Clinical Investigation, and members of the National Black Nurses Association, to understand best practices for engaging with diverse patient populations.
To measure the magnitude of associations for the community outreach analysis, the difference between the percent of Black patients in prostate cancer trials and in non-prostate cancer trials for which such community outreach was not done was measured per bootstrap sampling. Enrollment was also compared to a measure of Black patient enrollment in prostate cancer trials identified from the ClinicalTrials.gov electronic registry. The mean difference per bootstrap distribution was taken to be the magnitude of the association, as with the other analyses of magnitudes of association.
Results
BMS-sponsored US oncology clinical trials
A total of 208 BMS-sponsored oncology trials were identified in the parent database during the prespecified analysis period; 100 were excluded due to non-US geography or missing PI-related information required to conduct the analysis. The remaining 108 (52%) trials were conducted for solid tumors at 416 US study sites with matched PIs in the study-site management platform and included in the analysis. These 108 trials included 16,140 patients, 15,763 (98%) of whom had available race/ethnicity information, and 834 unique PIs (Table 1). Most trials were either phase 3 (42%), or phase 1/2 and phase 2 (45%), and 46% (± 23.3%) of the study sites were located in counties where the non-White population was ≥50% (Figure 1). Out of 416 sites, 148 (36%) were academic centers and 268 (64%) were community centers.
BMS-sponsored US oncology clinical trials included in the race/ethnicity analysis.
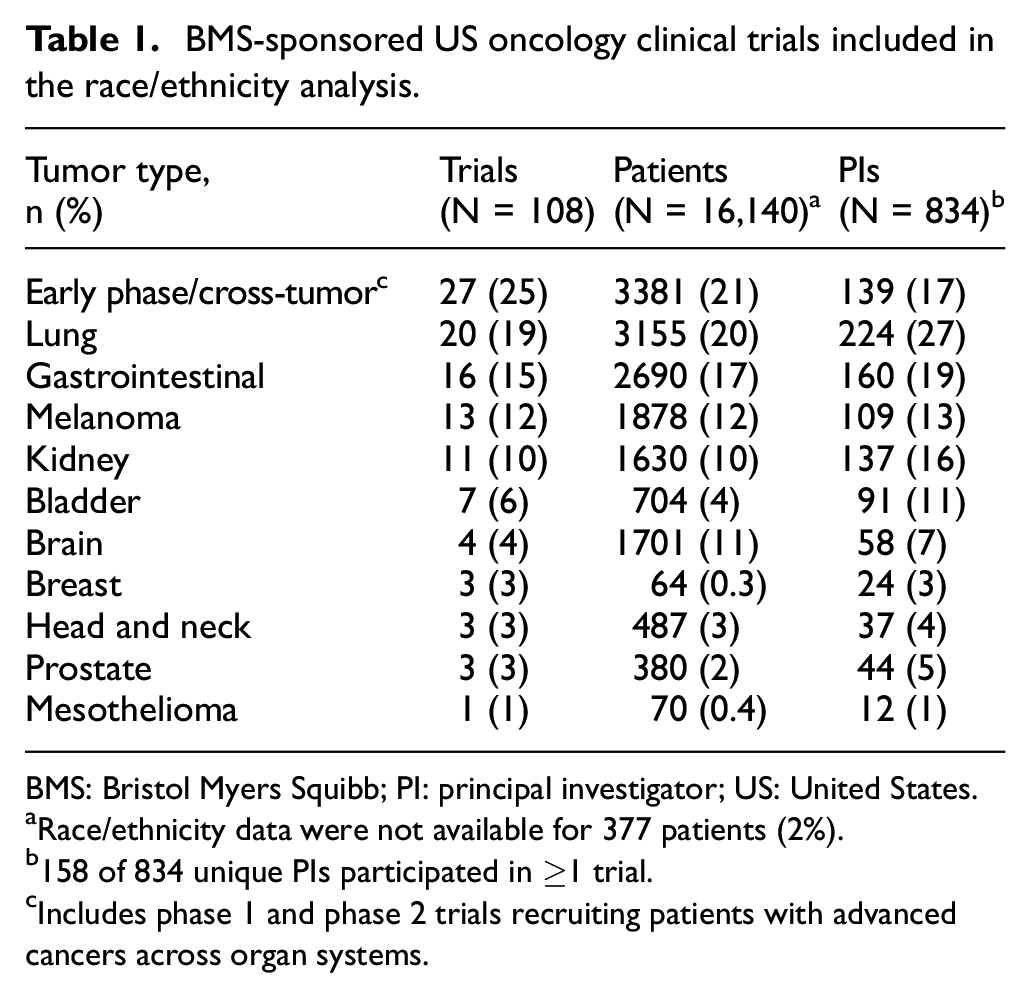
BMS: Bristol Myers Squibb; PI: principal investigator; US: United States.
