Abstract
Objective:
Our study aimed to observe and evaluate the clinical value of interleukin (IL)-11 in the serum and exhaled breath condensate of patients with non-small cell lung cancer (NSCLC).
Methods:
A total of 91 patients with NSCLC and 72 healthy volunteers were included in this study. IL-11 concentration was determined by ELISA, and the relationship between IL-11 expression in serum and exhaled breath condensate specimens, and the clinicopathological characteristics of patients with NSCLC were analyzed. The relationship between serum IL-11 expression and traditional tumor markers and inflammation indicators of NSCLC was also analyzed. The correlation between serum IL-11 and exhaled breath condensate IL-11 level was determined. The receiver operating characteristic curve was used to evaluate the diagnostic value of IL-11 and carcinoembryonic antigen single and combined detection for NSCLC. The published data from online databases were used to analyze the relationship between the expression of IL-11 and the prognosis of NSCLC.
Results:
IL-11 concentration in serum and exhaled breath condensate specimens of patients with NSCLC were significantly increased. IL-11 expression was positively correlated with lymph node metastasis, distant metastasis, tumor node metastasis stage, and tumor differentiation degree of NSCLC. The expression of IL-11 in serum was positively correlated with that in exhaled breath condensate specimens. IL-11 expression was closely related to that of neutrophil-to-lymphocyte ratio and carcinoembryonic antigen. The combination of serum IL-11 with exhaled breath condensate IL-11 and carcinoembryonic antigen showed significantly higher diagnostic value than any one marker alone. Besides, the high IL-11 expression was closely related to the poor prognosis of NSCLC.
Conclusion:
IL-11 can be used as a potential diagnostic and prognostic biomarker for NSCLC.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related deaths in the world with the highest death rate among all malignant tumors. 1 Non-small cell lung cancer (NSCLC), including adenocarcinoma, squamous cell carcinoma, and large cell carcinoma, accounts for approximately 85% of all lung cancer cases. 2 Compared with small cell lung cancer, NSCLC involves cells that grow and divide more slowly and spread and metastasize relatively later. 3 Therefore, the vast majority of patients with NSCLC are diagnosed in the middle and advanced stages, and their 5-year overall survival rate is less than 20%. 4
Inflammation is regarded as the eighth feature of malignant tumors. 5 Considering that human lungs are susceptible to a variety of toxins and pathogens, chronic injuries and inflammation may occur, which then contribute to the development of lung cancer. 6 Cytokines are the bridge between the inflammatory environment and cancer cells. Chronic inflammatory response mediated by cytokines in the tumor microenvironment is an important factor in the occurrence and development of tumors. 7 Cytokines are not only key participants in tumorigenesis but also potential targets for cancer treatment.
According to previous studies, interleukin (IL), as an important member of inflammation-related cytokines, plays an irreplaceable role in the occurrence and development of lung cancer and the proliferation and migration of cancer cells.8,9 Therefore, the biological detection of interleukin in the serum, exhaled breath condensate (EBC), cancer tissues, and cancer cell supernatants may be useful in the early diagnosis and prognosis evaluation of lung cancer; this strategy is also predicted to bring new breakthroughs to clinical chemotherapy and targeted treatment of lung cancer.
As an important member of the IL-6 family of cytokines, IL-11 was initially considered to play a role in promoting platelet production. 10 However, many recent studies have shown that IL-11 is also mostly involved in inflammation-associated gastrointestinal and breast tumors.11,12 IL-11 expression is increased in the body through the inflammatory stimulation of cancer cells and is closely related to the poor prognosis of cancer. 13 At present, research on the role of IL-6 in lung cancer has increasingly advanced, but the role of IL-11 in lung cancer is not yet fully elucidated. Therefore, in this study, we systematically evaluated the clinical value of the inflammatory factor IL-11 in NSCLC.
Materials and methods
Patients
A total of 91 patients with NSCLC admitted to the Second Affiliated Hospital of Nantong University from November 2018 to October 2019 were diagnosed through lung tissue or lymph node biopsy, pleural effusion cytology, and pathological examination. The participants had not received radiochemotherapy, immune-targeted therapy, or surgery prior to selection. In addition, 72 healthy volunteers who underwent physical examination in the same period were selected as the control group. The healthy volunteers had no acute and chronic infectious diseases, no vital organ diseases, and no genetic family tumor history. Tumor staging was performed in accordance with the lung cancer tumor node metastasis (TNM) staging guidelines published by the Union for International Cancer Control in 2017. 14 The details of the number of people in each group are shown in Figure 1. All patients signed the informed written consent. This study was approved by the Medical Ethics Committee of the Second Affiliated Hospital of Nantong University (approval no. 2019KY139).
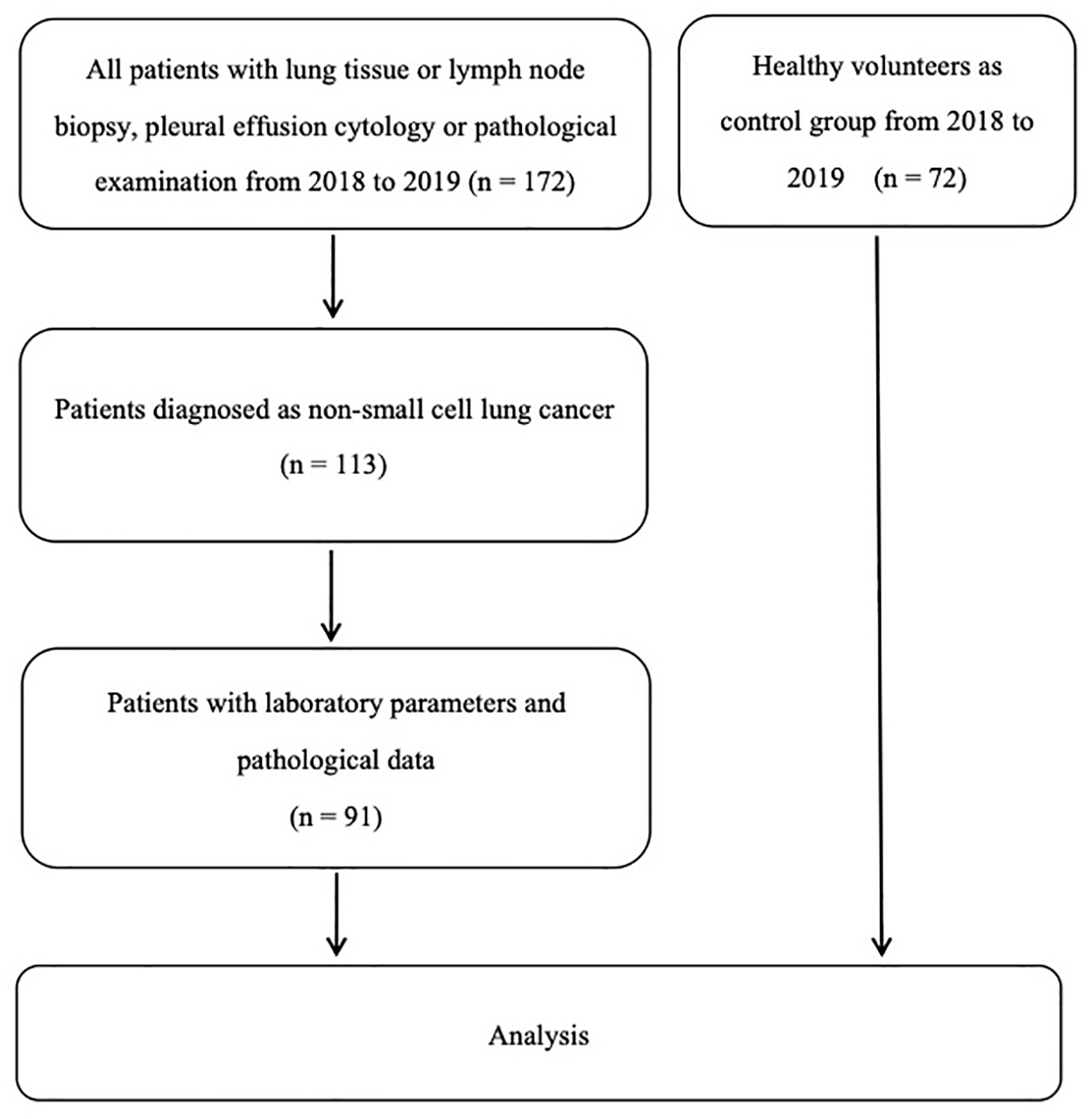
The flow chart of the study.
