Abstract
The implementation of an upper-limb rehabilitation simulator replicating patient-specific physical characteristics has garnered increasing interest as an effective alternative to human subjects for medical training and research. However, human upper-limb motion involves complex multi-joint coordination, whereas most existing patient simulators reproduce simplified mechanical properties of individual joints and often fail to mimic critical pathological characteristics, such as rigidity and physiological tremors. This study addresses these limitations by proposing a novel simulator that incorporates both compound joint coordination and patient-specific symptoms (e.g. lead-pipe rigidity and physiological tremors). A motion control system based on a linear quadratic regulator was designed, with patient-specific reference torques derived from adult male motion-capture data. The tremor parameters were selected within the range reported in neurophysiological literature, and rigidity was implemented based on spasticity-inspired torque models. The proposed controller achieved torque-tracking errors below 3%, while reproducing clinically relevant peak torque levels of 18–20 Nm at the shoulder and 3–7 Nm at the elbow under manual muscle testing–grade scaling. The experimental evaluations across rigidity levels highlighted the reliability and effectiveness of the proposed simulator in accurately mimicking patient characteristics, making it a practical alternative for patients in rehabilitation training. Additionally, a clinical similarity survey was conducted with eight rehabilitation specialists and 10 students following a standardized, blinded protocol, yielding a mean rating of
Keywords
Introduction
In medical training, robotic simulators replicating patient-specific physical characteristics have gained significant attention as effective substitutes for patients. Rehabilitation simulators designed to emulate spasticity or joint rigidity observed in hemiparetic patients serve as valuable training tools for rehabilitation therapists. However, most existing systems have focused on passive reproduction of joint resistance or on simplified stiffness without accounting for pathological conditions. Achieving a high degree of realism in these simulators requires advances in robotic system control to mimic patient characteristics accurately. 1
Patient-specific replicas are critical in rehabilitation therapy, where the precise replication of limb movements can substantially enhance treatment efficacy. The ability to reproduce pathological movement patterns, such as the joint rigidity and spasticity commonly observed in hemiplegic patients, is vital for creating an effective training environment. By simulating these motor impairments, rehabilitation therapists can refine therapeutic techniques in a controlled setting that closely mimics real-world clinical conditions.1–4
A gap exists in the realistic replication of simultaneous multi-joint interactions with pathological characteristics (e.g. tremors), which are often experienced in clinical practice. Accurate replication of joint rigidity, physiological tremors, and other abnormal motion patterns offers rehabilitation professionals invaluable opportunities for hands-on practice and clinical assessment. These simulators enable a controlled environment where therapists can hone their skills without risk to the patient. Furthermore, advanced robotic systems that simulate patient-specific impairments provide a platform for standardized and objective clinical training and evaluation (e.g. rigidity and spasticity scoring).1,2
Robotic replicas have increasingly been focused on the demand for systems capable of simulating multi-joint and full-limb motions. Such systems are necessary to represent the coordinated motions of the shoulder, elbow, and wrist accurately, which are vital for comprehensive rehabilitation training. Furthermore, upper-limb patient simulators must incorporate varying resistance, stiffness, and abnormal muscle behavior. Several approaches have been explored to achieve a high-fidelity robotic replica, including integrating magnetorheological dampers, electronic brakes, and tension cables.1–14
Numerous studies have explored upper-limb rehabilitation using robotic replicas that simulate joint behavior via control strategies to assist in clinical training. Takahashi et al. 6 developed an elbow-joint robotic replica incorporating a magnetorheological brake and direct current motor. The system featured a feedback mechanism that modulated the joint resistance torque in real time to emulate rigidity levels during physical therapy exercises. Park et al. 7 introduced a haptic elbow spasticity robotic replica using a cable-driven system and an electronic brake, where a proportional derivative (PD)-based feedback loop controls the cable tension to reproduce clinical spasticity profiles. Zakaria et al. 8 proposed a two-degree-of-freedom (two-DOF) robotic system addressing the shoulder and elbow joints. By applying impedance control, the robot replicated varying joint resistance in response to external manipulation, supporting therapist training. Liang 9 constructed a passive hydraulic robotic replica incorporating a Scotch-yoke structure with adjustable internal damping. In Table 1, previous research on upper-limb rigidity robotic replicas10–14 has rarely explored control strategies, especially in multi-joint rehabilitation robots that incorporate compound multi-joint coordination and simultaneous patient-specific symptoms (e.g. lead-pipe rigidity and physiological tremors).
Robotic replicas replicating joint rigidity in rehabilitation patients.

DOF: degree-of-freedom; PD: proportional derivative.
Integrating joint rigidity and physiological tremors is critical in developing a realistic surrogate for upper-limb rehabilitation patients. Takanokura and Sakamoto 15 analyzed the magnitude of physiological tremors in rigid shoulder, elbow, and wrist joints, and explored the relationship between tremor intensity and joint rigidity. Morrison and Newell 16 investigated the intensity of physiological tremors in static, rigid upper-limb joints. Similarly, Taheri et al. 17 explored tremor characteristics associated with the motion of a rigid wrist joint in a defined range. Despite these contributions to understanding medical symptoms, no existing research has replicated joint rigidity and physiological tremors in robotic simulators designed for rehabilitation applications. This lack of integration limits their clinical utility, especially for training that requires compound symptom expression. To bridge this gap, the present study implements an optimal control scheme for a multi-DOF upper-limb replica that accurately reproduces physiological tremor and joint rigidity, enhancing its applicability to rehabilitation training.
