Abstract
Introduction:
Clinical trial designs based on the assumption of independent observations are well established. Clustered clinical trial designs, where all observational units belong to a cluster and outcomes within clusters are expected to be correlated, have also received considerable attention. However, many clinical trials involve partially clustered data, where only some observational units belong to a cluster. Examples of such trials occur in neonatology, where participants include infants from both singleton and multiple births, and ophthalmology, where one or two eyes per participant may need treatment. Partial clustering can also arise in trials of group-based treatments (e.g. group education or counselling sessions) or treatments administered individually by a discrete number of health care professionals (e.g. surgeons or physical therapists), when this is compared to an unclustered control arm. Trials involving partially clustered data have received limited attention in the literature and the current lack of standardised terminology may be hampering the development and dissemination of methods for designing and analysing these trials.
Methods and examples:
In this article, we present an overarching definition of partially clustered trials, bringing together several existing trial designs including those for group-based treatments, clustering due to facilitator effects and the re-randomisation design. We define and describe four types of partially clustered trial designs, characterised by whether the clustering occurs pre-randomisation or post-randomisation and, in the case of pre-randomisation clustering, by the method of randomisation that is used for the clustered observations (individual randomisation, cluster randomisation or balanced randomisation within clusters). Real life examples are provided to highlight the occurrence of partially clustered trials across a variety of fields. To assess how partially clustered trials are currently reported, we review published reports of partially clustered trials.
Discussion:
Our findings demonstrate that the description of these trials is often incomplete and the terminology used to describe the trial designs is inconsistent, restricting the ability to identify these trials in the literature. By adopting the definitions and terminology presented in this article, the reporting of partially clustered trials can be substantially improved, and we present several recommendations for reporting these trial designs in practice. Greater awareness of partially clustered trials will facilitate more methodological research into their design and analysis, ultimately improving the quality of these trials.
Keywords
Introduction
Statistical methods for clinical trials are frequently based on an assumption of independence, such that each participant contributes a single outcome and the outcome of one participant is unrelated to the outcome of another. The independence assumption is violated in trials that involve some form of clustering, such as when an intervention is delivered to groups of participants. When clustering is present, outcomes measured on participants from the same group or cluster are expected to be similar or correlated. 1 This correlation may be due to existing similarities among individuals, for example, due to being located in the same geographical region or belonging to the same family. Alternatively, the correlation may be induced by the experimental design, for example, when otherwise independent individuals are allocated to receive treatment from the same clinician. Ignoring the clustering in such a trial can lead to it being over or under powered 2 and incorrect type I error rates, 3 and may therefore result in misleading conclusions.
There are many randomised trial designs that result in clustered data, broadly referred to hereafter as clustered trials. Common clustered trial designs are well described in the literature and include individually randomised cross-over trials (where multiple measurements are taken on each participant under different treatment conditions) 4 , parallel cluster randomised trials (where entire clusters are randomised, so that all participants within a cluster are allocated to the same treatment condition) 5 and longitudinal variants such as stepped wedge trials and cluster randomised cross-over trials.6,7 Design and analysis approaches for clustered trials have been developed that account for the resulting non-independent data.7–12 Reporting guidelines have also been developed that provide a standardised framework for the description and reporting of such trials.13–17
While fully clustered trials where each individual or observation is included in a cluster have been extensively studied, limited attention has been given to partially clustered trials, where only some observations belong to a cluster. We define a partially clustered trial as any trial in which, by design, some observations are independent from all others, while other observations belong to a cluster. These trials occur across a range of health areas including mental health (where intervention arm participants receive treatment in group sessions, compared to an ungrouped control arm),18,19 neonatology (where participants include infants from both singleton and multiple births) 20 and ophthalmology (where one or two eyes per participant may need treatment).21,22 Allowing inclusion of both independent and clustered observations can be advantageous. By widening the eligible population, a partially clustered trial may be more generalisable and result in faster recruitment, shorter trial duration and lower costs than other trial designs. Under certain scenarios, partially clustered trials can also have greater statistical power than a fully independent design. 23
Although partially clustered trials arise in many settings, the lack of standardised terminology to refer to these trials may be hampering the development and dissemination of methods for their design and analysis. The aims of this article are to highlight the occurrence of partially clustered trials in practice, describe the different types of partially clustered trial designs and propose a standardised approach for describing these trials in the scientific literature. Our overall goal is to bring together the somewhat disparate research that has been presented for specific types of partially clustered trials or in specific health fields under the general umbrella of partially clustered trials.
