Abstract
Purpose:
Genomic signatures, such as EndoPredict®, may help clinicians to decide which adjuvant treatment is the most appropriate.
Methods:
We propose the EndoPredict® assay for unclear cases of adjuvant treatment in patients treated in our comprehensive cancer center. We prospectively and retrospectively report the decision of adjuvant treatment before and after the EndoPredict® assay, respectively, compared to the PREDICT’s tool scores.
Results:
From November 2016 to March 2019, 159 breast cancer tumors were analyzed and presented before and after the EndoPredict® assay. Before the EndoPredict® results, clinicians recommended chemotherapy for 57 patients (57/159, 36%). A total of 108 patients (108/159, 68%) were classified as EPclin high-risk score. There was only a slight agreement between clinicians’ decisions and EPclin risk score. The EPclin score led to 37% changes in treatment (59/159); chemotherapy was favored in 80% of cases (47/59). The PREDICT tool recommended chemotherapy for 16 high-risk patients (16/159, 10%).
Conclusion:
Although genomic tests were developed in order to de-escalate adjuvant treatment, in our comprehensive cancer center the use of the EndoPredict® assay led to an increase in prescribed chemotherapy.
Introduction
Early-stage endocrine-receptor (ER)-positive HER2 negative breast cancer remains a heterogeneous disease. In this population, hormonal therapy is a standard of care irrespective of tumor size, node involvement, or grade,1–3 to decrease the risk of local and distant relapse.
The addition of adjuvant chemotherapy to endocrine therapy in this population remains an unfilled gap. Significant toxicities are associated with chemotherapy with a clinically significant 2%–3% of grade 3/4 adverse events, which requires selection patients with a high-risk of recurrence. Selection of these patients was, until a few years ago, solely based on prognostic factors such as tumor size, histological grade, vascular invasion, and nodal status.4–6 Despite molecular classification, luminal cancers are a heterogeneous entity with different clinical outcomes and benefits from chemotherapy or endocrine therapy. 7
Some models—using known clinical or pathological prognostic factors (tumor grade, size, proliferation, nodal status)—estimate survival and adjuvant treatment benefit. The British PREDICT online tool classified patients in three groups: (a) low risk (the absolute 10-year survival benefit from chemotherapy is under 3%, and chemotherapy is not recommended); (b) intermediate risk (chemotherapy has to be discussed); and (c) high risk (the absolute 10-year survival benefit from chemotherapy is more than 5%, and chemotherapy is recommended).8–10
In recent decades, genomic tests have been developed to help oncologist with chemotherapy decisions (Mamma-print®, OncotypeDX™, Genomic Grade Index®, Prosigna®, EndoPredict®, PAM50™, Breast Cancer Index™, UPA-PAI1). International consensus recommends the use of one of them in unclear cases.1,11
EndoPredict® is a second-generation genomic test that combines gene expression (eight functional genes (AZGP1, BIRC5, DHCR7, IL6ST, MGP, RBBP8, STC2, UBE2C) and three normalized genes (CALM2, OAZ1, RPL37A) (EP score), and clinical information (nodal status and tumor size). The EP risk score ranges from 0 to 15 with the higher values relating to a higher risk of relapse. An EP score < 5 was considered as low risk, and an EP score ⩾ 5 was categorized as high risk. An EPclin risk score was obtained with the combination of the molecular EP risk score with clinical factors; it was retrospectively validated as a prognostic marker of distant relapse at 10 years in two independent trials (ABCSG-6 and 8). Patients with an EPclin risk score < 3.32867 were identified as low risk, with an estimated risk of distant relapse < 10% with endocrine therapy during 5 years. Patients with an EPclin risk score ⩾ 3.32867 were classified as being at high 10-year risk of distant recurrence with endocrine therapy during 5 years. 12 In another retrospective analysis, patients with a high EPclin score who received chemotherapy had a significantly lower 10-year distance recurrence risk, compared to patients receiving endocrine therapy alone. 13
The test is recommended by the American Society of Clinical Oncology 14 as an option for guiding oncologists in adjuvant systemic therapy for post-menopausal patients with invasive pT1b/c or pT2, pN0-N1 breast cancer, ER-positive, HER2-negative with no evidence of distant metastasis. 15 Following the European Society for Medical Oncology (ESMO) recommendations, for patients with low-risk tumors (pT1a, pT1b, grade 1, ER high, pN0) endocrine therapy has to be the only adjuvant treatment. For high-risk tumors (one to three positive nodes with other risk factors, or with more than four nodes), chemotherapy has to be associated with endocrine therapy; for intermediate risk, genomic signatures could be proposed. 16
Genomic tests were initially designed in order to decrease the prescription of chemotherapy widely used even for stage I ER-positive HER2 negative breast cancer. Some institutions17–20 have reported their real-life experience, and demonstrated that the use of the EP assay has helped them to decrease their prescription of chemotherapy from 7% to 33%.