Race/ethnicity data were not available for 377 patients (2%).
158 of 834 unique PIs participated in ≥1 trial.
Includes phase 1 and phase 2 trials recruiting patients with advanced cancers across organ systems.
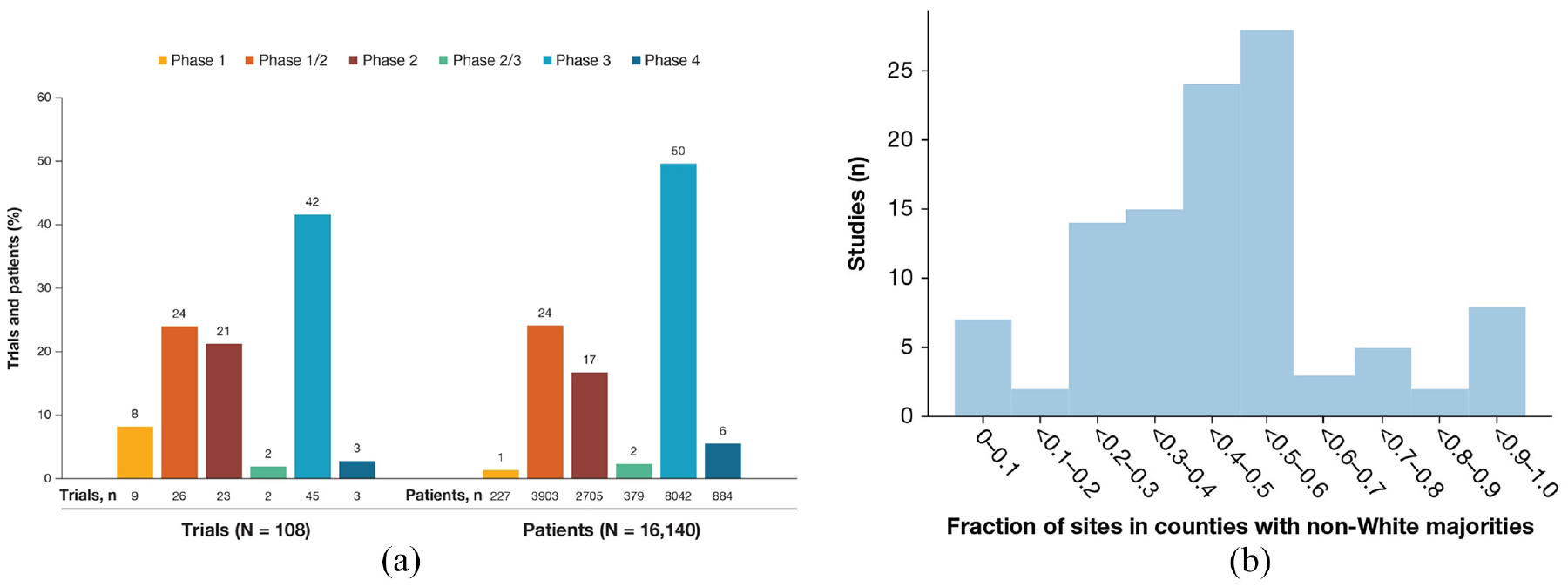
BMS-sponsored US oncology trials. (a) Number of trials and patients per phase. (b) Number of studies per fraction of sites based in counties where the non-White population is ≥50%. Each bar represents a bin and shows how many studies there are in the analysis in which the fraction of sites in majority non-White counties is within a given value.
