Abstract
Background:
MicroRNAs (miRNAs), a class of small non-coding, highly stable RNAs, have been reported to have diagnostic value for variety types of cancers.
Objectives:
To assess the diagnostic value of circulating miR-145 for non-small cell lung cancer (NSCLC) by using systemic review and meta-analysis.
Methods:
A systematic literature search was conducted in five databases until 20 February 2020 to identify diagnostic trials of miR-145 in the diagnosis of NSCLC. The quality of included studies was assessed by the QUADAS-2 tool with Review Manager 5.3, and the summary receiver operating characteristic (SROC) curve was plotted by STATA 13.1 software.
Results:
A total of 1394 patients from 11 data sets in trials (published in nine studies) were recruited. The area under the curve of the SROC was 0.83. According to the meta regression, the specimen selection was considered the source of heterogeneity, the SROC in serum (0.90 (95% CI 0.87, 0.92), the sensitivity was 0.84 (95% CI 0.79, 0.89), and the specificity was 0.80 (95% CI 0.71, 0.89)) was obviously higher than that in plasma (SROC=0.75).
Conclusion:
Serum miR-145 might be served as a potentially useful biomarker for NSCLC diagnosis. However, due to the existing limited-quality research, more large-scale and multicenter studies are required for further verification.
Introduction
Lung cancer is a malignant tumor with extremely high morbidity and mortality and has been a major burden due to many years of global cancer death. 1 Epidemiological research has indicated that lung cancer will remain the main public health issue in major countries before the 2050s. 2 In China, lung cancer is the leading cause of cancer death among both men and women, suggesting that it has become a major public health challenge. 3
Non-small cell lung cancer (NSCLC) is the most common histological type, comprising as high as 85% of lung cancer. 4 Currently, treatment technologies are being continuously developed, whereas control of NSCLC is still not optimistic, the diagnostic technique is considered a vital reason. 5 To the best of our knowledge, some new invasive diagnosing techniques are currently available, including endobronchial ultrasound-guided transbronchial needle aspirate and electromagnetic navigation bronchoscopy, which enhance the accuracy of NSCLC diagnosis. However, these techniques should still focus on technical factors including limited accuracy, high risk of adverse events, or safety considerations. 6 Researchers have focused more on minimally invasive sampling to optimize the diagnostic approach and the treatment of patients with NSCLC.7,8
Highly conserved non-coding RNA molecules known as microRNAs (miRNAs) participate in regulating gene expression. 9 Like many other biomarkers, aberrant miRNA expression has been shown to play an important role in incidence of human malignancies, and in recent years, numerous studies have investigated miRNAs and risks of different kinds of cancer including lung cancer.10,11 A recent study has summarized the evidence of the functional roles of miRNAs in NSCLC; and highlighted that miRNAs might have a relatively high diagnostic value for NSCLC in their clinical utility as biomarkers. 12 The diagnostic role of miR-126 and the prognostic role of miR-21 have been discussed in previous research and future perspectives of these studies, notably the need for further research of more accurate and easily standardized non-invasive biomarkers.13-15 The RNA gene miRNA-145 is located at the 5p32 chromosomal region, the expression of which is regulated by p53 and other transcriptional factors. 16 As a tumor suppressor, it has been reported to be irregularly expressed in cancers such as breast cancer, 17 gastric cancer, 18 bladder cancer, 19 colorectal cancer, 20 and ovarian cancer. 21 Recent studies demonstrated that molecular profiling of miR-145 is emerging as a key non-invasive tool for monitoring NSCLC invasion and migration, indicating that it may serve as a non-invasive biomarker for clinical application. 22 However, although several studies have reported miR-145 for the diagnosis of NSCLC, the diagnostic accuracy of these studies varies. For instance, in a small sample research by Xi et al., 23 the area under the curve (AUC) was 0.71, sensitivity and specificity were 78.6% and 73.3%, while in recent research by Sui et al., 24 variety the AUC was 0.879, while sensitivity and specificity were 84.21% and 71.67%. The specificity and sensitivity from different studies have currently limited the clinical applications of miR-145. Given its variety, it remains unclear whether miR-145 acts as a tumor suppressor or an oncogene in NSCLC, and there is an increased need for further identification of miR-145 for diagnosing NSCLC. In this study, we conducted a systematic review and meta-analysis to evaluate the effectiveness of the circulating miR-145 in NSCLC diagnosis.