Mathematical model
Figure 1 presents the developed two-DOF upper-limb robotic replica. The variables
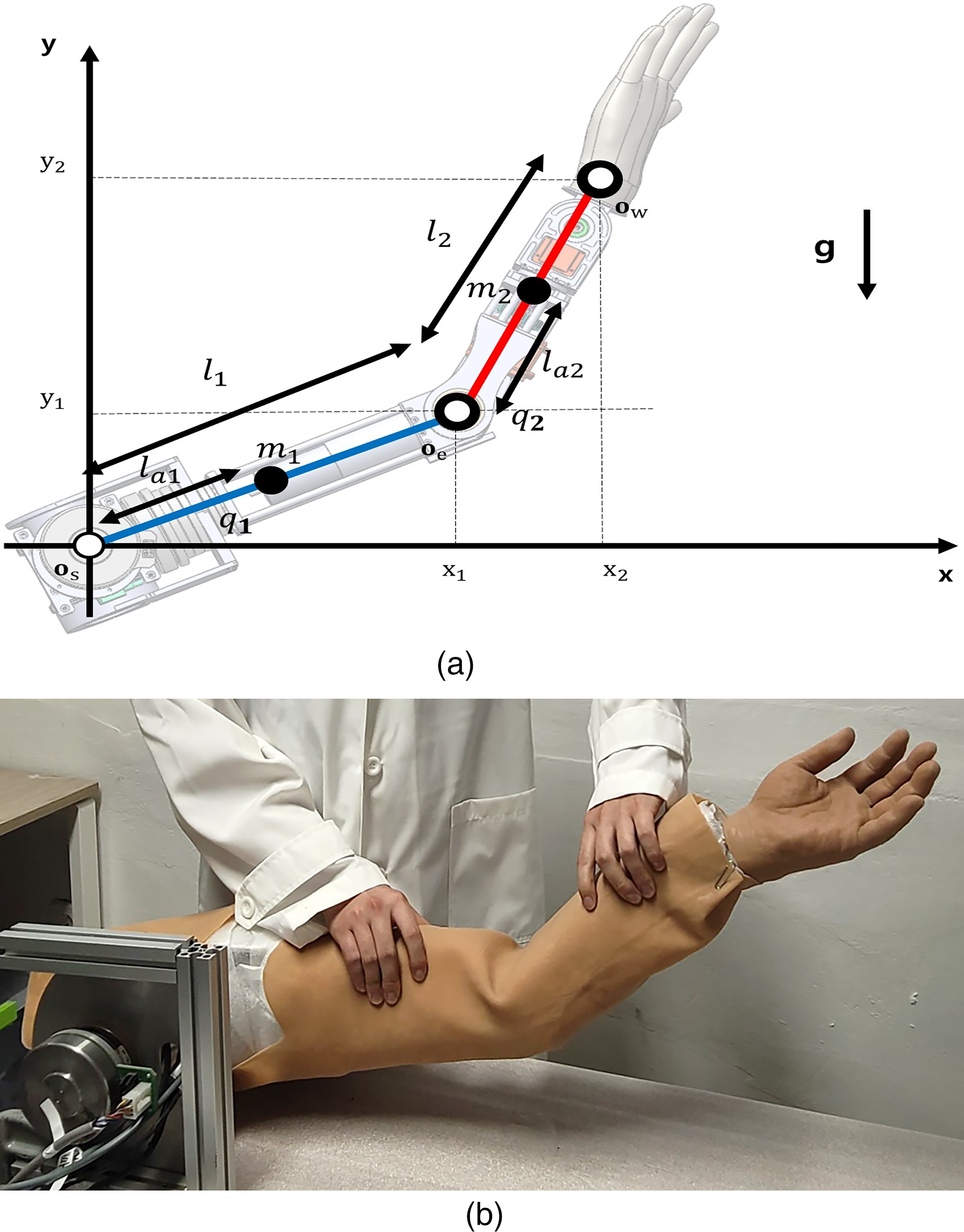
Developed upper-limb robotic replica with two-degree-of-freedom (two-DOF). (a) Mathematical model and (b) real model.
The coordinates of each joint are as follows:
The dynamics of the developed replica are as follows: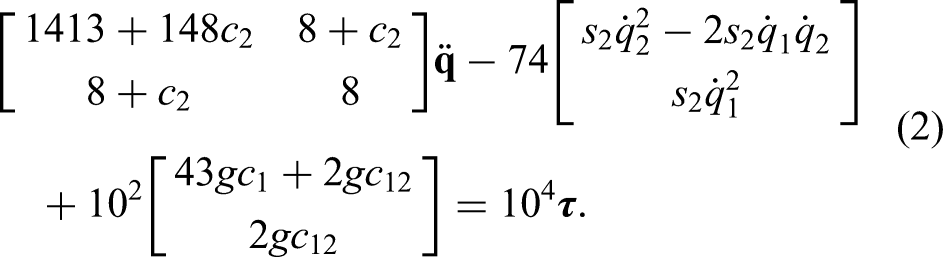
The state space form is as follows:

Motion control
The linear quadratic regulator (LQR) scheme is applied to achieve the desired angles of the elbow and shoulder joints in the presence of multiple inputs. This scheme updates the elasticity and damping parameters in the
From equation (4), the weight matrix of the inputs,
The proposed LQR-based tracking controller was evaluated by reproducing upper-limb joint motion measured during three repeated trials (approximately 30 s each). The reference of the driving torque of upper-limb joints for healthy individuals,
Adjusting
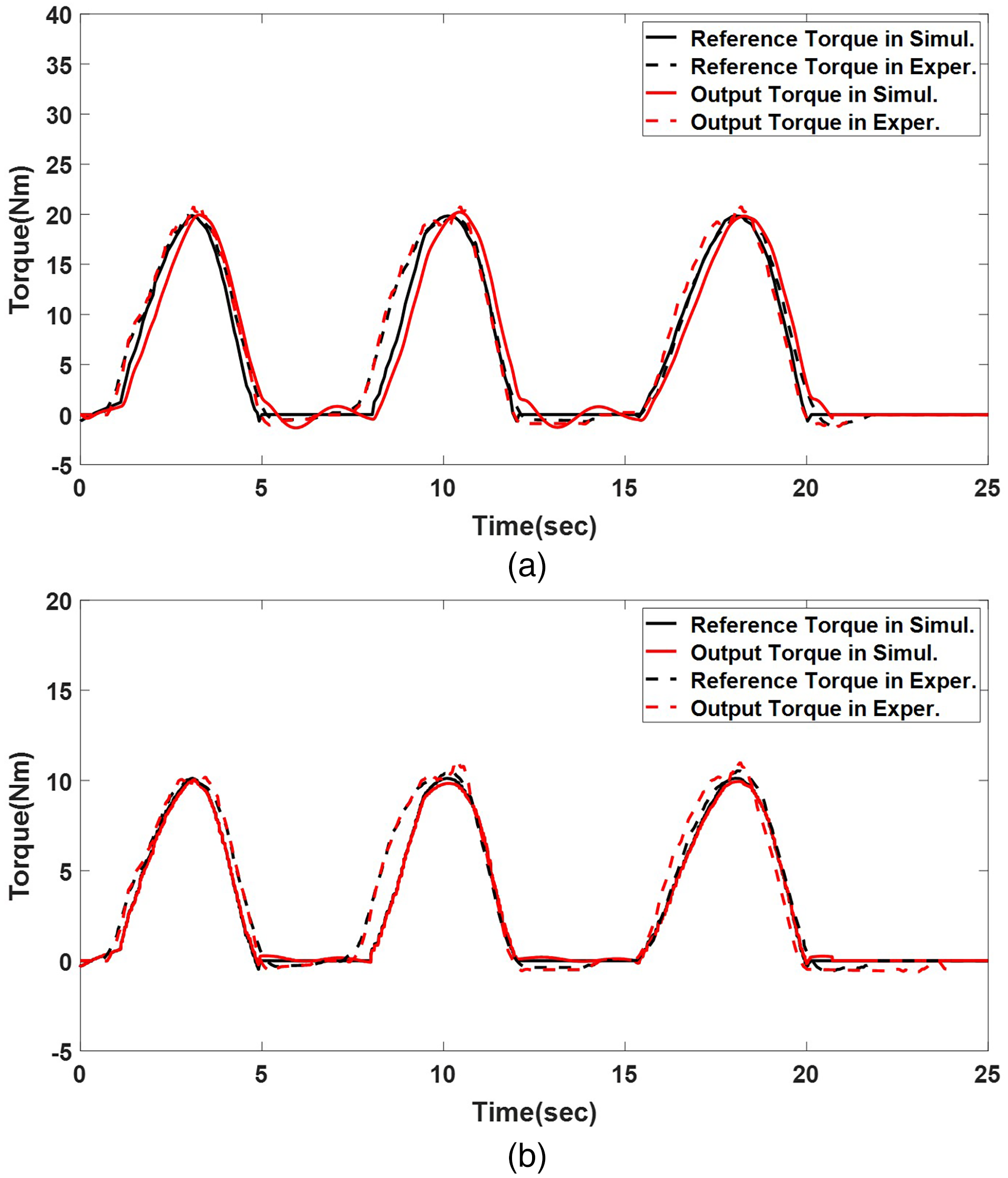
Torque tracking of three-peak motion on the developed upper-limb robot. (a) Shoulder joint and (b) elbow joint.
