Abstract
Background:
Bladder cancer is the ninth most common cancer worldwide and has high morbidity and mortality. We aimed to search for potential serum peptide biomarkers and establish a diagnostic model for early bladder cancer.
Methods:
A total of 67 bladder cancer patients and 64 healthy volunteers were randomly divided into a training set and testing set 1. There were 30 hematuria patients used as testing set 2. Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry based on weak cation exchange magnetic beads was used to obtain and analyze the serum peptide profiles between bladder cancer patients and healthy volunteers in the training set. Serum peptide diagnostic model through a k-nearest neighbor algorithm, was established and validated, and significantly differentially expressed protein biomarkers were ultimately identified.
Results:
We constructed a diagnostic model containing five peptides (m/z 1954.9, m/z 2081.0, m/z 3938.3, m/z 3946.5, and m/z 4268.8). In the training set, the area under the curve (AUC) value of the five-peptide model was 0.923, and the sensitivity and specificity was 93.75% and 96.77%, respectively. In testing set 1, the sensitivity and specificity was 91.43% and 90.91%, respectively, and the specificity of testing set 2 was 73.33%. For early-stage bladder cancer, the sensitivity and specificity was 92.31% and 93.75%, respectively; the sensitivity of early low-grade bladder cancer was 90.00%; and the AUC value was 0.944.
Conclusion:
The five-peptide diagnostic model established in this study had high sensitivity and specificity, especially in the diagnosis of early bladder cancer, and could differentiate between healthy volunteers and hematuria patients.
Introduction
Bladder cancer (BC) is the ninth most common cancer worldwide and has high morbidity and mortality, with approximately 549,000 new cases and 200,000 deaths worldwide in 2018. 1 It is a gender-specific disease, with 75% of all BC cases occurring in men. 2 According to the clinicopathologic features, urothelial carcinoma is the most common form of BC. 3 Based on current data, 60%–70% of non-muscle-invasive BC patients will experience recurrence; because of this, BC patients need to periodically monitored for tumor relapse and progression.4,5
For BC patients, early diagnosis and effective individualized treatment are very important for a favorable outcome. At present, cystoscopy and urine cytology are recommended tools for BC diagnosis. Cystoscopy is still considered the best approach, but it is invasive and uncomfortable. Above all, the cost of cystoscopy, especially when used to monitor recurrence, is the highest among all kinds of cancer. Since cystoscopy is both burdensome and expensive for patients, urine cytology often serves as a complementary tool for BC by detecting cancer cells in urine. It has high sensitivity for high-grade BC, but its sensitivity for low-grade BC is only 4%–31%. 6 For decades, researchers have been looking for noninvasive urological biomarkers that can improve the sensitivity and specificity of BC detection. Although a large number of detection methods have been reported in recent years, many of them lack external validation. 7 There is still an urgent need for highly sensitive and specific biomarkers of BC.
Low-molecular-weight serum peptides contain important information and can be used as markers for the early diagnosis and prognosis of many diseases. 8 It is of great clinical value to identify and quantify these peptides. However, due to the low molecular weight and low content of peptides, it is difficult to detect and identify them by traditional protein detection technology. The WCX-MB method can enrich and extract serum peptides in the mass range from 1000 to 10000 Da. Mass spectrometry techniques, which can be used to identify the composition of the blood proteome, have also emerged as a valuable tool for disease diagnosis. Among the variety of proteomic techniques, MALDI-TOF-MS is an advanced technology for analyzing protein profiles in clinical samples. Several papers have reported that peptide profiling is of great importance in the diagnosis of different kinds of cancer, such as lung cancer, gastric cancer, colorectal cancer, ovarian cancer, and leukemia.9-13 The purpose of this study was to use MALDI-TOF-MS technology based on WCX-MB to explore the serum peptide profiles of BC patients and healthy volunteers, to identify differentially expressed peptides, and to construct a classification model for the early diagnosis of BC.
Materials and methods
Ethics statement
Informed consent was signed by all subjects before the serum samples were collected, and the ethics committee of the First Affiliated Hospital of Dalian Medical University approved the study (PJ-KS-KY-2019-71). The study was conducted according to the Declaration of Helsinki.
