Abstract
Background:
Demographic diversity among clinical trials is required for representing the real-world populations intended for treatment and disease prevention. Moreover, genetic and environmental differences between ethnic and racial groups necessitate appropriately powered trials for relevant subgroups. We investigate the racial and ethnic demographic diversity of US-based participants in GSK-sponsored interventional trials. We also assess the evaluation of demographic diversity against US Census and epidemiologic data.
Methods:
GSK-sponsored interventional phase I–IV clinical trials conducted from 2002 to 2019 across three areas were analyzed: pharmaceutical (includes therapeutic medicines except for vaccines and human immunodeficiency virus (HIV)), vaccine (includes prophylactic and therapeutic vaccines), and ViiV (includes HIV therapies). A total of 1005 global trials encompassing 460,707 global participants were identified, of which 495 had US-based sites with a total of 108,261 (23.5% of global) US participants (pharmaceutical, n = 357 trials; vaccine, n = 45 trials; and ViiV, n = 93 trials). We evaluated how GSK US-based trial recruitment compares with US Census (in line with previously published studies from other groups) and with epidemiologic data.
Results:
GSK participant data for race and ethnicity combined across areas were generally similar to US Census levels (e.g. GSK versus census: White, 76.5% versus 76.3%; Black or African American, 15.1% versus 13.4%; Asian, 1.8% versus 5.9%; Hispanic or Latino, 14.0% versus 18.5%; Non-Hispanic White, 63.5% versus 60.1%). However, this was not the case for the individual pharmaceutical, vaccine, and ViiV data sets; least represented groups were Asian individuals for pharmaceutical and ViiV trials and American Indian or Alaskan Native individuals for vaccine trials (6.2%, 11.8%, and 11.1% of trials met/exceeded census level representation, respectively). The percentage of trials reaching/exceeding census levels also varied per trial phase for race and ethnicity. Furthermore, disparities in the percentage of trials reaching/exceeding census levels versus epidemiology-based prevalence levels have revealed opportunities to improve industry success metrics; in HIV trials, the proportion of Black or African American individuals (35.1%) exceeded census (13.4%) but not epidemiologic levels (55.3%).
Conclusion:
Further work is required to achieve demographic diversity across clinical trials. We conclude that US Census data are an inappropriate universal benchmark. A shift to epidemiology benchmarking will enable the consideration of global participants into US analyses for highly intrinsic (i.e. influenced by ancestry) diseases and more firm requirements for US-based participants into US analyses for extrinsic (i.e. influenced by location or culture) diseases. Benchmarking in line with epidemiologic data will allow us to set better trial enrollment goals, with the aim of conducting more demographically balanced, diverse, and representative clinical trials and enabling a better understanding of drug safety and efficacy per demographic group.
Introduction
Clinical trials are integral for evaluating the safety and efficacy of novel drugs and vaccines for different participant populations. 1 In recent years, there have been increasing discussions among the research community,2–6 and regulatory and government bodies, regarding the diversity landscape of clinical trial recruitment.7–9 Previous research and several recent meta-analyses have indicated that certain ethnic and racial groups have been underrepresented in clinical trials across different therapy areas.1,10–14
Trials should optimally reflect the real-world populations that are intended for treatment and disease prevention once the drug/vaccine is approved. 1 Certain diseases can disproportionately affect different ethnic and racial demographic subgroups due to both environmental and genetic factors, such as sickle cell disease in people of African descent, Tay-Sachs disease in Jewish people of Eastern European heritage, and human immunodeficiency virus (HIV) in people of African descent and Hispanic individuals.15–22 Thus, demographic representation of populations relevant to individual disease types must be improved to ensure that trial results are applicable to commonly underrepresented populations. 1 Key approaches to achieve this include generating a clear framework to guide and encourage diverse enrollment target goals and developing strategies to overcome recruitment-related barriers.1,10,23 Initiatives such as the INCLUDE Ethnicity Framework are currently underway to guide study teams to improve inclusion of underrepresented groups in health research. 23
In 2020, the US Food and Drug Administration (FDA) released a Drug Trials Snapshots Summary report, which provided an overview of the race, ethnicity, age, and sex compositions of individuals enrolled in past clinical trials conducted from 2015 to 2019. 24 Coupled with data from the US Census Bureau, it may be possible to benchmark clinical trial enrollment targets against local demographic census data. 25 However, in setting diverse enrollment targets, consideration must be given to how trial performance is assessed, for example, whether matching or exceeding census level heterogeneity is appropriate for the disease being studied. While census levels have previously been used as a benchmark for clinical trial diversity,2,26 it is important that we acknowledge and understand the limitations to this approach. Despite FDA guidance and the importance of accurate representation, it has been noted that proportions of patients enrolled in certain drug development programs are not always reflective of the intended patient population. 27 This provides further rationale to assess diversity against epidemiologic data in future clinical trials.
Through the results described herein using GSK (previously GlaxoSmithKline) data, we aim to complement and expand upon the Pfizer data set, which recently provided an important insight into demographic diversity of Pfizer-sponsored trials in the United States compared with census levels. 2 In addition to presenting GSK US trial data compared with census levels, we provide the number of global GSK participants by disease area, demographic, and trial phase, plus additional analyses by trial phase for US participants. Moreover, we aim to demonstrate the strengths of using epidemiologic data as a benchmark for assessing trial diversity in completed US trials, as opposed to using solely census data, in order to more accurately assess racial and ethnic representation.
Methods
Criteria for study/participant selection
The diversity of phase I–IV interventional clinical trials conducted by GSK and ViiV from 2002 to 2019 was assessed. Diversity data from more recent trials were not readily available due to ongoing data integration efforts and studies that had not converted to the Clinical Data International Standards Consortium data format. Definitions of races and ethnicities included in the study are reported in the Supplemental Appendix. Participants who selected more than one race in the case report form were counted in the “More Than One Race” category and were not counted under individual race categories to avoid duplication. In vaccine trials, participants self-reported the phrase “More Than One Race” under the “Other” case report form category, and these instances were programmatically corrected and moved to the “More Than One Race” category. Race and ethnicity terminology/categorization was based upon US Census reporting and FDA guidance (see Supplemental Notes).