Serum collection
Exactly 5 mL of fasting venous blood was collected from each group on the first day of admission. Blood was placed in a coagulation tube and immediately placed in a centrifuge at 3500 rpm for 5 minutes. Then, the serum in the tube was collected and transferred into a 2 mL centrifuge tube. All specimens were stored in a −80°C low-temperature refrigerator for the following inspection.
EBC collection
EBC specimens were collected from each group on the first day of admission. The EBC specimens were collected using the EcoScreen condenser produced by Eric Jaeger Company. The temperature was set to −20°C and the collection time was 20 minutes. 15 Given the limitations of the degree of cooperation of each group, the EBC specimens of 64 patients with NSCLC and 63 healthy people were collected.
Data collection
The electronic medical record system was used to collect baseline characteristics, including patient age, gender, smoking status, performance status, and medical history, as well as the statistics and analysis of clinical blood routines, tumor screening indicators, and other test data. The public data of online databases were used to collect the IL-11 mRNA level of patients with lung cancer and their overall survival (OS), progression-free survival (PFS), and first progression (FP).16,17
Neutrophil-to lymphocyte ratio, lymphocyte-to-monocyte ratio, and hemoglobin-to-red-blood-cell distribution width ratio calculations
The following equations were used: NLR = neutrophil count (×109/L)/lymphocyte count (×109/L), LMR = lymphocyte count (×109/L)/monocyte count (×109/L), HRR = hemoglobin (g/L)/red blood cell distribution width (RDW) (%).
IL-11 concentration
HRP conjugate is an enzyme conjugate that specifically binds to human IL-11 and anti-human IL-11 antibody in the sample to form an immune complex. Next, we calculated the concentration of IL-11 in the sample by detecting this immune complex. IL-11 concentration in serum and EBC was determined by the Human IL-11 Enzyme-Linked Immunosorbent Assay (ELISA) kit (Elabscience) following the manufacturer’s instructions. In brief, 100 μL of standard or 100 μL of sample per well was added to the microplate and incubated for 90 minutes. The liquid was removed from each well, and 100 μL of biotinylated detection antibody working solution was added to each well of the microplate and incubated for 60 minutes. After the microplate was washed three times, it was later incubated with 100 μL of HRP conjugate working solution for 30 minutes. After five washes, 90 μL of substrate reagent was added and incubated for 20 minutes. Then, 50 μL of stop solution was added to each well of the microplate, and absorbance was determined at 450 nm. Biochemical experiments were performed in triplicate, and a minimum of three independent experiments were evaluated.
Statistical analysis
All data were analyzed using the statistical software IBM SPSS Statistics 22.0 and the graphing software Graphpad Prism 8.0. All data were tested by Kolmogorov–Smirnov test or Shapiro–Wilk test to determine whether they were normally distributed. For normally distributed data, the t-test was used to compare the mean between two groups with equal variances. When the variance was not the same, the t-test (Satterthwaite method) was used. For non-normally distributed data, the non-parametric test (Mann–Whitney U test) was used, and the median value (interquartile range) [M (Q1–Q3)] was used. Count data were compared using the χ2 test. Linear correlation analysis was used to determine the correlation between the two variables. Pearson correlation analysis was used for data that corresponded to bivariate normal distribution, and Spearman correlation analysis was used for data that fitted non-parametric distribution. The diagnostic value of serum IL-11, EBC IL-11, and carcinoembryonic antigen (CEA) for NSCLC was assessed using receiver operating characteristic (ROC) curve analysis. The relationship between the level of IL-11 and OS, PFS and FP of NSCLC was calculated by the Kaplan–Meier method, and survival curves were compared with the log-rank test. P < 0.05 was considered statistically significant.
Results
Baseline characteristics of all participants
A total of 91 patients with NSCLC were eventually included in this study. Among these patients, 28 had squamous cell carcinoma and 63 had adenocarcinoma. In addition, 54.95% of the patients with NSCLC were in TNM stage III–IV, and 46.15% of the patients had poorly differentiated tumors (Table 1). In the same period, 72 healthy volunteers were included as healthy control group. The difference between the two groups in terms of age, gender, and smoking history was not significant (P > 0.05, Table 2).
Demographic and clinical characteristics of NSCLC patients (n = 91).
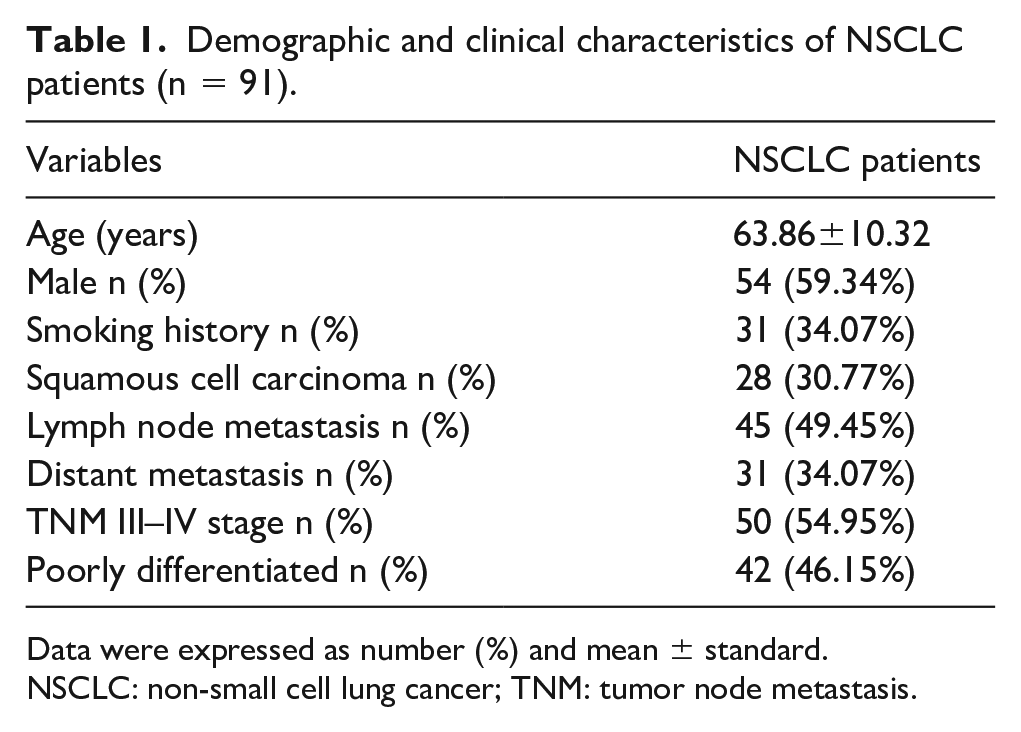
Data were expressed as number (%) and mean ± standard.
NSCLC: non-small cell lung cancer; TNM: tumor node metastasis.
Baseline characteristics of all participants.