Patients
A total of 161 serum samples were collected in this study: 67 patients with BC (51 males and 16 females; age range 27–89 years; mean age 66 years), 64 healthy volunteers (53 males and 11 females; age range 49–85 years; mean age 65 years) who underwent medical examination, and 30 patients of urolithiasis with gross hematuria (24 males and 6 females; age range 37–82 years; mean age 65 years). Serum samples were collected from the First Affiliated Hospital of Dalian Medical University, China, from September 2017 to March 2018. All of the BC patients and healthy volunteers were randomly divided into two groups. The training set included 32 preoperative serum samples from BC patients, and samples from 31 healthy volunteers were used as controls. Testing set 1 consisted of 35 preoperative serum samples from BC patients and 33 healthy volunteers. Another testing set (testing set 2) involved 30 hematuria patients. Details of the patients’ characteristics are displayed in Table 1.
Patient characteristics of bladder cancer.
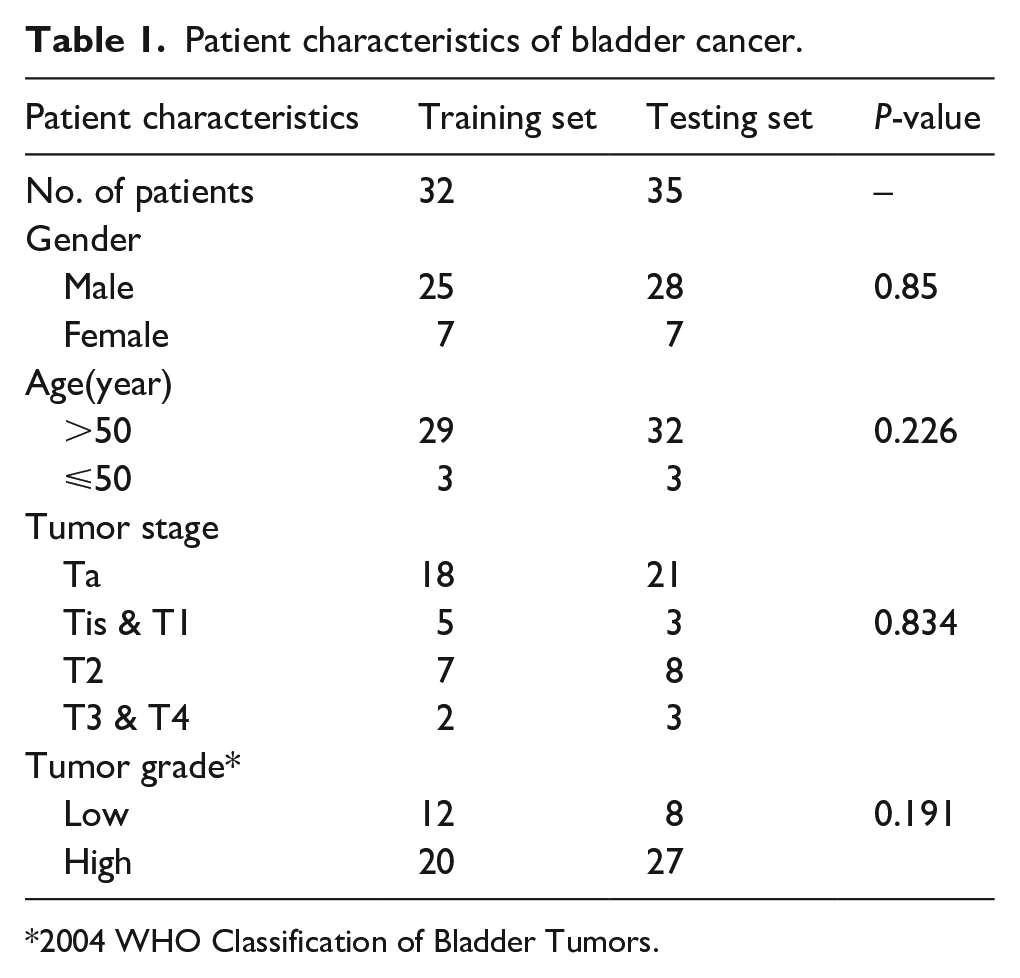
2004 WHO Classification of Bladder Tumors.
The inclusion criteria for this study were: (a) >18 years old; (b) all BC patients were diagnosed by two senior pathologists with no complications such as other malignant tumors, and serum samples from BC patients were collected before chemotherapy or radiotherapy; and (c) the subjects in the control group were age-matched with the cancer group with no history of bladder disease, and the medical examination results were normal.
Individuals with acute or chronic infections and malignant tumors were excluded from this study.
Serum processing
Fasting blood samples were collected from subjects, and all samples were left at room temperature for 1 hour to separate the serum, then centrifuged at 3000 rpm for 10 min, and stored at −80°C until detection.
