Abstract
Purpose
Serum bilirubin plays an important role in antioxidant and anticancer processes. The inverse association between serum bilirubin and cancer risk have been widely reported in multiple cancers. The aim of this retrospective study was to investigate the prognostic impact of serum bilirubin in colorectal cancer patients undergoing surgical resection.
Methods
The value of serum bilirubin including total bilirubin, direct bilirubin, and indirect bilirubin were tested at pre-operatively in 330 colorectal cancer patients. The optimal cut-off values for these three biomarkers were determined by X-tile program. The relationship between serum bilirubin and outcomes were examined using Kaplan–Meier curves log-rank test, univariate and multivariate cox regression. Moreover, a number of risk factors were used to form a nomogram for evaluating risk of survival.
Results
The optimal cut-off points of serum total bilirubin, direct bilirubin, and indirect bilirubin were 19.5 μmol/L, 5.0 μmol/L and 8.1 μmol/L, respectively. Elevated total bilirubin and direct bilirubin were significantly associated with overall survival in surgical colorectal cancer patients. Additionally, predictive nomogram including total bilirubin and direct bilirubin for overall survival was established for predicting overall survival in surgical colorectal cancer patients.
Conclusions
These findings indicated that preoperative elevated total bilirubin and direct bilirubin could be considered as independent prognostic biomarkers for poor overall survival of colorectal cancer patients.
Introduction
In developing countries, colorectal cancer (CRC) is not only the fifth most commonly diagnosed cancer in males and the fourth in females, but also one of the leading causes of cancer death. 1 It was reported that 376.3 thousand new CRC cases and 191.0 thousand CRC-related deaths occurred in 2015 in China. 2 Approximately 25% of CRC patients present with metastases at the initial diagnosis and 50% would develop metastases, 3 which might contribute to the poor 5-year overall survival (OS) outcome, though obvious improvements have been developed and applied in diagnosis and treatment of CRC. Thus, it is important for us to identify effective prognostic biomarkers for survival improvement of patients with CRC. Many prognostic biomarkers, such as tumor node metastasis (TNM) stage, cell differentiation grade, and tumor type, have been used for clinical decisions. However, different patients present different prognosis even with same TNM stage and cell differentiation grade.4,5 Therefore, it is urgent that other prognostic biomarkers for predicting the outcome of patients with CRC be explored.
Bilirubin, the end point product of heme catabolism, has been found to play a protective role in antioxidant 6 and anticancer, 7 giving a rise in the poor ability of patients with CRC scavenging plasma free radical.8,9 Currently, the inverse association between serum bilirubin and cancer risk has been widely reported in various cancers, including CRC.10–13 Bilirubin is mainly metabolized by the liver. In hepatocytes, unconjugated bilirubin can be converted into conjugated bilirubin through conjugation with two molecules of glucuronic acid by the action of uridine diphosphate glucuronosyltransferase 1A1 (UGT1A1), a specific hepatic bilirubin UDP-glucuronosyltransferase. 14 Therefore, it is indicated that UGT1A1 could play an essential role in the regulation of bilirubin levels.
It is well known that serum bilirubin concentrations are highly heritable, and UGT1A1, which was found to be associated with increased total serum bilirubin, was the most widely studied. It was reported that congenital under-expression of UGT1A1 results in mild, chronic unconjugated hyperbilirubinemia. 14 Jiraskova et al. revealed that UGT1A1 was associated with CRC risk, 15 and recent studies also suggest that the UGT1A1 gene polymorphism was related to the prognosis of irinotecan-based chemotherapy in patients with CRC.16,17 Additionally, the cytotoxicity of irinotecan to colorectal cancer cells has a negative correlation with UGT1A1 expression.18,19 However, upon consideration of the inconvenient and expensive detection of UGT1A1, it is speculated that serum bilirubin, which was tested routinely, might be an available prognostic biomarker for CRC. At the same time, serum bilirubin, including total bilirubin (TBIL), direct bilirubin (DBIL), and indirect bilirubin (IBI) have been reported by numerous studies as promising prognostic biomarkers in various cancers, including breast cancer, 20 non-small-cell lung cancer, 21 nasopharyngeal carcinoma, 22 pancreatic adenocarcinoma, and CRC.23,24 Until now, few studies have reported the prognostic value of serum bilirubin in CRC.23,24 In Cao's study, only two biomarkers (TBIL and DBIL) were investigated in rectal cancer and limited CRC patient samples under stage II and III were included in Zhang's study. Also, the cohort of their studies were from different populations (one from northern China and another from eastern China) and different cut-off values were used, which would lead to instability of the results. Hence, this retrospective study was conducted in the eastern Chinese population to comprehensively investigate the prognostic impacts of serum TBIL, DBIL, and IBI in 330 CRC patients under stage I–III.