This single-institution retrospective study aims to compare the EPclin risk score to PREDICT’s recommendations, and to analyze the impact of this score on the chemotherapy decisions in our comprehensive cancer center.
Material and methods
In French public institutions, the Multidisciplinary Breast Tumor Board (MBTB) meetings are held every week to validate the treatment plan of all early-stage breast cancer patients. Since November 2016, in our institution, EndoPredict® analysis has been proposed for ER-positive HER2-negative patients with intermediate risk of relapse based on usual prognostic factors following ESMO recommendations and the St. Gallen expert consensus.16,21 Therapeutic decisions were registered in the MBTB before and after the result of the test in order to evaluate the impact of the score on our clinical practice. Decisions were retrospectively compared to PREDICT v2.1; the results of PREDICT v2.1 were not given to oncologists before the treatment decision.
Molecular analyses were performed in the Department of Biology of the Centre Eugène Marquis (CEM). Total RNA was first molecule extracted from one 10-µM thick formalin-fixed paraffin-embedded (FFPE) slide with a minimal ratio of 30% between the invasive tumor and the total tissue. In case of a lower ratio, macrodissection was done by the pathologist. Each gene was analyzed by reverse transcription quantitative polymerase chain reaction (RT-qPCR) in triplicate in a 96-well plate using a VERSANT® kPCR module AD. EPclin scores were calculated as described previously, using a web-based implementation to process analytical polymerase chain reaction (PCR) results into EPclin test results.
Categorical and continuous variables were described using frequencies—n (%), and medians with extreme values—M (min–max), respectively. Concordance between low and high scores, according to either EndoPredict® risk and clinical decision or with PREDICT tool recommendations, were evaluated by Cohen’s Kappa coefficients. The 95% confidence intervals (CIs) were calculated using the adjusted bootstrap percentile (BCa) method based on 1000 bootstrap replicates. Statistical analyses were performed with the last available version of the R software and of these packages: “boot” (Angelo Canty and Brian Ripley (2019). boot: Bootstrap R (S-Plus) Functions. R package version 1.3-23), and “psy” (Bruno Falissard (2012). psy: Various procedures used in psychometry. R package version 1.1. https://CRAN.R-project.org/package=psy).
Results
From November 2016 to March 2019, 159 EPclin assays were performed on ER-positive HER2 negative breast cancer in our Department of Biology CEM. Patients’ characteristics are reported in Table 1. The delay between the validation of the pathological report and the EPclin results was clinically acceptable at about 3 weeks; and the delay between the first MBTB—where the indication of EndoPredict® was decided—and the EPclin results was 6 days. The median age at time of diagnosis was 55 years (range: 23–75). A total of 102 patients (102/159, 64%) had luminal B breast cancer defined by Ki67 ⩾ 14% or progesterone expression < 20%. Eight patients (5%) had pT1c tumor, and 34 patients (21%) had micrometastasis nodal involvement. A total of 57 patients (36%) had macrometastasis nodal involvement. Most patients (117/159) had grade SBR II breast cancer.