Patients’ self-reported race/ethnicity
Of 15,763 patients with available race/ethnicity information, 13,968 (89%) self-reported as White, 956 (6%) as Black, 466 (3%) as Asian, and 373 (2%) as Hispanic. Across tumor types, the proportion of patients self-reporting as White ranged from 60% to 99% and was the highest in a mesothelioma trial (one trial). The proportion of patients self-reporting as Black ranged from 0% to 18% and was the highest in prostate cancer trials (three trials), and the percentage of those self-reporting as Asian ranged from 0% to 7%, which was the highest in early-phase cross-tumor trials (27 trials). For Hispanics, this proportion ranged from 1% to 25%, being the highest in breast cancer trials (three trials), although the breast cancer cohort was small (57 patients) (Figure 2 and Supplemental Table E3).
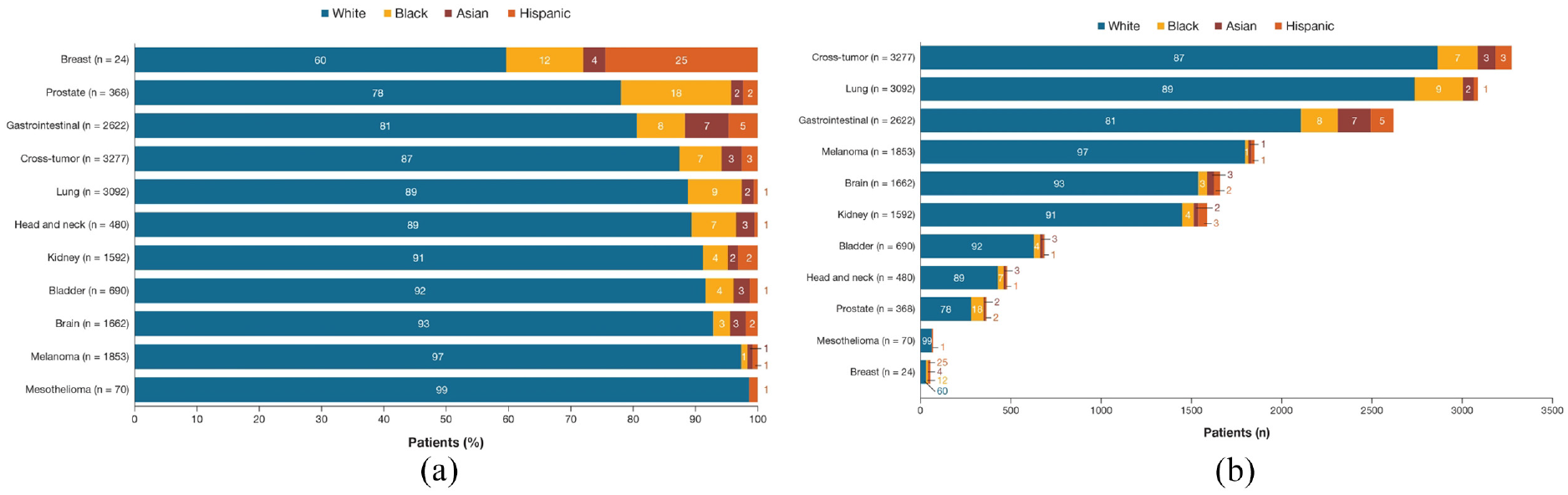
Patients’ self-reported race/ethnicity by tumor type (n = 15,763). (a) Proportion of patients by tumor type (presented as proportions of patients only). (b) Number and proportion of patients by tumor type (presented as number of patients). Proportions may not sum to 100% due to rounding.