Materials and methods
Study population
This retrospective analysis was conducted in patients with newly diagnosed CRC who underwent surgical resection in Jiangsu Cancer Hospital, Jiangsu, China, between 2007 and 2011. Patients with the following criteria were excluded: (a) preoperative anti-tumor therapy, such as chemotherapy or radiotherapy; (b) previous diagnosis of malignancy; (c) mingled with other cancer; (d) underlying liver disease; (e) elevated parameters in liver function tests (AST > 40 U/L; ALT > 50 U/L); (f) with any reasons resulting in jaundice; and (g) with pre-operative clinical parameter and laboratory results loss. A total of 330 patients were enrolled in this study and informed consents were obtained from all eligible patients. This study was approved by the ethics committee of Nanjing Medical University.
Clinical parameters and laboratory results
Eligible patients’ clinical parameters—including age, gender, tumor location, TNM classification, tumor cell differentiation grade, and treatment type—were retrieved from their medical records. At the same time, laboratory results—including biochemical data (TBIL, DBIL, and IBI) detected by Olympus AU5421 detection system (Olympus Co. Ltd, Tokyo, Japan)—were also collected from medical records. All enrolled patients’ blood samples were obtained at 6am to 8am before surgery.
Patients’ follow-up
Patients were followed-up regularly until July 1, 2016 or until death according to 7th edition of the TNM-UICC/AJCC classification for CRC (every 3–6 months for the first 2 years; every 6 months for the third to fifth years). The OS was defined as the time from the date of surgery to last follow-up or death, and disease-free survival (DFS) was defined as the time from the date of treatment to the first recurrence or metastasis, or last follow-up, or death. Follow-up data for patients were computed from medical records or telephone follow-up.
Statistical analysis
X-tile 3.6.1 software (Yale University, New Haven, CT, USA), IBM SPSS Statistical 20.0 (SPSS Inc. Chicago, IL, USA) and R 3.3.0 software (Institute for Statistics and Mathematics, Vienna, Austria) were used for statistical analysis. The optimal cut-off values of TBIL, DBIL, and IBI were determined by X-tile software (Yale University). 25 Patients were divided into the low group or the high group according to the cut-off value. The Kolmogorov-Smirnov test was selected to assess the normality of calculated parameters. The Student's t-test was used for normal distributed parameters, otherwise the Mann–Whitney U test was performed. The chi-square test was used to compare categorical variables, and the Kaplan–Meier log-rank test and the univariate Cox regression analysis were conducted to analyze the relationship of variables and survival of CRC patients. Age, gender, and other variables that were significant in the univariate Cox regression analysis were entered into the multivariate Cox regression model using the backward conditional method. The nomogram and Harrell's concordance index (c-index) evaluating the model performance for OS were established by R software (Institute for Statistics and Mathematics).26,27 P < 0.05 was considered to be statistically significant.
Results
The optimal cut-off values for TBIL, DBIL, and IBI
X-tile software (Yale University) was used to calculate the optimal cut-off values. For TBIL, DBIL, and IBI of OS these values were 19.5 μmol/L, 5.0 μmol/L, and 8.1 μmol/L, respectively, which are shown in Figure 1. Patients were subsequently divided into the high group and the low group according to the optimal cut-off levels.