Methods and examples
Partially clustered trials: a broad definition
We define an observational unit as the unit on which the primary outcome is measured, contributing one observation, and a cluster as a group of multiple related observational units. In some settings, people are observational units and clusters consist of multiple people (e.g. clusters of patients who are treated by the same physician). In other settings, people are clusters and contribute multiple observations (e.g. outcomes measured on two eyes from the same person). Fully independent data are defined as each observation being independent from all others, and fully clustered data as each observation belonging to a cluster of size greater than or equal to two, where outcomes of observational units from the same cluster are correlated. Partially clustered data are then defined as a mixture of independent and clustered observations, which may fall anywhere on a clustering continuum from fully independent data at one extreme and fully clustered data at the other extreme (Figure 1).
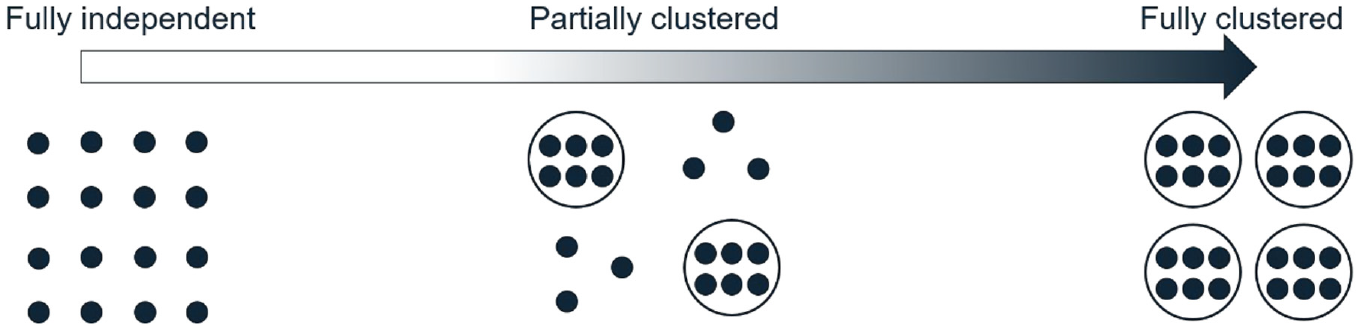
The clustering continuum from fully independent to fully clustered data. Each dot represents an observation. Dots grouped together in a circle represent a cluster of correlated observations. Ungrouped dots represent the independent observations.
Following on from these definitions, we broadly define a partially clustered trial as any trial that is designed to include at least one independent observation and one cluster of observations (Table 1). Our definition of partially clustered trials does not include trials that are intended to be fully clustered but end up including some independent observations due to missing data or intermediate events occurring to other cluster members. We restrict attention to designs with only one level of clustering and a single randomisation of observational units or clusters to treatments, since multiple levels of clustering or randomisation introduce additional complexities that are beyond the scope of this article. However, we recognise that such designs may be affected by partial clustering at one or more levels.24,25 Under our definition, partially clustered trials can be seen as an umbrella term used to encompass a wide range of specific designs, as described below.
A summary of terms.
Types of partially clustered trial designs
The specific type of partially clustered trial is determined by two factors. First, the clusters may exist before randomisation (e.g. body parts of a person) or may be induced only after participants have been randomised (e.g. by assigning people in the same treatment arm to group-based education classes). In fully clustered trials, this has been referred to as pre-randomisation and post-randomisation clustering, respectively, and we adopt these terms throughout, 26 although it is sometimes referred to as ‘recruitment-related’ and ‘treatment-related’ clustering, 27 or ‘natural’ and ‘imposed’ clustering, respectively. 28 Second, the method of randomisation for the clustered observations can vary when clusters exist prior to randomisation. For example, two eyes from the same person may be randomised independently (using individual randomisation of each observational unit), assigned to the same treatment arm (using cluster randomisation) or placed in different treatment arms (using balanced randomisation of observational units within clusters). Pre-randomisation clusters are typically small in many fields (e.g. two eyes in ophthalmology), but can be much larger (e.g. up to 19 warts per patient in a dermatology trial 29 ).
These two factors lead to four types of partially clustered designs: one design where clusters do not exist before randomisation and hence randomisation must be performed at the observational unit level, and three designs for clusters that exist before randomisation that differ based on the randomisation method used within clusters. These four designs are described in detail below and summarised in Table 2. Additional examples of each design type are provided in the Supplemental Material (Appendix 1).
Design features of different kinds of partially clustered trials.