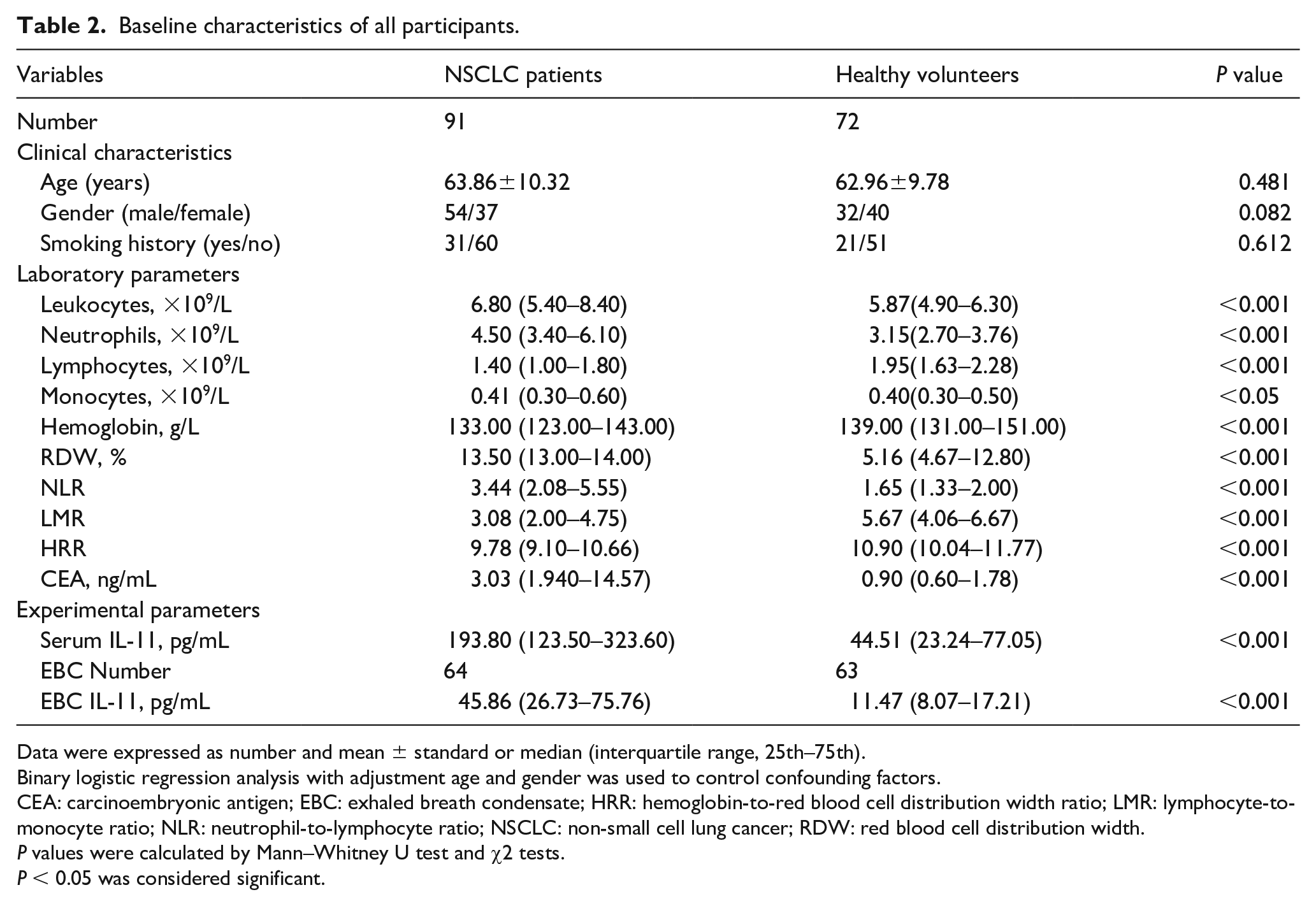
Data were expressed as number and mean ± standard or median (interquartile range, 25th–75th).
Binary logistic regression analysis with adjustment age and gender was used to control confounding factors.
CEA: carcinoembryonic antigen; EBC: exhaled breath condensate; HRR: hemoglobin-to-red blood cell distribution width ratio; LMR: lymphocyte-to-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; NSCLC: non-small cell lung cancer; RDW: red blood cell distribution width.
P values were calculated by Mann–Whitney U test and χ2 tests.
P < 0.05 was considered significant.
Expression levels of serum and EBC IL-11 in patients with NSCLC and healthy groups
IL-11 concentration in the serum and EBC of patients with NSCLC and healthy controls were detected using ELISA. The median concentration of serum IL-11 in the group of patients with NSCLC was 193.80 pg/mL, which was significantly higher than that in the healthy control group (44.51 pg/mL) (P < 0.001, Table 2). Similarly, compared with healthy controls, the concentration of IL-11 in EBC of patients with NSCLC was higher (P < 0.001, Table 2). Figures 2(a) and (b) show that IL-11 concentration in the serum and EBC of patients with NSCLC was significantly up-regulated with the increase of NSCLC severity, represented by TNM stage (P < 0.05).
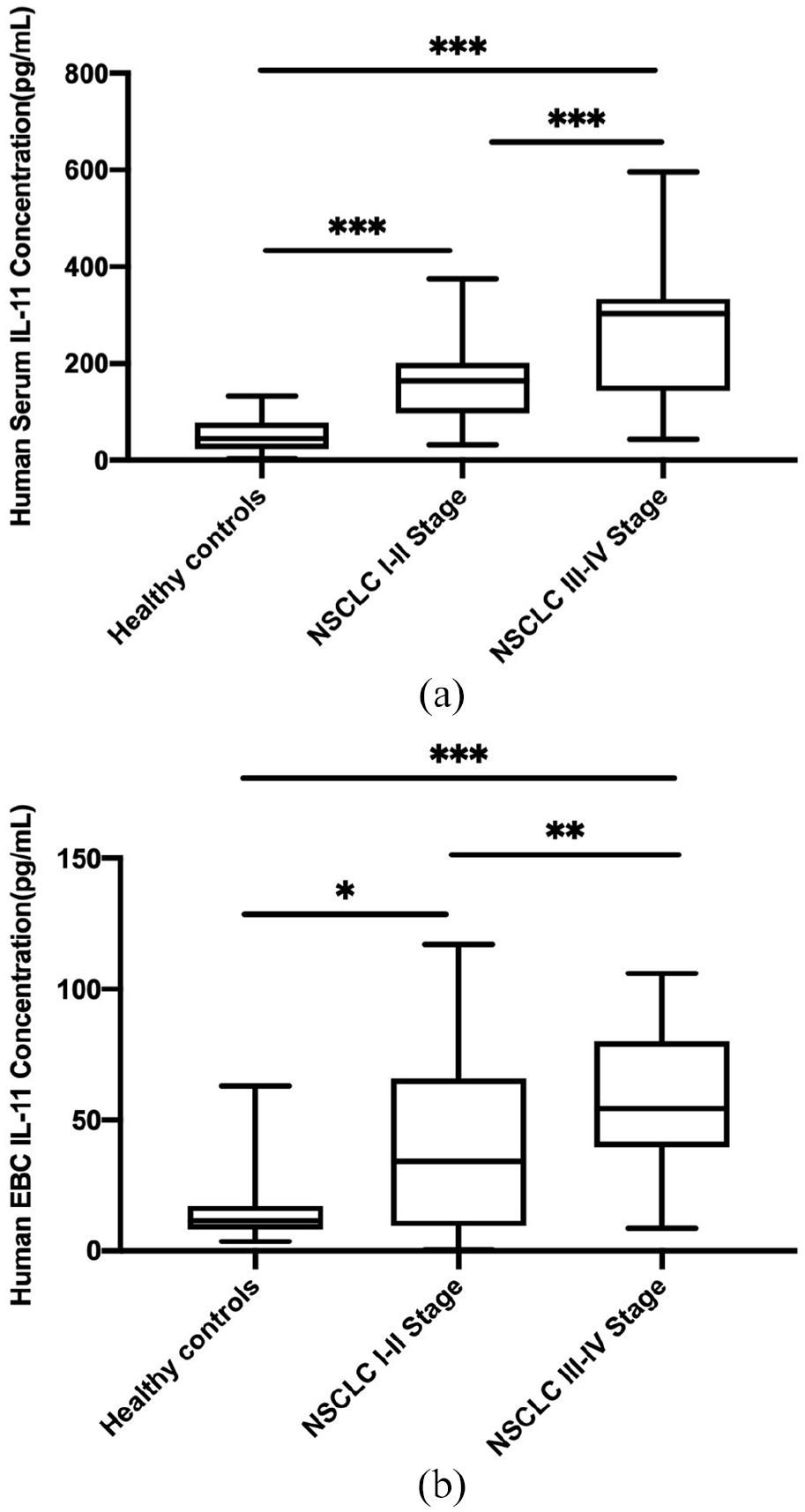
Expression levels of IL-11 in serum (a) and EBC (b) of NSCLC patients and healthy controls.
Correlation between IL-11 level in serum and clinicopathological features in 91 cases of all non-small cell lung cancers
We analyzed the relationship between IL-11 level in serum and clinicopathological characteristics of patients with NSCLC. Table 3 shows that the correlation among serum IL-11 level and age (P = 0.338), gender (P = 0.313), smoking history (P = 0.124), and histological type (P = 0.187) was not significant. When patients with NSCLC had a tumor size > 5 cm, lymph node metastasis, distant metastasis, high TNM stage, and poor tumor differentiation, the IL-11 expression level in serum was significantly increased (P < 0.05).