Clinical trials in the areas of pharmaceutical (therapeutic medicines except vaccines and HIV therapies), vaccine (both prophylactic and therapeutic vaccines), and ViiV (therapies for HIV) were eligible. Identified trials comprised those that were completed and archived (no active trials were included), collected both race and ethnicity data, and had data from demographic and disposition domains in the Study Data Tabulation Model (SDTM) format. Excluded trials included investigator-sponsored, extension and compassionate use trials, studies that had a non-defined therapeutic area, and consumer-healthcare business area trials. All participants who passed screening and entered an eligible study were included; participants comprised those from randomized and non-randomized studies and those who were assigned a “treatment” or “no treatment,”“randomized, but not dosed,” or “not treated.” Screening failures (individuals who never passed screening), run-in failures, and participants screened but never enrolled in a study were excluded from the enrolled population.
Analyses of diversity
Percent distribution of race, ethnicity, sex, and age categories was calculated for all available global data. US distribution values were compared with demographic distribution data reported by the US Census Bureau in 2019. US data only were further analyzed due to the availability of comparable census data. US data included those from US-based clinical trials and data from participants at US sites within global trials. Distribution values were also compared against real-world epidemiologic data where sample sizes were sufficient. The epidemiologic data were obtained from the publicly available Centers for Disease Control and Prevention data sets.28–31 Statistical analysis was primarily carried out using SAS (version 9.4). The displays in this article were created using R (version 4.0.2) and RStudio, and Workbench (version 4.1717-3), which was validated, and were quality checked against SAS.
Results
Clinical trial participants by therapy area
The full global analysis data set included 1005 trials comprising 460,707 participants, while the full US data set comprised 108,261 participants (23.5% of global) across 495 trials (Figure 1(a), Supplemental Table 2a). The pharmaceutical, vaccine, and ViiV data sets accounted for 75.0%, 14.7%, and 10.2% of global trials comprising 45.0%, 51.8%, and 3.2% of trial participants, respectively (Figure 1(b)). Most US trials were pharmaceutical (72.1%) and included just over half of all US participants (55.5%) (Figure 1(c)). US respiratory trials comprised the largest portion of US trials (33.9%). Only 10.0% of US trials were vaccine trials, most prophylactic (88.9%), although these included 39.3% of all US participants. US ViiV trials comprised 18.8% of all US trials, with 5.3% of US participants.
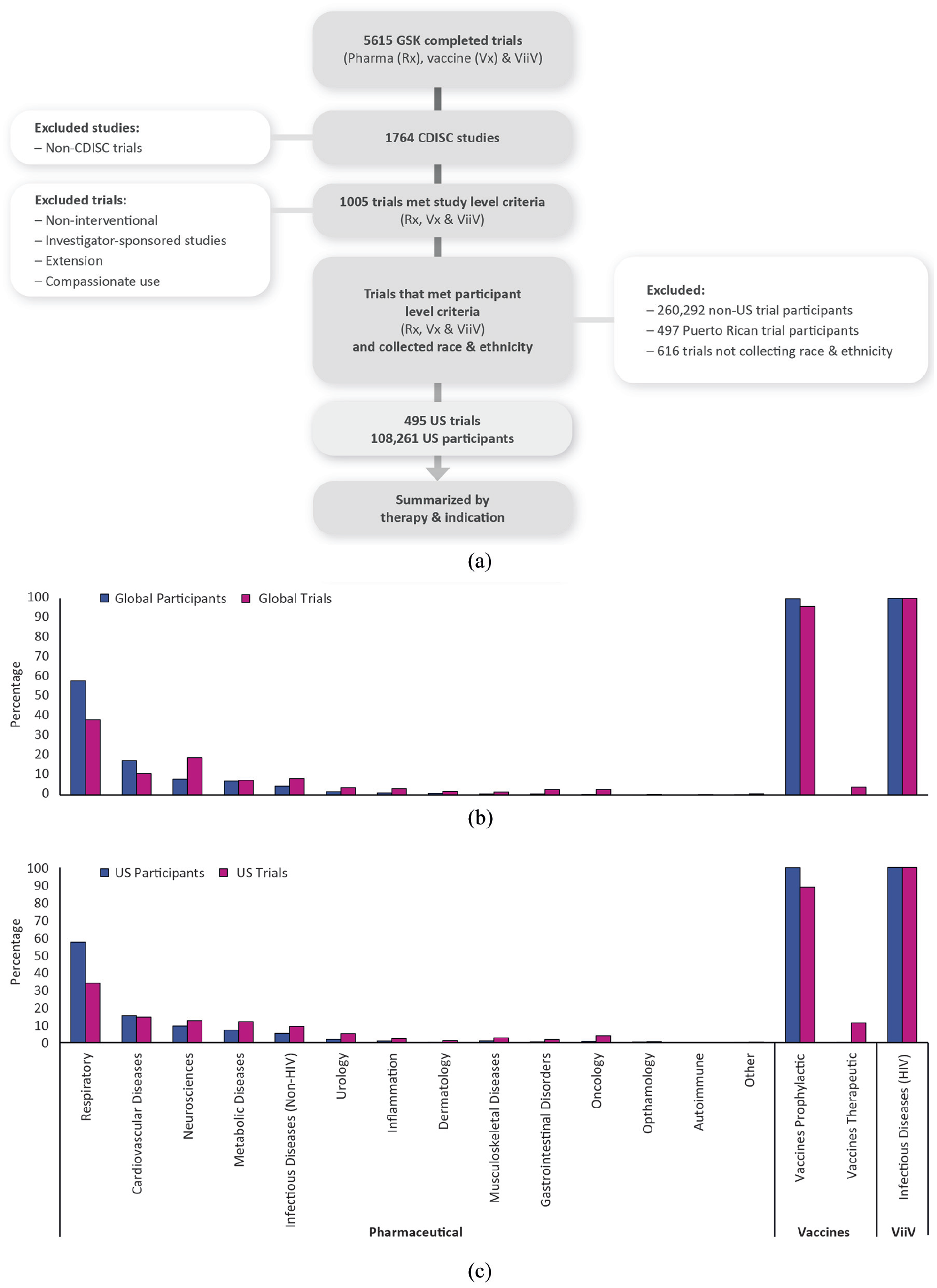
Summary of GSK’s analysis. (a) Composition of GSK’s demographic analysis. N = 759 trials excluded in total. (b) Summary of global pharmaceutical, vaccine, and ViiV trials by therapy area. GSK portfolio includes all therapeutic areas across all companies. Percentages are calculated by the total number of participants or trials by individual therapy areas per research area. Global trials include trials with US sites. Global participants: pharmaceutical, n = 207,415; vaccine, n = 238,648; and ViiV, n = 14,644. Global trials: pharmaceutical, n = 754; vaccine, n = 148; and ViiV, n = 103. Full data are provided in Supplemental Table 2a. (c) Summary of US pharmaceutical, vaccine, and ViiV trials by therapy area. GSK portfolio includes all therapeutic areas across all companies. Percentages are calculated by the total number of participants or trials by individual therapy areas per research area. US trials include global trials with a site in the United States. US participants: pharmaceutical, n = 60,038; vaccine, n = 42,503; and ViiV, n = 55. US trials: pharmaceutical, n = 357; vaccine, n = 45; and ViiV, n = 93. Full data are provided in Supplemental Table 2c.