Individual randomisation with post-randomisation clustering
Partial clustering can occur in a trial when cluster membership is established after individual randomisation (i.e. independent randomisation of each observational unit) to treatment arms, and clustering occurs in some, but not all, treatment arms (Figure 2(a)). In this design, clustering is induced by the implementation of the intervention, and we refer to this design as a partially clustered trial using individual randomisation with post-randomisation clustering. An example of this design is the widely discussed group-based treatment design for the assessment of interventions administered at the cluster level, such as education classes, group counselling sessions or group physical activity sessions, compared to an unclustered control arm.30,31 The intervention arm consists of fully clustered observations, while observations in the control arm are fully independent. Post-randomisation clustering can also arise from the interventions that are administered individually, but by a discrete number of health practitioners such as surgeons, counsellors and physical therapists.32,33 This clustering has been termed the ‘therapist effect’ or ‘facilitator effect’ 30 and typically results in fully clustered observations nested within the intervention arm, although partial clustering within this arm is possible if some facilitators administer treatment to only one participant, and designs where clusters are crossed with treatment are also possible. 24 The term ‘individually randomised group treatment’ design was also proposed for trials of group-based treatments, including both fully and partially clustered designs, 34 although it is now used more widely for both group-based treatment and facilitator effect designs. 35 Within some fields, the term ‘partial clustering’ or ‘partial nesting’ is used to exclusively refer to trials with individual randomisation and post-randomisation partial clustering;24,36 however, we consider this design to be just one of four possible partially clustered trial designs that fall under our broader definition.
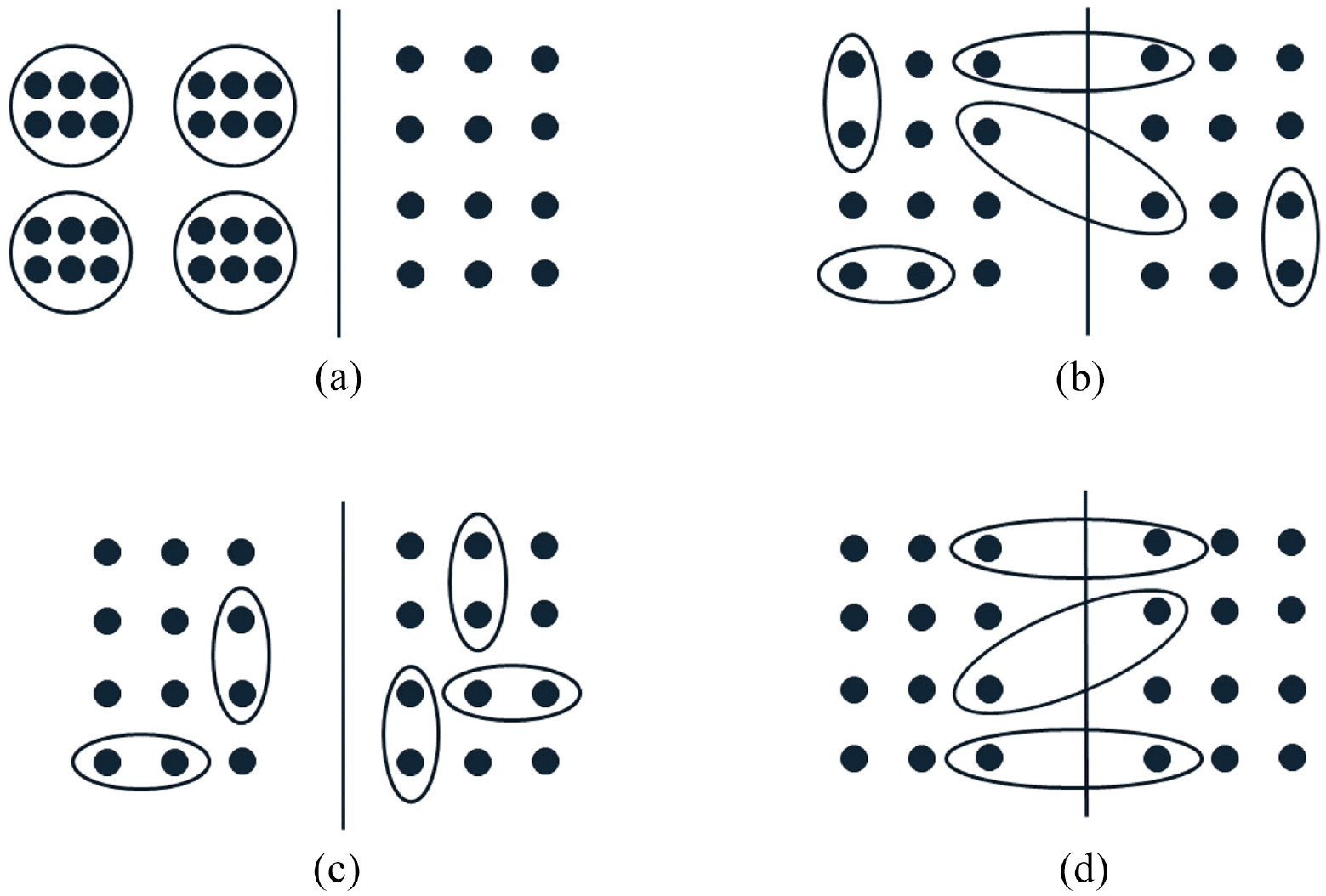
Graphical representations of different types of partially clustered trials. Each panel represents a two-arm trial with the left and right sides of the solid line depicting the two treatment arms. Each dot represents an observation. Dots grouped together in a circle/ellipse represent a cluster of correlated observations. Ungrouped dots represent the independent observations. (a) Individual randomisation with post-randomisation clustering. (b) Individual randomisation within pre-existing clusters. (c) Cluster randomisation of pre-existing clusters. (d) Balanced randomisation within pre-existing clusters.