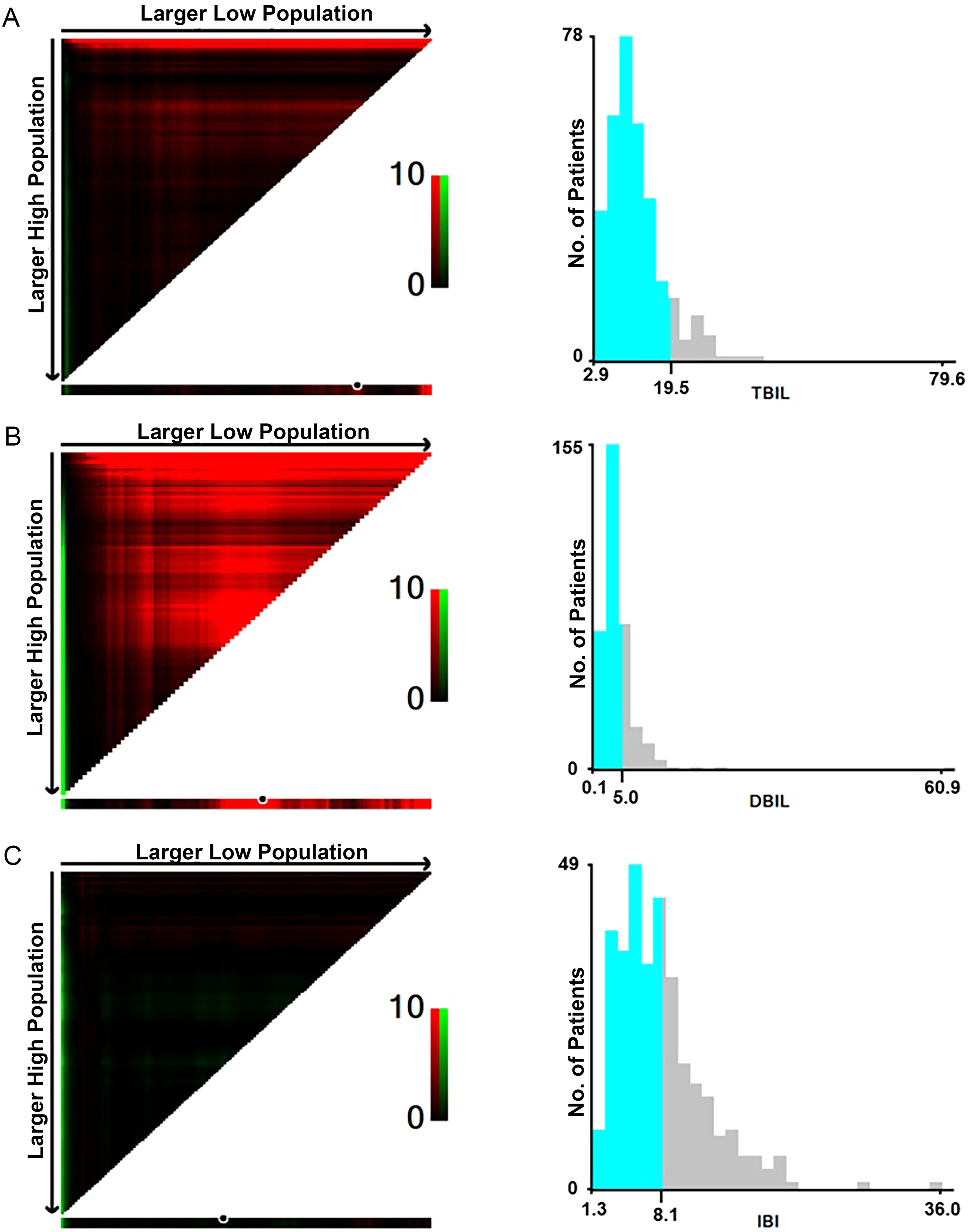
X-tile analyses for 5-year overall survival.
Baseline characteristics of patients based on TBIL, DBIL, and IBI
Baseline characteristics of patients are summarized in Table 1. A total of 330 CRC patients (median age = 65 years) with 221 males and 109 females were enrolled in this study. The median follow-up time was 60 months. There were 200 colon cases and 130 rectal cancer patients. Based on the 7th edition of the TNM-UICC/AJCC classification, the number of patients with T1/T2 and T3/T4 were 52 and 278, respectively. A total of 34 patients were diagnosed as poor cell differentiation and 204 were identified without lymph node metastasis (N = 0). Surgical resection was performed in all patients, and only 226 CRC patients underwent chemo-radiotherapy after surgery.
Baseline characteristics of patients based on TBIL, DBIL and IBI.