Correlation between IL-11 level in serum and clinicopathological features in 91 all non-small cell lung cancers.
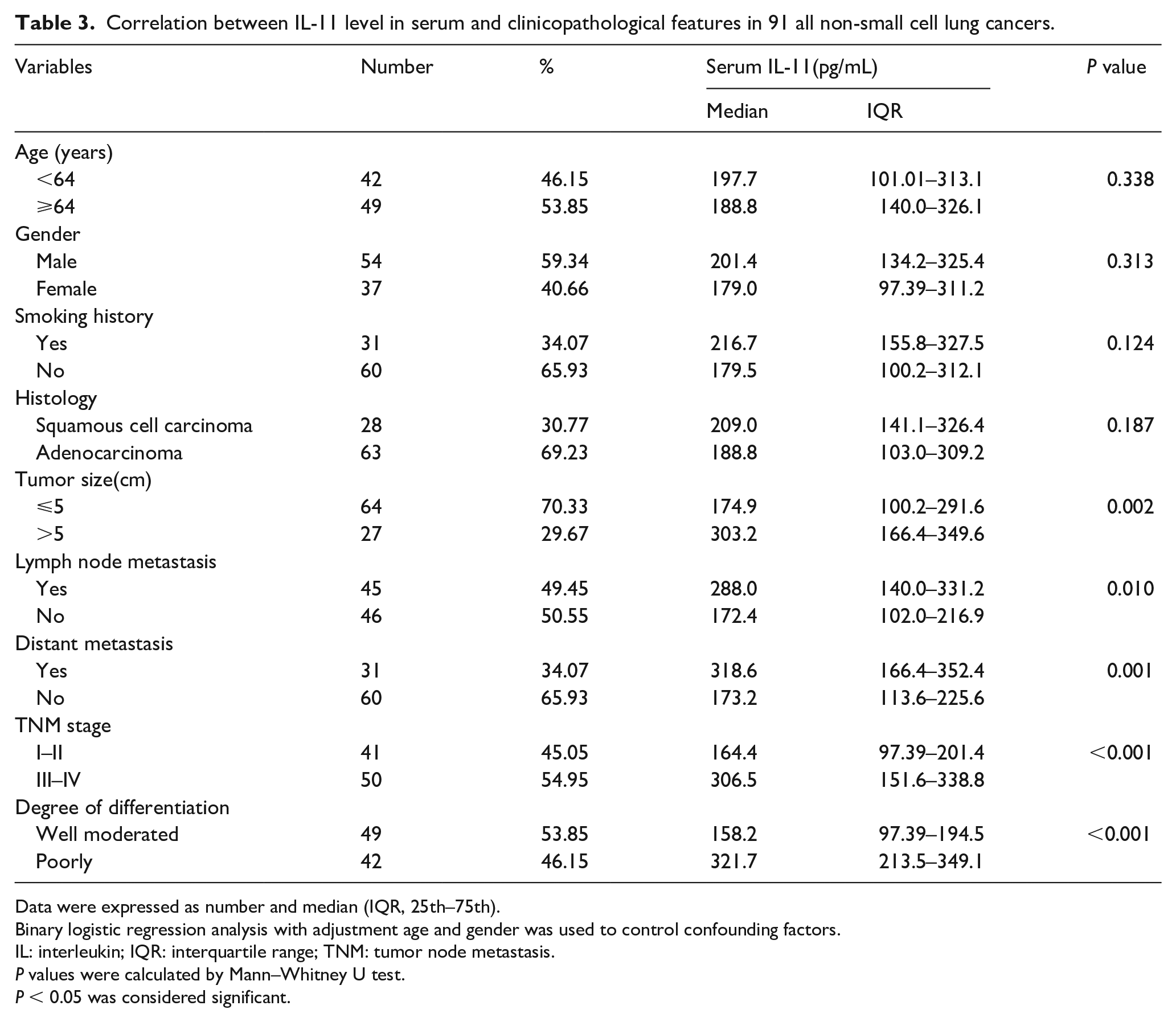
Data were expressed as number and median (IQR, 25th–75th).
Binary logistic regression analysis with adjustment age and gender was used to control confounding factors.
IL: interleukin; IQR: interquartile range; TNM: tumor node metastasis.
P values were calculated by Mann–Whitney U test.
P < 0.05 was considered significant.
Correlation between IL-11 level in EBC and clinicopathological features in 64 cases of all NSCLC
We also analyzed the relationship between IL-11 level in EBC and clinicopathological characteristics of patients with NSCLC. Table 4 shows that the IL-11 level in EBC was not significantly correlated with age (P = 0.142), gender (P = 0.091), smoking history (P = 0.459), histological type (P = 0.430), and tumor size (P = 0.162). Compared with the low expression level group of IL-11 in EBC, the high expression level group of IL-11 in EBC was closely associated with lymph node metastasis (P = 0.028), distant metastasis (P = 0.037), high TNM stage (P = 0.008), and poor tumor differentiation (P < 0.001).
Correlation between IL-11 level in EBC and clinicopathological features in 64 all non-small cell lung cancers.
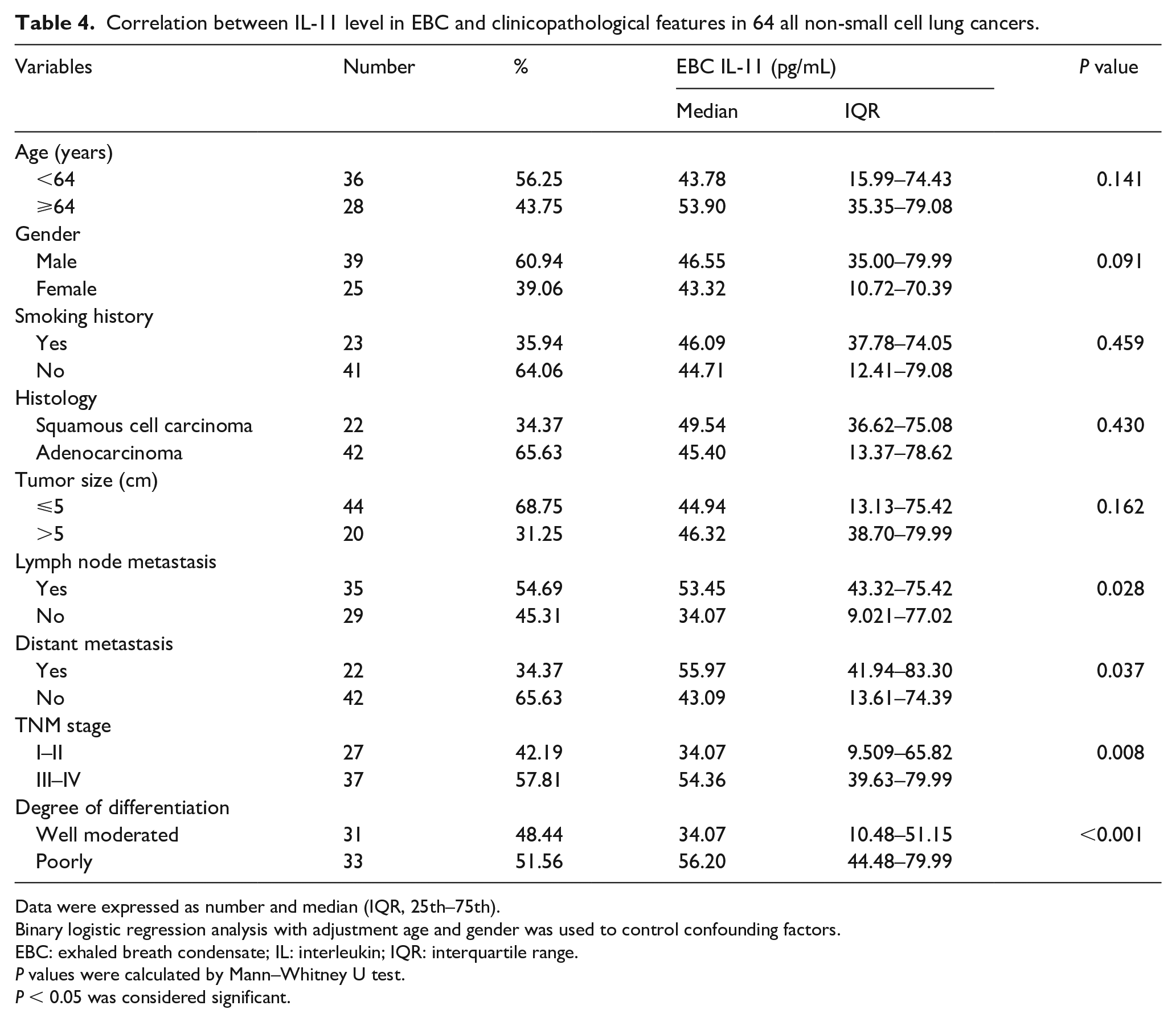
Data were expressed as number and median (IQR, 25th–75th).