Diversity of US clinical trial participants combined across pharmaceutical, vaccine, and ViiV trials
A summary of participant race, ethnicity, sex, and age by therapy area for global and US data sets is shown in Figure 2(a–d) and Supplemental Tables 2(b–e). For race, ethnicity, sex, and age categories, GSK participant US-based enrollment data were generally similar to US Census levels (Supplemental Figure 1). The percentages of people of African descent and non-Hispanic White individuals were slightly higher than US Census levels, while the percentages of Asian and Hispanic or Latino individuals were lower. Likewise, percentages of people in the 5–17 and 18–64 (years) age categories were lower than US Census levels, while the percentages of people in the <5 and ≥65 years categories were higher.
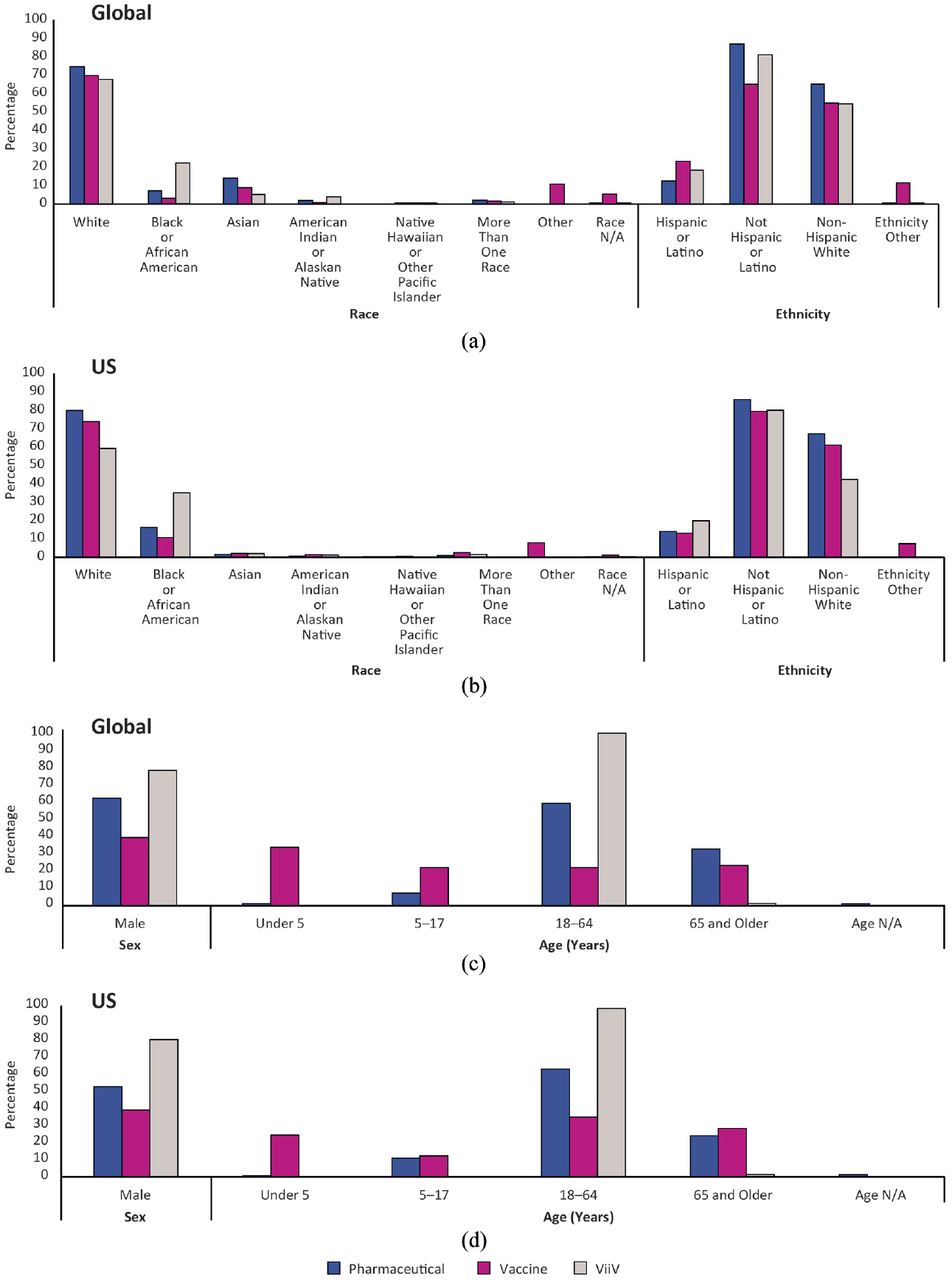
Summary of participant demographics for pharmaceutical, vaccine, and ViiV trials by therapy area. (a) Summary of participant race and ethnicity for global trials. Percentages are calculated by dividing the number of participants of a specified race or ethnicity per therapy area by the total number of participants enrolled to each individual therapy area per research area. Full data and breakdown of race and ethnicity into therapy area are provided in Supplemental Table 2b. For “Race, Other,” trial participants self-reported their race via “Free Text” option; for “Race, N/A,” trial participants’ race was not self-reported; for “Ethnicity, Other,” trial participants’ ethnicity was not self-reported. Individuals described as “person of African descent” are displayed as “Black or African American” and individuals described as “Indigenous” are displayed as “American Indian or Alaskan Native.” (b) Summary of participant race and ethnicity for US trials. Percentages are calculated by dividing the number of participants of a specified race or ethnicity per therapy area by the total number of participants enrolled to each individual therapy area per research area. Full data and breakdown of race and ethnicity into therapy area are provided in Supplemental Table 2c. For “Race, Other,” trial participants self-reported their race via “Free Text” option; for “Race, N/A,” trial participants’ race was not self-reported; for “Ethnicity, Other,” trial participants’ ethnicity was not self-reported. Individuals described as “person of African descent” are displayed as “Black or African American” and individuals described as “Indigenous” are displayed as “American Indian or Alaskan Native.” (c) Summary of participant sex and age for global trials. Percentages are calculated by dividing the number of participants of a specified sex or age by the total number of participants enrolled to each individual therapy area per research area. Full data and breakdown of sex/age into therapy area are detailed in Supplemental Table 2d. For “Age, N/A,” trial participant age was not reported. Three individuals did not self-report sex; however, they are included in the total number of participants in the GSK portfolio. (d) Summary of participant sex and age for US trials. Percentages are calculated by dividing the number of participants of a specified sex or age by the total number of participants enrolled to each individual therapy area per research area. Full data and breakdown of sex/age into therapy area are detailed in Supplemental Table 2e. For “Age, N/A,” trial participant age was not reported. One individual did not self-report sex; however, this individual is included in the total number of participants in the GSK portfolio.