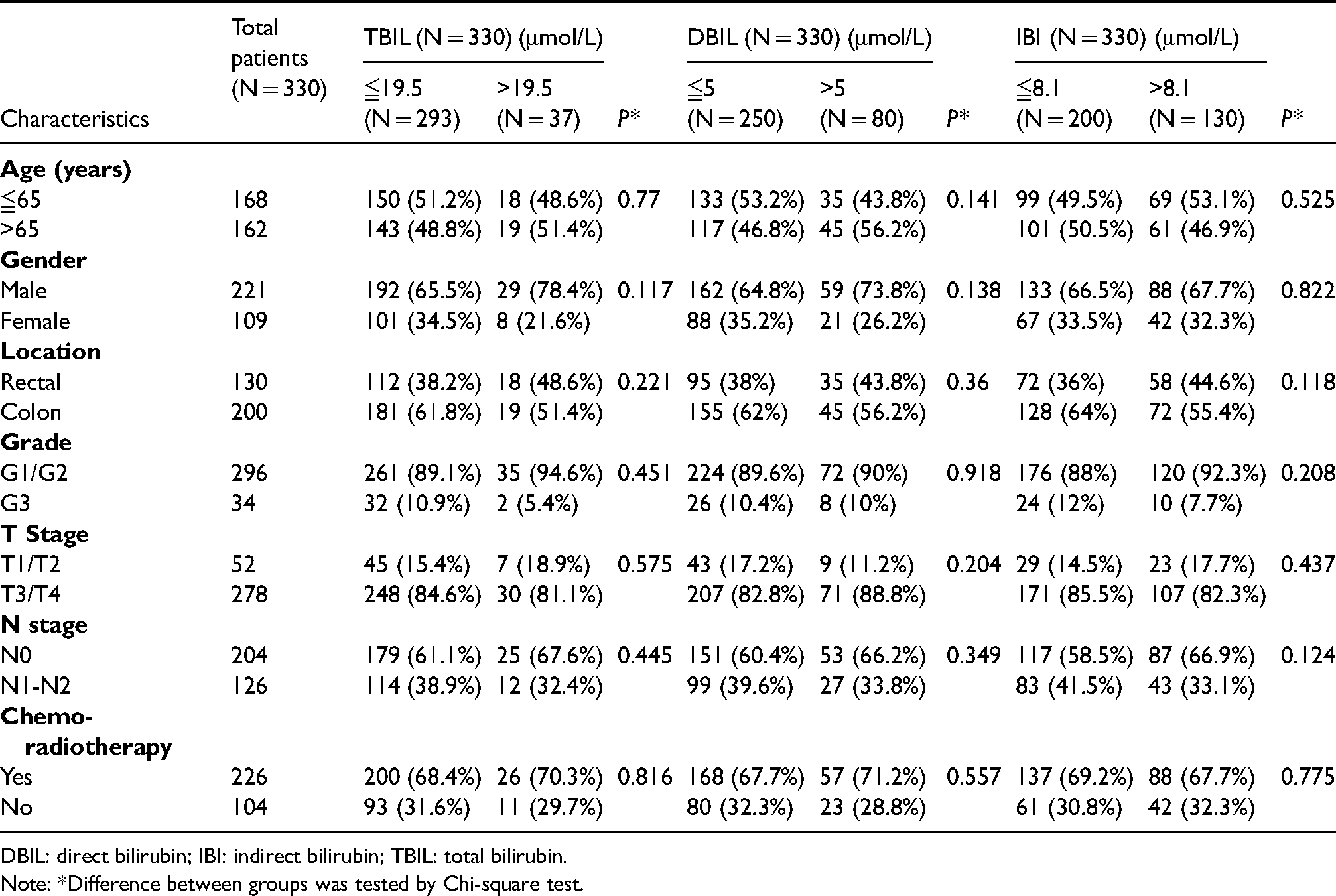
DBIL: direct bilirubin; IBI: indirect bilirubin; TBIL: total bilirubin.
Note: *Difference between groups was tested by Chi-square test.