Sample preparation and mass analysis
Serum samples were purified using weak cation exchange magnetic beads (WCX-MB, Bioyong, Beijing, China) before mass spectrometry analysis according to the kit instructions. 14 First, 7 µL of beads were added to a premixture of 10 µL of serum sample and 100 µL of binding solution in a tube, thoroughly mixed and incubated for 5 min, and then transferred to a magnetic bead separator and incubated for 1 min. Second, the supernatant was discarded, and the beads were washed three times with 100 µL of washing solution. After washing, 10 µL of eluting solution was added to the tube, thoroughly mixed and incubated for 5 min, and then transferred to a magnetic bead separator and incubated for 1 min. Finally, the MALDI-TOF matrix and eluent were mixed 1:1 (v/v), and 2 µL of protein solution was applied to a target plate, and all the samples were tested in triplicate to prevent deviation. The matrix solution was 8 mg/mL of αlpha-cyano-4-hydroxycinnamic acid (CHCA), which was soluble in acetonitrile, trifluoroacetic acid and deionized water. System quality control was performed before the detection of the samples, and the peptide peaks were calibrated with a mixture of three standard serum peptides (1533.8582 Da, 2465.1989 Da, and 5730.6087 Da, Product Numbers P2613, A8346, and I6279, respectively; Sigma) used as external standard. The profiles were normalized by applying the total ion current, and peak area was analyzed for quantitative standardization.
The air-dried target plate was detected using a Clin-TOFⅡ instrument (Bioyong, Beijing, China), and the mass profiles were collected in the m/z range of 1000–10000 Da with a signal-to-noise (S/N) ratio >5. Then, the raw data were processed and analyzed by BioExplorer software (Bioyong, Beijing, China). Each profile was normalized, baseline corrected and smoothed. The Wilcoxon test was used to compare the average peak intensities between the BC group and the healthy volunteers group, and the Benjamini method was used to correct each data to control false discovery rate; P <0.05 was considered statistically significant. An average peak intensity higher than 300 was considered significant. The k-nearest neighbor (KNN) algorithm was used to select peptide peaks and to set up a classification model to distinguish BC patients from healthy volunteers and hematuria patients. This method has been successfully used in previous published articles and has proved its reliability. 15
Identification of the serum peptides by LC-MS/MS
The sequences of the peptides in the BC diagnostic model were identified by using LC-MS/MS to detect and collect the analysis results, and searching Uniprot database with Peaks software version 8.5. Peptides were loaded onto a C18 column ( 50 um × 15 cm; 2 um particle size, C18 PepMap100) with a flow rate of 200 nL/min in 0.1% FA loading buffer for 2 min, and a gradient of 6%–22% solvent B over 40 min, 22%–32% solvent B over 5 min, held constant at 100% for 8 min in all. Nanospray was performed with a 10 um uncoated silica tip emitter (New Objective, FS360-20-10-N-20).
The MS was operated in data-dependent MS/MS mode in which each full MS scan was collected in the orbitrap (Q-Exactive Plus), precursor ion range of 350–2000 m/z, R = 70,000 for MS and 17,500 for MS2, followed by up to eight MS/MS scans performed in the linear ion trap where the most abundant peptide molecular ions were selected for High energy Collision Dissociation (HCD), using a normalized collision energy of 28%. Total MS acquisition time was 60 min.
Data were searched against the human proteome (UniprotKB, 10/2015, canonical and isoform, 92 035 entries) using Peaks (Version 8.5) employing following search settings as Oxidation (M) and Deamidation (NQ) as variable modification, unspecific digestion mode, with Parent Mass Error Tolerance of 10 ppm and a Fragment Mass Error Tolerance of 0.02 Da.
Statistical analysis
The Wilcoxon test was used to compare the average peak intensities between the BC group and the healthy volunteers group. P<0.05 was considered statistically significant. Receiver operating characteristic (ROC) curves were generated to assess the diagnostic value of the peptide biomarkers with SPSS software v20.0.
Results
Selection of significantly expressed serum peptide peaks between BC patients and healthy volunteers
To evaluate the reproducibility of MALDI-TOF-MS, six serum samples were tested in triplicate with MALDI-TOF-MS before the experiment, and the corresponding mass profiles were obtained. Five peptide peaks were randomly selected, and the average coefficient of variation (CV) was calculated to be 12.23%±4.36% (Supplementary Table 1). As shown in Figure 1(a), there was a significant difference in the serum peptide profile range from 1000 to 10,000 Da between BC patients and healthy volunteers. A total of 50 peptide peaks were identified; among them, 17 were significantly differentially expressed between the BC group and the healthy volunteer group (P<0.05), including 13 upregulated and 4 downregulated peptide peaks. All significant peaks are displayed in Table 2.