The mean and median number of participants required to meet census levels in each underrepresented demographic was relatively small compared with the total number of trial participants across pharmaceutical, vaccine, and ViiV trials (Figure 3).
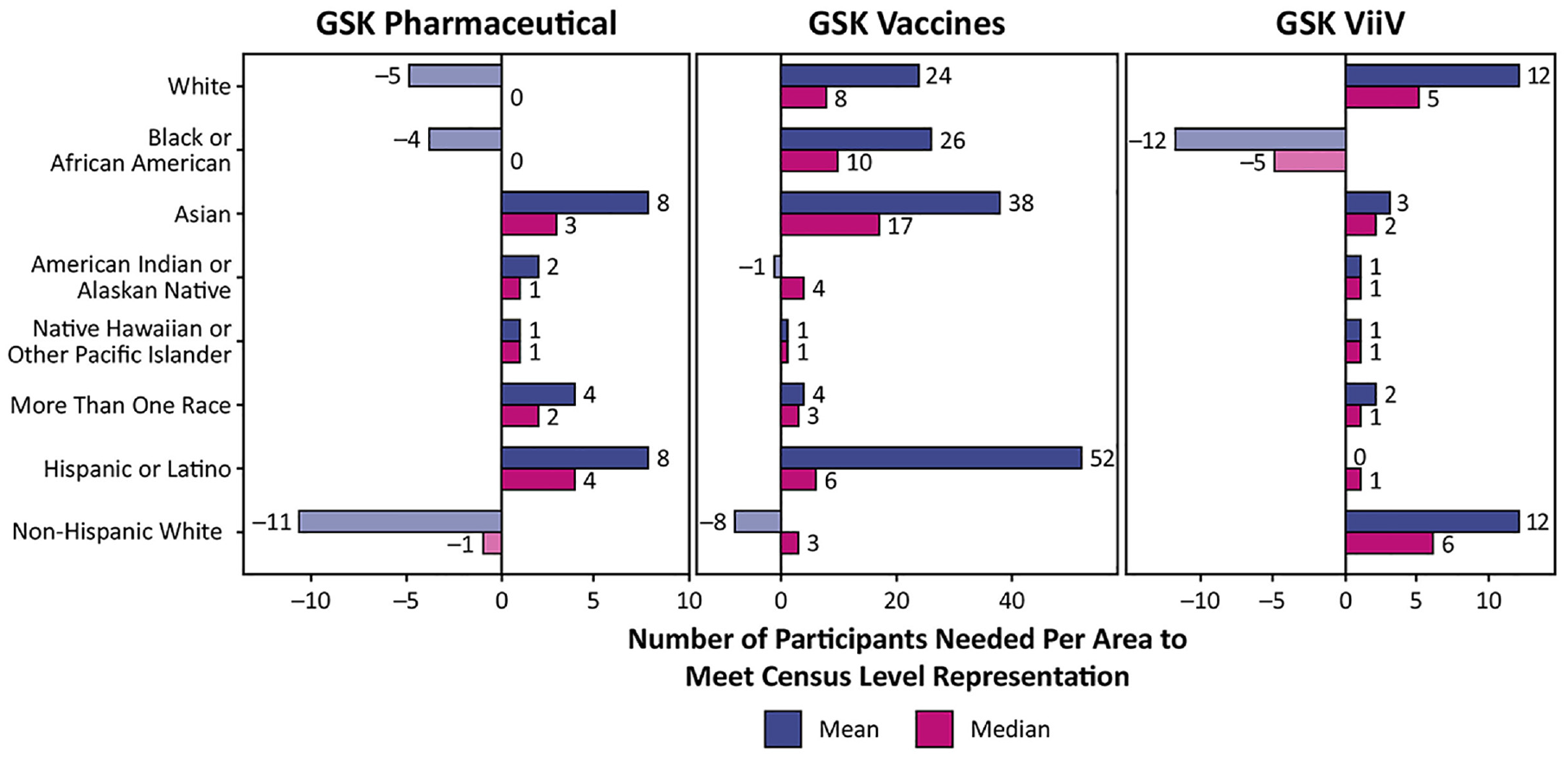
Mean and median number of participants required to meet census levels for each race and ethnicity. Target participant numbers for each trial were determined by multiplying the trial size by the census percentages for each demographic, and then rounded to the nearest whole number. The mean and median of these target values were calculated across all trials, with the mean also being rounded in order to report whole numbers of people. Negative values represent overrepresentation compared with census data. Positive values represent underrepresentation compared with census data. Individuals described as “person of African descent” are displayed as “Black or African American” and individuals described as “Indigenous” are displayed as “American Indian or Alaskan Native.”