Patient characteristics.

EP: EndoPredict®; MBTB: Multidisciplinary Breast Tumor Board.
The median EP and EPclin risk scores were 6.3 (range: 1.7–13.5), and 3.6 (range: 2.1–5.3), respectively (Table 2). Among the 159 patients, 38 (24%) were classified as EP low risk and 121 (76%, 121/159) as EP high risk. After integration of the clinical information to the molecular score, 51 patients (32%, 51/159) were considered EPclin low risk and 108 (68%, 109/159) EPclin high risk.
EP results for the whole cohort.
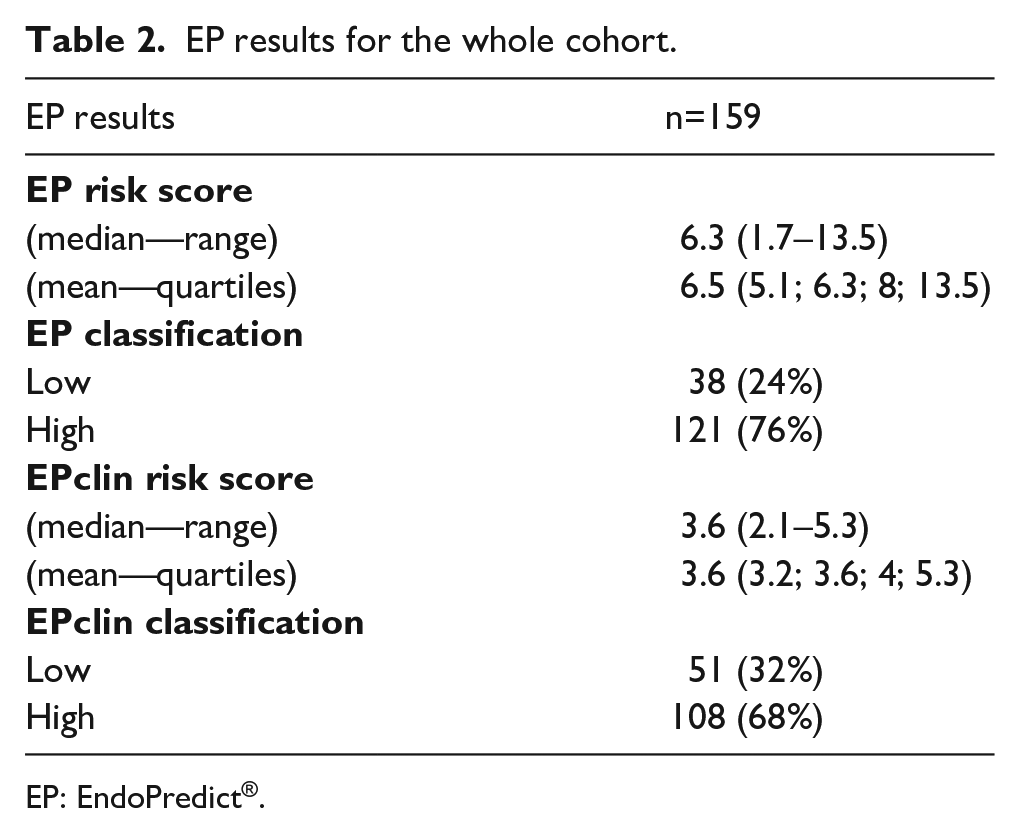
EP: EndoPredict®.
Before the EP assay, the oncologist recommended chemotherapy for 57 patients out of 159 (36%) (Table 3). For these patients, 45 were classified as high EPclin risk and 12 as low risk. For the 102 patients for whom oncologists recommended hormonotherapy, 63 were classified as high EPclin risk and 39 as low risk.
Comparison between clinical decision before results of EPclin risk score and the actual EPclin risk score.
