Abstract
Background:
A common intercurrent event affecting many trials is when some participants do not begin their assigned treatment. For example, in a double-blind drug trial, some participants may not receive any dose of study medication. Many trials use a ‘modified intention-to-treat’ approach, whereby participants who do not initiate treatment are excluded from the analysis. However, it is not clear (a) the estimand being targeted by such an approach and (b) the assumptions necessary for such an approach to be unbiased.
Methods:
Using potential outcome notation, we demonstrate that a modified intention-to-treat analysis which excludes participants who do not begin treatment is estimating a
Results:
We identify two key criteria in determining whether the modified intention-to-treat estimator is likely to be unbiased: first, we must be able to measure the participants in each treatment arm who experience the intercurrent event, and second, the assumption that treatment allocation will not affect whether the participant begins treatment must be reasonable. Most double-blind trials will satisfy these criteria, as the decision to start treatment cannot be influenced by the allocation, and we provide an example of an open-label trial where these criteria are likely to be satisfied as well, implying that a modified intention-to-treat analysis which excludes participants who do not begin treatment is an unbiased estimator for the principal stratum effect in these settings. We also give two examples where these criteria will not be satisfied (one comparing an active intervention vs usual care, where we cannot identify which usual care participants would have initiated the active intervention, and another comparing two active interventions in an unblinded manner, where knowledge of the assigned treatment arm may affect the participant’s choice to begin or not), implying that a modified intention-to-treat estimator will be biased in these settings.
Conclusion:
A modified intention-to-treat analysis which excludes participants who do not begin treatment can be an unbiased estimator for the principal stratum estimand. Our framework can help identify when the assumptions for unbiasedness are likely to hold, and thus whether modified intention-to-treat is appropriate or not.
Keywords
Background
A common intercurrent event affecting many randomised controlled trials (RCTs) is when some participants do not begin the treatment they were randomised to. 1 For example, the FLuid Optimisation in Emergency LAparotomy (FLO-ELA) trial (ISRCTN 14729158) compared the use of a cardiac output monitor against usual care in determining when and how much fluid to give to participants undergoing emergency laparotomy. Due to practical considerations, there is usually a small delay between randomisation and the start of surgery, and a small number of participants had their surgery cancelled after randomisation, either because they deteriorated and were too unwell to undergo surgery or because the underlying issue resolved itself. Here, a cancelled surgery precludes the participant from initiating treatment, as fluids are no longer relevant for participants who do not undergo their planned surgery. Similar issues may arise in other trials, where participants do not begin their study treatment or do not receive any dose of study drug.
Many trials use a ‘modified intention-to-treat’ (mITT) approach to handle these intercurrent events in the analysis, whereby participants who do not initiate treatment are excluded (i.e. the analysis is performed only on the participants who initiate treatment).1–4 However, this approach has been criticised on the basis that post-randomisation exclusions can induce bias.
1
Furthermore, it is not clear exactly
Given the widespread use of mITT in practice, there is urgent need to define the target estimand for such an approach, as well as the settings in which it will be unbiased. The purpose of this article is therefore to (a) demonstrate that an mITT estimator which excludes participants who do not begin treatment targets a
Methods
We begin by defining the principal stratum estimand, and then show how an mITT estimator can correspond to this estimand under certain assumptions. We then provide a simple framework for determining whether the mITT estimator is likely to be unbiased in practice for a given trial.
Principal stratum estimand
A principal stratum strategy to handle intercurrent events implies interest lies in the treatment effect in the subpopulation of participants who would (or would not) experience the intercurrent event.5–9 Here, we define the principal stratum estimand in terms of the intercurrent event of interest, which we refer to as ‘failure to initiate treatment’. Depending on the trial, this could be defined to include any cause of initiation failure (e.g. in a placebo-controlled drug trial where participants do not begin their course of study treatment, regardless of reason) or be defined based on a single cause of initiation failure, with other causes handled using alternate strategies (e.g. in FLO-ELA, where surgery cancellation precludes treatment initiation, but participants may also not initiate for other reasons).