US clinical trial diversity by research area
When viewed in more detail, the overall number of US trials meeting or exceeding census level representation varied among racial and ethnic groups. For pharmaceutical trials, White, people of African descent, and non-Hispanic White groups appeared to be well-represented in a large proportion of clinical trials. Asian individuals and individuals of more than one race were the least represented (Figure 4(a)). For vaccine trials, the most well-represented group was non-Hispanic White, while the least represented group was Indigenous individuals (Figure 4(b)). For ViiV trials, people of African descent (coded as Black and/or African American for FDA purposes) were most well-represented, and less than half of all trials met census levels for the other ethnic and racial groups of individuals (Figure 4(c)).
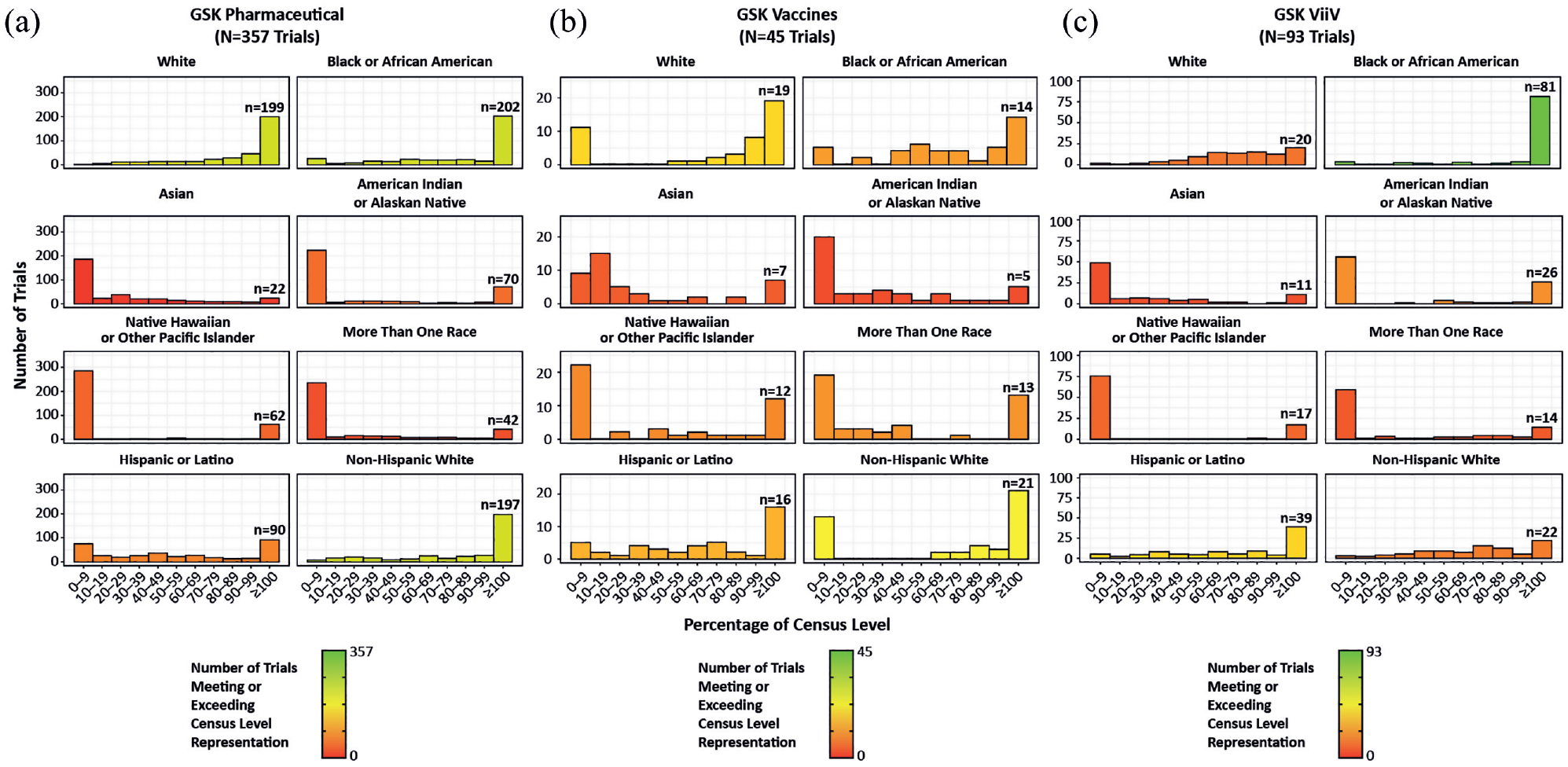
Histograms showing the percentage of trials meeting or exceeding census level representation. (a) Pharmaceutical trials. A total of 357 trials were included in this analysis. (b) Vaccine trials. A total of 45 trials were included in this analysis. (c) ViiV trials. A total of 93 trials were included in this analysis. Individuals described as “person of African descent” in the article are displayed as “Black or African American” and individuals described as “Indigenous” are displayed as “American Indian or Alaskan Native.” Trials meeting or exceeding census levels were grouped together because exceeding census levels does not equate to exceeding epidemiologic benchmarks.
US clinical trial diversity by development phase
A summary of race and ethnicity by trial phase for global and US data sets is shown in Supplemental Table 3. Phase I pharmaceutical trials had a smaller proportion of White individuals than phase II–IV pharmaceutical trials in both the global and US data sets. Conversely, phase I pharmaceutical trials had a greater proportion of people of African descent than phase II–IV pharmaceutical trials in both the global and US data sets. A similar pattern was seen when evaluating the percentage of US pharmaceutical trials meeting census levels across trial phases (Supplemental Figure 2). For White individuals, the percentage of trials meeting census levels increased from phases I to IV. Conversely, while census levels are being met for people of African descent in phase I trials, the percentage of trials meeting census levels decreased throughout the phases, with a slight increase from phases III and IV. Census levels were not met in a large proportion of Asian individuals or individuals of more than one race across all trial phases. The highest representation of Indigenous individuals was in phase I trials, while the highest representation of Native Hawaiian or other Pacific Islander was reached in phase IV trials. A higher percentage of trials reached census levels, particularly in later phases, for non-Hispanic White individuals than Hispanic or Latino individuals.