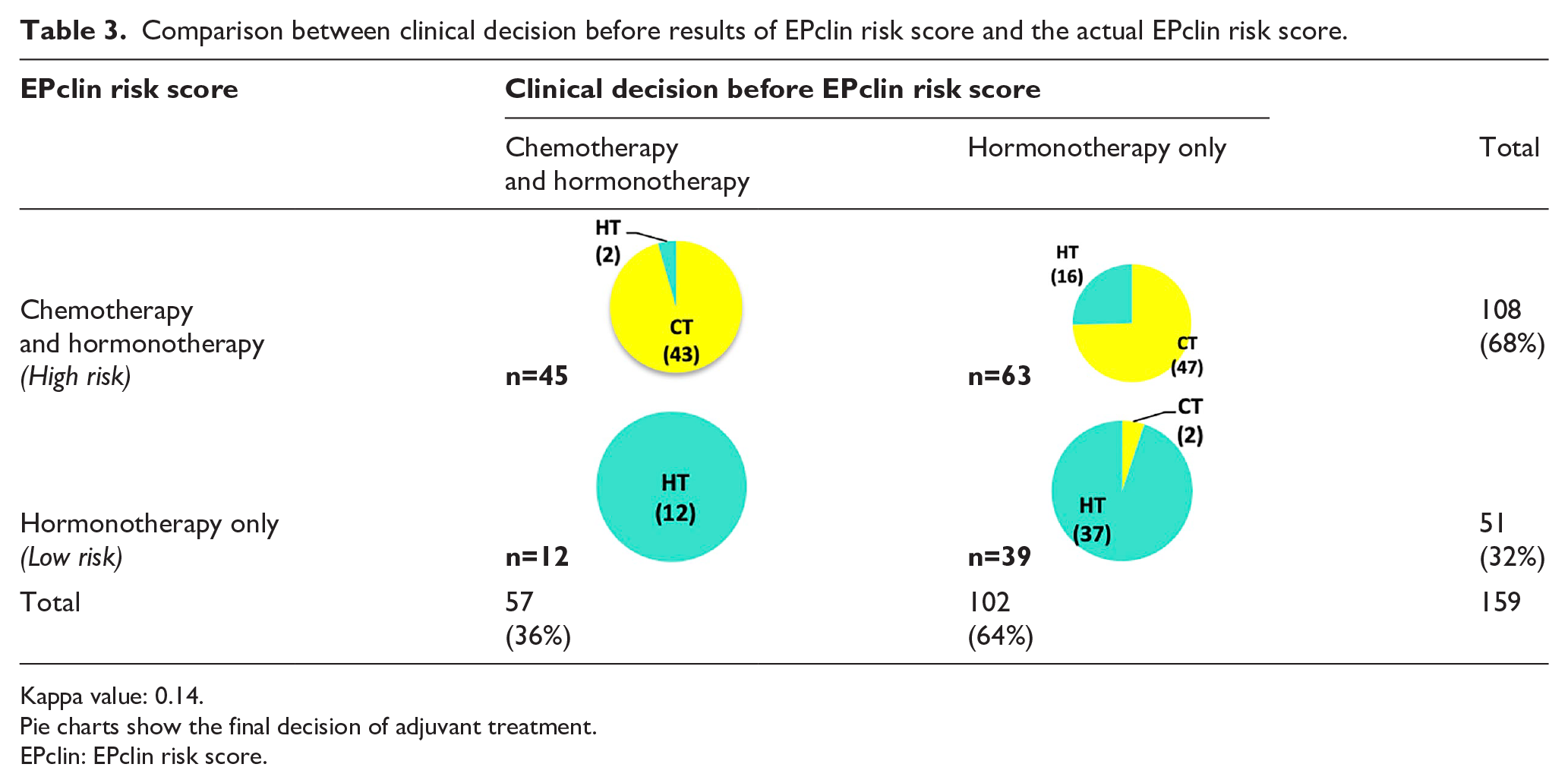
Kappa value: 0.14.
Pie charts show the final decision of adjuvant treatment.
EPclin: EPclin risk score.