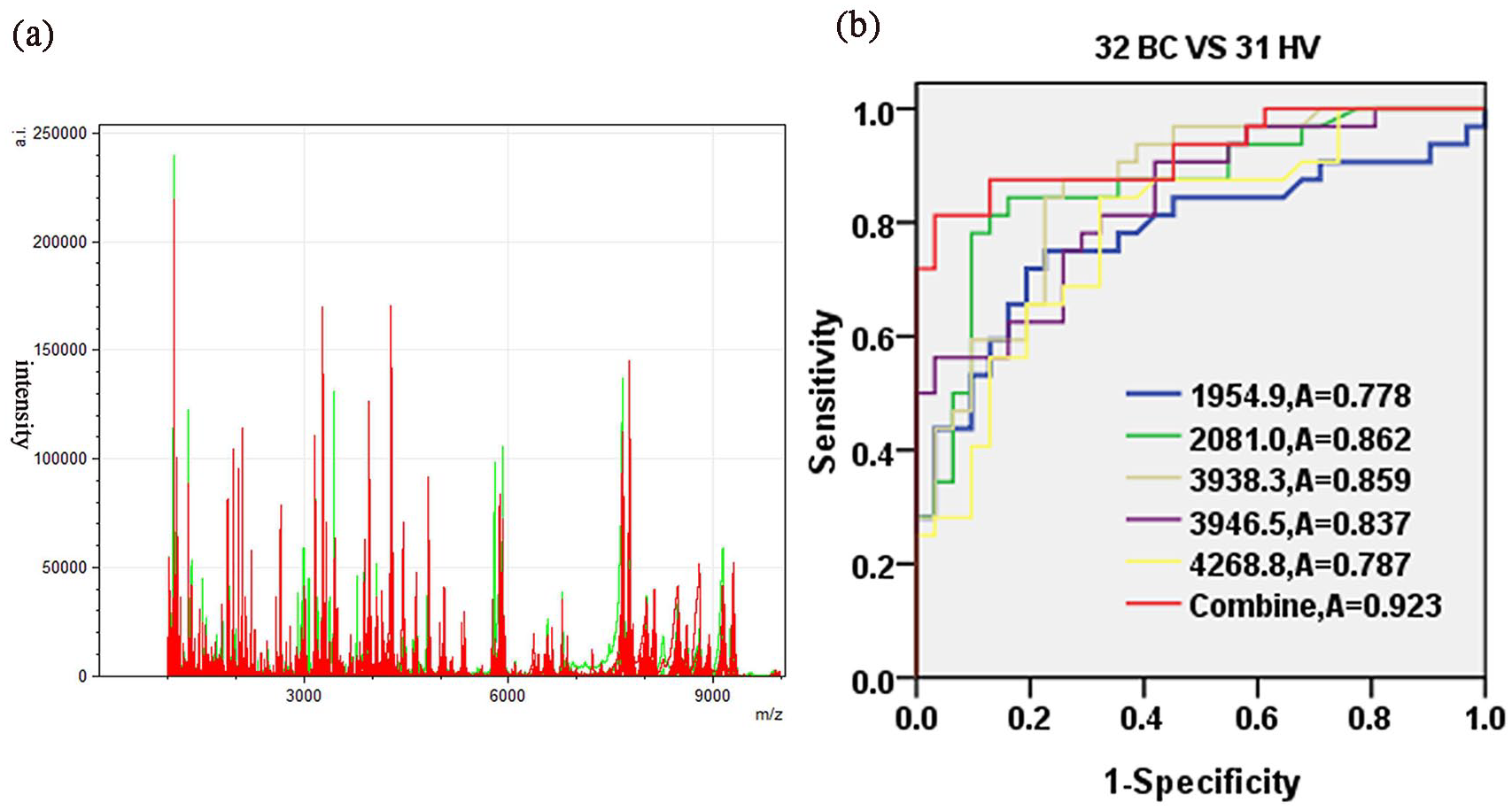
(a) Overall serum peptide profiling in the mass range from 1000 to 10,000 Da obtained from all BC patients (red) and healthy controls (green) in the training set. (b) Evaluation of the diagnostic ability of the five peptides by ROC curve analysis.
A total of 17 significant peptide peaks between bladder cancer patients and healthy volunteers in the training set.