We compared our results to those of the general population. The 5-year 2019 ACS reported 60% of the US population to be White (BMS patient cohort, 89%), 13% Black (BMS patients, 6%), 6% Asian (BMS patients, 3%), and 18% Hispanic (BMS patients, 2%). The US epidemiology data from SEER demonstrated that the incidence of lung and bronchus cancer in the US was 13% and 10% for Blacks and Hispanics, respectively. Smaller proportions of these groups were enrolled in BMS lung cancer trials, with a greater difference for Hispanics (9% Blacks, 1% Hispanic). According to the SEER data, 15% and 18% of patients with gastrointestinal cancer were Black and Hispanic, respectively, and 8% and 5% of patients enrolled in BMS gastrointestinal trials were Black and Hispanic, respectively. Whites were at the greatest risk for melanoma (95%), with a low incidence of melanoma across other groups (0% Asian, 1% Black, 4% Hispanic) (Supplementary Table E4). The population enrolled in BMS melanoma trials was similar to that of SEER, with 97% White, 1% Asian, 1% Black, and 1% Hispanic. Proportions of Black patients in BMS prostate cancer trials were similar to the SEER estimates (18% BMS vs 20% SEER), but lower for Hispanic men with prostate cancer (2% BMS vs 16% SEER).
Predicted PI race/ethnicity
In the prediction analysis of race/ethnicity for the 834 unique PIs, manual labeling of 40 sample names compared to the algorithm predictions showed 75% accuracy, in line with the 74%–82% precision for race (US Census) and 50%–91% precision for ethnicity models (Wikipedia) reported by the authors of the ethnicolr package. 16 A total of 607 (73%) PIs were predicted to be White, 17 (2%) Black, 161 (19%) Asian, and 49 (6%) Hispanic. Among the 1035 PIs across all trials and tumor types (including 158 PIs supporting ≥1 tumor type), the proportion predicted to be White was 64%–81% and the highest for brain cancer trials (four trials). The predicted proportion of Black PIs was 0%–6% and the highest for melanoma trials (13 trials). The percentage of predicted Asian PIs was 4%–33%, the highest for mesothelioma trials (one trial), and of predicted Hispanic PIs, 0%–25%, being the highest for breast cancer trials (three trials) (Figure 3). Results of the validation study showed that the algorithm had as good or better precision for Asian and White PIs in this study compared with the published precision of algorithm, slightly worse precision for Hispanic PIs, and worse precision for Black PIs (Supplementary Tables E1 and E2).
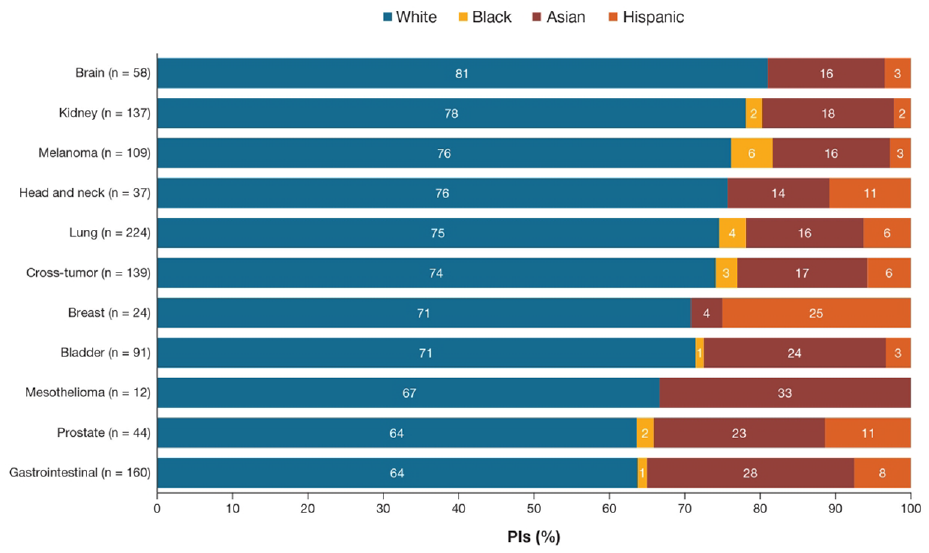
PI-predicted race/ethnicity by tumor type (n = 1035). Proportions may not sum to 100% due to rounding.