Materials and methods
Data sources and search strategy
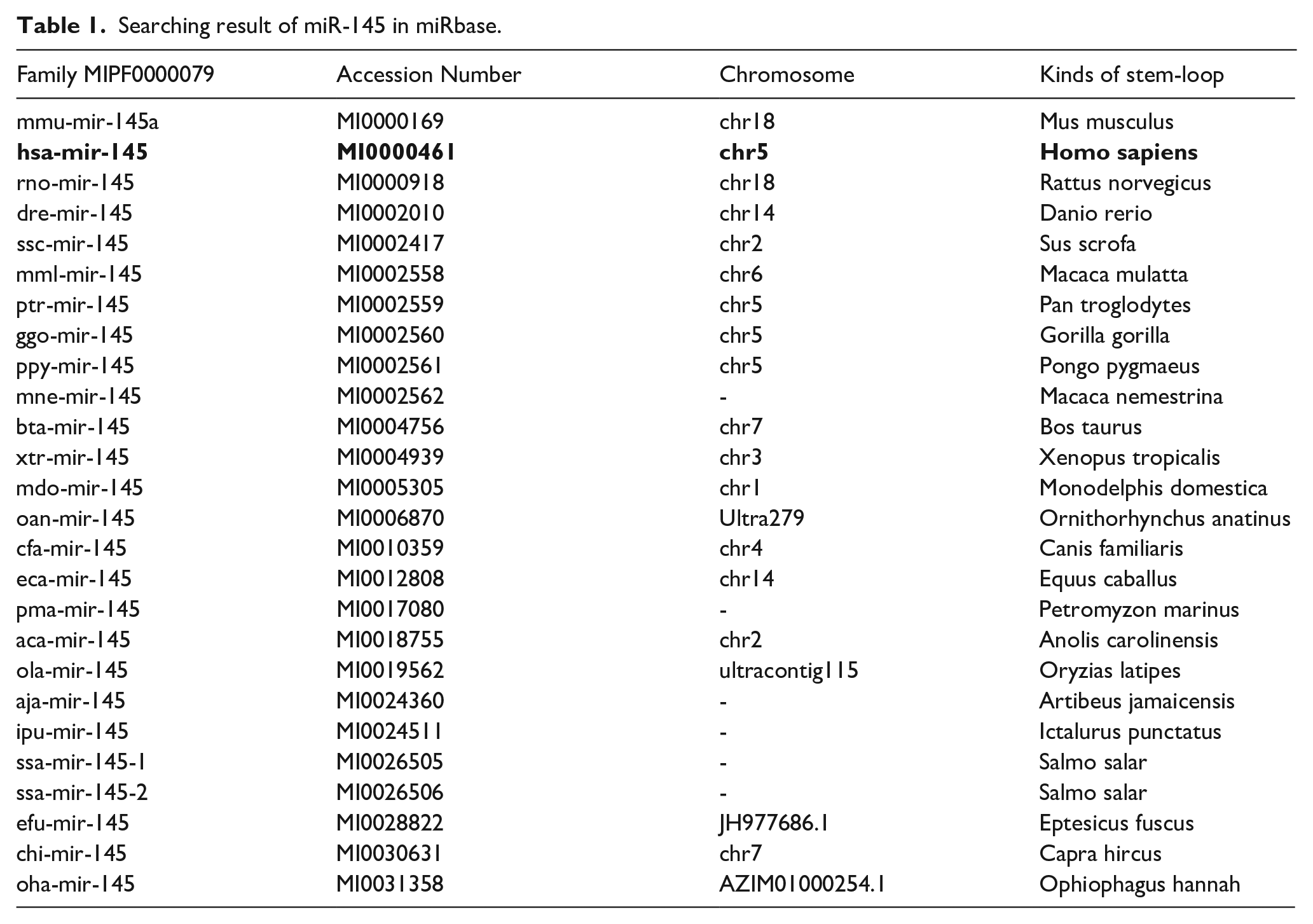
The study was conducted and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (the PRISMA 2009 Checklist is provided in the Supplementary Table 1). 25 Before the literature review, we identified miR-145 species via the miRbase (http://www.mirbase.org/) to ensure that only information regarding human miR-145 (homo sapiens miR-145) could be collected. 26 According to miRbase searching, the gene family MIPF0000079 contains 26 GENE ID entries (Table 1), which matches miR-145, hsa-mir-145 (including two mature sequences: miR-145-5p and miR-145-3p), which were finally screened as human miR-145. 27 We searched PubMed, Embase (via Ovid), Cochrane Library, Chinese National Knowledge Infrastructure (CNKI) and Wan Fang databases using the search terms: “MIRN145 microRNA, human”, “miRNA-145, human”, “miR-145, human”, “hsa-miR-145 microRNA”, “microRNA-145, human”, “miR-145-3p, human”, “miR-145-5p, human” and NSCLC (“Carcinoma, Non-Small-Cell Lung”, “Carcinoma, Non Small Cell Lung”, “Lung Carcinoma, Non-Small-Cell”, “Lung Carcinomas, Non-Small-Cell”, “Lung Carcinomas, Non-Small-Cell”, “Nonsmall Cell Lung Cancer”, “Non-Small-Cell Lung Carcinoma”, “Non-Small Cell Lung Carcinoma”, “Carcinoma, Non-Small Cell Lung” and “ Non-Small Cell Lung Cancer”), up until February 20. Subsequently, we screened further information by using manual retrieval of references from recent literature.
Searching result of miR-145 in miRbase.
Inclusion and exclusion criteria
Inclusion criteria: (a) published as complete article and studied issues including expression of miR-145 and diagnosing value of NSCLC; (b) objects of study: suspected confirmed NSCLC patients; (c) diagnostic method: all NSCLC patients were diagnosed with NSCLC based on the clinical gold standard; (d) evaluating indicators: provide enough data for sensitivity, specificity in order to calculate true positive (TP), false positive (FP), false negative (FN) and true negative (TN) results for each investigation.
Exclusion criteria: (a) the gold standard of diagnosis of NSCLC was not mentioned; (b) there is no receiver operating characteristic (ROC) curve, single sensitivity and specificity of the miR-145 during diagnosis; (c) the literature could not be used because of repeated reporting (if a study reported on a previously reported dataset, the larger and better-quality paper was used for analysis) and poor quality; (d) review literature, abstracts, and lectures, etc. relating to non-original research and basic research such as animal experiments.