The estimand can be written as
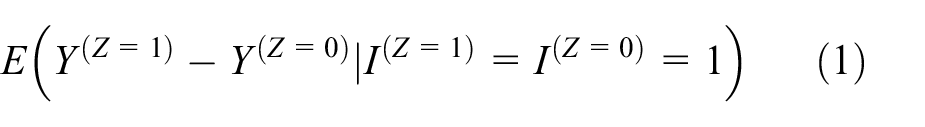
where
We note that the definition above primarily focuses on how the intercurrent event ‘failure to initiate treatment’ is handled; the other aspects which make up the estimand (population, treatment conditions, endpoint, summary measure, and handling of other intercurrent events) would also need to be specified in order for the estimand to be complete. 5
MITT estimator
The mITT estimator, which excludes participants who do not begin treatment from the analysis population, can be written as
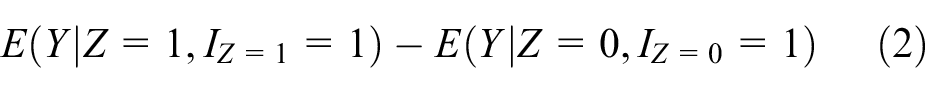
where
To evaluate the properties of this estimator, we begin by outlining the four principal strata defined by the intercurrent event ‘failure to initiate treatment’ (Table 1). 9
The four principal strata defined by the intercurrent event ‘failure to initiate treatment’.
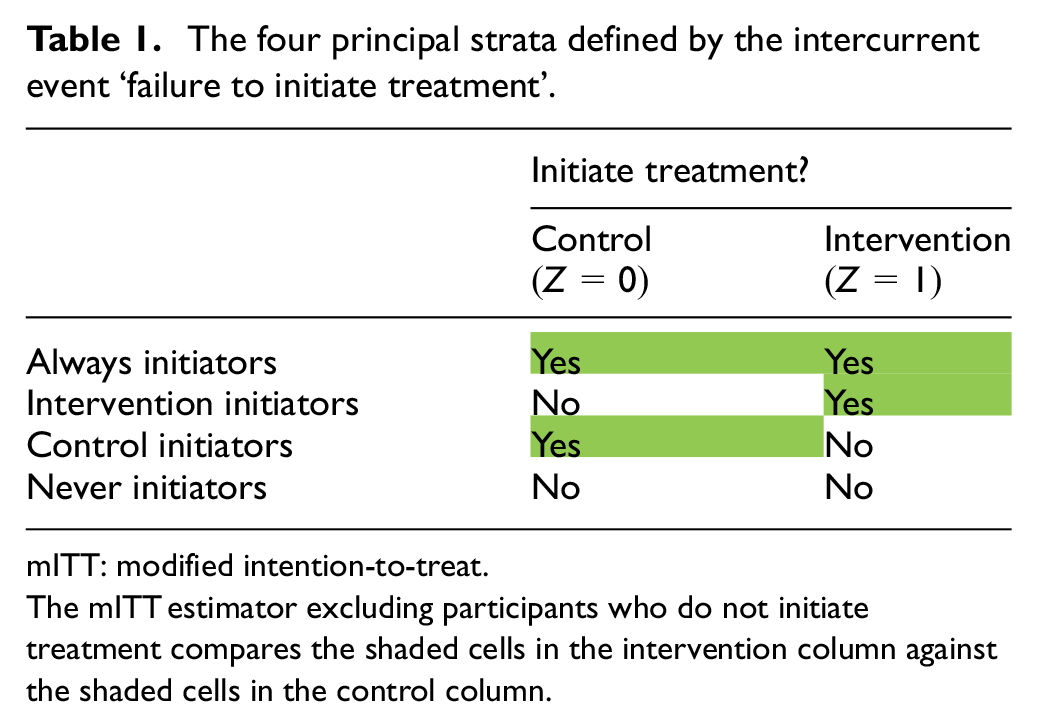
mITT: modified intention-to-treat.
The mITT estimator excluding participants who do not initiate treatment compares the shaded cells in the intervention column against the shaded cells in the control column.