Binary logistic regression analysis with adjustment age and gender was used to control confounding factors.
EBC: exhaled breath condensate; IL: interleukin; IQR: interquartile range.
P values were calculated by Mann–Whitney U test.
P < 0.05 was considered significant.
Correlation between IL-11 level in serum, inflammatory parameters in blood routine, and traditional tumor biomarkers in 91 cases of all NSCLCs
Leukocyte count, neutrophil count, monocyte count, red blood cell distribution width, and CEA in patients with NSCLC were significantly higher than those in the healthy control group (P < 0.05), whereas lymphocyte count and hemoglobin were significantly lower than those in the healthy control group (P < 0.001, Table 2). We then further calculated the neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), and hemoglobin-to-red blood cell distribution width ratio (HRR) values of each group. Table 2 shows that the NLR of the patients with NSCLC was significantly higher than that of the healthy controls (P < 0.001). By contrast, compared with the healthy controls, the LMR and HRR levels of patients with NSCLC were significantly lower (P < 0.001).
The ROC curves were drawn on the basis of the NLR, LMR, and HRR values of patients with NSCLC and healthy volunteers. The Youden’s index (sensitivity + specificity −1) was used to determine the cut-off value. The optimal cut-off values of NLR, LMR, and HRR were 2.332, 3.367, and 10.23, respectively (Table 5). Therefore, the patients with NSCLC were grouped into two in accordance with the optimal cut-off values of NLR, LMR, and HRR, and the relationship among these parameters with IL-11 level in serum of patients with NSCLC was analyzed. Table 5 shows that the NLR ⩾ 2.332 group had higher IL-11 level in serum of patients with NSCLC than the NLR < 2.332 group (P < 0.001), while the IL-11 level in serum of patients with NSCLC was not significantly correlated with inflammation indicators LMR and HRR (P > 0.05).
Correlation between IL-11 level in serum, inflammatory parameters in blood routine, and traditional tumor biomarkers in 91 non-small cell lung cancers.
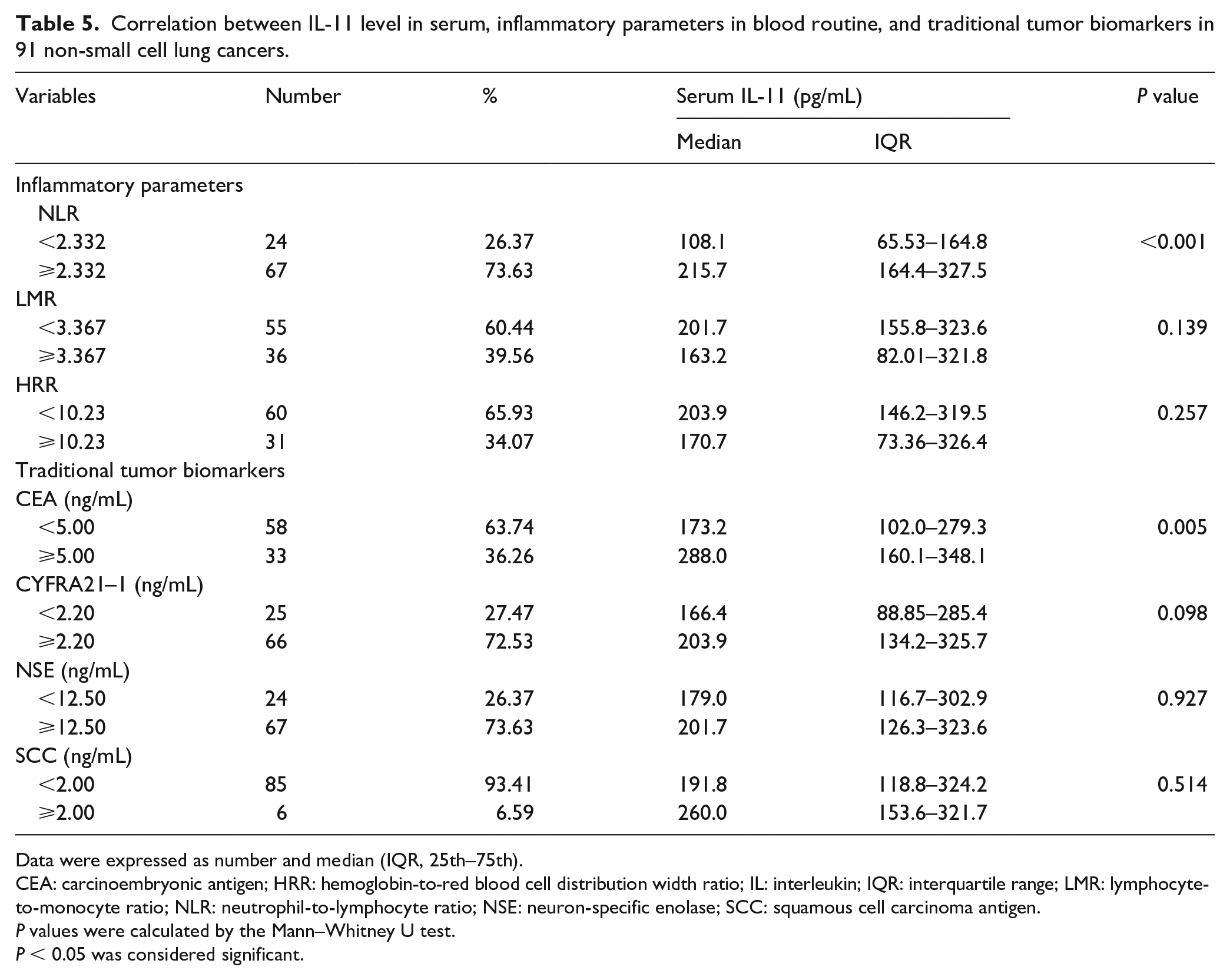
Data were expressed as number and median (IQR, 25th–75th).
CEA: carcinoembryonic antigen; HRR: hemoglobin-to-red blood cell distribution width ratio; IL: interleukin; IQR: interquartile range; LMR: lymphocyte-to-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; NSE: neuron-specific enolase; SCC: squamous cell carcinoma antigen.
P values were calculated by the Mann–Whitney U test.
P < 0.05 was considered significant.
The traditional tumor screening indicators for patients with lung cancer in clinical practice are CEA, CYFRA21-1, neuron-specific enolase (NSE), and squamous cell carcinoma antigen (SCC). Therefore, we divided the patients with NSCLC into two groups in accordance with the most suggestive cut-off value of each indicator, and compared the relationship of these parameters with IL-11 level in serum of patients with NSCLC. Table 5 shows that when CEA ⩾ 5.00 ng/mL, the IL-11 expression level in the serum of patients with NSCLC increased significantly (P = 0.005). When CYFRA21-1 is ⩾ 2.20 ng/mL, NSE is ⩾ 12.50 ng/mL, and SCC is ⩾ 2.00 ng/mL, the IL-11 expression level in the serum of patients with NSCLC increased; however, the difference was not statistically significant (P > 0.05).
Correlation analysis of IL-11 in serum and EBC (n = 64)
We evaluated the correlation between the expression levels of IL-11 in the serum and EBC of 64 patients with NSCLC. Figure 3 shows the strong positive correlation between the expression levels of IL-11 in serum and EBC, r = 0.728, which was statistically significant (95% CI 0.664, 0.862; P < 0.001).