Data extraction and quality assessment
The following information was extracted for enrolled studies: first author, year of publication, study design, country, and sample sizes (e.g. numbers of experimental groups and control groups), sample types, major experimental methods, the gold standard, the AUC, sensitivity and specificity. The numbers of TP, FP, FN, and TN were recorded directly or calculated indirectly according to the original data of enrolled studies. Two investigators (ST and XJ) independently performed the literature search and data extraction to ensure the quality of the meta-analysis. If necessary, a third author (QZ) was asked to resolve disagreements between the investigators. Study quality was independently assessed by using a tool for the quality assessment of diagnostic accuracy studies, band-2 (QUADAS-2), provided by the Cochrane center. 28
Statistical methods
The quality of included studies was assessed by using the QUADAS-2 tool from RevMan 5.3 (Cochrane Collaboration, Oxford, UK). The meta-analysis was conducted by STATA 13.1 (Stata Corporation, College Station, TX, USA). Heterogeneity caused by influencing factors was primarily tested by I2 as a testing standard. I2 ⩽ 50% suggested no statistical heterogeneity, while I2 > 50% indicated statistical heterogeneity. If statistically significant evidence of heterogeneity was found in the overall sensitivity and specificity analysis, the meta-regression analysis was further performed by verifying potential influencing factors including sample size (more than 100 or not), blindness (blinded or not), history of publication (>3 years or <3 years), specimen selection (plasma or serum), and stage of disease (early or otherwise). The Cochrane’s Q statistic (α=0.05) was also used for the heterogeneity test. The Fagan nomogram was used to test the clinical utility of diagnostic indicators by setting different pretest probabilities. The likelihood ratios were utilized to simulate three clinical scenarios by implementing different pretest probabilities, 25% indicating relatively low clinical suspicion, 50% indicating moderate clinical suspicion, and 75% suggesting a relatively high clinical suspicion. 29
Results
A total of 350 citations, including reports in English and Chinese, were primarily retrieved after the initial database search, complying with inclusion and exclusion criteria, and 11 data sets in trials from nine studies (seven studies in English23,24,30-34 and two studies in Chinese35,36 were finally enrolled for further analysis (Figure 1). The basic characteristics included authors, publication years, countries, specimens, and testing methods; the results of their QUADAS-2 evaluation are presented in Table 2 and Figure 2.
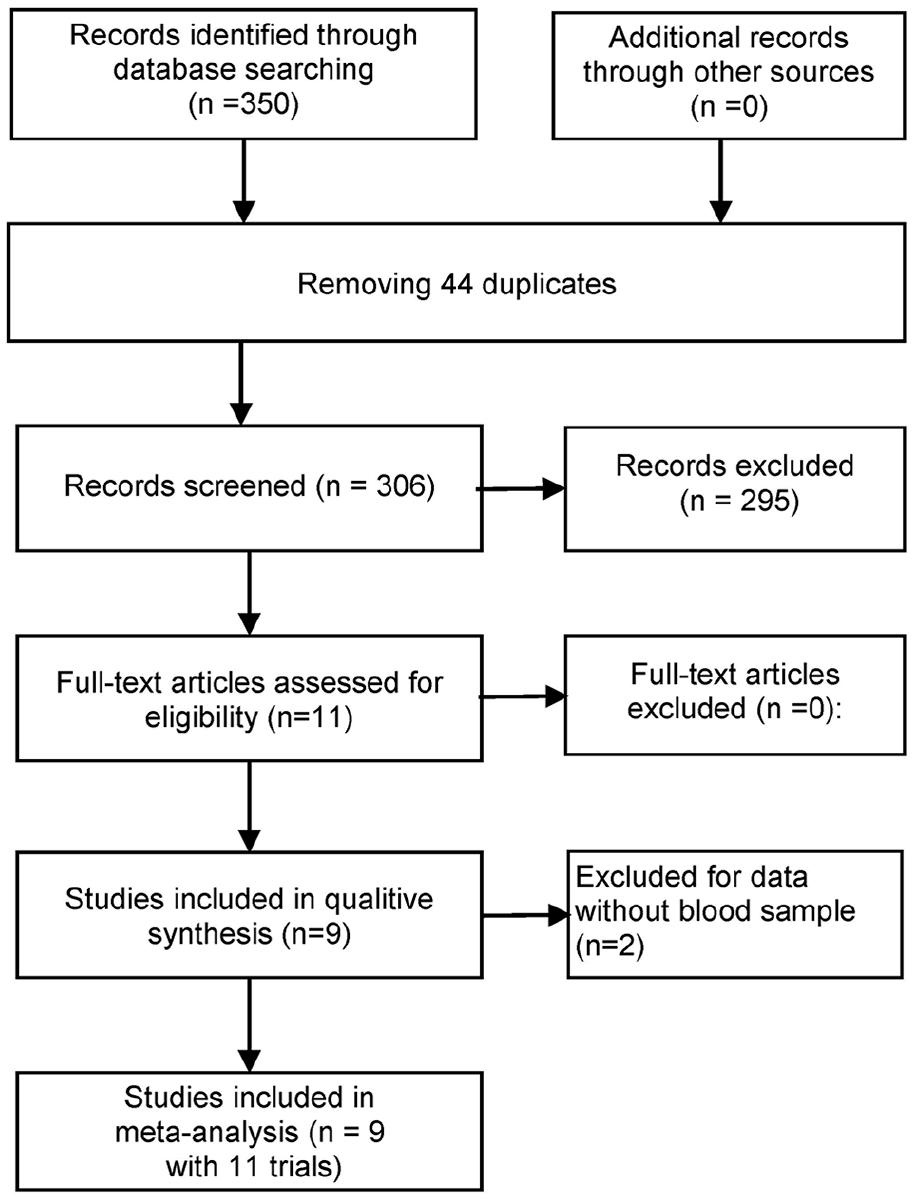
Flow diagram of the meta-analysis of miR-145 for NSCLC.
Characteristics of trials included in the meta-analysis.