The ‘always initiators’ are participants who would initiate treatment in either treatment group (i.e. for whom
In an RCT, randomisation can be seen to balance the treatment groups within each stratum in expectation. Hence, if we knew the principal stratum membership of each participant, we could perform the analysis in the stratum of interest and obtain an unbiased estimator based on a randomised comparison. However, in practice, we do not know the principal stratum membership, as this requires knowledge of participants’ initiation status under both treatment conditions, but we can only observe a participant’s initiation status under the treatment arm to which they were randomised. For instance, if a participant allocated to the intervention initiated treatment, we have no way of telling whether they are an ‘always initiator’ or an ‘intervention initiator’.
We can see from Table 1 that the estimator described in equation (2) is not based on a randomised comparison, as it compares different principal strata between the treatment arms (i.e. the analysis population differs between treatment groups). 10 The intervention arm includes the ‘always initiators’ and the ‘intervention initiators’, while the control group consists of the ‘always initiators’ and the ‘control initiators’. Hence, if these two strata (the ‘intervention initiators’ and ‘control initiators’) differ in terms of their potential outcomes, this estimator will be biased.
Assumptions for unbiasedness
Ensuring the mITT estimator in equation (2) is unbiased for the principal stratum estimand in equation (1) requires a way of identifying the principal stratum membership of participants. This is possible under the assumption that there are no ‘intervention initiators’ and no ‘control initiators’ in the trial, that is, that
This assumption implies there are only two principal strata in the trial, ‘never initiators’ and ‘always initiators’; hence, if we observe a participant’s initiation status in one treatment arm, we can infer their initiation status had they been allocated to the alternative treatment, and identify the principal stratum to which they belong (i.e. if we observe
Then, the mITT estimator in equation (2), which only includes participants who initiated treatment in their assigned arm, will in fact be a comparison within the ‘always initiators’ stratum, which, as discussed above, is based on a randomised comparison (Table 2). As such, this estimator will be unbiased for the principal stratum estimand in equation (1).
The mITT principal stratum estimator under the assumption of no ‘intervention initiators’ and no ‘control initiators’.
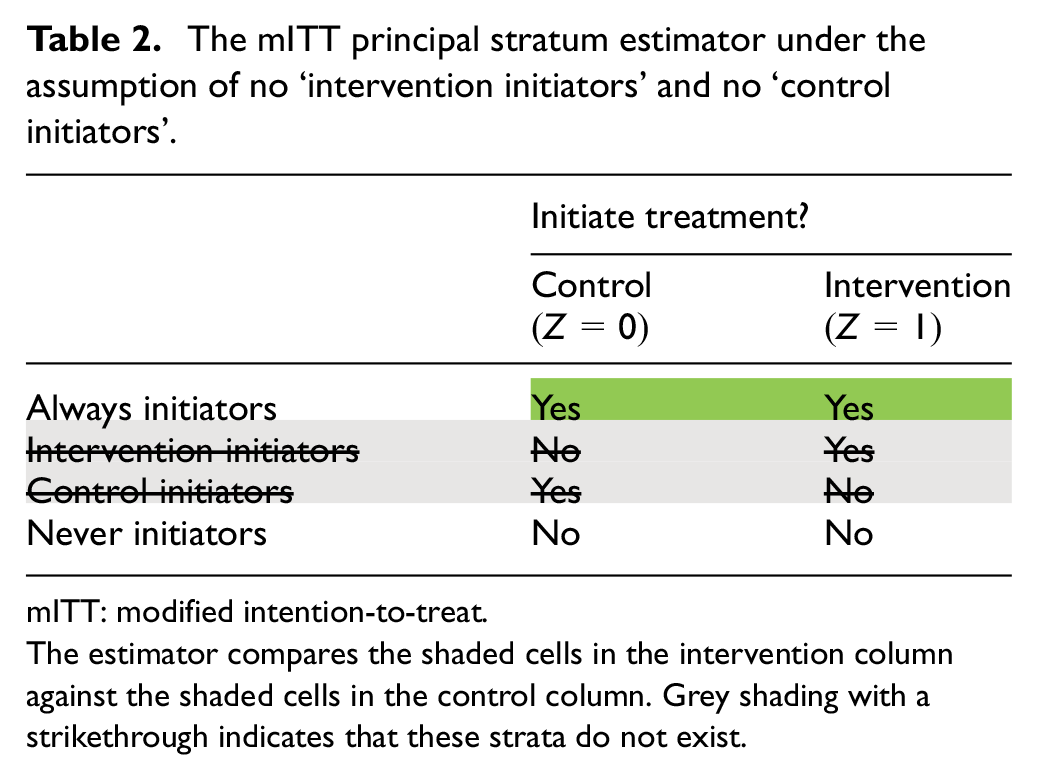
mITT: modified intention-to-treat.