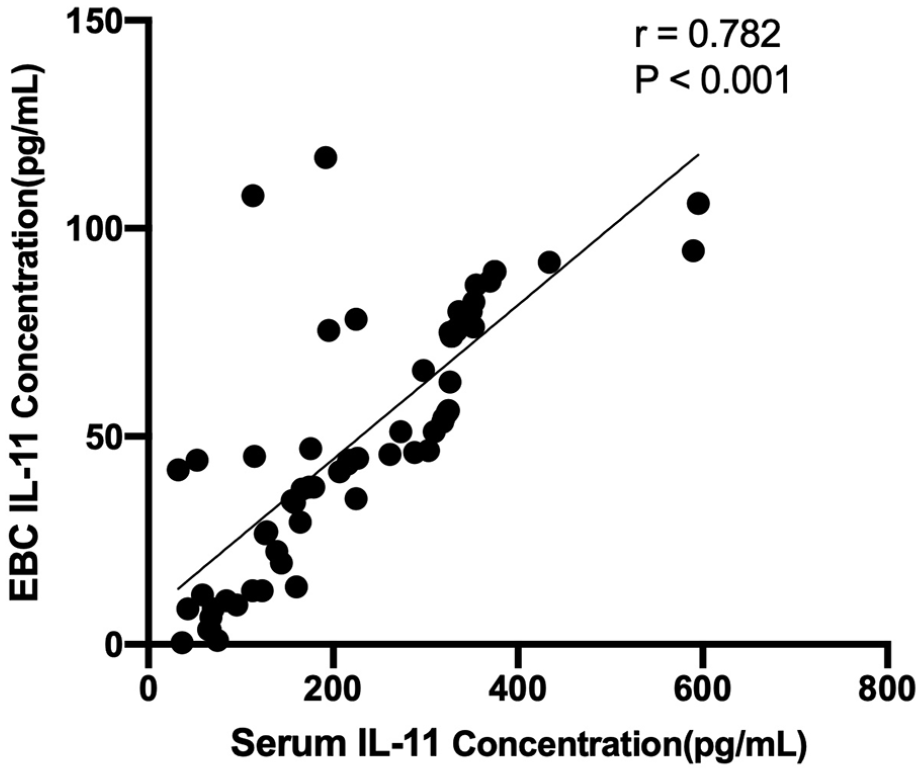
Correlation analysis of IL-11 in serum and EBC (n = 64). EBC exhaled breath condensate. Correlation analysis was performed using the Pearson correlation.
Diagnostic values of serum IL-11, EBC IL-11, and CEA alone and combined biomarkers in NSCLC (n = 64)
We used ROC curves to analyze and evaluate the diagnostic value (equal to the area under the curve, AUC) of individual and combined biomarker (Figure 4 and Table 6). In terms of a single indicator, serum IL-11 had the highest diagnostic value, with the AUC of 0.926 (95% CI 0.883, 0.970; P < 0.001), and sensitivity and specificity of 75.00% and 100.00%, respectively. However, CEA has the highest sensitivity to distinguish patients with NSCLC from healthy volunteers, which is 92.19%. The combined detection of serum IL-11 and EBC IL-11, the AUC was 0.929 (95% CI 0.883, 0.974), which was higher than the diagnostic value of a single indicator (P < 0.001), the sensitivity and specificity were 76.60% and 98.40%, respectively. In addition, the combination of serum IL-11 with EBC IL-11, and CEA could show significantly higher diagnostic value compared with the use of any one marker alone, with an AUC of 0.944 (95% CI 0.908, 0.981; P < 0.001); sensitivity and specificity were 78.10% and 100.00%, respectively.
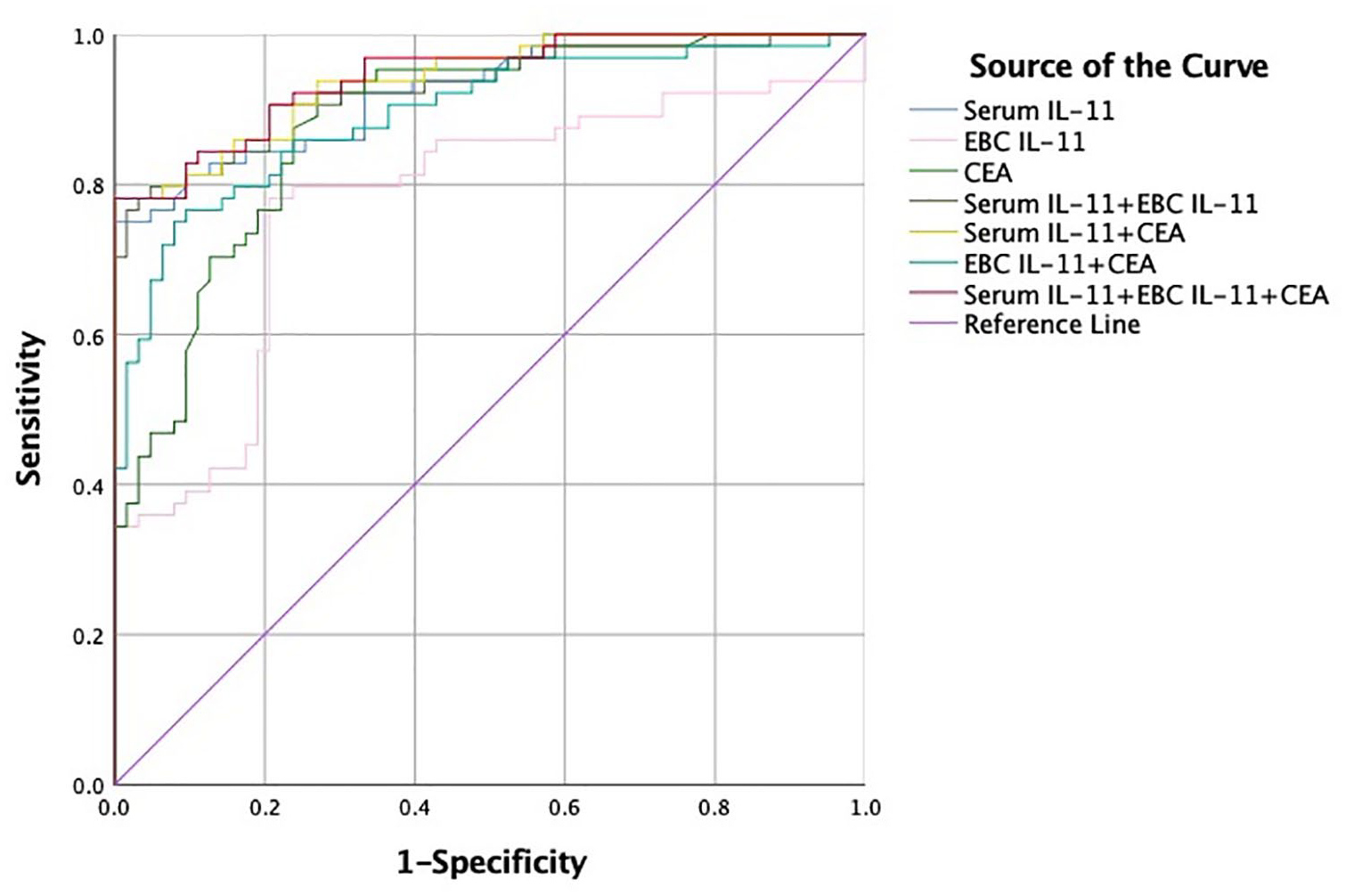
The ROC curve analysis for serum IL-11, EBC IL-11, and CEA alone and combined biomarkers in non-small cell lung cancers.
The diagnostic values of serum IL-11, EBC IL-11 and CEA alone and combined biomarkers in non-small cell lung cancers (n = 64).