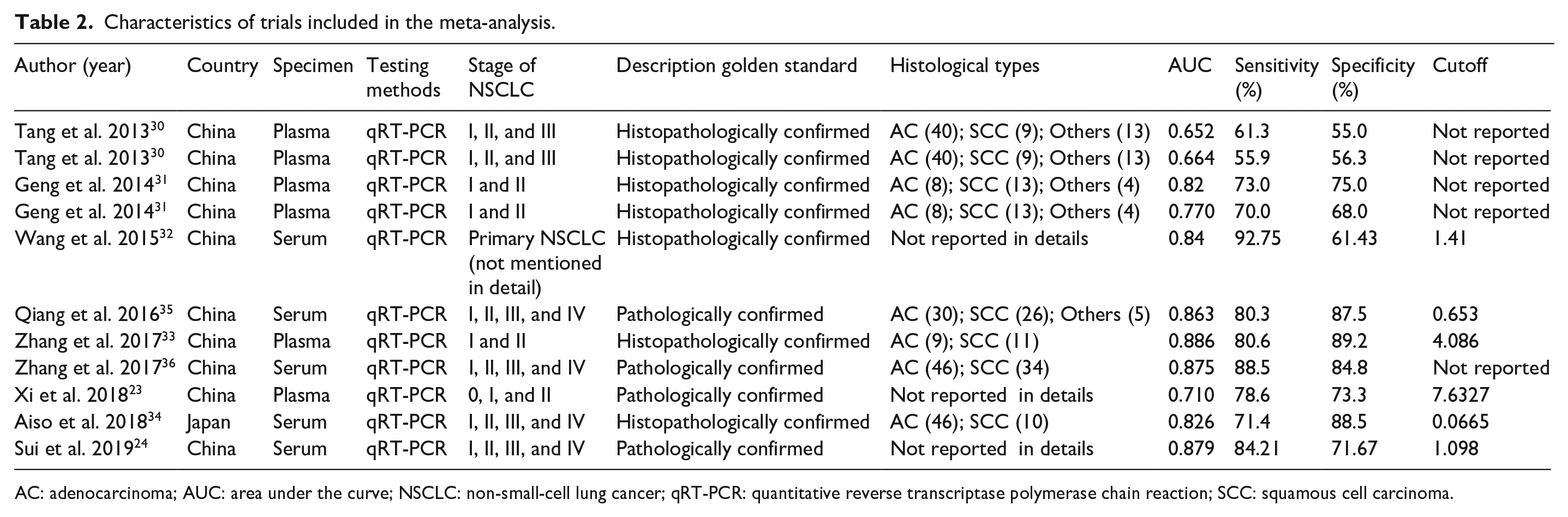
AC: adenocarcinoma; AUC: area under the curve; NSCLC: non-small-cell lung cancer; qRT-PCR: quantitative reverse transcriptase polymerase chain reaction; SCC: squamous cell carcinoma.
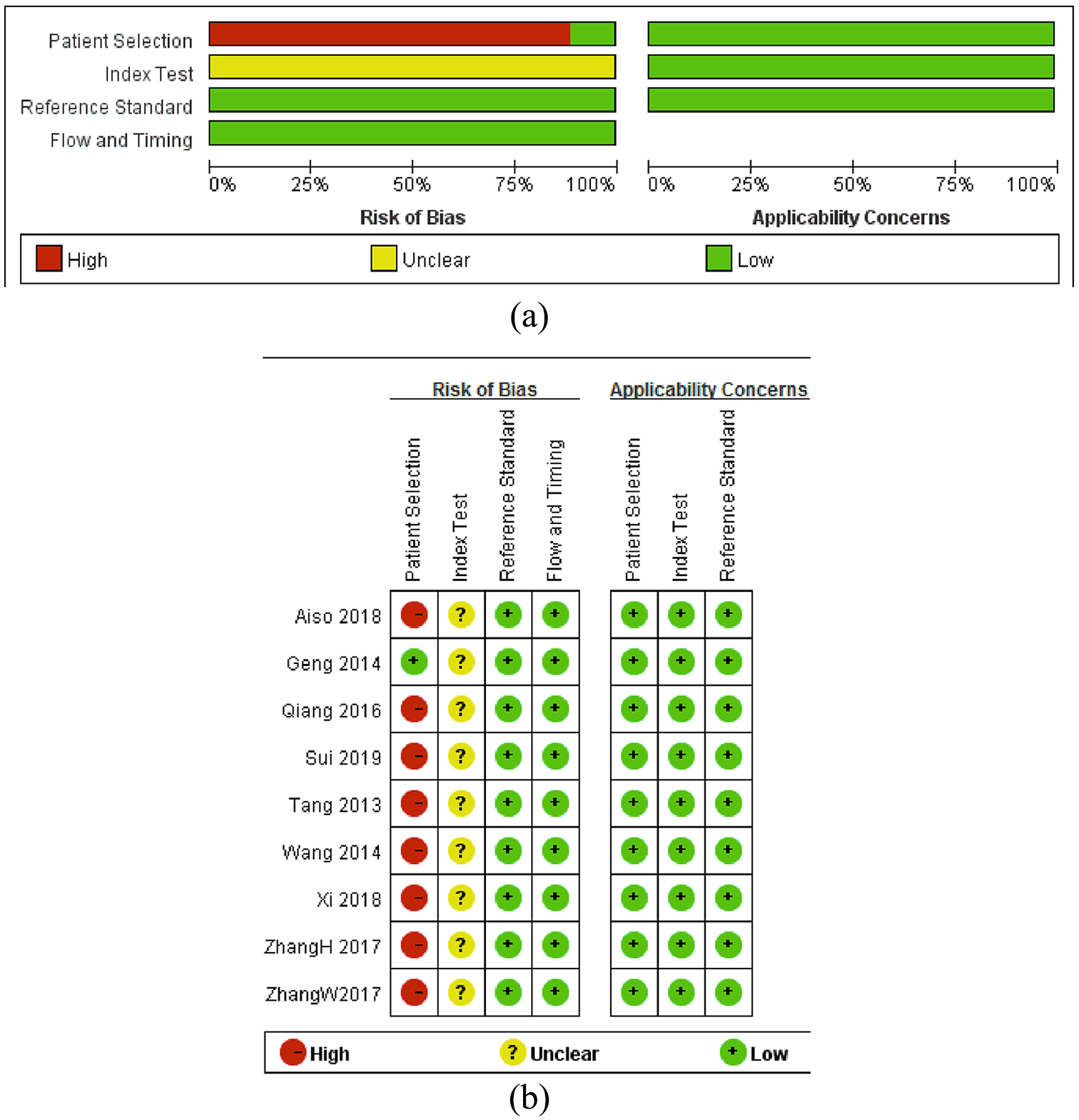
Quality assessment of the included studies by QUADAS-2. (a) Methodological quality graph. (b) Methodological quality summary.