Proposed control algorithm
Motion capture
This study employs motion capture to collect the trajectories of normal upper-limb motion, which are applied to the upper-limb robotic replica. Typical Asian male participants in their 30s performed a drinking motion using a cup. The starting position is with the shoulder in a neutral position, the elbow at 90
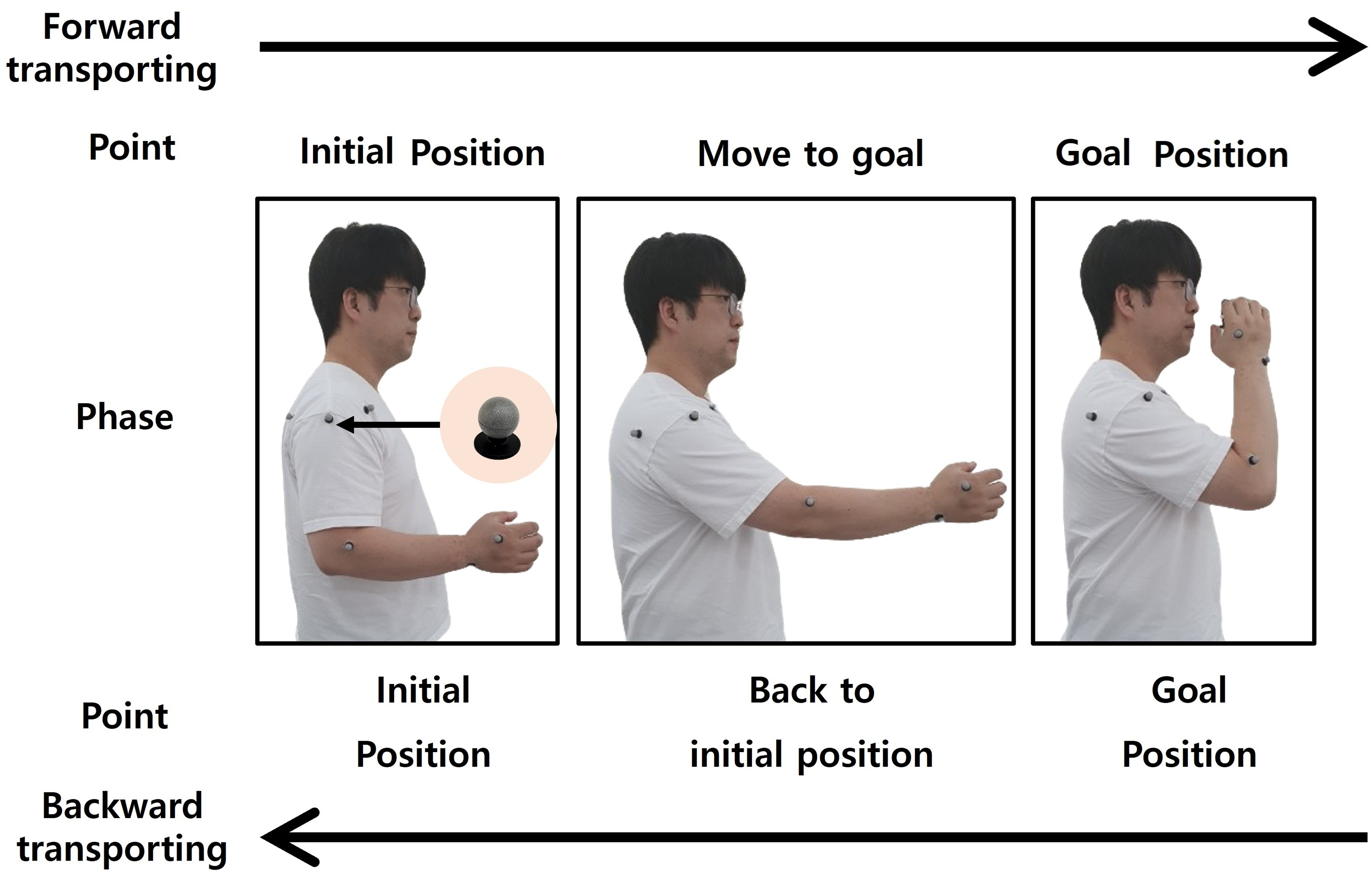
Phase state of upper-limb motion.
Eleven markers were attached to anatomical landmarks according to the ISB guidelines.