In 2018, 52% of US hematologists/oncologists were White (compared with 73% of predicted BMS PIs), 3% were Black (2% of predicted BMS PIs), 27% were Asian (19% of predicted BMS PIs), and 5% were Hispanic (6% of predicted BMS PIs) (Supplementary Table E5).
Magnitude of association of patient–PI race/ethnicity
When patient race/ethnicity was analyzed by the race/ethnicity of PIs at their study sites, the highest proportions of patients recruited by all PIs were White (range = 81%–90%), consistent with the primary findings. Among non-White patients, the highest proportion of non-White patients in study sites run by Black PIs was also Black (7%), and likewise for Asian patients and PIs (6%); by contrast, Hispanic PIs enrolled a similar number of Hispanic (8%) and Black (7%) patients (Supplemental Table E6). The concordance analysis showed a positive concordance between Hispanic patients and Hispanic PIs, with a mean 5.9% (95% CI = 2.4, 8.9) more Hispanic patients with Hispanic PIs than with all other PIs (Figure 4). A smaller magnitude of concordance was observed between Black patients and Black PIs, with a mean 1.0% (95% CI = −2.7, 5.5) more Black patients with Black PIs than all other PIs. The analysis did not suggest a meaningful concordance for Asian patients and Asian PIs (mean = 0.2%; 95% CI = −0.8, 1.2). Results of the algorithm validation study showed that magnitudes of association were not substantively different pre- and post-validation (Supplementary Tables E1 and E2).
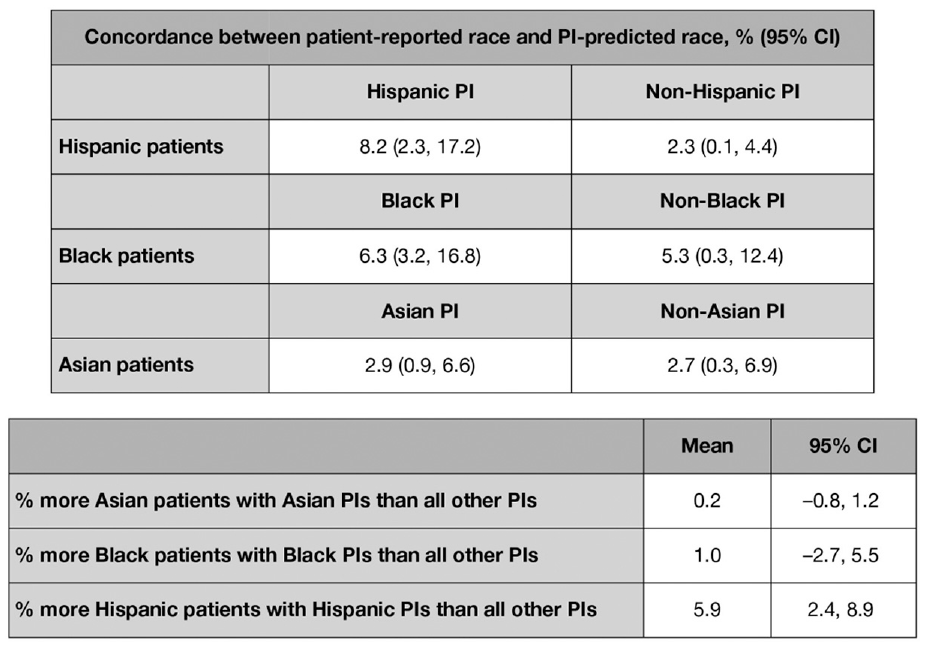
Concordance analysis of non-White patients and PIs. Each bar indicates, for a given PI’s race/ethnicity, the proportion of patients of a given race/ethnicity. The error bars represent the 95% CI from bootstrapping.