The percentage of vaccine trials achieving census levels was also mixed across trial phases for each race and ethnicity (Supplemental Figure 2). For all races and ethnicities apart from White and non-Hispanic White, 0% of trials met census levels in phase IV trials. However, only two trials were included in this phase IV trial analysis.
Enrollment of participants in ViiV trials generally did not reach census levels across the different races and ethnicities; however, was close to or met census levels for people of African descent across all phases (Supplemental Figure 2). No trials in this US-specific data set met census levels for Asian individuals in phase II, III, or IV trials. Representation of White and Indigenous individuals decreased across phases. Census levels of Native Hawaiian or other Pacific Islander participants were not met in the majority of phase I, II, or III trials; however, were met in the single phase IV trial analyzed. Representation of individuals of more than one race was highest for phase II trials. Conversely to pharmaceutical and vaccine trials, a larger number of trials met census levels for Hispanic or Latino individuals compared with non-Hispanic White individuals.
Trial enrollment versus epidemiologic data and US Census data
Comparisons of asthma, chronic obstructive pulmonary disease (COPD), HIV, and influenza vaccine trial enrollment versus real-world epidemiologic data and census data are shown in Figure 5. Differences in the percentage of ethnic groups, and differences in the comparisons to epidemiologic and census data, were seen across all four therapy areas. Enrolled Asian individuals and individuals of more than one race did not reach epidemiologic or census levels in any of the four therapy areas. For HIV trials, the proportion of enrolled White individuals exceeded epidemiologic levels but did not reach population proportions as described by the US Census; in contrast, the proportion of people of African descent enrolled did not reach epidemiologic levels but exceeded census levels.
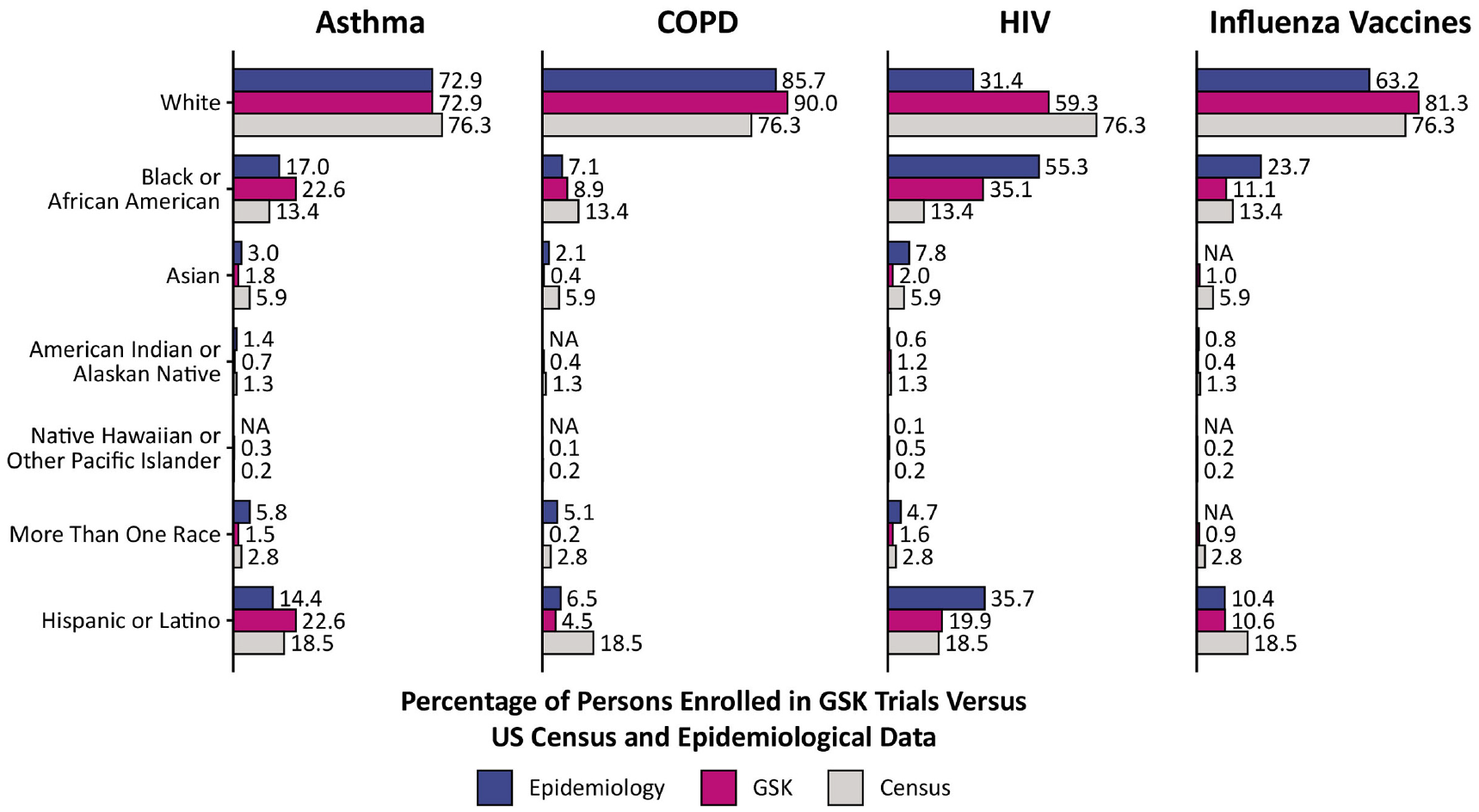
Comparison of asthma, COPD, influenza vaccines, and HIV enrollment demographics versus real-world epidemiologic data and census data. Due to the way race and ethnicity is interpreted by the CDC, the following apply: for influenza and HIV, people with Hispanic or Latino ethnicity could be of any race; however, persons of each individual race were not classified as Hispanic or Latino. for COPD and asthma, people with Hispanic or Latino ethnicity could be of any race and persons of each individual race could be either Hispanic or Latino or non-Hispanic or Latino. In order to determine the proportion of each indication (asthma, COPD, influenza, and HIV) by race, those categorized by ethnicity were removed from the population sum or denominator. Each race represents a proportion of the population sum and therefore, for persons categorized by ethnicity as Hispanic or Latino, the race is not reported but they could be of any race. Epidemiology for influenza is derived from CDC influenza-associated hospitalization data from 2016 to 2020; no data were available for correlation directly to individuals who are defined as “Asian,”“Native Hawaiian or Other Pacific Islander,” or “More Than One Race.” Individuals described as “person of African descent” in the article are displayed as “Black or African American” and individuals described as “Indigenous” are displayed as “American Indian or Alaskan Native.”