The association between baseline characteristics and clinical prognosis
To evaluate the association of baseline characteristics with clinical prognosis, the Kaplan–Meier log-rank survival analysis (Table 2, Figures 2(a) and (b)) and the univariate cox regression analysis (Table 3) were performed based on the pathological data and the postoperative survival time of patients. Our results revealed that lymph node metastasis was significantly correlated with OS (HR = 2.323, 95% CI = 1.456, 3.704, P < 0.001) and DFS (HR = 2.078, 95% CI = 1.458, 2.961, P < 0.001). At the same time, tumor invasion depth (HR = 2.712, 95% CI = 1.093, 6.729, P = 0.031), elevated TBIL (HR = 1.939, 95% CI = 1.064, 3.536, P = 0.031), and DBIL (HR = 2.407, 95% CI = 1.502, 3.857, P < 0.001) were also associated with poor OS (Table 3, Figures 2(a) and (b)). Furthermore, age, gender, and the significant factors in univariate analysis were included to further determine the influence on OS and DFS by multivariate analysis. Table 3 shows that lymph node metastasis was associated with poor OS (HR = 2.337, 95% CI = 1.454, 3.757, P < 0.001) and DFS (HR = 2.078, 95% CI = 1.458, 2.961, P < 0.001), indicating that lymph node metastasis could be regarded as an independent prognostic biomarker for OS and DFS of patients with CRC. More importantly, elevated TBIL (HR = 2.282, 95% CI = 1.244, 4.187, P = 0.008) and DBIL (HR = 2.561, 95% CI = 1.593, 4.118, P < 0.001) were also associated with poor OS, indicating that TBIL and DBIL could be considered as independent factors for the OS of CRC patients.
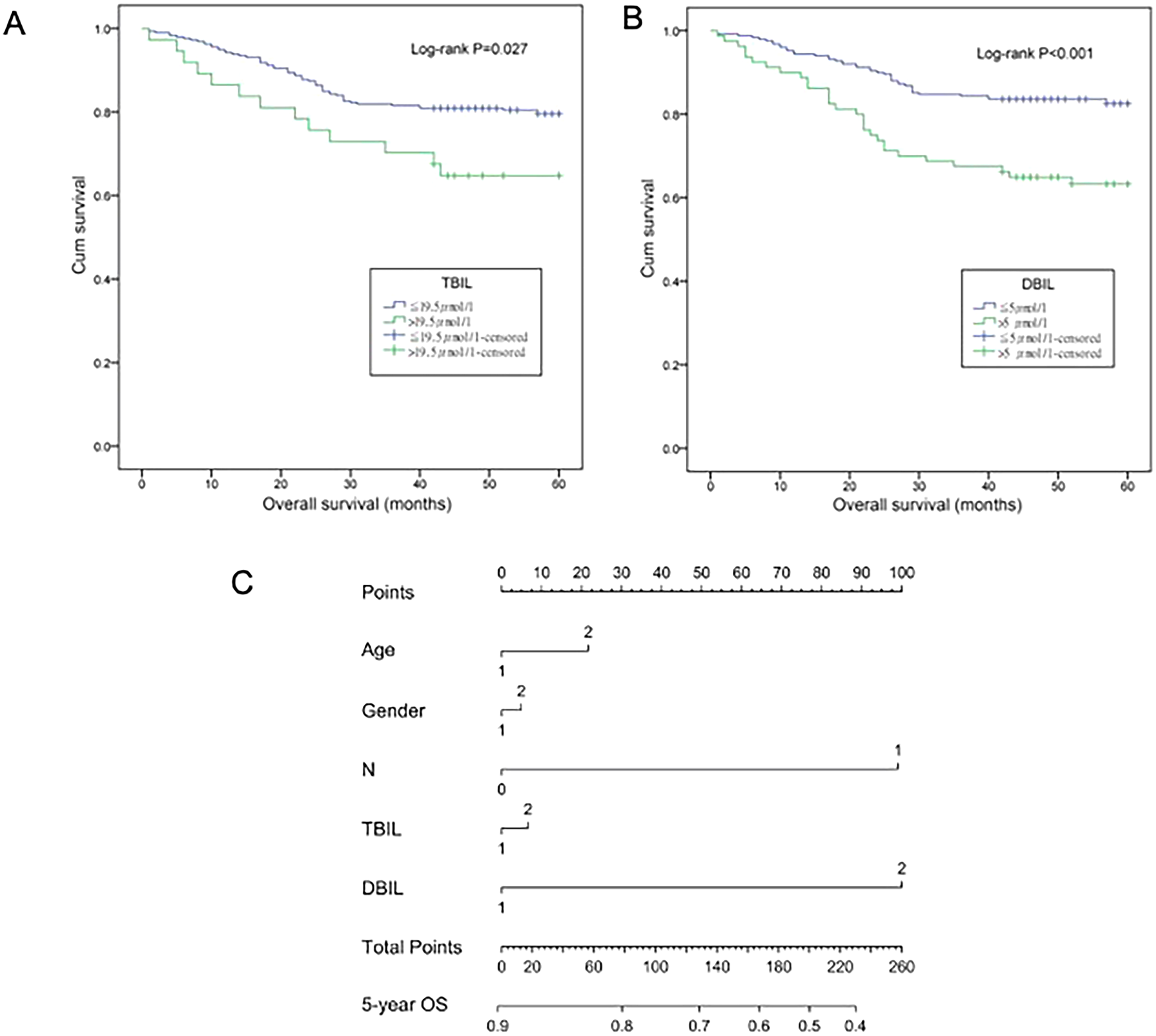
Kaplan–Meier curves and predictable prognostic nomograms of TBIL and DBIL for OS.