Quality assessment of methodology
According to the literature quality assessment using the QUADAS-2 tool, 10 trials were identified as case-control studies (n=10), whereas only one study was identified as a prospective clinical trial. 31 In patient sampling quality assessment, we found that patient sampling in most studies was unclearly described, which was also one of the biases of this study. During the flow and timing part of quality assessment, no literature was found regarding the assignment interval between miR-145 testing and the golden standard. However, we considered that the time of miR-145 testing and intervals of the gold standard were low-risk events since this study was only about NSCLC patients, the length of intervals did not change the risks of disease.
Meta-analysis results
Meta-analysis was performed and the results showed the bivariate summary receiver operating characteristic (SROC) graph with 95% confidence, and the AUC was 0.83 (95% CI 0.80, 0.86) (Figure 3). Additionally, miR-145 had a pooled sensitivity of 0.78 (95% CI 0.71, 0.83) and a pooled specificity of 0.75 (95% CI 0.67, 0.82) in its ability to diagnose NSCLC (Figure 4). miR-145 had a pooled positive likelihood ratio of 3.1 (95% CI 2.2, 4.4) and a pooled negative likelihood ratio of 0.30 (95% CI 0.21, 0.41); the diagnostic odds ratio was 11 (95% CI 6, 20).
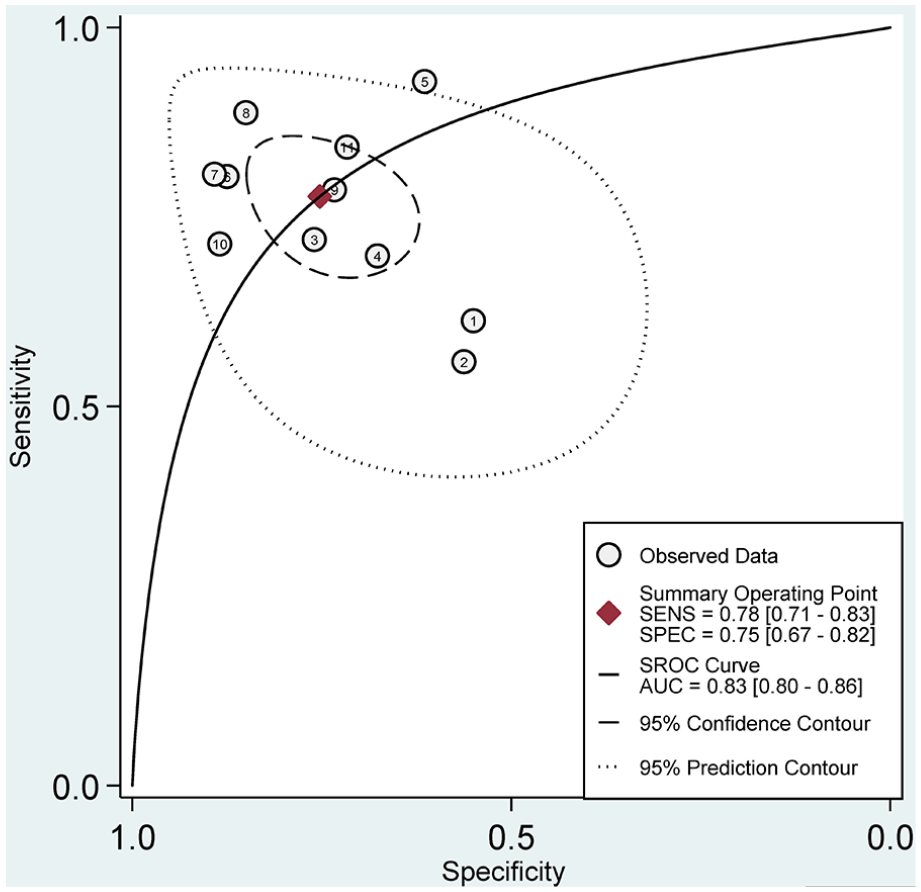
Summary receiver operating characteristic curve of miR-145 for diagnosing NSCLC.