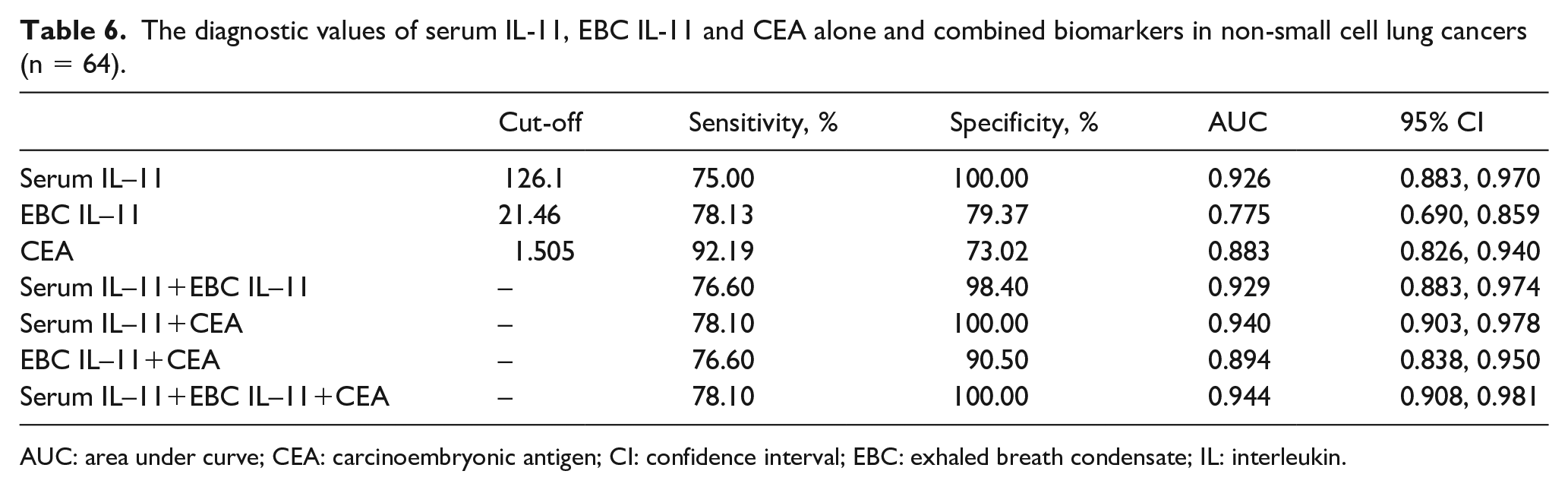
AUC: area under curve; CEA: carcinoembryonic antigen; CI: confidence interval; EBC: exhaled breath condensate; IL: interleukin.
Correlation between IL-11 level and poor prognosis in NSCLC
We analyzed the relationship between the IL-11 expression level and the prognosis of NSCLC from public data in online databases by using the Kaplan–Meier overall survival analysis. The log-rank test demonstrated that the median OS was 78 months in the low IL-11 expression group and 66.47 months in the high IL-11 expression group, respectively (P = 0.025, Figure 5(a)). 16 In addition, compared with the low IL-11 expression group, patients in the high IL-11 expression group had a significantly poorer prognosis in terms of FP (P < 0.001, Figure 5(b)). However, as shown in Figure 5(c), the difference between the IL-11 expression level and the PFS of NSCLC was not significant (P = 0.052).
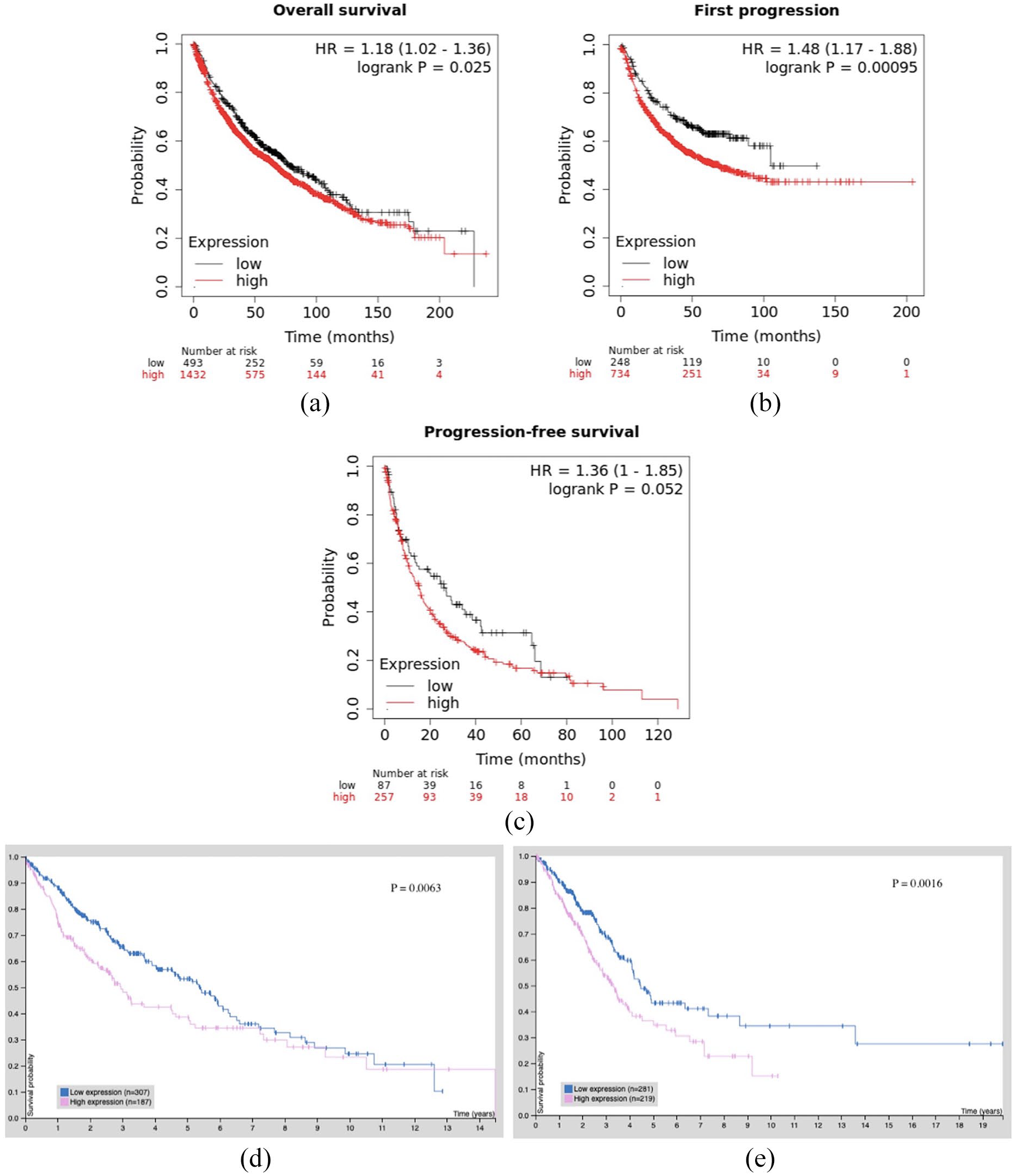
Correlation between IL-11 mRNA level and poor prognosis in non–small cell lung cancer. Kaplan–Meier overall survival analysis (a), progression-free survival (b), and first progression (c) of IL-11 expression in NSCLC from a public data set (http://kmplot.com/analysis). Kaplan–Meier overall survival analysis of IL-11 expression in squamous cell carcinoma (d) and adenocarcinoma (e) from Human Protein Atlas (http://www.proteinatlas.org).
Then, we analyzed the relationship between the IL-11 expression level and the OS of different histological types of NSCLC. 17 In the lung squamous cell carcinoma group, the median OS was 1.82 years, and the OS of the low IL-11 expression level group was significantly higher than that of the high IL-11 expression level group (P = 0.0063, Figure 5(d)). Similarly, in the lung adenocarcinoma group, the median OS was 1.79 years, and the OS of the high IL-11 expression group was significantly lower than that of the low IL-11 expression group (P = 0.0016, Figure 5(e)).
Discussion
Global cancer epidemiology has revealed that lung cancer is the main cause of cancer-related deaths worldwide. 1 Therefore, actively searching for relatively simple and rapid biological markers for early lung cancer diagnosis is necessary. In this work, we studied the expression levels of inflammatory factor IL-11 in the serum and EBC of patients with NSCLC, analyzed the relationship between IL-11 and the clinical biological characteristics of NSCLC, and analyzed the diagnostic value and prognostic significance of IL-11 in NSCLC to explore and determine whether the inflammatory factor IL-11 could be used as an effective biomarker for the diagnosis and prognosis of NSCLC.