The estimator compares the shaded cells in the intervention column against the shaded cells in the control column. Grey shading with a strikethrough indicates that these strata do not exist.
This assumption can be partly assessed, on the basis that if it is true, then the proportion of non-initiators should, on average, be the same across randomised groups. Hence, if there are large discrepancies in the proportion of non-initiators between groups, this may provide evidence that this assumption has been violated. However, even if this proportion is the same across groups, this is not a guarantee the assumption is true; hence, this assumption cannot be fully tested, and therefore must rely primarily on contextual information around its underlying plausibility.
We demonstrate the unbiasedness of the mITT estimator under the assumptions given above as follows. First, under consistency assumption
11
(which states that if
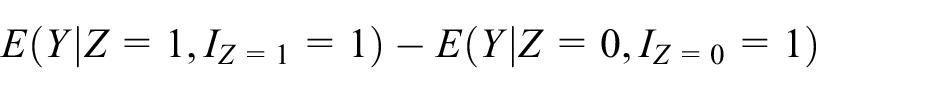
can be written as
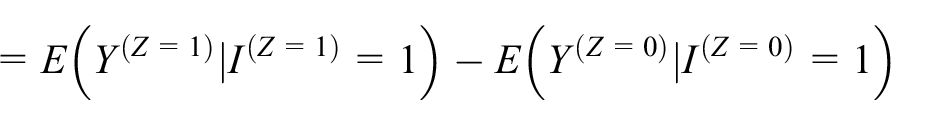
Then, under the assumption that there are no ‘intervention initiators’ and no ‘control initiators’,
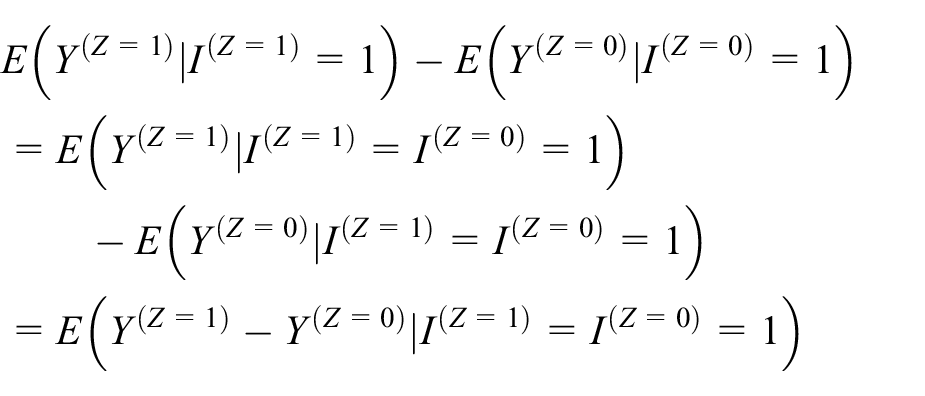
which is the principal stratum estimand in equation (1).
We note that this estimator is unbiased regardless of what happens to ‘never initiators’ (e.g. regardless of whether they receive no treatment at all or receive a non-trial treatment instead).