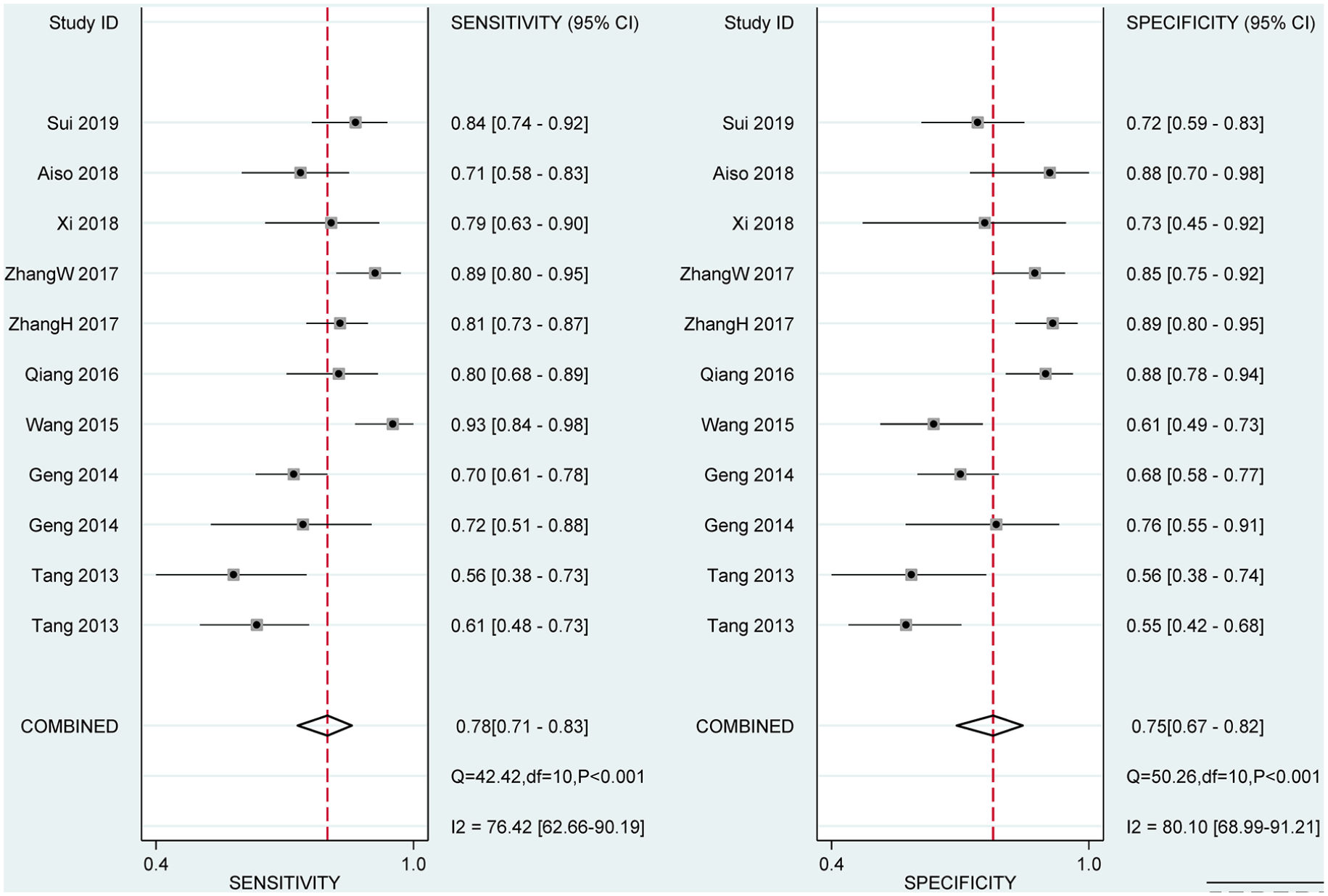
Forest plot showing trial-specific and mean sensitivity and specificity of miR-145 for NSCLC with corresponding heterogeneity statistics.
Meta-regression and subgroup analysis results
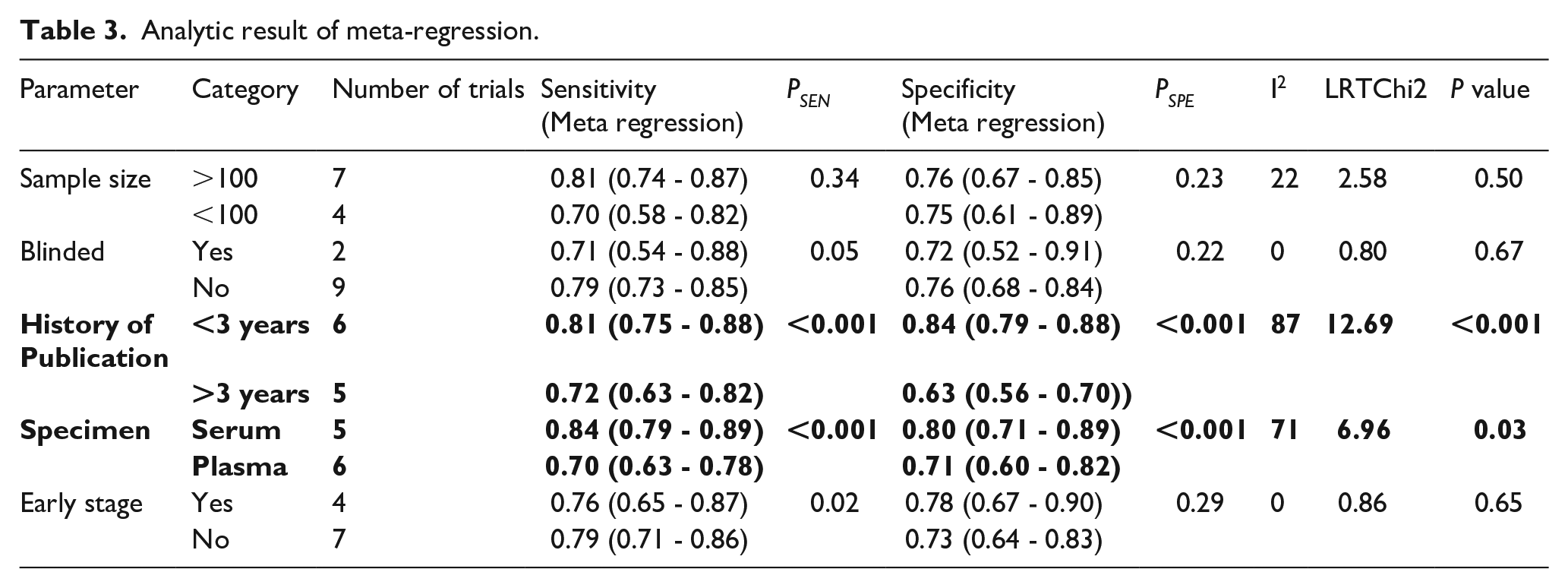
The Deeks’ funnel plot asymmetry test recognized no evidence of notable publication bias (t=0.73, P=0.48). According to the Cochran Q test and I2 of the pooled sensitivity (Cochran Q test =42.42, P<0.001, I2 = 76.42%) and specificity results (Cochran Q test = 50.26, P<0.01, I2 = 80.10%), heterogeneity was found among the included articles, and the meta regression was performed to screen for influencing factors. The results indicated that the history of publication could be the source of heterogeneity (LRTChI2=12.69, P<0.01); for research published in the past 3 years, the SROC of miR-145 for diagnosing NSCLC was 0.88 (95% CI 0.85, 0.90), with the pooled sensitivity of 0.81 (I2=30.71) and the pooled specificity of 0.84 (I2=54.02). For research published more than 3 years ago, the SROC was 0.64 (95% CI 0.60, 0.68); the pooled sensitivity was 0.72 (I2=82.76); and the specificity is 0.63 (I2=21.56). Specimen selection was also an important source of heterogeneity (LRTChI2=6.96, P=0.03); the AUC of miR-145 for diagnosing NSCLC in serum was 0.90 (95% CI 0.87, 0.92); the sensitivity was 0.84 (I2=68.81); and the specificity was 0.80 (I2=82.48). For plasma the AUC was 0.75 (95% CI 0.68, 0.82); the sensitivity was 0.70 (I2=50.6); and the specificity was 0.71 (I2=78.4) (see Table 3).