The association of clinical baseline characteristics with overall and recurrence-free survival in all patients.
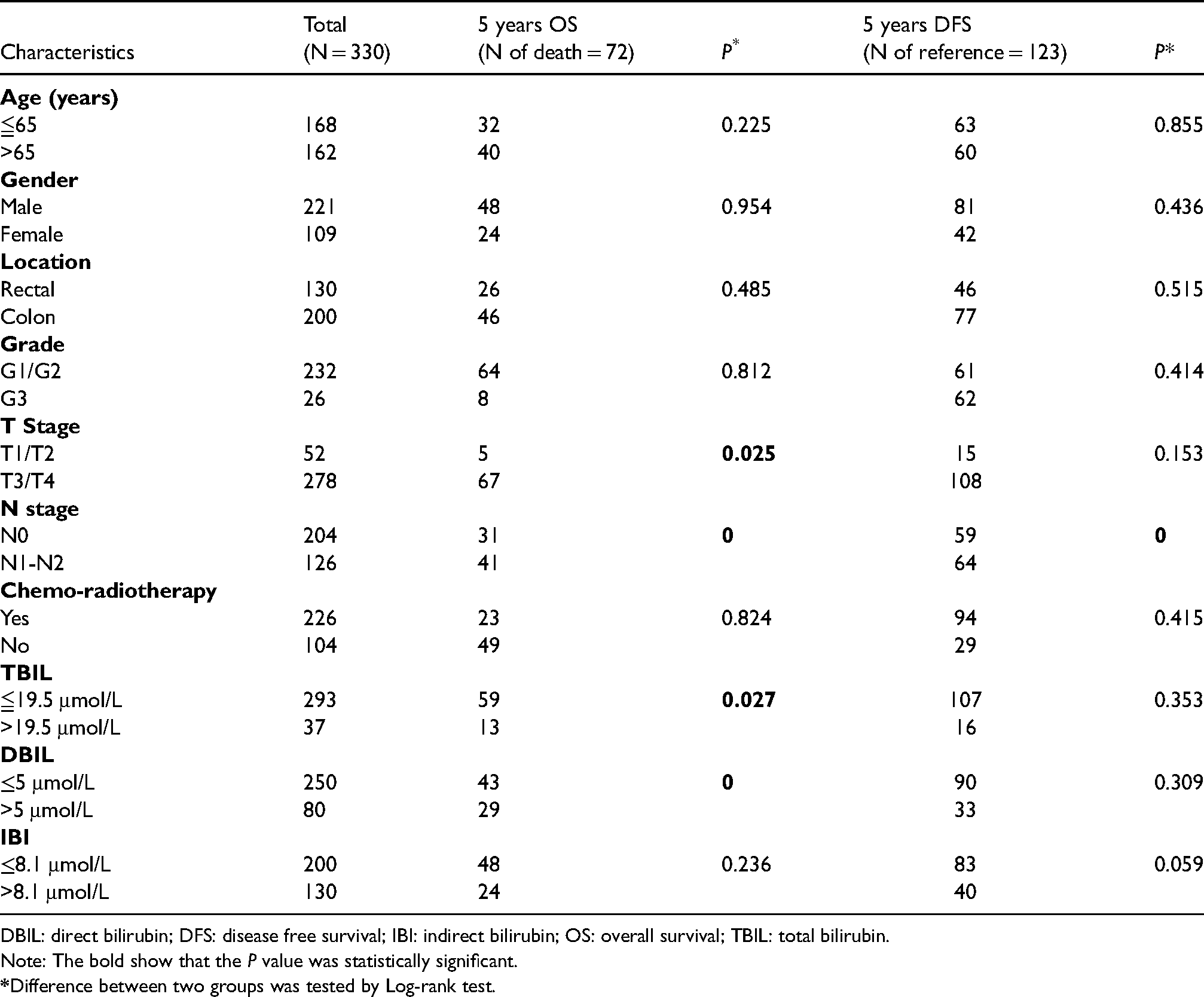
DBIL: direct bilirubin; DFS: disease free survival; IBI: indirect bilirubin; OS: overall survival; TBIL: total bilirubin.