22
The three-dimensional trajectories of the markers were recorded using a motion capture system (OptiTrack, Flex13) comprising six cameras calibrated at a sampling frequency of 120 Hz. The occlusion gaps of the frames (
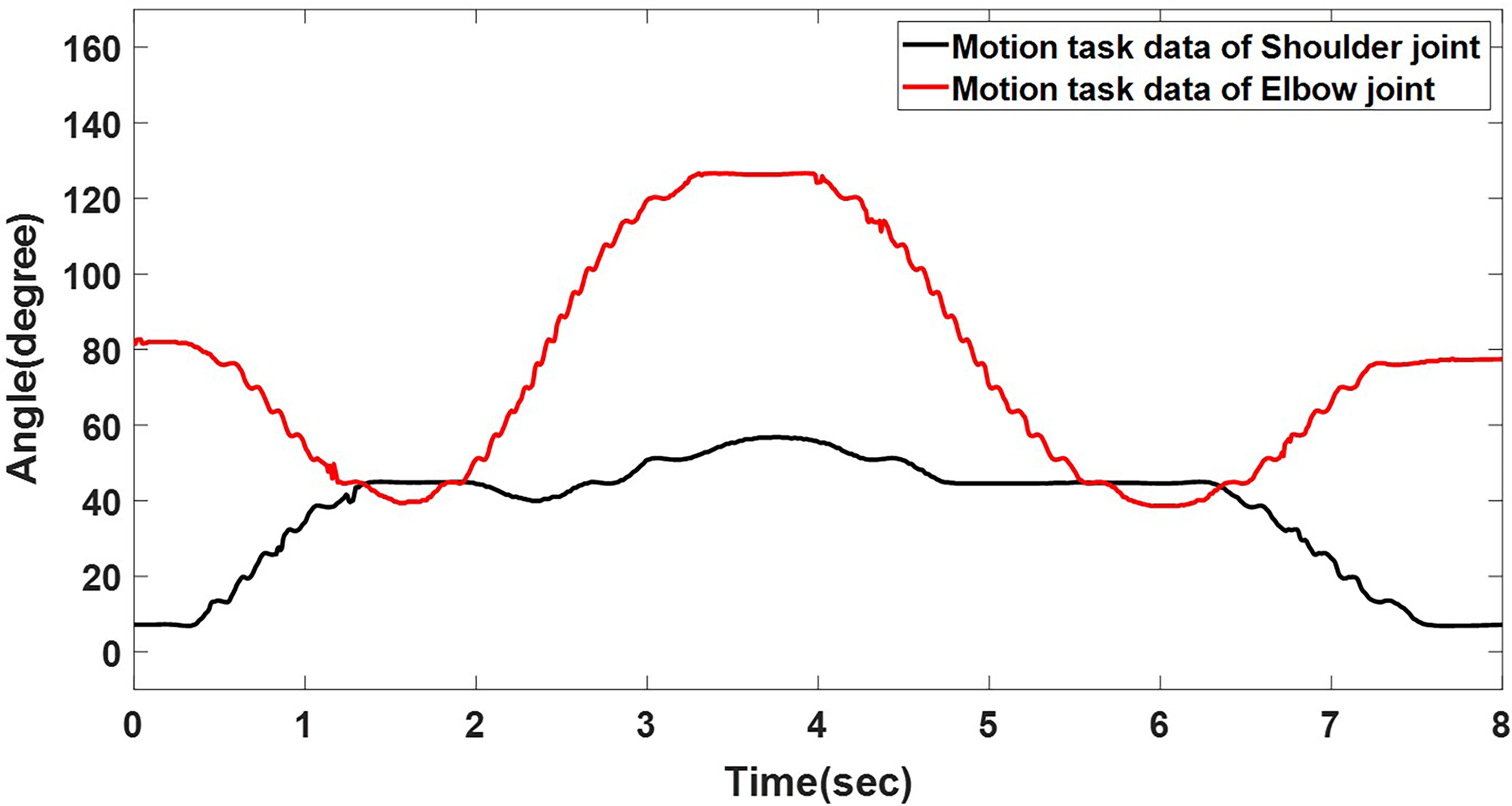
Mean of the joint angle in human upper-limb motion.
The upper-limb kinematic data collected from healthy individuals serve as input for the motion controller, which was adapted to accommodate patient-specific rigidity constraints. Although only data from one subject were used, the recorded joint angles were cross-checked against established physiological ranges reported in biomechanics literature.15,16,23 Therefore, the motion data are regarded as representative of typical upper-limb kinematics of healthy individuals while drinking water.
Human rigidity
The proposed motion controller reproduces joint rigidity observed in hemiplegic patients by constraining the driving torque during the prescribed motion (Figure 5). The desired rigid-joint torque,
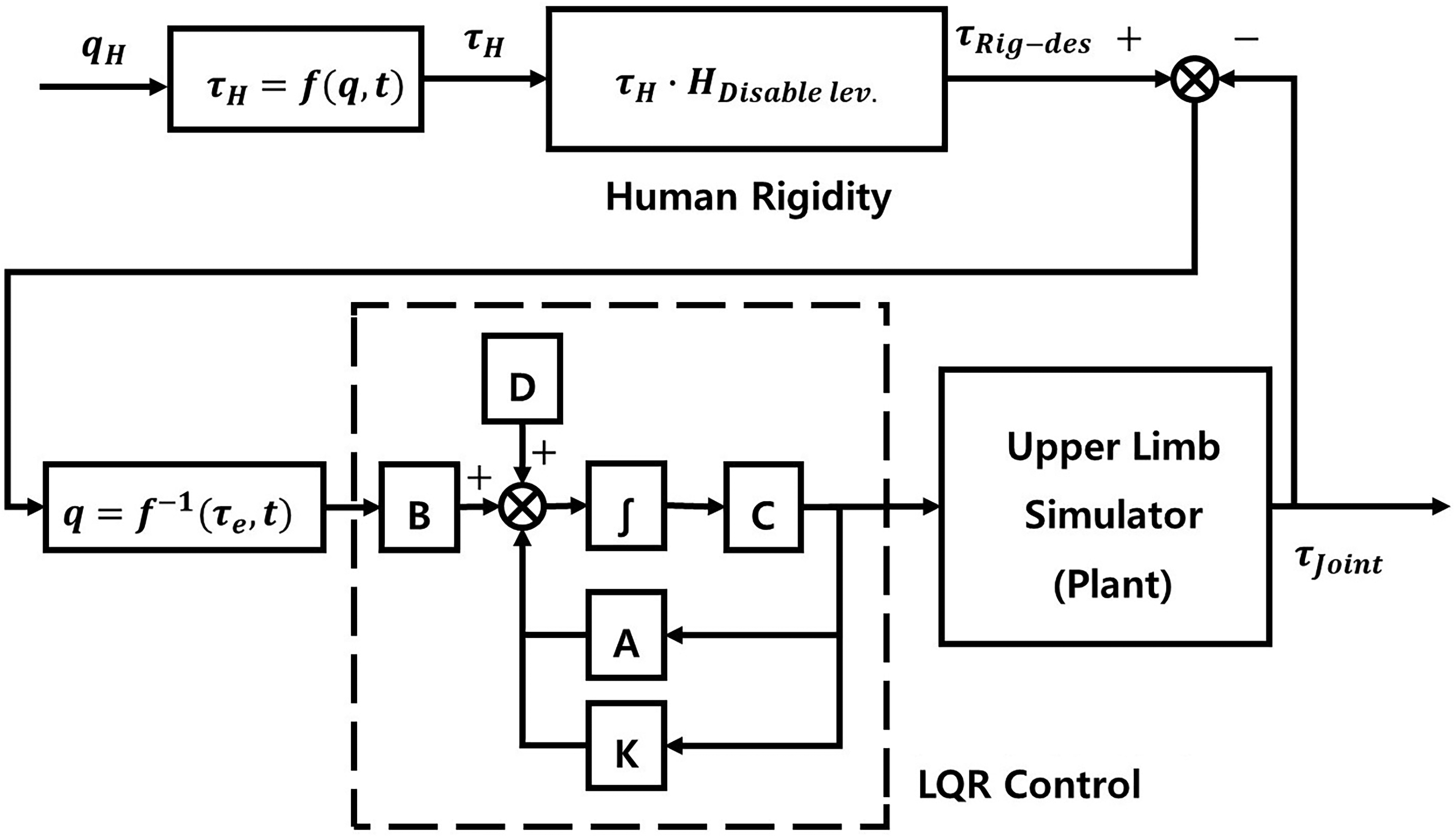
Block diagram of the control scheme on the robotic replica.