Discussion
Diversity of participants in clinical trials
Combined data from pharmaceutical, vaccine, and ViiV trials indicated that percentage GSK participant enrollment at US sites was generally similar to census level data across race, ethnicity, age, and sex categories. However, when looking at individual research areas, few trials met census proportions for Asian, Indigenous, Native Hawaiian or other Pacific Islander, Hispanic or Latino individuals, or individuals of more than one race. In contrast, a large number of pharmaceutical and vaccine trials met or exceeded census levels for White and non-Hispanic White individuals. Many pharmaceutical and ViiV trials met or exceeded census levels for people of African descent. This may reflect that, in the United States, people of African descent are significantly more affected by HIV and therefore more likely to be enrolled in ViiV trials compared with other ethnicities, reinforcing the need for consideration of epidemiologic benchmarks. 32 We also identified that the mean and median number of participants required to meet a representation disparity in line with census levels were surprisingly small, and we expect that this gap would remain similarly small when shifted to an epidemiologic reference point. This suggests that the effort required to close the gap and achieve census/epidemiologic levels may be less than expected.
The two largest US minority groups (people of African descent and Hispanic or Latino) showed notable changes across study phases. People of African descent met representation near census levels in 78% of pharmaceutical and 60% of vaccine phase I trials–this steadily declined toward phase III, with a small increase to phase IV in pharmaceutical trials. Vaccine and ViiV trials have limited phase IV data, with only two and one trial(s) analyzed, respectively. However, given the targeted nature of phase IV trials, 0% population representations are expected. US phase I healthy volunteer trials are known to disproportionately enroll individuals from racial and ethnic minority groups, such as those that are economically and educationally disadvantaged, 33 possibly driven by the financial incentives around participation. 34 The demography of phase I trial populations may also reflect the tendency to locate phase I study centers in urban areas or around universities, which have higher proportions of certain ethnic minorities. This raises ethical questions given the high risk–benefit ratio associated with lower phase trials. The decline in participation as phases progress warrants further analyses of operational data with respect to phase and phase-specific site characteristics and overall study accessibility.
GSK enrollment compared with epidemiologic and census levels for asthma, COPD, HIV, and influenza differs per race and ethnicity for each condition. In trials of each therapeutic area, the proportion of White individuals enrolled varied between 59% and 90%, while Black or African American individuals ranged from 9% to 35%. Furthermore, our data demonstrated that across GSK asthma, COPD, HIV, and influenza vaccine therapy areas, trials were differentially representative of certain ethnic or racial populations depending on the reference population (i.e. US real-world epidemiologic data or census data). For example, the enrollment of individuals of African origin for HIV markedly exceeded census levels, but this group of individuals were underrepresented when compared with epidemiologic values.
Challenges related to using census data as a benchmark
Census data largely fail to reflect disease epidemiology and we conclude that alone these data are not a scientifically valid way to develop clinical trial enrollment goals. For ethnic or racial groups comprised of few individuals, it may be necessary to sample a larger proportion compared with census levels to appropriately represent the relevant groups of individuals. For a given disease, certain populations of individuals may be more severely affected due to various intrinsic (genetic and biologic) and extrinsic (societal disease burden and health equity) factors that may influence treatment, disease disparity, disease risk, and the availability of, and access to, therapeutic agents.1,35–38 The COVID-19 pandemic is a clear and recent example of how the impact of a disease may vary depending on sociodemographic factors (see Supplemental Notes). In order to provide appropriate access to clinical trials across relevant demographic groups, it is important to understand and quantify disease burden and advocate for health equity.39,40 In addition, utilizing medical anthropology may provide further understanding of how global, historical, and political forces contribute to health and clinical trial diversity challenges. 41
Our position on limiting the use of the US Census as a benchmark aligns with efforts of others within the research community. The National Institutes of Health has incorporated related guidelines in its policy on Inclusion of Women and Minorities as Subjects in Clinical Research. 42 In addition, Abdel-Rahman et al. 43 have identified an alternative measure of trial diversity, developing a “representation quotient,” which utilizes reference populations determined by a sample of the general population at the same geographic frequency as the research participant population. Furthermore, a recent study assessing racial and ethnic diversity in precision oncology trials for prostate, lung, breast, and colorectal cancers identified that relative to cancer in the US population, minority groups were markedly underrepresented. 14 The move toward using epidemiologic data as a benchmark is a promising development and requires significant attention moving forwards. A shift to epidemiology benchmarking will enable the consideration of global participants into US analyses for highly intrinsic diseases, and more firm requirements for US-based participants into US analyses for extrinsic diseases.
A key challenge remains regarding the limited reporting of race and ethnicity data in clinical trials. In a recent cohort study of 20,692 US-based clinical trials, collection of race and ethnicity data moderately increased from March 2000 to March 2020 but less than 44% of trials reported any race and ethnicity data, highlighting the need for consistent reporting. 44 To combat this, recent FDA draft guidance recommends that pharmaceutical sponsors provide race and ethnicity diversity plans to ensure appropriate representation across clinical development programs based on epidemiologic disease prevalence. 45 Updated FDA guidance is an important future direction that will enable transparent and uniform reporting across trials, in addition to allowing readers to assess the representativeness of a trial population.