Example trial
The Scleroderma Patient-centred Intervention Network COVID-19 Home-isolation Activities Together (SPIN-CHAT) Program is a support programme for people with an existing medical condition during COVID-19. The impact of the programme on anxiety symptoms after the 4-week intervention period was assessed in a partially clustered trial. 18 Eligible participants were individually randomised to the intervention or a waitlist control arm. The intervention was delivered virtually to clusters of participants, whereas the waitlist control arm received no contact during the intervention period resulting in a group-based treatment design. The trial randomly assigned 172 participants to intervention or control (n = 86 in each arm), and intervention participants were allocated to 1 of 11 clusters of 6–10 participants for delivery of the intervention.
Individual randomisation within pre-existing clusters
If clusters exist before randomisation (i.e. pre-randomisation clustering) and observational units are independently assigned to treatment arms using individual randomisation, all members within some clusters may be randomised to the same treatment arm, while other clusters may be split across treatment arms (Figure 2(b)). We refer to this design as a partially clustered trial with individual randomisation within pre-existing clusters. Since observational units within the same cluster may be allocated to different treatment arms, the intervention must be administered at the observational unit level. Trials utilising individual randomisation of pre-existing clusters have been used in various fields, including neonatal health, 37 antenatal health, 38 fertility treatment 39 and chronic liver failure. 40 The re-randomisation design is a specific example of this type of design, where participants may enrol into the trial on multiple occasions and are randomised independently at each enrolment. 41 Re-randomisation designs can increase the recruitment rate and decrease time to trial completion. 42
Example trial
The Omega-3 (N-3) Fatty Acids for Improvement in Respiratory Outcomes (N3RO) trial was a randomised controlled trial involving preterm infants who were randomised to either an omega-3 or control emulsion that was administered enterally. 37 The primary outcome was the incidence of physiological bronchopulmonary dysplasia measured at 36 weeks postmenstrual age or discharge home, whichever occurred first. Infants from single or multiple births were eligible for the trial and infants from the same birth were randomised independently, resulting in a partially clustered design with individual randomisation within pre-existing clusters (families). A total of 1273 infants from 1098 families were included in the trial.
Cluster randomisation of pre-existing clusters
If cluster-level randomisation is used with pre-existing, pre-randomisation, clusters, the result is a partially clustered trial where clusters may occur in all treatment arms and all members of the same cluster will be allocated to the same treatment arm. Non-clustered observational units are randomised independently, leading to each treatment arm containing a mix of independent and clustered observations (Figure 2(c)). We refer to this design as a partially clustered trial with cluster randomisation of pre-existing clusters. Under this design, the intervention may be administered at either the cluster level or observational unit level. Cluster randomisation is the only choice for treatments administered at the cluster level (e.g. a systemic medication that affects both eyes in an ophthalmology trial). Partially clustered trials with cluster randomisation of pre-existing clusters have been reported in fields including pregnancy and neonatal health,43–49 sports medicine, 50 ophthalmology 51 and surgery. 52
Example trial
A surgical trial compared two mesh prosthetics placed during minimally invasive inguinal hernia repair. 52 Patients were randomised to surgery with either a flat lightweight polypropylene mesh or a preformed lightweight mesh. Patients with both unilateral and bilateral inguinal hernias were eligible, and patients with bilateral hernias received the same mesh treatment for both procedures, resulting in a partially clustered trial with cluster randomisation of pre-existing clusters (patients). The primary outcome was the time for the mesh placement during surgery, measured on each hernia. Forty-eight patients were enrolled for a total of 65 hernia repairs.