Magnitude of association of patient, PI, and county-level diversity
The geographic spread of BMS oncology trials showed that most patients were from New York, Pennsylvania, California, Texas, and Florida, with a smaller proportion coming from the upper Midwest (Supplemental Figure E3). When the race/ethnicity of patients was analyzed against county-level race/ethnicity for the study sites, a greater county prevalence of non-White patients correlated with a higher proportion of non-White patients enrolled in the trials (Supplemental Table E7). For example, when 5%–30% of a county’s population was Black, 7%–14% more Black patients enrolled in study sites in those counties (Supplemental Table E7). The same patterns were observed for Asian and Hispanic counties and trial patients. However, a decrease in enrollment was seen for proportions >30% of the Black population. When the concordance of patient–PI diversity was also analyzed by county-level demographic information, the proportions of Hispanic patients enrolled by Hispanic PIs increased with the proportion of Hispanic people living in the county of the study site; counties with ≥30% Hispanic population had 14% more Hispanic patients enrolled with Hispanic PIs, for example. A similar but lower in magnitude trend was observed for Black patients, PIs, and counties, but not for Asian patients, PIs, and county demographics (Supplemental Table E8).
Increasing diversity with proactive partnerships: the case of prostate cancer clinical trials
As of this analysis, the CheckMate 9KD trial was completed, and a “data snapshot” of interim patient demographic information was used from the ongoing CheckMate 650 and CheckMate 7DX trials. Following purposeful recruitment efforts in prostate cancer trials, 18% of men enrolled in the CheckMate 9KD trial were Black. Based on the data snapshot, 18% of men enrolled in the three US prostate cancer trials were Black. Incorporating community partnerships resulted in an increase of 11.0% (95% CI = 7.7, 15.3) more Black patients than trials for other tumor types to date (Figure 5). 17 A previously published analysis of the prostate cancer trials in the ClinicalTrials.gov electronic registry was used as a historical comparison and showed that, among the trials reporting demographic data, 9.8% of the patients were Black.
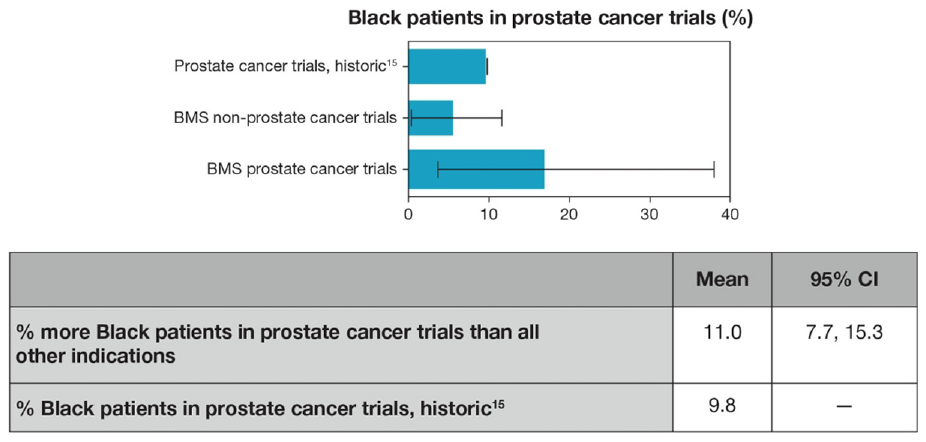
Enrollment of Black patients in BMS prostate versus non-prostate cancer trials.
Discussion
This analysis solidifies a preliminary but essential step in reporting race/ethnicity benchmarks among patients participating in BMS-sponsored US clinical trials. Improving diversity in clinical trial populations will improve the relevance of trial results without compromising recruitment or examination of other subgroups based on demographic or clinical characteristics. The insights from this work will inform further data-driven efforts to improve BMS clinical trial diversity. These efforts are being undertaken across therapeutic areas and demographic characteristics and will be oriented to the epidemiology of the conditions being investigated.