Analytic result of meta-regression.
Clinical utility
The clinical utility analysis was used in Figure 5 to show the relations between pre-test and post-test probabilities and likelihood ratio. Due to a pretest probability of 25%, the post probability positive (PPP) and post probability negative (PPN) were 51% and 9%, respectively; with a pretest probability of 50%, the PPP and PPN reached 76% and 23%, respectively; with a pretest probability of 75%, the PPP and PPN were 90% and 47%, respectively, which demonstrated a good clinical performance of miR-145 for a NSCLC diagnosis.
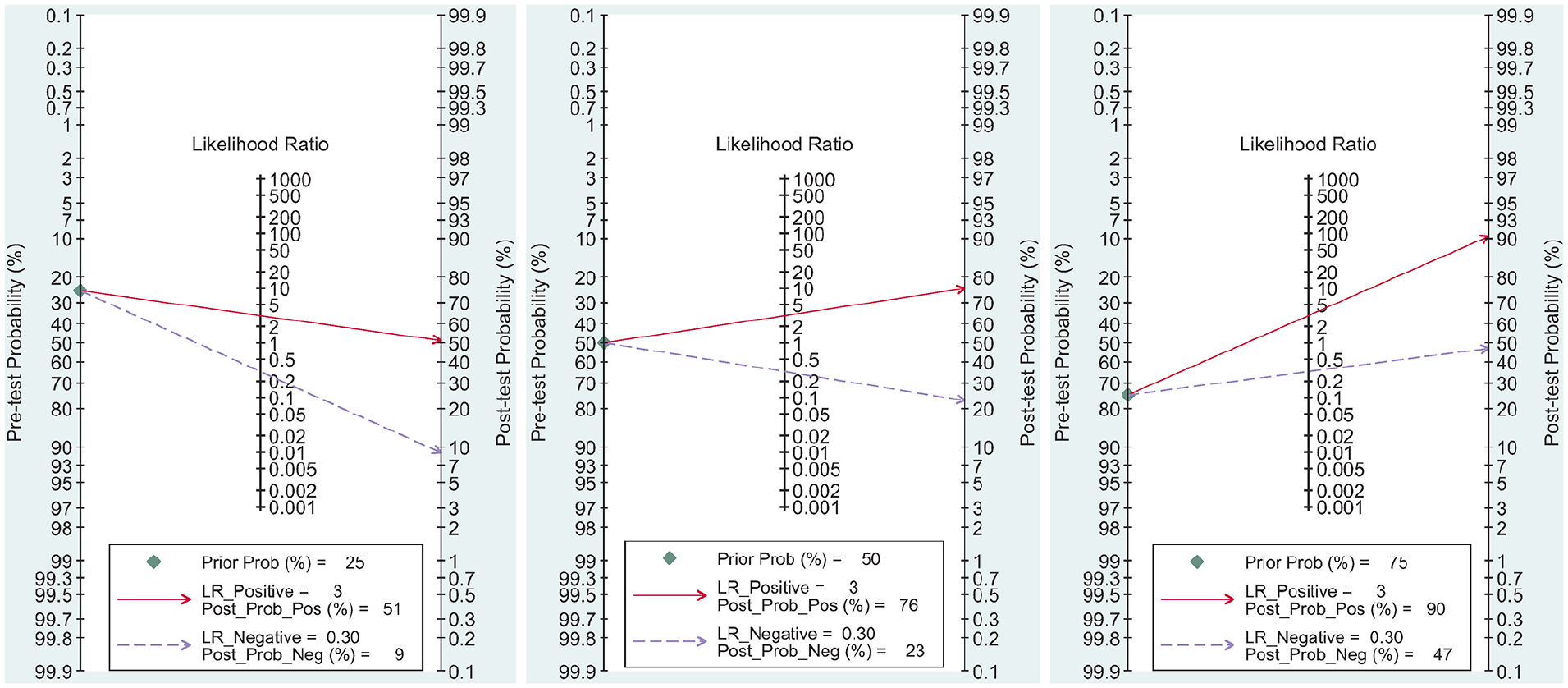
Fagan plots for miR-145 with 25%, 50%, and 75% pre-test probability of diagnosing NSCLC.