Balanced randomisation within pre-existing clusters
When clusters exist pre-randomisation, a trial may be designed to balance treatment allocations across observational units within clusters. For example, if clusters have a maximum size of two and there are two treatment arms, one cluster member is randomised to a treatment arm and the other cluster member is then automatically assigned to the other treatment arm. Each cluster therefore receives both treatments (Figure 2(d)) as occurs in matched-pair designs (e.g. crossover trials) in the fully clustered setting. We refer to this design as a partially clustered trial with balanced randomisation within pre-existing clusters. The intervention must be administered at the observational unit level under this design. Balanced randomisation is most commonly seen in settings involving paired body parts (e.g. eyes, joints) where clusters naturally have a maximum size of two,53–56 although it can also be used with larger cluster sizes or more than two treatment arms. 57
Example trial
An ophthalmology trial evaluated the effectiveness of ciliary neurotrophic factor (CNTF) for the treatment of macular telangiectasia type 2, a degenerative disease of the retina. 53 Participants were eligible if one or both eyes met the inclusion criteria, leading to a partially clustered trial. The intervention was administered via a surgical implant, or a sham surgery if in the control group, into the study eye. The primary outcome was the change in the area of the ellipsoid zone disruption of the study eye at 24 months. For participants with both eyes eligible, the right eye was randomised to CNTF or sham surgery, and the left eye was allocated to the alternate treatment, resulting in balanced treatment allocations within clusters. A total of 99 eyes from 67 participants (clusters) were randomised into the study.
Choice of partially clustered trial design
Some characteristics of a partially clustered trial will be determined by the population under study, the type of intervention, and logistical issues around the recruitment of participants and the delivery of the intervention. If the intervention is delivered in groups formed after randomisation or by one of several facilitators and an ungrouped comparator arm is to be used, then researchers will be implementing a partially clustered trial using individual randomisation with post-randomisation clustering. When clusters exist before randomisation, researchers may be able to choose the randomisation method for the clustered observations (cluster, individual or balanced), and this will dictate which partially clustered trial design is used. Individual randomisation can be used when the treatment is assigned at the observational unit level, and there is minimal risk of contamination of the treatment between cluster members. Cluster randomisation may be preferred due to logistical reasons, 8 when the treatment of one cluster member has the potential to influence other cluster members, 58 or participant preferences, especially when clusters are twins. 59 Balanced randomisation within clusters may have ethical advantages in trials where clusters are participants, as it ensures each participant will be exposed to the novel or gold standard treatment, 60 and can result in faster recruitment and lower trial costs. 61 The method of randomisation within pre-existing clusters can impact the statistical power and target sample size of a trial, with balanced randomisation providing the greatest power. 23 The feasibility, pros and cons of using each method of randomisation for pre-existing clusters should be carefully considered in the context of the individual trial before determining which type of partially clustered design to use.
Problems identifying partially clustered trials
There are currently no reporting guidelines specifically for partially clustered trials. With no consistent terminology in place, it is difficult to identify reports of partially clustered trials and hence conduct a systematic review of current reporting practices to inform recommendations for describing partially clustered trial designs. Instead, we identified a convenience sample of protocols and reports of partially clustered trials and examined how the designs were reported to provide some initial insight. Reports were identified from citations of the small number of existing methodological papers on partially clustered trials, a literature search for ‘partial clustering’ or ‘partial nesting’, and by ad hoc inspection of recent issues of clinical trial and medical journals. Given the way these trials were identified, they are not expected to be representative of all published partially clustered trials and may overestimate the quality of reporting, as they likely represent some of the more easily identified partially clustered trials.
Our convenience sample included 40 articles (10 protocols and 30 results papers) covering all four types of partially clustered trials across a range of research areas, including education, psychology and medicine (Table 3). While most trials utilising individual randomisation with post-randomisation partial clustering used the terms ‘partially clustered’ or ‘partially nested’ within the publication (7/11), these terms were not used to describe trials of any other design type. Eleven re-randomisation trials were identified, only one of which was described as using a ‘re-randomisation design’. 39 The use of re-randomisation in the other examples could only be inferred from the description of the design, 62 patient population, 63 eligibility criteria, 64 or the randomisation process 65 or results. 66 For the other 18 partially clustered trials involving randomisation of pre-existing clusters but not using a re-randomisation design, the presence of clustering was not included in the primary description of the study design, even when other important design features were explicitly included (e.g. multicentre, double-blind, randomised controlled trial). Importantly, there was no consistency on where or how the partial clustering was described. Instead, the relevant information was variously included in the description of the study design, 57 inclusion criteria, 45 or the randomisation43,53 or treatment allocation 56 processes. Overall, these findings suggest that current practices for describing partially clustered trial designs are inadequate.
Reporting practices in reports of 40 partially clustered trials.