In equation (6),
Manual muscle testing grade.

ROM: range of motion.
The state-weighting matrix,
Scaling coefficients reflecting the correlation between MMT (strength) and MAS (tone).
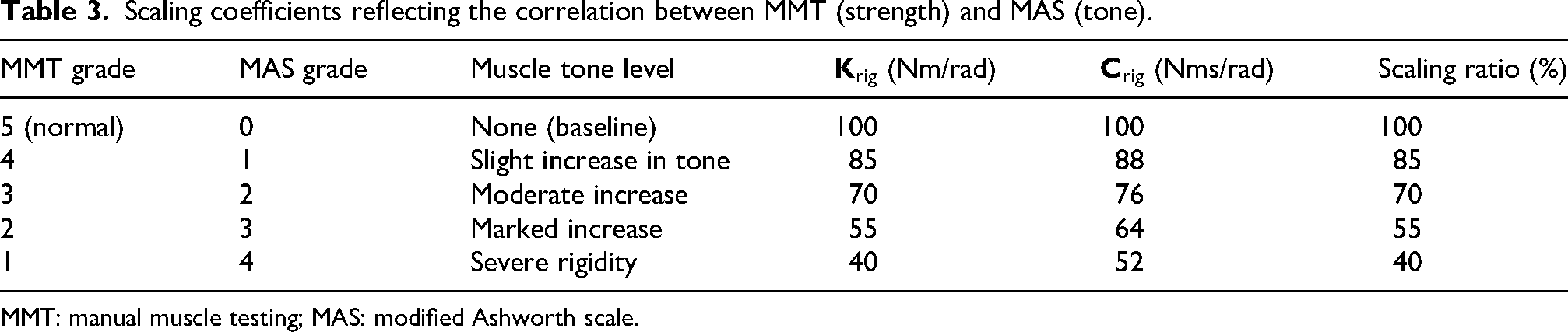
MMT: manual muscle testing; MAS: modified Ashworth scale.
To enhance physiological validity, joint torque values for healthy adults,
Based on the described scaled torque profiles, the effectiveness of the proposed tracking control strategy incorporating joint rigidity was evaluated on a two-DOF robotic replica via simulation and experimental validation. Figure 6(a) and (b) displays the performance of the rigidity-incorporated robotic replica under the proposed control scheme, compared with that for conventional proportional-integral-derivative (PID) and