Considering the EPclin risk score, 108 patients (108/159, 68%) were classified as being at a high risk of relapse and should have received chemotherapy following EP assay recommendations. The concordance rate between clinical decision and EPclin risk score was 53% with κ = 0.14 (95% CI, 0.014, 0.263), indicating a slight agreement.
Finally, 92 patients received chemotherapy: 90 with high EPclin risk, 43 (48%, 43/90) had chemotherapy recommended before testing, and 2 were classified as low risk by EPclin and clinicians before complementary axillary dissection that revealed a secondary, massive nodal involvement leading to a chemotherapy decision (Table 3). A total of 67 patients were treated with endocrine therapy: 49 patients with a low EPclin risk score (37 had this treatment planned before testing) and 18 with a high EPclin risk score. For 16 of these 18 patients, oncologists had recommended hormonotherapy before the EPclin results.
Overall, 18 patients classified as high risk did not receive chemotherapy: (a) for the 2 patients for whom oncologists recommended chemotherapy, 1 declined treatment and 1 was not eligible due to comorbidities; (b) 2 had micrometastasis nodal involvement (EPclin would have been low without nodal invasion); 1 was tested before axillary node dissection and final negative results made the clinician decide against chemotherapy; and (c) for the other 13 patients, the clinician and patient decided together to only have endocrine therapy because of a low EP score.
Following the EPclin results, chemotherapy was withdrawn in 12 patients (8%, 12/159) and added in 47 (30%, 47/159). The EPclin score led to 37% of changes in treatment (59/159), and in 80% of cases (47/59) chemotherapy was favored.