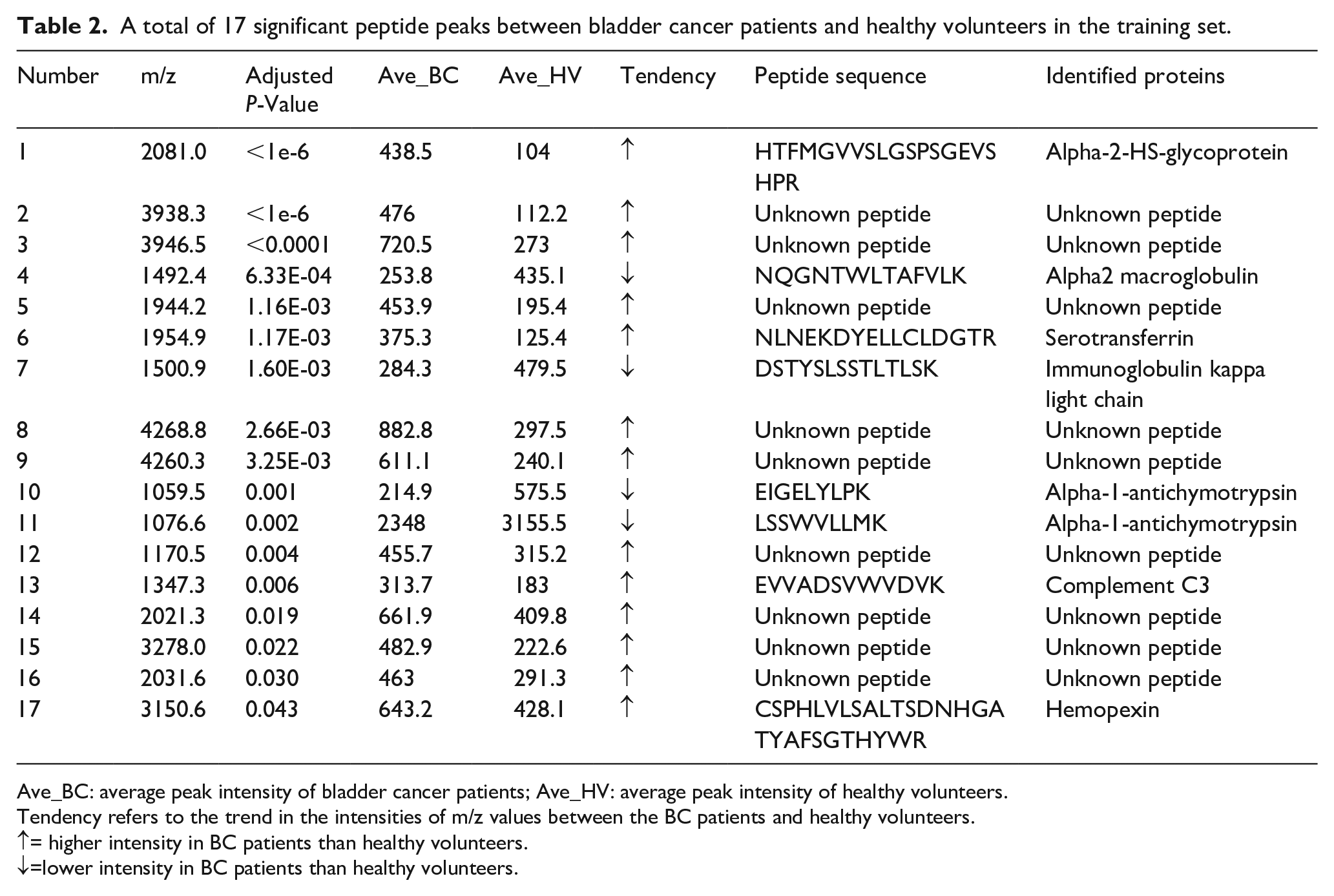
Ave_BC: average peak intensity of bladder cancer patients; Ave_HV: average peak intensity of healthy volunteers.
Tendency refers to the trend in the intensities of m/z values between the BC patients and healthy volunteers.
↑= higher intensity in BC patients than healthy volunteers.
↓=lower intensity in BC patients than healthy volunteers.
Establishment of a diagnostic model for BC and further validation
We selected 5 peptides of 17 peptide peaks with higher peak intensities in BC patients and significant differences compared with healthy volunteers (>2.5 average fold change, P<0.001, Table 2). The 5 peptides (m/z 1954.9, m/z 2081.0, m/z 3938.3, m/z 3946.5 and m/z 4268.8) that had higher peak intensities in BC patients were significantly different compared with healthy volunteers. The average peak intensities of all five peptides were significantly upregulated in BC patients, compared with healthy volunteers (HV) (Supplementary Figure 1). The diagnostic value was determined by the area under the ROC area under the curve (AUC) analysis. As shown in Figure 1(b), the ROC curves showed that the AUC values of the five peptide peaks were 0.778 (95% CI 0.659, 0.898; P< 0.001), 0.862 (95% CI 0.768, 0.955; P< 0.001), 0.859 (95% CI 0.769, 0.950; P< 0.001), 0.837 (95% CI 0.741, 0.933; P < 0.001) and 0.787 (95% CI 0.675, 0.900; P< 0.001), respectively. The AUC value of the five-peptide combination was 0.923 (95% CI 0.857, 0.990; P < 0.001).