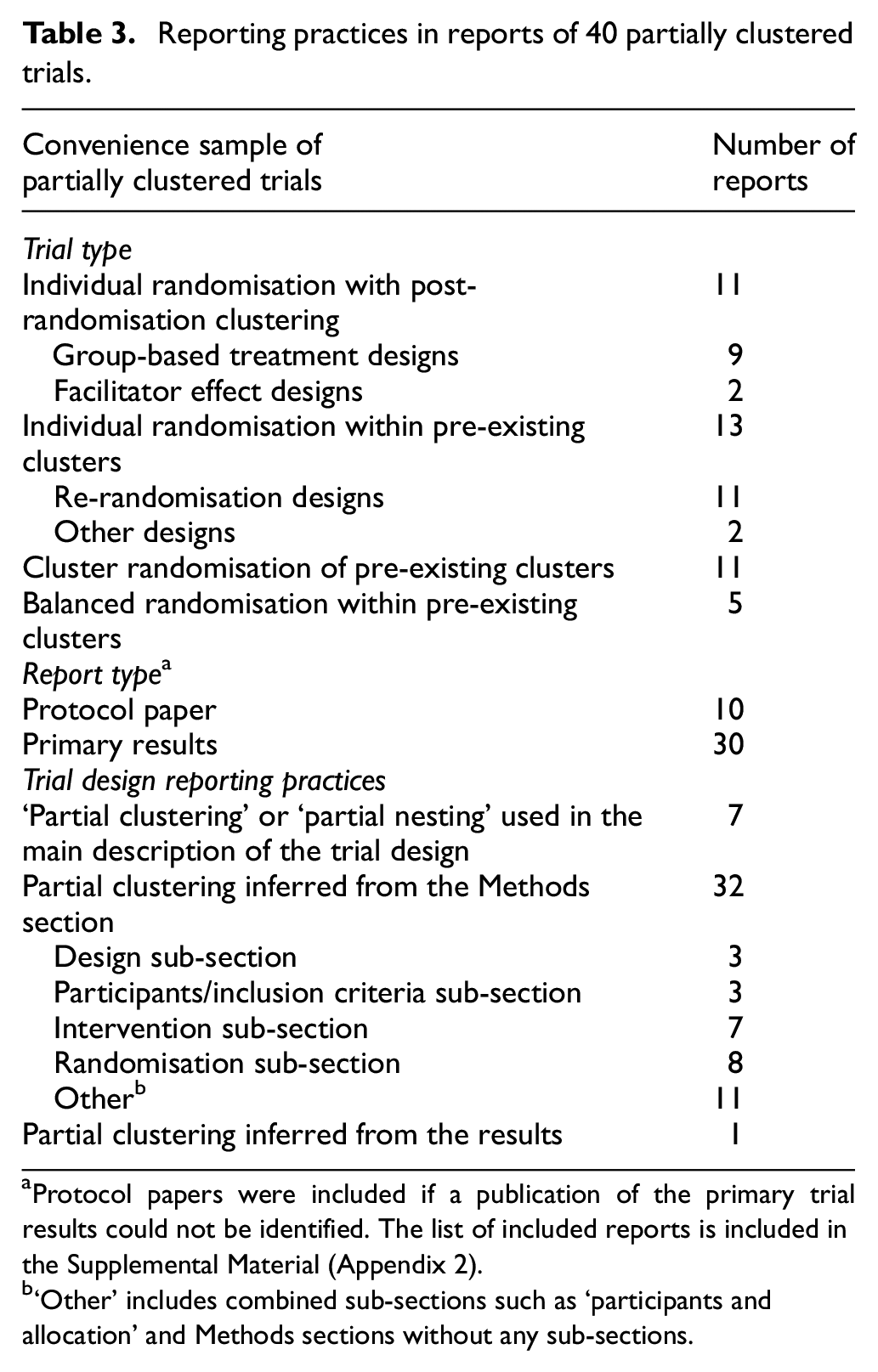
Protocol papers were included if a publication of the primary trial results could not be identified. The list of included reports is included in the Supplemental Material (Appendix 2).
‘Other’ includes combined sub-sections such as ‘participants and allocation’ and Methods sections without any sub-sections.
Recommendations for describing partially clustered trial designs
To facilitate the identification of partially clustered trials in the literature, clear reporting of the trial design is required. Reporting guidelines acknowledge the importance of clearly identifying the specific trial design in reports of protocols, 67 trial results13–17,68,69 and in abstracts. 70 We therefore recommend that the type of partially clustered trial should be described in reports of such trials using the terminology and design types described in this article (or other established terms, where applicable). While current guidelines recommend identifying the trial design in the title, the length of the partially clustered trial descriptions and the limited number of clusters in some settings suggest that it may be more useful to: (a) describe a partially clustered trial as a ‘randomised trial’ in the title, (b) describe the trial as ‘partially clustered’ or involving ‘partial clustering’, in both the abstract and methods and (c) describe the specific type of partially clustered design clearly in the methods, such that the method of randomisation and what constitutes the cluster-level and observation-level units are clear. Addressing this final recommendation may involve using the terminology for the trial designs introduced in this article, established terms for specific designs (such as re-randomisation or individually randomised group treatment designs) or language appropriate for the context of the particular trial. Examples of adequately described design descriptions for each type of partially clustered trial are provided in Table 4. Such detail provides transparency and context for interpreting trial details such as the level of treatment administration, the method of randomisation, sample sizes at the observation and cluster level and the statistical analysis methods.