Determining whether the mITT estimator is appropriate
Two key factors can be used to determine whether the ‘always initiators’ principal stratum can be identified, thus ensuring the mITT estimator in equation (2) is appropriate. First, the intercurrent event of interest must be identifiable in each treatment arm (i.e. we must be able to measure which participants in each treatment group experience the intercurrent event).
Second, we must be able to assume that the occurrence of the intercurrent event is not affected by treatment allocation (i.e. that if the intercurrent event occurs for a participant under one treatment condition, it would also occur under the other treatment condition). Together, these two factors justify the assumption of no ‘intervention initiators’ and no ‘control initiators’.
Results
Below we give several examples of trials both where the mITT estimator is and is not appropriate (Table 3).
Examples of trials where the mITT principal stratum estimator both is and is not appropriate.
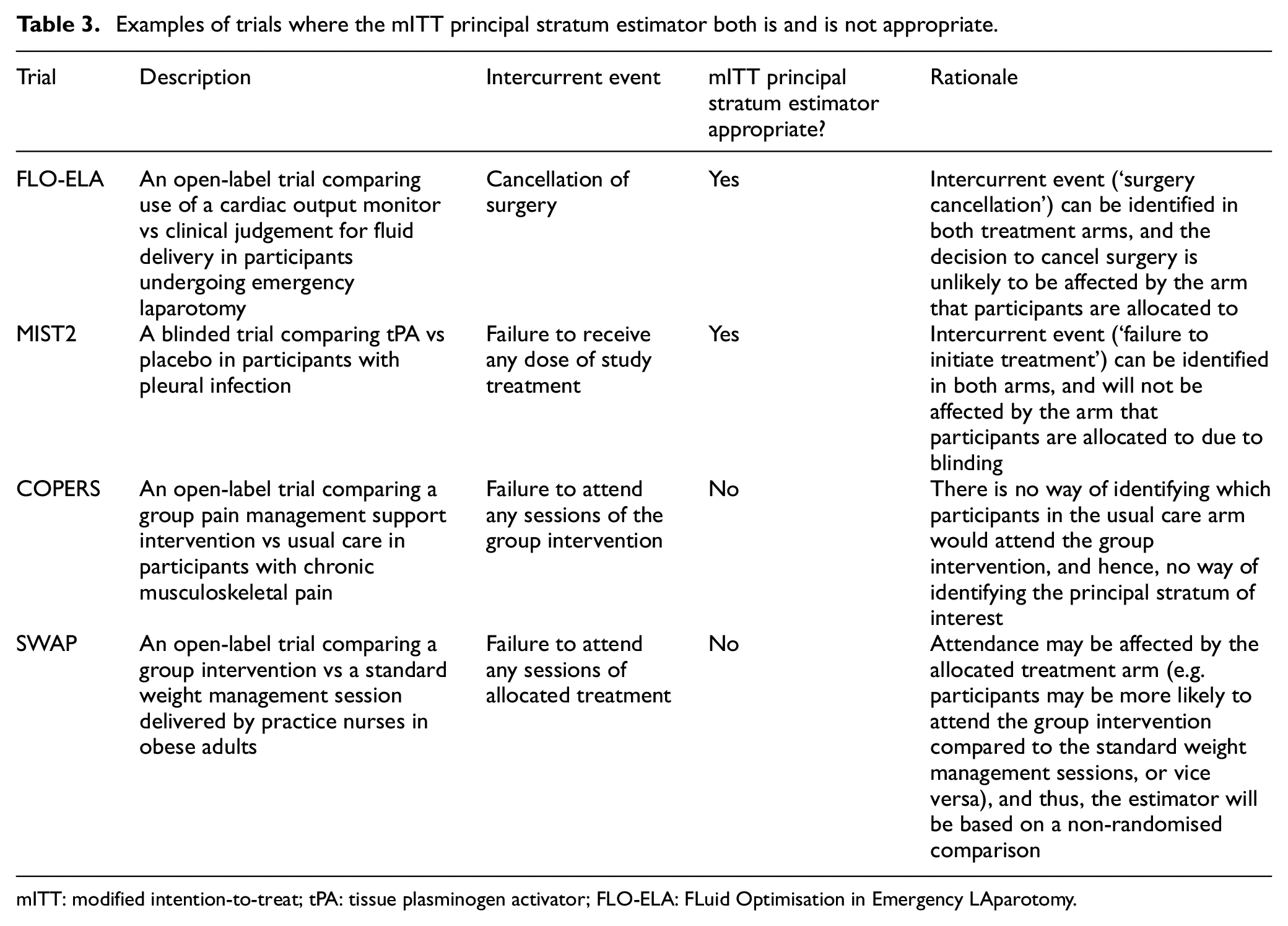
mITT: modified intention-to-treat; tPA: tissue plasminogen activator; FLO-ELA: FLuid Optimisation in Emergency LAparotomy.