IL-11 is an important member of the IL-6 cytokine family, which has biologically active functions. It is mainly produced by interstitial cells in the body. As a signal molecule, IL-11 must bind to its specific transmembrane receptor, IL-11 receptor (IL-11Rα), to mediate a variety of cell transmission pathways. According to research on the IL-6-mediated signal transduction pathway, it can be said that the classical IL-11-mediated signal transduction pathway is induced by the binding of soluble IL-11 to the transmembrane receptor IL-11Rα, which interacts with membrane-bound gp130 to form a trimeric complex. Then, this trimer is combined with the second IL-11–IL-11Rα–gp130 trimer to form a functional hexameric signaling complex. In this case, the Janus kinase–signal transducer and activator of transcription (JAK–STAT) signaling pathway is activated, and it participates in various biological processes, such as cell proliferation, migration, differentiation, apoptosis, and immune regulation in the body.18,19 According to recent studies, IL-11 expression in gastric cancer, breast cancer, and other tumors have a significant correlation with STAT3 activation in epithelial cells.11–12,20–21 Thus, IL-11 may participate in the occurrence and development of a variety of tumors through the gp130/JAK/STAT signaling pathway.
The expression level of inflammatory factor IL-11 is increased in a variety of tumors, including liver cancer, gastric cancer, colon cancer, and breast cancer.11–12,20–21 In addition, many recent studies have revealed that patients with gastric and colon cancer whose IL-11 expression level was high have a poor prognosis.11,20 Existing studies indicate that the inflammatory factor IL-11 is a key cytokine in the development of inflammation-associated gastrointestinal tumors. 11 Inhibiting the IL-11 signaling pathway can reduce the proliferation of cancer cells and induce the apoptosis of cancer cells, thereby inhibiting the development of a tumor.18,22 Our present study revealed that the inflammatory factor IL-11 is up-regulated in NSCLC and is closely related to the malignant progression and poor prognosis of NSCLC.
In this study, the expression levels of IL-11 in serum and EBC can significantly distinguish patients with NSCLC from healthy controls. Moreover, IL-11 expression in NSCLC patients with III–IV stage was remarkably higher than that in patients with I–II stage. This result suggests that IL-11 might act as an oncogene in NSCLC and is related to tumor progression. In addition, the correlation analysis showed a significant correlation between serum IL-11 and EBC IL-11. Our study proved for the first time that the inflammatory factor IL-11 can be detected from EBC specimens collected from patients with NSCLC, and its level was up-regulated with the increase of NSCLC severity.
For patients with respiratory diseases, traditional sample collection methods include sputum collection, induced sputum and bronchoalveolar lavage fluid (BALF). Sputum specimens are susceptible to a variety of internal and external environment, resulting in a marked decline in sensitivity and specificity. Induced sputum requires patients to inhale hypertonic saline by atomization, which can stimulate the bronchus and aggravate the original respiratory symptoms. 23 BALF is an invasive examination and cannot be widely used in clinical practice. Therefore, EBC detection is a new method. The content and dynamic changes of biomarkers in EBC specimens have important clinical significance in the diagnosis and prognosis evaluation. 24 Therefore, detecting IL-11 up-regulation in EBC specimens of patients with NSCLC is of great significance for clinicians in disease assessment and clinical diagnosis and treatment.
The “tumor microenvironment” concept can be traced back to the “seed and soil” hypothesis proposed by Stephen Paget in 1889. 25 This hypothesis states that tumor cells can remodel the tumor microenvironment, and in turn, tumor microenvironment remodeling can further affect the biological behavior of tumor cells. A growing body of evidence shows that the host’s systemic response to tumor cells can cause inflammation, and systemic inflammation plays an indispensable role in the occurrence and development of various tumors. 26 On the one hand, systemic inflammatory response stimulates the immune process of the body, thereby inhibiting tumor development. On the other hand, it may promote the growth, malignancy, and metastasis of tumor cells by disrupting the balance of the internal environment, suppressing immune response, inhibiting apoptosis, and activating angiogenesis. 27 In this study, we further analyzed the close relationship between the expression level of IL-11 and the systemic inflammatory response in patients with NSCLC.
We found that the peripheral blood inflammation indicators of patients with NSCLC were significantly higher than those of healthy controls, and NLR was closely related to the expression level of IL-11. NLR represents the quantitative relationship between neutrophils and lymphocytes in the body, which can fully reflect the systemic inflammation of patients. Some studies have found that NLR can be used as a biomarker to assess the progress of cancer in the inflammatory microenvironment. 28 Neutrophils are derived from the body’s bone marrow hematopoietic stem cells, accounting for approximately 50%–70% of the total number of leukocytes in the peripheral blood; they are considered as the main immune cells that protect the body from pathogenic infections and eliminate pathogens. 29 Numerous studies have shown that under the interaction of tumor cells and the tumor microenvironment, neutrophils can undergo remodeling of their own phenotype, and function and participate in the occurrence and development of tumors through various mechanisms, including the promotion of tumor cell proliferation, migration, and invasion and the formation of new blood vessels.30,31 In contrast to neutrophils, lymphocytes play a key role in the protective immunity of the body by inhibiting the proliferation and migration of tumor cells. Lymphocytes can induce the death of cytotoxic cells and produce cytokines, such as IL-4 and IL-12, which inhibit tumor cell proliferation and metastatic activity.32,33 Therefore, a decrease in the number of lymphocytes can cause a decrease or disappearance of the body’s immune response to tumors, thereby promoting tumorigenesis and development. Therefore, NLR can be used as a reliable biomarker to reflect the degree of systemic inflammatory response during tumor progression in the body. Our research confirmed that the expression level of inflammatory factor IL-11 is closely related to the expression of NLR in the body, indicating that the inflammatory factor IL-11 can change the tumor microenvironment in patients with NSCLC through systemic inflammatory response and participate in the occurrence and development of NSCLC.
CEA, a classic tumor marker, not only indicates the occurrence and development of lung cancer but also is directly related to tumor infiltration and metastasis. CEA can well reflect the biological activities of tumor cells, such as proliferation, infiltration, invasion, and migration.34–36 However, we found that the specificity of CEA is not high, which has a certain interference effect in the diagnosis of lung cancer. Therefore, we considered combining multiple biomarkers to improve the effective diagnosis rate of NSCLC. Through the analysis of ROC curves, we found that the combination of serum IL-11 with EBC IL-11 and CEA could show significantly higher diagnostic value compared with the use of any one marker alone, with a larger AUC area and higher specificity. In addition, public data from online databases showed that high IL-11 expression is closely related to the poor prognosis of NSCLC. Therefore, the inflammatory factor IL-11 can better diagnose NSCLC and assess the progress and prognosis of cancer.
This study has some limitations. First, the sample size was too small and must be expanded in further studies. Second, although IL-11, an inflammatory factor, is closely related to the occurrence and development of NSCLC and provides a new insight into the inflammatory microenvironment to promote tumorigenesis, the specific mechanism of IL-11 on NSCLC is still unclear, and relevant basic experiments are still needed. Third, this work is a retrospective analysis, which included manual extraction and input of clinical data. Although data on laboratory values or basic patient information were complete and reliable, the risk of patient selection bias is possible. Therefore, a large-scale prospective study and basic experiments are needed to confirm the results of the current study.
In conclusion, the expression levels of IL-11 in serum and EBC are up-regulated in NSCLC and are closely related to the malignant progression and poor prognosis of NSCLC. Therefore, the inflammatory factor IL-11 can be used as a potential diagnostic and prognostic biomarker for NSCLC and is expected to become a potential therapeutic target for lung cancer.
Footnotes
Abbreviations
AUC: area under the curve; BALF: bronchoalveolar lavage fluid; CEA: carcinoembryonic antigen; CI: confidence interval; EBC: exhaled breath condensate; ELISA: enzyme-linked immunosorbent assay; FP: first progression; HRR: hemoglobin-to-red blood cell distribution width ratio; IL-11: interleukin-11; JAK–STAT: Janus kinase–signal transducer and activator of transcription; LMR: lymphocyte-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; NSCLC: non-small cell lung cancer; NSE: neuron-specific enolase; OS: overall survival; PFS: progression-free survival; RDW: red blood cell distribution width; ROC: receiver operating characteristic; SCC: squamous cell carcinoma antigen.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants: Postgraduate Research & Practice Innovation Program of Jiangsu Province (Grant No. SJCX19_0873); the Natural Science Foundation of Jiangsu Province (Grant No. BK20191207); the Scientific Research Project of Health Commission of Jiangsu Province (Grant No. H2018035); the Scientific Research Project of “333 Project” in Jiangsu Province (Grant No. BRA2018224); and the Science and Technology Program of Nantong City (Grant No. HS2018002 and JCZ18130).