For comparison purposes, we also retrospectively calculated PREDICT scores. These results were not known at the time of MBTB. The PREDICT tool would recommend chemotherapy for 16 patients out of 159 (10%); endocrine therapy for 82 patients (52%, 82/159); and for one-third of patients (38%, 61/159)—classified as intermediate risk—chemotherapy had to be considered as an option (Table 4). According to EPclin, 32% patients (51/159) were categorized low risk and 68% (108/159) were considered high risk. For the 38% of patients classified as intermediate risk with the PREDICT tool, it was difficult to identify which ones would have a high genomic score and need chemotherapy.
Comparison between PREDICT tool risk score and the EPclin risk score.
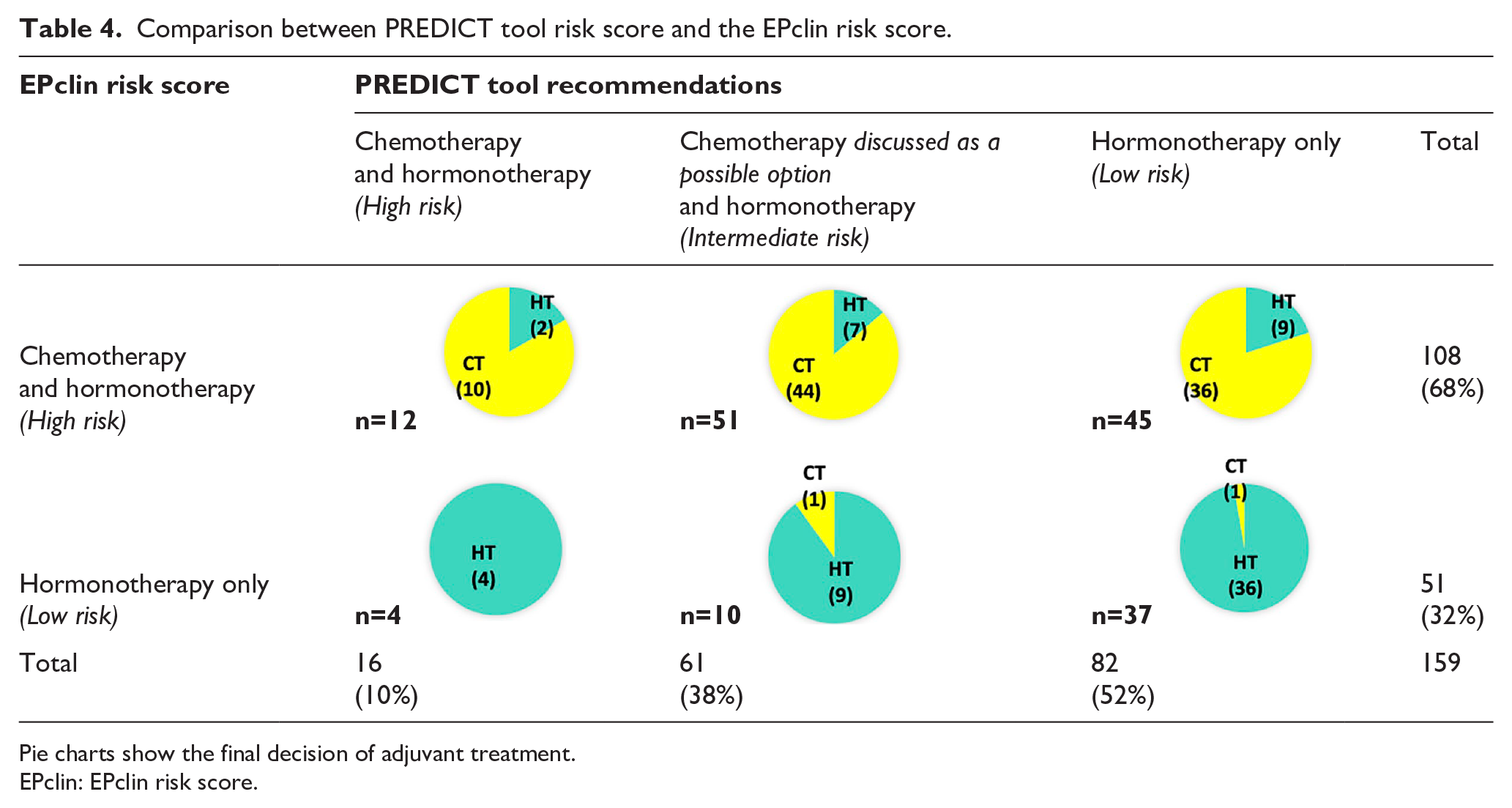
Pie charts show the final decision of adjuvant treatment.
EPclin: EPclin risk score.
Discussion
Our study demonstrates that the EP assay is reliably feasible in real life with an acceptable delay to see results.
In our cohort, the EPclin risk score led to a 37% change of planned adjuvant treatment (59 of 159 patients). For 47 of these patients (47/59, 80%) chemotherapy was added to the initially planned endocrine therapy based on EP results. There was a slight agreement between the clinical decisions on the first MBTB meeting and the EPclin risk score. Initially, following the oncologists’ decisions, chemotherapy was planned for 36% of patients (57/159); in the end, 58% of patients (92/159) were treated with chemotherapy. Our results are not those attempted; genomic signatures have been developed to decrease the prescription of chemotherapy. Our indications of the EndoPredict® assay followed the ESMO recommendations. 16 In our population, 21% (34/159) and 36% (57/159) patients had micrometastasis and pN1 nodal involvement, respectively. This relative high rate of nodal involvement could be explained by molecular analysis of sentinel lymph node by OSNA (one-step nucleic acid amplification) in our institution. Moreover, EndoPredict assay does not distinguish micrometastasis from macrometastasis nodal involvement.