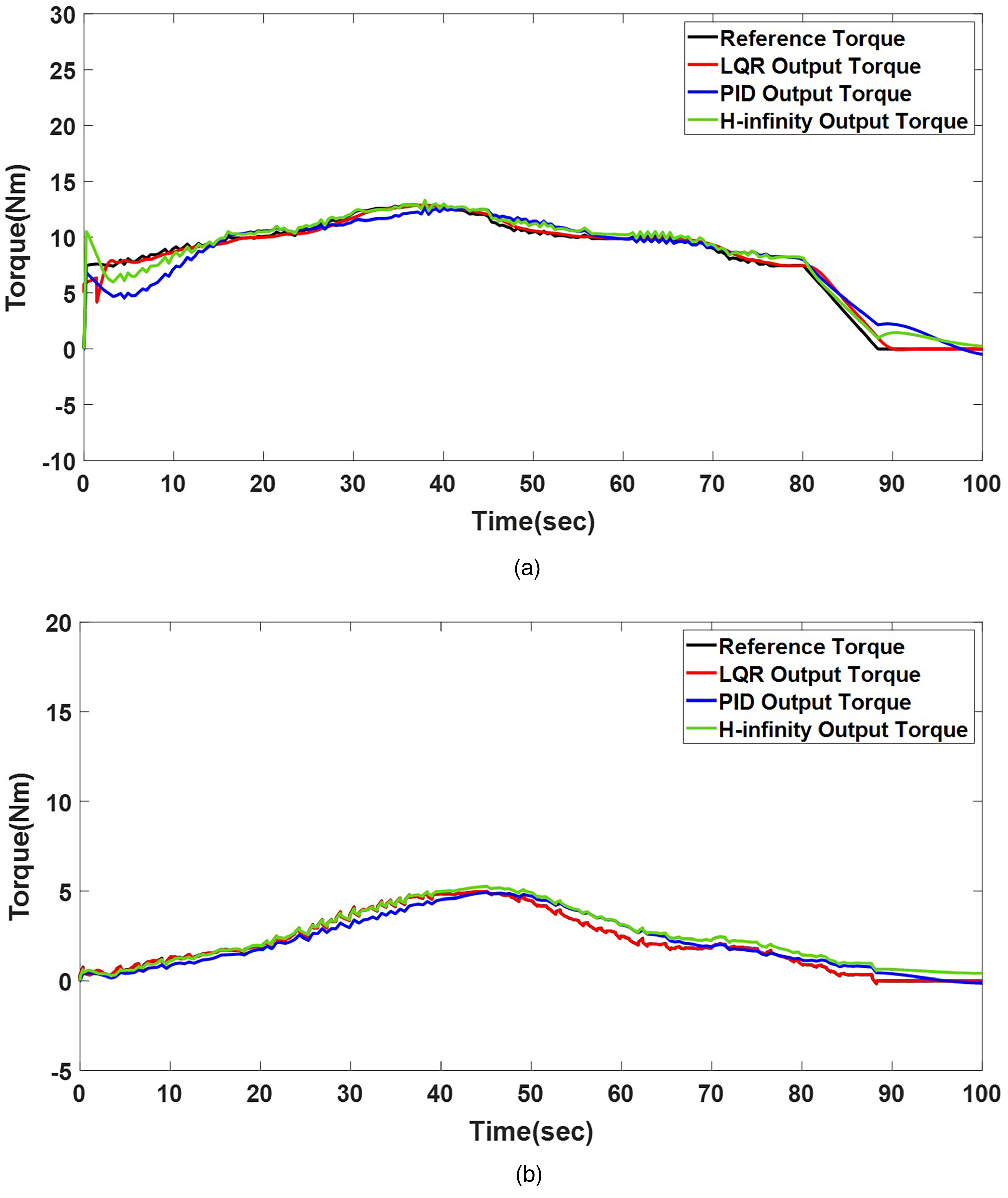
Torque-tracking in LQR, PID, and
Figure 7(a) and (b) reveals that the LQR controller tracked the reference trajectory (Grade 3) for both joints in the experiment, maintaining an error margin of 3%. In contrast, the PID controller exhibited a tracking error of approximately 5% for the same reference trajectory, while the
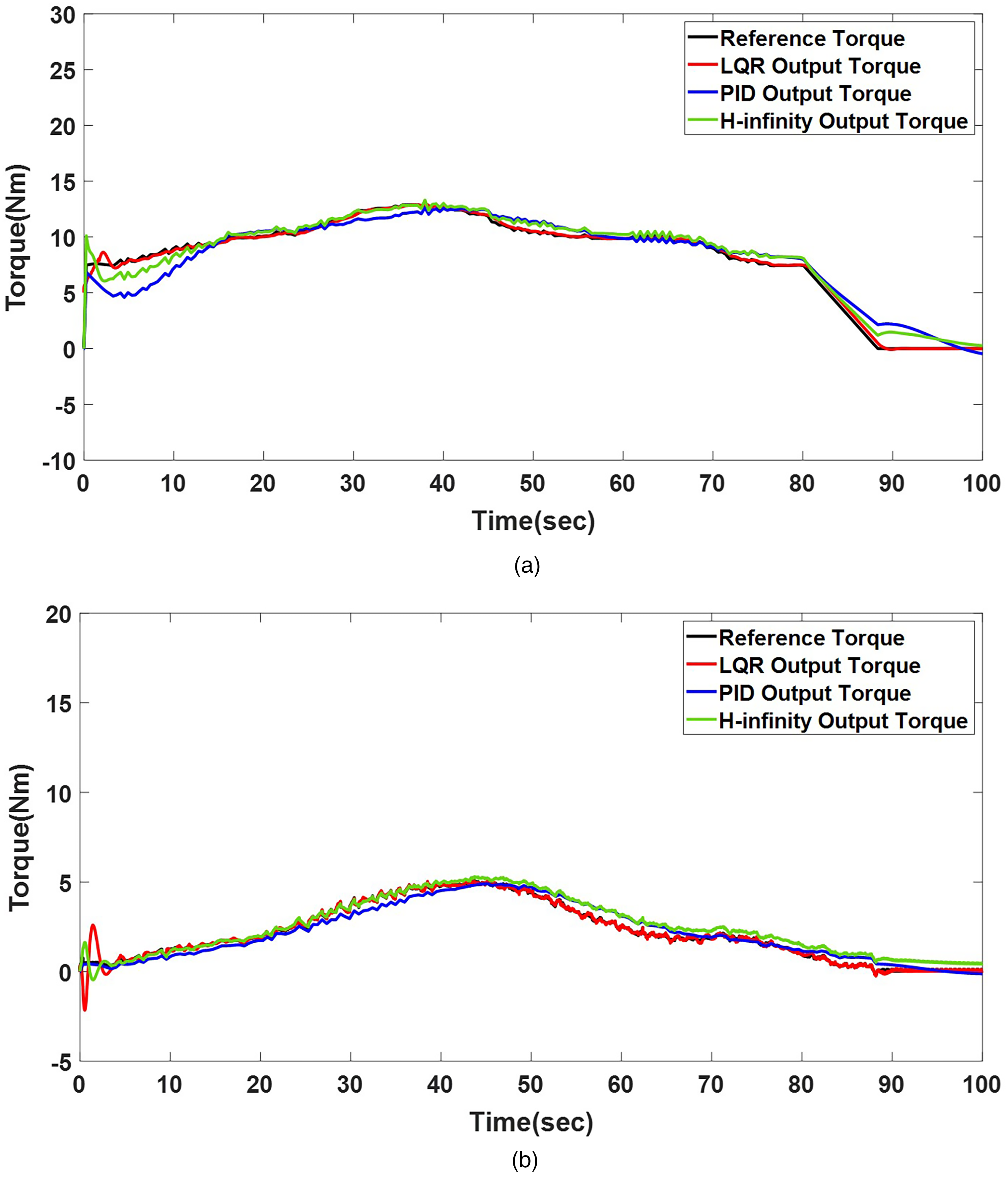
Torque-tracking in LQR, PID, and
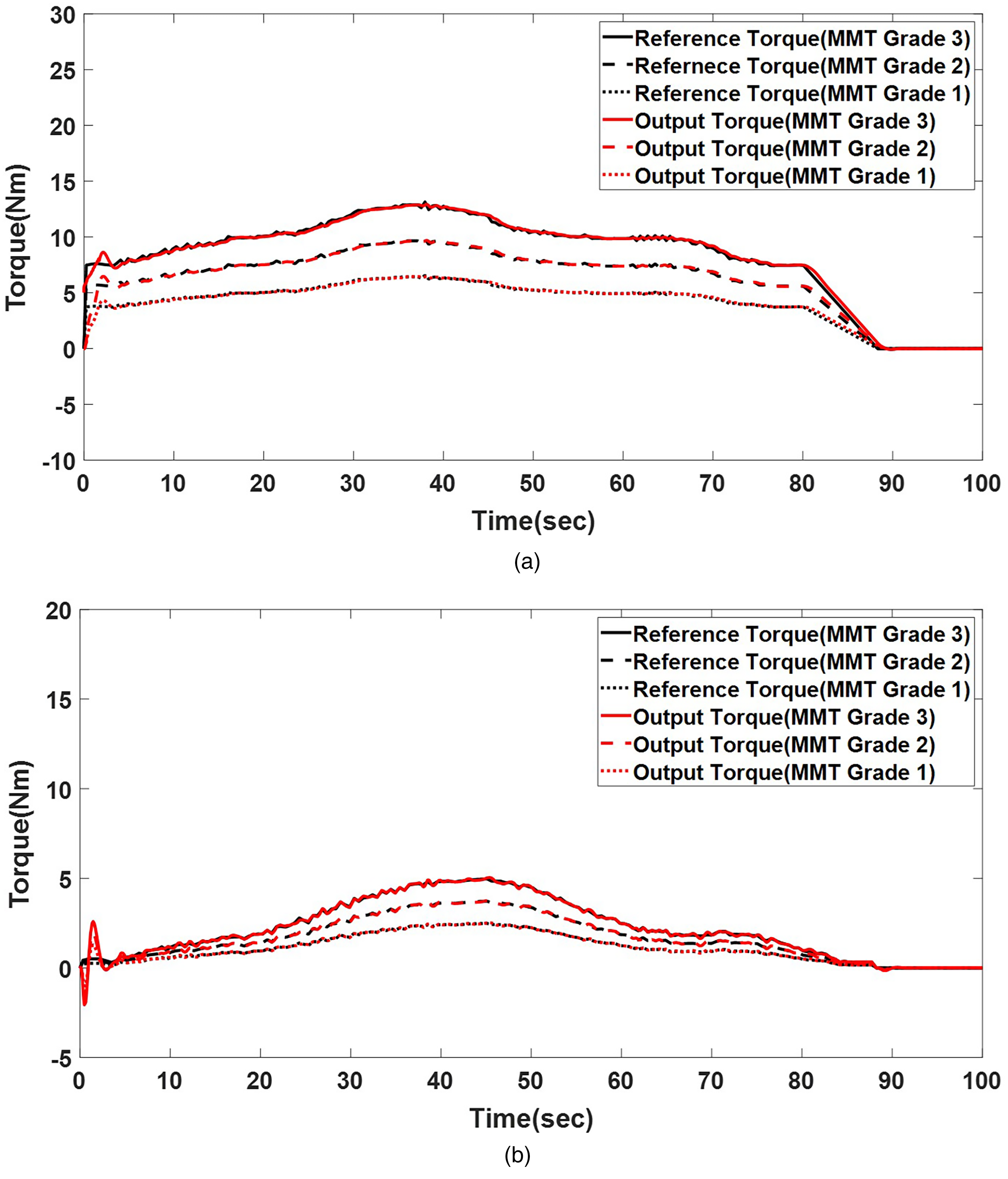
Torque-tracking results by rigidity level corresponding to MMT–MAS mapping (MMT Grades 1–3). (a) Shoulder joint torque tracking by rigidity level and (b) elbow joint torque tracking by rigidity level. MMT: manual muscle testing; MAS: modified Ashworth scale.