Based on the KNN algorithm, the diagnostic sensitivity and specificity of this model was 93.75% and 96.77%, respectively, in the training set. In testing set 1, the sensitivity and specificity of the model was 91.43% and 90.91%, respectively, and the diagnostic specificity of testing set 2 was 73.33%. The sensitivity and specificity for early BC (Ta stage) was 92.31% and 93.75%, respectively. The sensitivities in the diagnosis of early low-grade BC and early high-grade BC were 90.00% and 94.74%, respectively, and the specificity was 93.75% (Table 3).
Sensitivity and specificity in the diagnosis of bladder cancer patients.
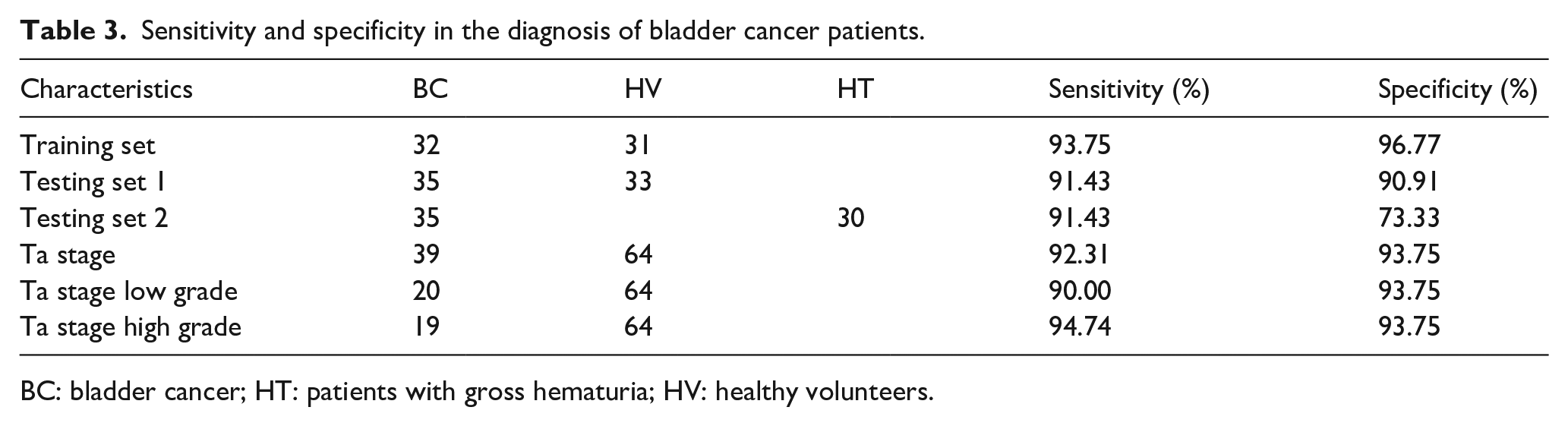
BC: bladder cancer; HT: patients with gross hematuria; HV: healthy volunteers.
Diagnostic value of the five-peptide model in early BC
The ROC curves of early low-grade BC patients and healthy volunteers and of early low-grade BC patients and hematuria patients were analyzed, and the AUCs were 0.944 and 0.927, respectively (Figure 2(a) and (b)). The ROC curves of early high-grade BC patients and healthy volunteers and of early high-grade BC patients and healthy volunteers were analyzed, and the AUC values were 0.963 and 0.949, respectively (Figure 2(c) and (d)).