FLO-ELA
As described earlier, FLO-ELA is an open-label trial comparing two methods of fluid delivery (cardiac output monitor vs clinician judgement) in participants undergoing emergency laparotomy. The primary outcome measure is the number of days that participants are alive and out of hospital within 90 days of randomisation. The intercurrent event of interest is the cancellation of surgery, which precludes treatment from being initiated (as fluid delivery is only relevant once participants begin surgery). This is an example of a ‘cause-specific’ initiation failure, as some participants who do undergo surgery may also not initiate treatment for other reasons. We note that ideally randomisation would be done at the point surgery was due to begin, to minimise the number of cancellations; however, in practice, this is not always feasible due to the time required to prepare the intervention combined with the emergency setting.
In this trial, the mITT estimator is appropriate. First, the intercurrent event can be easily identified and measured in both treatment groups, and second, the decision on whether to cancel surgery or not will almost certainly not be affected by the allocated treatment arm. This is because in most cases, the relevant decision-makers (surgeons) will be unaware of trial group allocation until surgery starts (i.e. at the point the decision is made). Furthermore, the decision not to proceed with surgery has large health implications for the patient and is only undertaken in response to a major change in the patient’s clinical condition since surgery was initially planned, and it is implausible that such a fundamental change in patient care would be undertaken on the basis of the planned method of fluid delivery.
MIST2
The Second Multicenter Intrapleural Sepsis Trial (MIST2) was a 2 × 2 factorial trial evaluating the use of tPA and deoxyribonuclease (DNase) in participants with pleural infection. 12 We focus on the tPA comparison here. tPA was compared in a double-blind fashion against a matching placebo, so that neither participants nor clinicians were aware of whether participants were receiving tPA or placebo. Treatment was to be given for 3 days. The intercurrent event of interest is failure to begin treatment (i.e. if participants did not receive any dose of study drug), regardless of the reason.
In this trial, the mITT estimator is appropriate. First, the intercurrent event (‘failure to begin study treatment’) can be identified and measured in each treatment arm. Second, treatment allocation should not influence the occurrence of the intercurrent event, as the trial is double-blinded, so those deciding on whether to initiate treatment will not be aware of whether it is tPA or placebo being initiated.
COPERS
The Coping with Persistent Pain, Effectiveness Research into Self-management (COPERS) trial evaluated the use of a group pain management support intervention for participants with chronic musculoskeletal pain. 13 The intervention consisted of a group intervention delivered over 3 days with a follow-up session at 2 weeks. The control consisted of usual care. The intercurrent event of interest is failure to begin the group intervention session (i.e. failure to attend any sessions).
Here, the mITT estimator is
SWAP
The Peer-support weight action programme (SWAP) trial evaluated the use of a multi-modal group intervention to reduce weight in obese adults. 14 The intervention consisted of eight group sessions delivered by trained advisors, while the control consisted of four standard weight management sessions delivered by a practice nurse. Due to the nature of the interventions, the trial was open-label, with participants and healthcare professionals aware of the allocated group. The intercurrent event of interest is failure to attend any treatment sessions.
In this trial, the mITT estimator is
Reporting of this estimator
Given that the mITT estimator in equation (2) relies on certain assumptions for unbiasedness, its use must be transparently reported so that readers may evaluate whether the assumptions underpinning the study results are reasonable.