Discussion
By 2035, the estimated mortality of NSCLC will have increased globally by 86% compared with that in 2012, indicating that the preventive and control situations of NSCLC in most countries remains pessimistic in the near future. 37 Despite remarkable progress in diagnostic technologies, the usefulness of molecular indexes for diagnosing NSCLC remains controversial. Therefore, it has been increasingly recognized that accurate prognostic factors are particularly valuable in its screening, such as epidermal growth factor receptor mutation lung tissues, 38 circulating tumor cells, 39 carcinoembryonic antigen, and cyfra21-1 in serums. 40 However, conflicting results have been reported in different biomarkers, populations, and studies on the predictive role of the mutations identified in disease, whereas there is still a lack of widely used diagnostic biomarkers for NSCLC with satisfactory sensitivity and specificity. 12
miR-145 has been detected in peripheral blood and other body fluids. 41 Previous studies have indicated that miR-145 has been reported to be associated with cell apoptosis, proliferation, neural development, and stem cell differentiation. 42 In stem cell differentiation, miR-145 is induced during differentiation, and it directly silences the stem cell’s self-renewal and pluripotency program by suppressing multiple pluripotent genes such as OCT4, SOX2, and KLF4.43,44 In NSCLC, miR-145 is capable of inhibiting tumor cell growth and invasion by focusing on several targets, such as c-Myc and mucin 1, and it acts as an oncogene by suppressing the migration and invasion capacity of A549 cells. 45
Several studies have investigated the potential correlation between miR-145 profile expression in the circulation and its possible clinical use to diagnose NSCLC, as the experimental results varied from different studies may influence diagnostic capabilities. This research conducted a meta-analysis to ascertain the overall accuracy of miR-145 in detecting NSCLC. Using meta-analysis, we found that the summary ROC of the miR-145 for detection of NSCLC was 0.83. As heterogeneity existed in the pooled analysis of sensitivity and specificity, a meta-regression analysis was further performed to incorporate specimen selection (plasma or serum). SROC increased to 0.90 of miR-145 for the diagnosis of NSCLC in serum as compared to plasma (0.75), which demonstrated the potential role of serum miR-145 with promised diagnostic efficiency in NSCLC, higher than the miR-126 previously reported by Grimolizzi et al., 46 as well as carcinoembryonic antigen (AUC=0.834) and cyfra21-1 (AUC=0.711) in serums. 40
Compared with previous studies, our present study has systematically summarized the diagnostic value of miR-145 for NSCLC in recent years and has several advantages. First, we concentrated on the detection of NSCLC by using miRNA-145 as a non-invasive biomarker, which was a novel research field aiming at a single miRNA that has never been previously studied. Second, miRNA-145 was comprehensively and systematically analyzed in this research, which expanded the investigation number of miRNAs for screening NSCLC. Furthermore, we conducted stratified analyses in different subgroups, according to public years (studies published in the past 3 years vs more than 3 years) and specimen selection (serum vs plasma). Our results indicated that miR-145 showed better diagnostic performance in studies published in serum.
We fitted clinical scenarios by using the likelihood ratios. The results found that for a relative low pretest probability set to 25%, the positive posttest probability increased to 51% and the negative posttest probability reduced to 9%. For a pretest probability set to 50%, the positive posttest probability increased to 76% and the negative posttest probability reduced to 23%. For a high pretest probability set to 75%, the positive posttest probability was elevated to 90%, and the negative posttest probability reduced to 47%. We can confirm the performance of circulating miR-145 in differential diagnosis between NSCLC and non-NSCLC patients, indicating a good clinical utility of miR-145 to improve the diagnosis of NSCLC.
Considerations regarding molecular analysis of NSCLC may include the substrate not only for the convenience of implementation, but also the effective identification of new diagnostic molecular biomarkers. In this study, all cases in enrolled studies received blood tests as convenient noninvasive biomarkers. Meanwhile, the results of a systematic review and meta-analysis also demonstrated a level of promised diagnostic accuracy. However, some limitations still exist in this study. First, there was only one study that provided continuous retrospective analysis, while the other studies were all case controls. Second, time intervals between miR-145 testing and the gold standard were not mentioned in all studies. Third, miR-145 in different histological types of NSCLC should be analyzed. Unfortunately, the subgroup analysis of different histological types is still unclear as not enough information is currently available. Fourth, as other biomarkers in the diagnosis of NSCLC, a possible threat to the clinical use of miR-145 relates to threshold effects, which might be related to the cutoff values in different studies. We found that different studies had different cutoff values; subgroup classifications of cutoff value analysis based on the different backgrounds were not conducted because some studies lack detailed information. Finally, sample sizes in individual trials were relatively small, which may reduce reliability in statistical analysis.
Conclusions
A total of 350 articles were retrieved according to the search strategy and data collection methods, and 11 data sets in trials were ultimately included by referencing the inclusion and exclusion criteria. The results indicated that serum miR-145 may serve as a noninvasive biomarker in NSCLC diagnosis. However, given the limited amount of relevant literature, more large-scale and multicenter studies are required for further verification.
Supplemental Material
Supplementary_Table_1_2 – Supplemental material for Circulating microRNA-145 as a diagnostic biomarker for non-small-cell lung cancer: A systemic review and meta-analysis
Supplemental material, Supplementary_Table_1_2 for Circulating microRNA-145 as a diagnostic biomarker for non-small-cell lung cancer: A systemic review and meta-analysis by Shaohua Tao, Xuegui Ju, Hui Zhou and Qianglin Zeng in The International Journal of Biological Markers
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
ST and QZ conceived and designed the study; XJ collected and analyzed the data; QZ, ST, and QZ prepared the original draft; HZ and QZ finally approved the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Data availability
The datasets during the current study are available from the corresponding author on reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the Health Commission of Sichuan Province (NO. 18PJ405).
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