Note: The bold show that the P value was statistically significant.
Univariate and multivariate analysis of prognostic factors of overall and disease-free survival by Cox regression model.
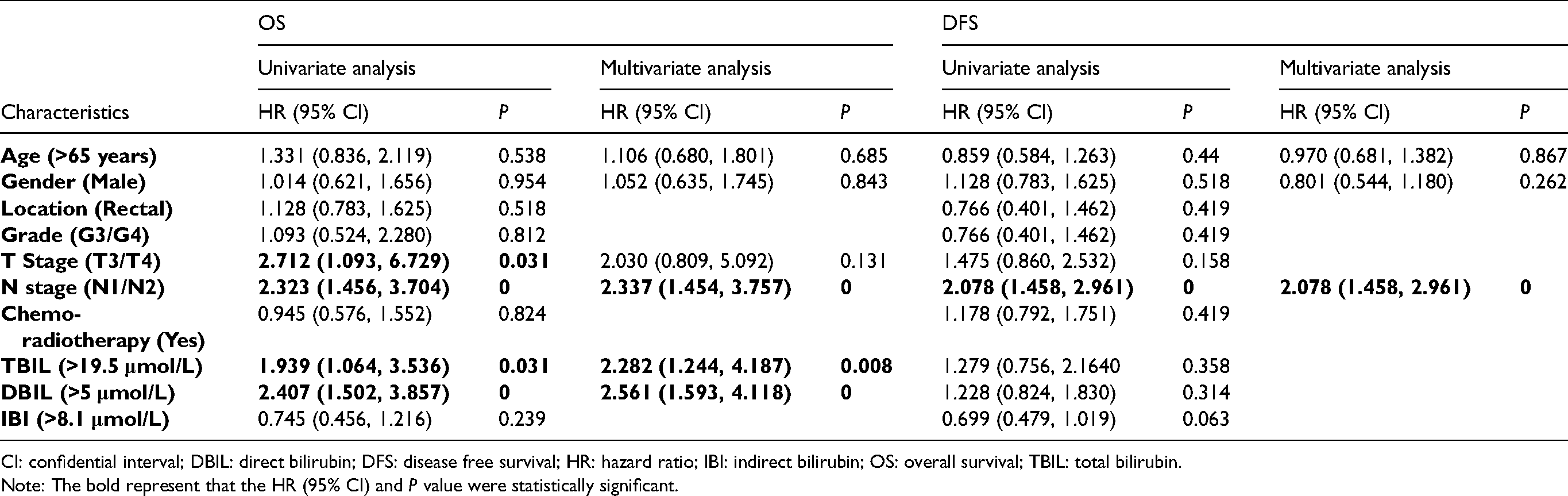
CI: confidential interval; DBIL: direct bilirubin; DFS: disease free survival; HR: hazard ratio; IBI: indirect bilirubin; OS: overall survival; TBIL: total bilirubin.
Note: The bold represent that the HR (95% CI) and P value were statistically significant.
Prognostic nomogram for OS
In order to predict OS for CRC patients, nomograms—which were estimated by multivariate cox regression model—were produced in R software (Institute for Statistics and Mathematics) (Figure 2(c)). Discrimination using Harrell's C-index was used to estimate the predicted accuracy of the model. 28 The Harrell's C-index of the Cox model, including TBIL and DBIL for OS, was 0.669 (95% CI = 0.603, 0.735). However, the C-index of the nomogram without TBIL and DBIL for OS was only 0.624 (95% CI = 0.562, 0.686), indicating that the former nomogram had a superior discrimination ability to predict clinical outcomes in surgical CRC patients and nomogram including TBIL and DBIL for OS, which could be recommended to predict OS for CRC patients.