Two institutions in Germany (in Berlin and in Munich) and one in Mexico17–19 have reported their experiences with the EP assay, with a change in adjuvant therapy in one-third of patients, and the decision not to have the originally planned chemotherapy in one-quarter of patients. Similar results have been observed with the Oncotype® DX 21-gene assay.22,23 More recently, a French team, Nicolaï et al., 24 reported their experience of Prosigna/PAM50. The results of the genomic signatures led to a change in adjuvant treatment in one-third of patients (21/68). Before the test, chemotherapy had been planned for 31 patients (31/68, 45%); the genomic signature classified 31 patients as high risk, and in the end 42 patients (42/68, 62%) received chemotherapy. Prosigna/PAM50 increased the prescription of chemotherapy to 23.5%. These results are similar to ours. This difference of the genomic signature impacting the therapeutic decision could be explained by heterogeneous practice not only at an international level but also at a national or regional level. Chemotherapy decisions could be different depending on geographical localization 25 ; for example, before EPclin results, chemotherapy had been planned for 63.8% patients (83/130) in the Müller et al. cohort, 17 whereas in our cohort, only 36% of patients (57/159) had been scheduled for chemotherapy (Table 3). In a prospective French multicentric trial, ADENDOM, 20 inclusion criteria were strictly defined as: fully resected localized ER positive HER2 negative, node-negative or pN1mi breast cancer grade II or grade III and pT < 2cm or lobular histology; chemotherapy was initially proposed in 48% of patients (96/201). In the end, 26% of patients (53/201) received chemotherapy after the EndoPredict® assay.
The indication of adjuvant therapy in early breast cancer is based on clinical, histological features—and in some cases, genomic data. In clinical practice, therapeutic decisions are discussed at MBTB meetings, following international, national, or regional guidelines; however, there is no standardized recommendation. 26 For instance, a survey was conducted at the 2013 Annual St-Paul-de-Vence Breast Congress on decision of adjuvant therapy for patient with early endocrine positive, Her2 negative breast cancer. 27 . For two out of four cases, 69% (217/270) and 52% (136/260) clinicians opted for chemotherapy, whereas 31% (83/270) and 48% (124/260) did not. Following National Comprehensive Cancer Network recommendations, all patients with early breast cancer with nodal involvement—whatever tumor size or grade—should receive adjuvant chemotherapy 28 ; however, the St. Gallen Expert Panel recommends chemotherapy for stage III early breast cancer and is against chemotherapy for stage I/II luminal A breast cancer. 21 In the prospective MINDACT study, 29 patients with pN1 nodal involvement could be included (21% patients (1405/6693) had node-positive disease). However, in node-positive patients, the use of a gene-expression signature is still an ongoing question. 21 The RxPONDER trial (NCT01272037) could help us in our treatment decisions. Before gene-expression signatures, the 10th St. Gallen Expert Consensus recommended chemotherapy for pN1 patients with incomplete endocrine response (a low expression of the endocrine receptor) and for pN2 patients. 30 In our population, more than a half (91/159, 57%) had nodal involvement (micro or macro metastasis).
Following the PREDICT tool recommendations for patients classified as intermediate risk, chemotherapy could be an option; however, this does not help the clinician in his/her therapeutic decision. The PREDICT tool could be useful for clinicians or patients with free online access, and give overall survival at 5 and 10 years plus the benefit of each adjuvant treatment. However, one of its major inconveniences is the intermediate risk with no clear recommendation and the absence of prospective validation. The EndoPredict® assay could help to classify patients with intermediate risk with the predict tool.
The limitations of our study include the monocentric and retrospective analysis of adjuvant decisions. The use of genomic signatures modifies our clinical practice and adds information to standard clinicopathological features. However, the changes observed are intrinsically linked to current chemotherapy recommendations, which are variable worldwide. The selection of patients that benefit more from this genomic information remains an unmet need.
Conclusion
The EndoPredict® assay could be routinely used. The EPclin score is used to help with adjuvant therapy decisions for breast cancer patients. Genomic signatures were initially designed in order to decrease chemotherapy prescription. In our population, the EPclin score led to 37% changes in treatment, and in 80% of cases, chemotherapy was favored, because most of our patients (108/159, 68%) were categorized as high risk. Genomic signatures help clinicians to make therapy decisions, but the final decision is based on multifactor parameters. Molecular tests could help oncologists to standardize their prescription of adjuvant treatment for breast cancer patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