Figure 8 displays the torque tracking of the LQR controller for upper-limb joints with varying rigidity levels, corresponding to MMT Grades 1 to 3. For the shoulder joint, rigidity at MMT Grade 3 resulted in a peak torque of 13 Nm at 40 s, whereas MMT Grades 2 and 1 produced peak torques of 10 Nm at 41 s and 6.5 Nm at 39 s, respectively. For the elbow joint, rigidity at MMT Grades 3, 2, and 1 resulted in peak torques of 5.8 Nm at 47 s, 4.1 Nm at 45 s, and 2.7 Nm at 43 s, respectively. These results demonstrate the LQR controller’s capability of tracking torque trajectories effectively across rigidity levels in the shoulder and elbow joints.
Physiological tremor
Patients with hemiparesis may experience tremors when subjected to resistance exceeding the desired joint torque during rehabilitation exercises.15–17 To replicate the tremor characteristics, this study incorporates a physiological tremor,
The proposed model enables the robotic replica to reproduce involuntary oscillatory responses, providing a platform for evaluating tremor-suppression strategies in robotic rehabilitation. The time-dependent oscillatory torque patterns are characteristic of upper motor neuron lesions and integrate rigidity and tremor-related parameters into a unified representation of spastic resistance and involuntary joint instability. Mechanical parameters were scaled according to the MMT grades to reflect subject-specific impairments. For example, in MMT Grade 3, the forearm spring constant (
The physiological-tremor transfer function
In equation (7),
Figure 9 presents the computed physiological-tremor result for the shoulder and elbow joints under rigidity conditions corresponding to MMT Grade 3 (dominant tremor frequency: the 4–7 Hz). During 100 s of complex movement, the shoulder joint (Figure 9(a)) exhibits an average physiological-tremor amplitude of 0.21 Nm, with a peak tremor of 0.58 Nm. Similarly, the elbow joint (Figure 9(b)) demonstrates an average tremor amplitude of 0.17 Nm, with a maximum tremor of 0.44 Nm. These results with the parameterized stiffness and damping adjustments (
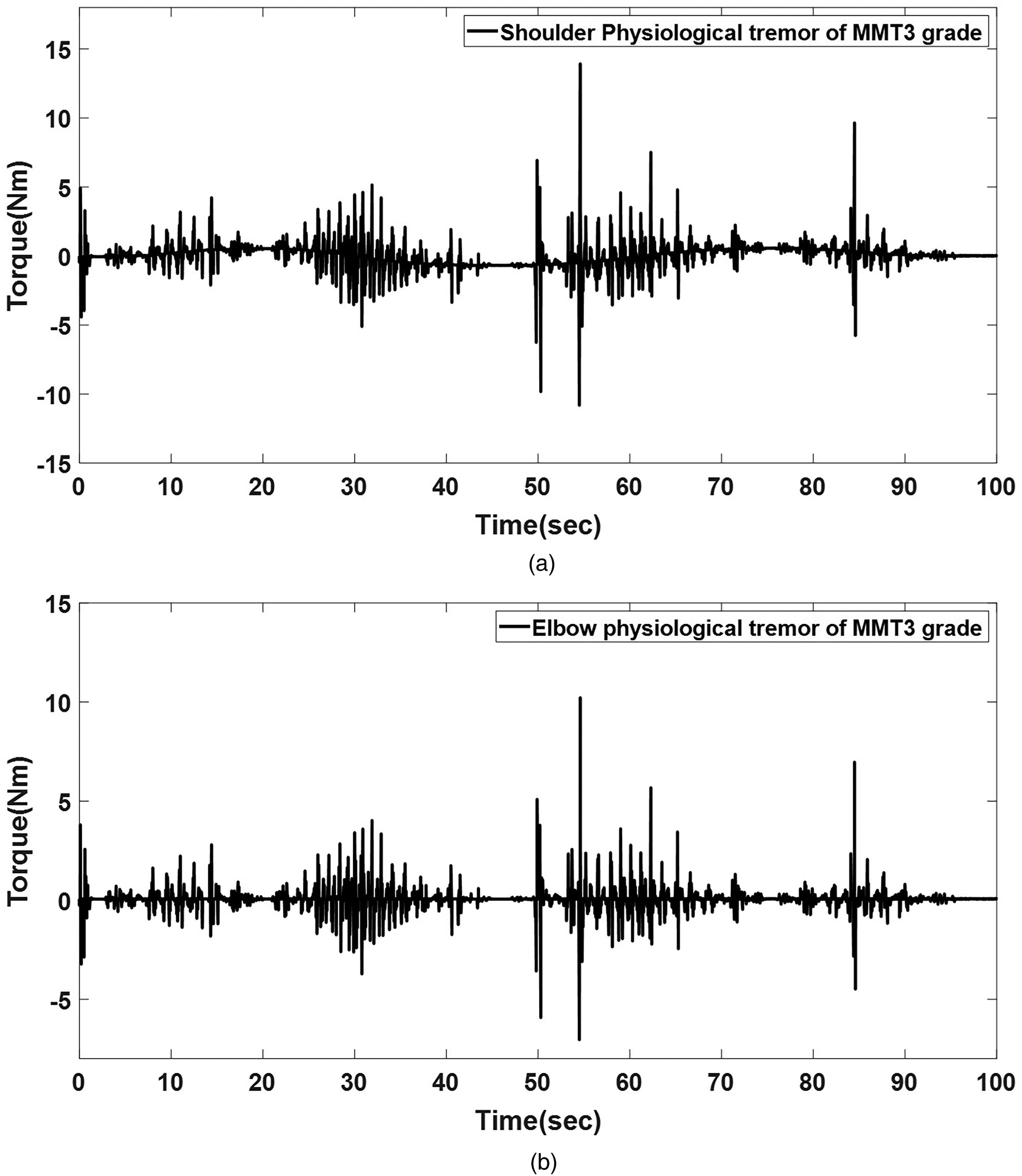
Multiple rigid joints with physiological tremor (manual muscle testing (MMT) Grade 3) in the simulation. (a) Shoulder joint and (b) elbow joint.