A limitation of this study is that it exclusively analyzed US-based participants, which inherently provides a larger proportion of non-White participants compared with global populations. Global demographic representation varies from these analyses, especially in European and Asian countries. In addition, due to the lack of easily accessible metadata, we were unable to universally collect demographics of the screened populations, or present baseline characteristics stratified by enrolled, retained, and completed–it may be beneficial for future studies to report this as an additional measure of equity. However, our analyses comprised intent-to-treat trial populations, for which results from trials are reported and include data from enrolled participants regardless of whether they withdraw from, or do not complete, the study. Furthermore, we did not assess median year of enrollment for the different trials, which may have been valuable to evaluate progress in trial recruitment over time. Our data set included a marked number of respiratory trials compared with trials for other conditions; hence, the results of the analysis are skewed toward respiratory conditions. Another limitation is that there were inconsistencies in how race and ethnicity were reported and categorized across studies; however, this is widely applicable to similar diversity studies. As discussed, the comparison to census rather than epidemiologic data comes with its own limitations, in particular the disparities in ethnic and racial trial enrollment that differ across a number of different therapy areas (see Supplemental Notes). In addition, the US population, and therefore census proportions, are constantly changing over time and as trials may span several years, the accuracy of comparisons may be hindered by time mismatches. We chose the most recent US Census figures (highest minority proportions) to be conservative. Finally, some of the analyses by trial phase involved a small number of trials.
Future directions
Continued evaluation of clinical trial diversity will be important to shape and refine recruitment efforts, and further analyses of GSK’s global clinical trial data are underway. Determination of an appropriate benchmarking procedure to allow consistent and universal comparisons with other clinical research sponsors is a key challenge and should involve industry partnership organizations and patient community partnerships dedicated to increasing awareness of clinical trials.6,46 The creation of disease- or ethnic-specific data sets will also allow the comprehensive analyses of clinical studies and may help to address a lack of diversity in early genetic studies that subsequently influence later trials (see Supplemental Notes). A lack of adequate race or ethnicity categorization in many real-world data sets presents a major barrier to data set generation 47 although concentrated efforts are underway to address this, including the 23andMe Black Representation in Genetic Research study 48 and the All of Us Research Program. 49 This is supported by work to achieve global consistency across definitions and terminology of different races and ethnicities, such as the recent Journal of the American Medical Association guidelines; 50 GSK intends to lead ongoing industry efforts. Despite advances in reference populations, restrictive inclusion and exclusion criteria across clinical trials may still eliminate certain populations of individuals,11,12,51 and there remains fundamental and significant health equity work to be carried out alongside advances in genome sequencing. 52 Moving forward, adopting several key strategies to reach epidemiologic benchmarks will be required, together with process changes required to enroll higher proportions of underrepresented groups in clinical trials (see Supplemental Notes). The GSK Clinical Trial Diversity initiative, established in 2010, aims to optimize the diversity of clinical trials at GSK and ViiV Healthcare (ViiV) and includes a corporate level commitment to ensure that 75% of phase III trials initiated in 2022 will have proactive plans in place designed to enroll appropriately diverse trial participants consistent with the disease epidemiology.
Conclusion
These data have highlighted groups of individuals among GSK trials that are over- or underrepresented when compared with US Census data, and for a selection of disease states, when compared with epidemiologic data. Acknowledging these data is important; however, we must use care in reinforcing local census as a scientifically appropriate benchmark for clinical trial recruitment targets. In disease areas where study recruitment efforts were strongly aligned to epidemiologic prevalence, such as in HIV, COPD, and asthma, a potentially more accurate scientific understanding of the affected population was generated. GSK is currently adopting these per-disease and per-study benchmarks based upon epidemiologic data and sociodemographic factors. Our data can be used to support future meta-analyses, to present additional methodologies, and to offer guidance and insight to ensure diverse clinical trial enrollment. An infographic-based summary of this study can be found in Supplemental File 2.
Supplemental Material
sj-pdf-1-ctj-10.1177_17407745221149118 – Supplemental material for Demographic diversity of US-based participants in GSK-sponsored interventional clinical trials
Supplemental material, sj-pdf-1-ctj-10.1177_17407745221149118 for Demographic diversity of US-based participants in GSK-sponsored interventional clinical trials by Michel M Reid, Scott P Davis, Ouzama N Henry, Ashwin A Mathew, Scott McCallister, Taj T Nero, Sanjit A Rabheru, Shani H Sampson, Tracy F Vanderslice and Danaé T Williams in Clinical Trials
Supplemental Material
sj-pdf-2-ctj-10.1177_17407745221149118 – Supplemental material for Demographic diversity of US-based participants in GSK-sponsored interventional clinical trials
Supplemental material, sj-pdf-2-ctj-10.1177_17407745221149118 for Demographic diversity of US-based participants in GSK-sponsored interventional clinical trials by Michel M Reid, Scott P Davis, Ouzama N Henry, Ashwin A Mathew, Scott McCallister, Taj T Nero, Sanjit A Rabheru, Shani H Sampson, Tracy F Vanderslice and Danaé T Williams in Clinical Trials
Footnotes
Acknowledgements
The authors are grateful for the input and support provided by Alberto Fernandez, Annette Gross, Anup R Patel, Apoorva Chaloori, Carletha Blanding, Carol Lee, Catherine Leeming-Price, Chris Leach, Christine Clifton, Christopher Corsico, Fangfang Du, Fanny Mitrani-Gold, Harrison James, Justin Koteff, Kieran Rothnie, Kimberly Smith, Lucy Hoch, Matt Marino, Margaret G Ehm, Michael Aboud, Mohammad Ali, Nicki Kyriacou, Rabia Ahmad, Rachel Williams, Rob Wartenhorst, Sabine Luik, Sara Hughes, Sherene Min, and Vincent Barnett. GSK provided financial support for editorial assistance from Clare Davis of Articulate Science, UK.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.A.M., D.T.W., M.M.R., S.P.D., S.A.R., and T.T.N. are employees of GSK and own shares in GSK. O.N.H., S.H.S., and T.F.V. were previously employed at GSK; O.N.H. is now an employee of Dynavax Technologies; S.H.S. is an independent project management consultant for the pharmaceutical industry; and T.F.V. is an employee of Gilead Sciences. S.M. is an employee of ViiV Healthcare and owns shares in ViiV Healthcare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the research described herein and editorial assistance with the article was provided by GSK.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