Examples of adequately detailed descriptions of partially clustered trial designs for published trial reports.
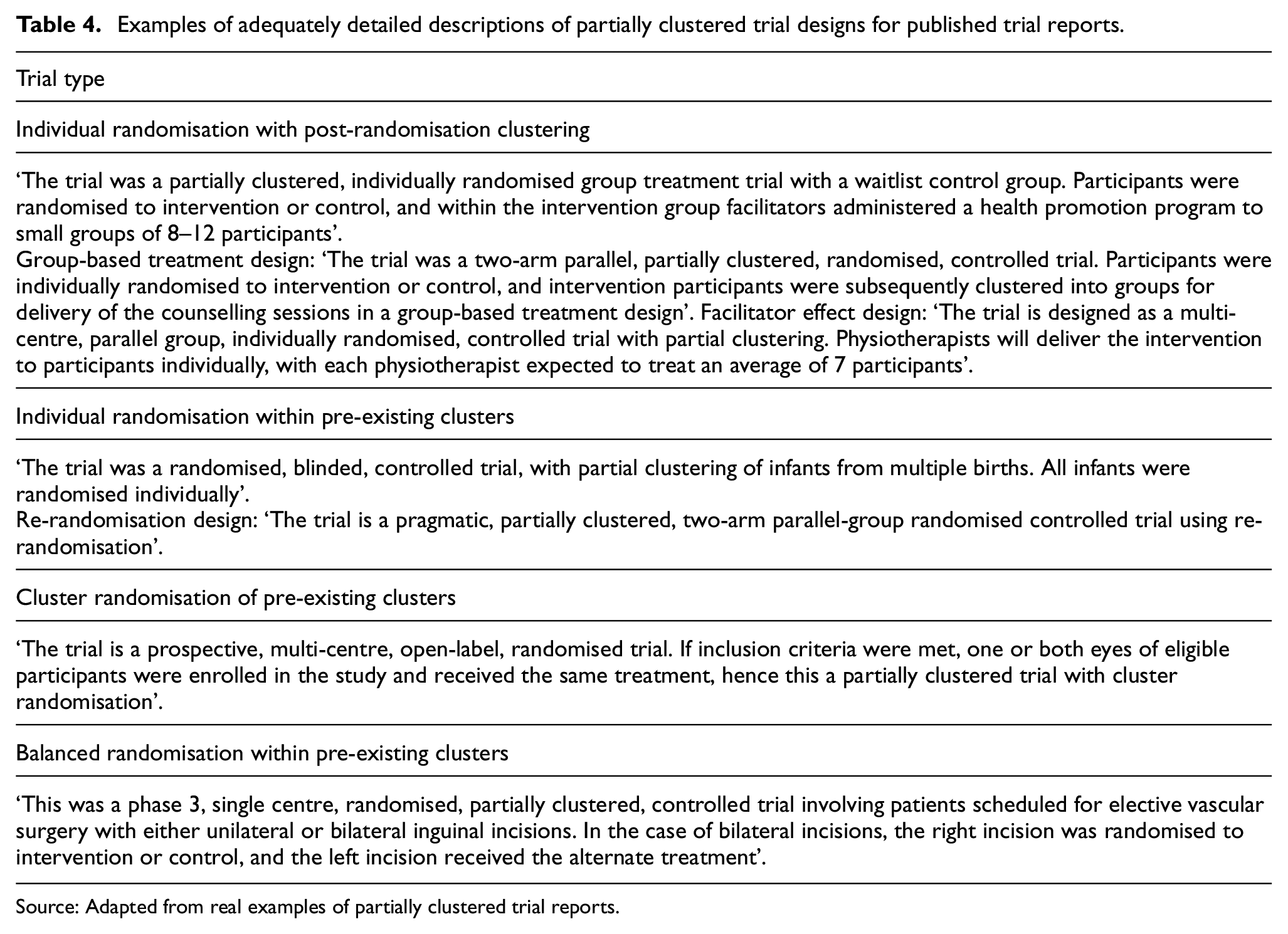
Source: Adapted from real examples of partially clustered trial reports.