Discussion
This retrospective study first indicated that pre-operative increased TBIL and DBIL within the relatively normal range and lymph node metastasis could be identified as independent prognostic biomarkers for I–III stage CRC patients with surgical resection. Patients with stage IV CRC are excluded because the condition is usually inoperable. The study also revealed that elevated IBI was a protective factor for survival, but no statistical significance was observed, which was consistent with the previous observations that bilirubin (specifically, unconjugated bilirubin) could trigger mitochondrial depolarization and then induce apoptosis in colon cancer cell lines in vitro. 29 However, the relationship between IBI and longer survival of patients with non-small-cell lung cancer was verified by Li et al. 21 The above inconsistent results may be mainly caused by the different kinds of cancer, suggesting that the roles of IBI might vary by cancer type, and the correlation of IBI and survival should be confirmed by a large size sample study.
In addition, our study also observed that elevated TBIL and DBIL were associated with poor outcome, which was consistent with previous published studies.20,23 UGT1A1 plays an essential role in the level of bilirubin. UGT1A1 gene polymorphism with (TA) n repeat variations are commonly seen in patients with CRC leads to the under-expression of UGT1A1. Therefore, the conversion of IBI to DBIL was blocked due to the abnormal metabolism of bilirubin regulated by UGT1A1, 30 resulting in elevated IBI and decreased DBIL in CRC. At the same time, Jiraskova et al. 15 revealed that UGT1A1*28 was a protective factor against the development of CRC, and elevated serum bilirubin levels were associated with a decreased risk of CRC. Xu et al. 16 found that allele variants of UGT1A1*28/*6 were associated with short OS of patients with CRC. Therefore, elevated IBI and decreased DBIL would be associated with a favorable prognosis of CRC. The high serum DBIL level, which was reported to be associated with an increased risk of lymph node metastasis, may also explain why DBIL was correlated with a poor survival outcome. Our findings showed that lymph node metastasis was also an independent unfavorable factor for both OS and DFS, which is consistent with a previously published article. 23 Notably, our study provided valid evidence that the evaluation of preoperative biomarkers may contribute to predicting the prognosis of patients with CRC.
Our study observed that the optimal cut-off value of 19.5 μmol/L for TBIL and 5 μmol/L for DBIL were complicated by a time-dependent assessment of outcome using the X-tile program, a robust graphical tool verified by Yale University. 25 However, the optimal cut-off level for TBIL and DBIL in this study were inconsistent with the results of previous studies20,21,23 (data are presented in Supplementary Table S1). This may be due to multiple variations in our investigation, including the assays used in measuring bilirubin and calculating the optimal cut-off value, as well as different population, race, sample size, cancer type, and survival end-point, especially patients enrolled in the research under stage I–III, which should be verified by further studies. Moreover, the 330 cases enrolled in our study may have limited statistical power and cannot be representative of all CRC patients in general. Thus, more samples should be collected for more accurate outcomes regarding the clinical advantage of TBIL and DBIL detection as prognostic biomarkers in CRC patients. In conclusion, our study demonstrates that pre-operative levels of TBIL and DBIL were independent prognostic biomarkers in stage I–III CRC patients with surgical resection, which provides a promising clinical approach to predict the OS of patients with CRC.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008211036128 - Supplemental material for The prognostic value of serum bilirubin in colorectal cancer patients with surgical resection
Supplemental material, sj-docx-1-jbm-10.1177_17246008211036128 for The prognostic value of serum bilirubin in colorectal cancer patients with surgical resection by Zhangjun Jia, Zeyu Zhu, Ying Wang, Jing Ding, Zhenzhong Lin, Yanyan Zhang and Zhipeng Li in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the China Postdoctoral Science Foundation (No. 2017M621681) to YZ and National Nature Science Foundation of China Young Investigator (No.81602681) to YZ.
Author contributions
Zhangjun Jia, Zeyu Zhu, and Ying Wang have contributed equally to this work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