Our contextual analysis was performed to highlight that diversity goals for clinical trials must be oriented to the epidemiology of tumor types. The epidemiology of lung and gastrointestinal tumors highlighted opportunities to increase diversity among clinical trials. However, the population enrolled in BMS melanoma trials was fairly representative of the expected US melanoma population. The proportions of Black patients in BMS prostate cancer trials were similar to the SEER estimates, but highlighted a specific need to enroll more Hispanic patients. While methodological differences between the CDC, SEER, and BMS data sets are important to recognize, this informal view provides an initial epidemiological comparison to understand the differences in incidence rates of various cancers across race and ethnic populations. Disease-specific racial and ethnic epidemiologic data will be critical to establish appropriate patient diversity recruitment goals for clinical trials and should ideally be utilized to determine if a clinical trial has successfully recruited an appropriately diverse patient population reflective of patients in real-world clinical practice.
The spread of predicted race/ethnicity of PIs was generally similar to that of patients, but with a greater proportion of Asian PIs. Black and Hispanic PIs appeared to be under-represented. The greatest proportions of non-White patients in study sites run by Hispanic PIs were Hispanic as well as Black. The patient–PI concordance analysis showed a positive concordance between Hispanic patients and Hispanic PIs, suggesting that intentional selection of diverse PIs may contribute to improved recruitment of historically under-represented patient populations. Since this is BMS’s first work examining the impact of racial and ethnic spread of PIs on patient diversity, additional research is planned to further bolster the information supporting PI diversification efforts. Deeper investigation of the drivers of patient–PI concordance would also be of interest, particularly to understand the positive concordance observed with the Hispanic populations, the weaker concordance among Black populations, and the lack of concordance observed with the Asian population. Furthermore, future work examining the association of diversity of study site staff, including sub-investigators, research nurses, patient ambassadors, and community navigators, and patient diversity is needed, as these important partners interact with patients often and are well positioned to facilitate diversity and inclusion efforts in the daily activities of the trials. Achieving greater diversity among the clinical staff may indeed be essential to encouraging non-White patients to participate in trials and to helping them overcome expressed barriers.18,19 The observed patient–PI concordance highlights that concerted efforts to increase the diversity of site research staff could potentially positively impact patient diversity.
In this analysis, patients were most often enrolled in the most populous states, and non-White patients were enrolled with higher frequencies where the local population reflected a greater proportion of the same non-White race/ethnicity. As a result, BMS is committed to locating 25% of US research sites in highly diverse geographic areas with ≥30% non-White population.
Finally, the increase in enrollment of Black patients in the BMS-sponsored US prostate cancer trials suggests that multifaceted community outreach and collaboration approaches can have an impact and should continue to be leveraged. Based on these preliminary results from the initial data snapshot, BMS will continue to partner with advocacy and community outreach organizations as well as research sites to facilitate educational initiatives about clinical trials and develop public service announcements and digital campaigns for certain diseases.
As part of the BMS commitment, the BMS Clinical Trials Operations team is trained on the need for greater diversity in clinical trials, and the site feasibility process prioritizes information related to diversity and inclusion. The cultural appropriateness of study materials is also reviewed routinely by the BMS People and Business Resource Groups Advisory Committee as well as patients and caregivers.
While this analysis focused on patient race and ethnic diversity in BMS-sponsored US oncology solid tumor trials, benchmarking and improving diversity in clinical trials is a global initiative across all therapeutic areas. The results reported here may not be generalizable to other therapeutic areas or geographies as patient and PI populations may be heterogeneous based on these factors, where the epidemiology of disease and characteristics of environments and healthcare settings will inherently vary. Patient diversity in trials of other therapeutic areas and in other countries will be evaluated in the future. Future work will also focus on dimensions of patient diversity related to sex, gender orientation, sexual identity, age, and presence of disability.