Discussion
We have provided a unifying, broad definition of partially clustered trials and proposed standardised terminology for describing different partially clustered trial designs. Currently, there is a lack of consistent terminology used to describe partially clustered trials, which makes it difficult to systematically search the literature to identify published examples of such trials. Therefore, little is known about how they are designed, described and analysed in practice, including whether the clustering has been appropriately accounted for. Previous reviews on the use of partially clustered trials have been conducted within individual fields by searching complete issues of specialist journals.21,71,72 A similar review could be conducted in general medical journals, potentially identifying other applied fields where these trials are used, and this is a suggested area for future research. While the framework presented here is restricted to simple designs that involve a single level of clustering and one stage of randomisation, more complex designs involving partial clustering (including partial crossing of treatments and facilitators, 24 and multitier designs that include more than one stage of random allocation24,25) are possible and our work could be extended to these scenarios in the future.
Limited research has been conducted to date on the design and analysis of partially clustered trials. Most methodological literature relates to partially clustered trials using individual randomisation with post-randomisation clustering.30,36,73 The statistical issues that arise in partially clustered trials involving pre-existing clusters have been discussed in specialty fields including ophthalmology, 21 orthopaedics71,72 and surgery, 74 but without the use of a specific term for this design. Recent methodological work has been conducted on sample size 23 and multiple imputation 75 for trials involving a combination of independent and paired data, but without reference to partial clustering. Sample size methods exist for some types of partially clustered trials.23,76,77 Research has been conducted on the analysis of partially clustered trials that arise in neonatology due to multiple births78–81 and of re-randomisation designs;41,82 however, reviews in specific subject-matter areas have found that many partially clustered trials are analysed using potentially inappropriate methods,71,72,83,84 suggesting that the importance of clustering in these designs is under-recognised. Appropriate analysis of partially clustered data is important to ensure correct estimates of variability and type I error rates, 3 and partial clustering should be accounted for when determining sample size to avoid trials being over or under powered. 2 Further development of sample size methods, assessment of appropriate analysis methods and analysis recommendations that are accessible to applied researchers are needed for the full range of partially clustered trial designs. While each design could be considered in isolation, we believe there is benefit in considering similarities across designs and how methods developed for one partially clustered trial design could be applied to or modified to suit other designs. Such development should be conducted with consideration of the estimand framework to ensure an appropriate definition of the desired treatment effect. 85 Estimands have recently been defined for fully clustered designs86–88 and re-randomisation designs, 89 and extensions to partially clustered designs more generally are a logical next step. We encourage researchers to follow the recommendations provided in this article when referring to different partially clustered designs in future methodological work to aid the dissemination of relevant methodological advances across applied areas.
Poor and inconsistent reporting of trial results, including unclear randomisation methods, is associated with bias in reported treatment effects.90,91 This is of particular relevance to partially clustered trials, where there are multiple methods available for randomisation of observational units within pre-existing clusters, and this may not always be clearly described. Critically assessing the quality and validity of trial results requires complete reporting of the trial design and implementation. Development of detailed reporting guidelines for partially clustered trials is thus an important area for future research. In the absence of specific guidelines, we encourage researchers and journals to refer to the relevant recommendations for fully clustered trials, 13 non-pharmacologic treatments16,17 (in the case of individual randomisation with post-randomisation clustering) and within-person trials 69 (in the case of balanced randomisation within pre-existing clusters), and the specific advice for partially clustered trials that we have presented in this article. However, following all recommendations for fully clustered trials may be challenging for some partially clustered trials, such as reporting an intraclass correlation coefficient for outcomes collected in partially clustered trials with few clusters, and hence some reporting discretion may be necessary based on the specific trial.
In conclusion, partially clustered trials occur across a range of applied areas. The lack of a unified definition of partially clustered trials and consistent terminology for describing the various types of designs limits the ability of researchers to accurately assess their use and develop methods for their design and analysis. By adopting standard definitions and terminology, such as those presented in this article, the reporting of partially clustered trials can be substantially improved. Greater awareness of these trials will facilitate more methodological research into their design and analysis for the benefit of clinical researchers.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745221146987 – Supplemental material for Partially clustered designs for clinical trials: Unifying existing designs using consistent terminology
Supplemental material, sj-docx-1-ctj-10.1177_17407745221146987 for Partially clustered designs for clinical trials: Unifying existing designs using consistent terminology by Kylie M Lange, Jessica Kasza, Thomas R Sullivan and Lisa N Yelland in Clinical Trials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: K.M.L. is supported through an Australian Government Training Programme (RTP) Scholarship administered by The University of Adelaide, Australia, an Australian Trials Methodology (AusTriM) Research Network supplementary scholarship and a Statistical Society of Australia PhD top-up award. T.R.S. is supported by a National Health and Medical Research Council (NHMRC) emerging leadership fellowship APP1173576. This research was supported by a Centre of Research Excellence grant from the NHMRC, ID #1171422, to the AusTriM Research Network.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