Integrated control algorithm
The tremor component is incorporated into the two-DOF upper-limb robotic replica by reformulating the previously computed matrix,
By adjusting the elastic and damping forces based on an individual’s muscle mass, the rigid joint of the two DOF upper-limb robotic replica can replicate changes in muscle elasticity and damping characteristics observed in rehabilitation patients. Figure 10 presents the implementation of complex rigid-joint motions incorporating physiological tremors in the proposed robotic replica. The experimental trials were conducted with a driving torque corresponding to MMT Grade 3, where tremor inputs were applied from the initial posture to the target position of each joint. The experiments were designed to evaluate peak torque tracking performance under compound upper-limb motion.

Proposed control system architecture of the two-degree-of-freedom (two-DOF) upper-limb robotic replica.
Figure 11 presents the torque-tracking performance of the three controllers at the shoulder and elbow joints. Quantitatively, the RMS tracking errors for LQR control in Figure 11(a) and (b), PID control in Figure 11(c) and (d), and

Torque tracking with physiological tremor (MMT Grade 3) for LQR, PID, and
To evaluate the performance of the proposed controller further across varying joint rigidity conditions, experimental trials were conducted by MMT grade. The shoulder joint was assessed under MMT Grades 4 and 2. Figure 12(a) and (b) illustrates the effective performance of the controller in simulating joint behavior across rigidity levels, validating its adaptability to various patient conditions. Consistent with previous experimental results, the shoulder joint, rigidified to MMT Grade 4, and the elbow joint, rigidified to MMT Grade 2, followed the reference trajectory with a tracking error of <5%.

Torque tracking of multiple rigid joints by rigidity condition. (a) Shoulder joint (manual muscle testing (MMT) Grade 4) and (b) elbow joint (MMT Grade 2).
To substantiate the physiological-tremor behavior observed in the experiments, rigidity modeling was quantitatively parameterized according to clinically defined MMT grades. The inertial (
The torque profiles in Figures 10 and 11 reflect the joint responses under the rigidity-adjusted conditions and confirm the validity of the selected tremor parameters. For instance, in the shoulder joint modeled with MMT Grade 3 characteristics (
The integrated LQR-based control framework establishes a robust foundation for replicating upper-limb tremor and rigidity phenomena in rehabilitation robots. The proposed control method presented the most stable tradeoff between tracking fidelity and physiological realism, offering clinically relevant torque outputs with RMS errors below 3%.
Survey
A satisfaction survey was conducted to evaluate the similarity of the developed upper-limb robotic replica that incorporates joint rigidity to patient movement. The evaluators comprised a group of rehabilitation professionals, including eight therapists and 10 students from the therapeutic department. The participants assessed the robotic replica using a 10-point similarity scale, ranging from 1 (not similar at all) to 10 (very similar). Prior to the evaluation, the functionality of the upper-limb robotic replica was explained to the participants. After testing the degree of joint rigidity in the robotic replica, the participants completed the questionnaire to provide assessments (KNU Institutional Review Board No. 2024-0517).
A standardized protocol was implemented to enhance the methodological rigor of the similarity evaluation. Prior to assessment, participants underwent a brief exposure protocol consisting of 2 min of passive mobilization with an actual patient exhibiting upper-limb rigidity, followed by 2 min of interaction with the developed robotic replica; exposure order was randomized (patient-first vs. robot-first), and evaluators were blinded to the controller type. The order of exposure was randomized, and evaluator blinding was applied to minimize potential bias. The concept of similarity was assessed across three distinct domains: joint rigidity, tremor rhythmicity, and overall movement impression. Each domain was rated on a 5-point Likert scale, and the average score was normalized to a 10-point scale for consistency with the previous literature. Fleiss’s kappa coefficient was calculated to verify inter-rater reliability based on evaluations from four physicians selected from the participant group. These therapists, each with substantial clinical experience in neurorehabilitation, independently assessed all experimental conditions following the standardized evaluation protocol. The resulting Fleiss kappa value was 0.73, corresponding to substantial agreement according to conventional criteria.27,28
The objective of this evaluation was to assess the similarity between composite rigid joint movements in upper-limb rehabilitation robots and those observed in patients. The evaluation is categorized into three groups: Group_PID refers to the application of a PID controller, Group LQR refers to the application of an LQR controller, and Group_TR refers to the application of an LQR controller with physiological tremor. This classification enables a comparative analysis of the control strategies in replicating patient-specific joint movements with rigidity.
Figure 13 reveals that the rigid joint movements of the upper-limb robotic replica controlled by the PID controller were rated with an average similarity score of 7.25 (

Similarity survey on joint rigidity between a robotic replica and patient.
A one-way analysis of variance was conducted to determine the effect of the controller type on the perceived similarity.29,30 This classification enables a comparative analysis of control strategies in replicating patient-specific joint movements with rigidity. The results indicated a statistically significant main effect,
Conclusion
This study presents an LQR-based control approach for implementing complex rigid joint movements in a two-DOF upper-limb patient robotic replica, incorporating physiological tremors in the shoulder and elbow joints. The joint driving torque was computed based on the motion capture data for the upper-limb motion of an adult male. Torque tracking across varying rigidity levels by joint maintains an error of
The evaluations were conducted to assess the clinical relevance and accuracy of the proposed robotic replica. A similarity assessment involving rehabilitation experts revealed that the LQR controller incorporating physiological tremor dynamics achieved the highest similarity scores for patient-specific joint behavior. The mean similarity ratings were
To replicate patient-specific torque characteristics, the upper-limb rehabilitation robotic replica integrates joint stiffness, damping properties, and physiological-tremor parameters to emulate varying levels of motor impairment. These parameters were linearly scaled by combining Winters’ muscle-tone–strength proportionality model with MMT grading, establishing an initial modeling framework that concurrently reflects increased muscle tone and reduced voluntary strength. Nevertheless, this linear scaling approach cannot fully capture time-dependent viscoelastic changes or nonlinear fatigue effects in musculoskeletal dynamics. Therefore, future work should extend this framework to an adaptive control architecture in which stiffness and damping parameters are continuously updated in real time using EMG-based feedback from muscle activation.
Footnotes
Ethical approval
The Institutional Review Board Ethics Committee of Kyungpook National University approved the study (approval number: KNU-2024-0517), which was conducted in accordance with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Regional Innovation System & Education (RISE) Glocal 30 Program through the Daegu RISE Center, funded by the Ministry of Education (MOE) and Daegu, Republic of Korea (Grant No. 2025-RISE-03-001), and by the Translational Research Center for Rehabilitation Robots, South Korea (NRCTR-EX22005), National Rehabilitation Center, Ministry of Health and Welfare.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