Interest in increasing diversity extends beyond patients to include diverse PIs and study site staff. There is a particular focus on increasing participation among Black and Hispanic PIs and sub-investigators in both across all phases of clinical development worldwide. These efforts are being made in the context of overall decreases in enrollment of Black patients associated with the globalization of cancer clinical trials, which exacerbate existing enrollment disparities. 20
Certain limitations of the analysis should be taken into account in the interpretation of these findings. Almost half of the trials identified in the parent database were excluded from the analysis as they did not focus on solid tumor trials and lacked availability of PI information; future work will aim to analyze all retrieved trials. As there is no mechanism for PIs to self-report race or ethnicity, an algorithm was used. This algorithm fit each name string into one of the four categories and may perform unevenly across the four categories; thus, some imprecision in assigned racial and ethnic groups was inherent to the analysis. The validation sub-study conducted as part of this work (Supplementary Tables E1 and E2) showed that prediction of Black PI race/ethnicity was poor, suggesting PI and study staff self-reported demographic characteristics would be preferable. Until processes to capture PI and study staff demographic information are in place, BMS is leveraging the report and analytic systems of the field-based medical and clinical operation teams regarding PI and study staff race/ethnicity.
The analysis of the impact of purposeful recruitment efforts used an interim “data snapshot” from two of the three prostate cancer trials. Additional analyses will be needed when enrollment for these trials is complete in order to determine the final impact of the community partnerships on patient diversity in these trials.
Conclusion
This investigation of diversity within BMS-sponsored US oncology clinical trials showed an imbalance of race/ethnicity among patients. The advanced analytics provide a data-driven approach to inform strategies that can impact clinical trial patient diversity. The positive concordance results observed with PI race/ethnicity, county-level diversity of trial sites, and engagement initiatives are hypothesis-generating in nature. BMS is implementing measures to improve diversity among PIs, increase study sites in areas of high diversity, and to further engage with advocacy and community organizations. Further analyses are needed to confirm whether these interventions have a positive, sustained impact on clinical trial patient diversity. Additional interrogation of BMS clinical trial data across geographies and therapeutic areas will continue to define efforts to include patients that reflect real-world epidemiology and medical practice worldwide.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745231180506 – Supplemental material for Data-driven strategies for increasing patient diversity in Bristol Myers Squibb–sponsored US oncology clinical trials
Supplemental material, sj-docx-1-ctj-10.1177_17407745231180506 for Data-driven strategies for increasing patient diversity in Bristol Myers Squibb–sponsored US oncology clinical trials by Lorena Kuri, Sagar Setru, Gengyuan Liu, Diane Moniz Reed, David Weigand, Aparna Surampudi, Susan Berger, David Paulucci, Angshu Rai, Venkat Sethuraman, Blythe Vito, Helen Kellar-Wood and Mariann Micsinai Balan in Clinical Trials
Footnotes
Acknowledgements
The authors thank The Prostate Health Education Network, FORCE—Facing Hereditary Cancer EMPOWERED, ZERO—The End of Prostate Cancer, Black Health Matters, and members of the Clinical Trial Ambassadors Program from the Yale Center for Clinical Investigation for their collaboration in this initiative. Writing assistance was provided by Jeff Frimpter, MPH, of Excerpta Medica, funded by Bristol Myers Squibb.
Correction (June 2023):
This article has been updated with minor grammatical or style corrections since its original publication.
Author contributions
L.K. and D.M.R. contributed to study conception and design and data interpretation. S.S., G.L., D.W., A.S., D.P., and A.R. were involved in data acquisition, analysis, and interpretation. S.B. contributed to data interpretation, and B.V. and H.K-W. to data acquisition and interpretation. V.S. and M.M.B. contributed to study conception and design and data acquisition, analysis, and interpretation. All authors reviewed all drafts of the manuscript and approved the final version of the publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.K., G.L., D.W., A.S., S.B., D.P., V.S., H.K-W., and M.M.B. are employees of Bristol Myers Squibb; S.S., A.R., B.V., and D.M.R. were employees of Bristol Myers Squibb at the time the analysis was conducted. L.K., S.S., D.M.R., S.B., and V.S. are stockholders of Bristol Myers Squibb; A.R. is a stockholder of Amgen and Bristol Myers Squibb.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Bristol Myers Squibb.
Third-party submission
The authors received editorial support from Patricia Fonseca, PhD, of Excerpta Medica. The authors have authorized the submission of their manuscript by Excerpta Medica and approved all statements and declarations.
Availability of data and material
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