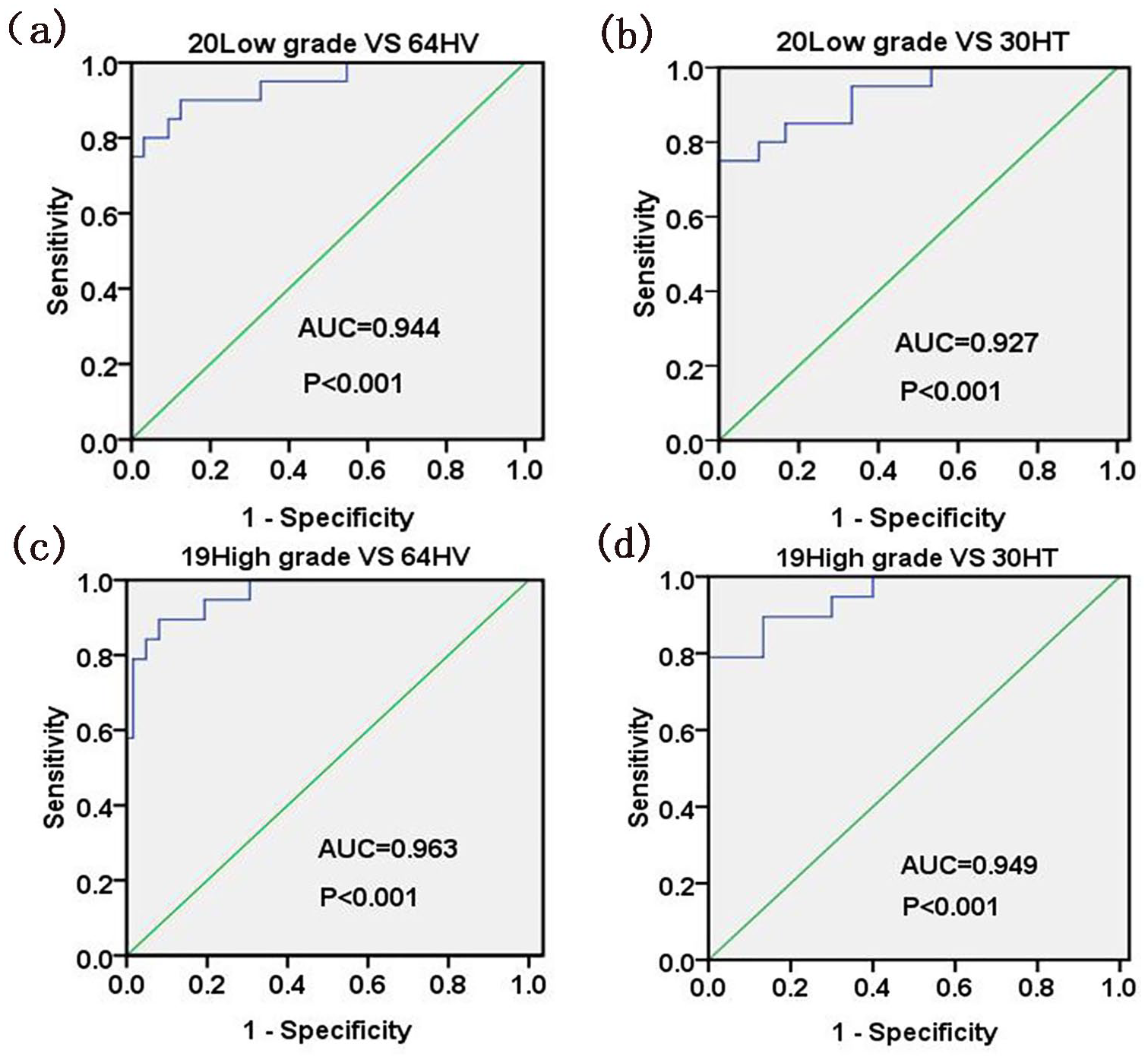
Evaluation of ROC curves for five-peptide model in the diagnosis of early BC. (a) For 20 patients with early low-grade BC and 64 HV, the AUC value was 0.944 (95% CI 0.871, 1.000; P < 0.001). (b) For 20 patients with early low-grade BC and 30 HT, the AUC value was 0.927 (95% CI 0.852, 1.000; P < 0.001). (c) For 19 patients with early high-grade BC and 64 HV, the AUC value was 0.963 (95% CI 0.916, 1.000; P < 0.001). (d) For 19 patients with early high-grade BC and 30 HT, the AUC value was 0.949 (95% CI 0.874, 1.000; P < 0.001).
Identification of the peptide biomarkers for BC
LC-MS/MS successfully identified 8 of the 17 peptides that were significantly differentially expressed between the BC patients and healthy volunteers. Among them, two upregulated peptides in the diagnostic model, m/z 1954.9 and m/z 2081.0, revealed the sequences as NLNEKDYELLCLDGTR and HTFMGVVSLGSPSGEVSHPR (Table 2); these sequences correspond to fragments of serotransferrin and alpha-2-HS-glycoprotein, respectively.