To provide full clarity, we suggest the methods be reported according to Table 4. This provides the target of estimation (the estimand), which alerts readers a principal stratum effect is of interest; explicitly states that participants who experience the intercurrent event are excluded from the analysis population, along with the reason for doing so; provides the assumptions required for this estimator to be unbiased; and finally, justifies those assumptions for the specific trial. Together, this provides readers with an understanding of
Reporting recommendations for using the mITT principal stratum estimator.

mITT: modified intention-to-treat.
Recommendations are based on the mITT principal stratum estimator defined in formula (2) for the intercurrent event ‘failure to initiate treatment’.
Discussion
Failure to initiate treatment is a common intercurrent event in randomised trials. An mITT approach is frequently used, whereby participants who do not begin treatment are excluded from the analysis; however, it is not clear what estimand is being targeted by such an approach or the assumptions required for unbiasedness. In this article, we show that the mITT estimator which excludes participants who do not begin treatment targets a principal stratum estimand, and is unbiased under the assumption that if a participant experiences the intercurrent event under one treatment condition, they would also experience it under the other conditions.
This assumption is likely to be fulfilled in many settings, including most double-blind trials, where the decision to initiate treatment will not be affected by the treatment arm. This assumption may also be plausible in many open-label trials as well; for instance, in FLO-ELA, there is strong justification for expecting cancellation of surgery not to be affected by treatment group. However, these assumptions are not always appropriate, and when they are not, the mITT estimator will be biased. The results in this article can help investigators determine whether an mITT approach is appropriate for their trial. When the underlying assumptions cannot be justified, alternative methods to estimate the principal stratum estimand can be used (see Bowden et al. 6 and Lipkovich et al. 7 for an overview of approaches to estimate principal stratum effects in more challenging settings).
In addition to determining whether the mITT estimator will be unbiased, investigators also need to decide whether a principal stratum estimand is appropriate to use. This decision will depend on the specific aims of each individual trial; however, we argue that for failure to initiate treatment, a principal stratum strategy often is the most clinically relevant choice. For example, in the FLO-ELA trial, cancellation of surgery renders the participant no longer part of the population of interest (which is participants undergoing emergency laparotomy); hence, a principal stratum approach may be more reflective of the treatment effect in routine clinical practice compared to other strategies, such as treatment policy (which compares two different strategies of fluid delivery during surgery,
Despite the appeal of the principal stratum estimand, it has rarely been used in practice. 15 This may, in part, be because in more complex settings, principal stratum estimators require both more complex estimation procedures as well as more complex underlying assumptions which cannot always be verified based on contextual information about the trial. One benefit of our approach is that it is far more straightforward, both conceptually and to estimate, and so may be simpler to implement in practice.
mITT analyses have been criticised on the basis of ambiguity, that is, the term has been used by different researchers to mean different things.2–4,16 If using this term to describe the estimator, we recommend it be described as in Table 4 (i.e. that it is explicitly stated which intercurrent events are excluded), to avoid potential misunderstandings. It is also not always clear which estimand such mITT analyses have targeted in the past. The results in this article can serve to clarify the target of estimation for these analyses, as well as the assumptions required to justify their validity.
Although this article has focused on the intercurrent event ‘failure to initiate treatment’, the results could also apply to other intercurrent events where the assumptions discussed here are likely to be fulfilled. For instance, in trials where mortality is an intercurrent event but the treatments are known to not affect mortality (for instance, in certain palliative care trials), then (if clinically appropriate) the estimator described in equation (2) could also be used.
Conclusion
An mITT analysis which excludes participants who do not begin treatment can be an unbiased estimator for the principal stratum estimand. Our framework can help identify when the assumptions for unbiasedness are likely to hold, and thus whether mITT is appropriate or not.
Footnotes
Author contributions
B.C.K. wrote the first draft of the manuscript. I.R.W., M.E., and M.H. revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B.C.K. and I.R.W. are funded by the UK Medical Research Council (MRC) (grants MC_UU_00004/07 and MC_UU_00004/09).
Data availability
This article contains no data.