Discussion
Some urinary markers of BC, such as nuclear matrix protein 22 (NMP22) and bladder tumor antigen (BTA), have been approved by the Food and Drug Administration of the United States (FDA). However, these biomarkers have not been widely used in the clinic because of a lack of sensitivity and specificity and an increase in the false positive rate in urinary tract infections.16,17 The infrequent use of these biomarkers may be because most cancer diagnostic methods rely on only one marker, and when combined with multiple markers, they tend to be more sensitive. 18 Considering the heterogeneity of BC, it seems unlikely that a single marker could be used for accurate diagnosis, and the MALDI-TOF-MS technology used in this study makes it possible to simultaneously evaluate a panel of different markers.
The MALDI-TOF-MS technique has the advantage of recognizing minor differences between all of the peptide samples, and is highly sensitive in detecting small molecular weight protein. 19 In fact, in recent years, it has continued to develop and served as a high-throughput method to search for protein biomarkers for different kinds of diseases. Urine samples are commonly used to study markers of BC; however, the protein composition in urine is complex and variable, which is greatly affected by diet and exercise. 20 The protein in serum is relatively stable; therefore, we selected serum samples from BC patients for this study.
In this study, the WCX-MB method combined with MALDI-TOF-MS was used to detect the serum peptide profiles of 67 BC patients, 64 healthy volunteers, and 30 hematuria patients. In order to evaluate the reproducibility of MALDI-TOF-MS in peptide studies, six serum samples were tested in triplicate with MALDI-TOF-MS to determine reproducibility before the experiment. The average CV of five randomly selected peptide peaks was 12.23%±4.36% (less than 20%), suggesting that the platform had a high reproducibility. Seventeen peptide peaks were significantly differentially expressed between the BC group and the healthy volunteers group. Among them, we selected five peptides (m/z 1954.9, m/z 2081.0, m/z 3938.3, m/z 3946.5, and m/z 4268.8); these had higher peak intensities in BC patients and were significantly different compared with healthy volunteers. Compared with healthy volunteers and hematuria patients, these five peptides are highly expressed in BC patients, even those with early BC. Furthermore, ROC curves were used to assess the diagnostic ability of the five peptides, and the results showed that the AUC values of each peptide peak were 0.778, 0.862, 0.859, 0.837, and 0.787, respectively. However, when the five peptides were combined, the AUC value was 0.923, which was higher than that of each peptide alone, indicating that the combination of the five peptides was more accurate in the diagnosis of BC. We finally established a diagnostic model consisting of these five peptides, with a sensitivity of 93.75% and a specificity of 96.77%. In test 1, the sensitivity reached 91.43% and the specificity reached 90.91%. When the 30 hematuria patients were used as testing set 2 to validate the model, the diagnostic specificity was 73.33%. The results of the two testing sets showed that the model could not only accurately distinguish BC patients from healthy volunteers but also distinguish BC patients from hematuria patients. Schwamborn et al. 21 first reported that they established a serum diagnostic model consisting of five peptides (m/z 3525.45, m/z 4281.66, m/z 4963.10, m/z 5804.12, and m/z 5903.43) for BC based on MALDI-TOF-MS. The sensitivity and specificity were 94.1% and 89.2%, respectively, but the sequences of these peptides were not identified in their experiment. The peptides in our model were significantly different from those in their research, which may be due to different ethnic groups and populations. In addition, the differences of the results may be related to sample preparation. According to a study, even at −80°C, sample preparation details and retention time may affect the peptide profiling. 22 More importantly, we further identified eight different serum proteins between BC patients and healthy volunteers. The study of the serum peptide diagnostic model for BC by MALDI-TOF-MS has not been previously reported in China. Therefore, it has clinical value for the diagnosis of BC in the Chinese population.
The five-peptide diagnostic model had an advantage in early BC, with a sensitivity of 92.31% and a specificity of 93.75%. The sensitivity of early high-grade BC was 94.74%. In addition, early low-grade BC could be distinguished from healthy volunteers and hematuria patients, with a sensitivity of 90.00%, and the AUC values were higher than 0.9. These results showed that the model had a good diagnostic ability for early BC, which was of great significance for the early screening, early detection and early treatment of BC. BC patients often have no obvious symptoms in the early stage, and hematuria is sometimes the only symptom. It is difficult to distinguish BC from other diseases that cause hematuria, such as urinary calculi. In this study, the five-peptide model of BC was shown to accurately identify early BC and differentiate these diseases. For the early diagnosis of BC, Schwamborn et al. 21 were able to distinguish early stage BC patients from Germany with their diagnostic model, with a sensitivity of 95.5%. Jia et al. 23 also published a similar study in esophageal squamous cell carcinoma, and their diagnostic model presented a sensitivity of 96.94% for early stage esophageal squamous cell carcinoma. Changes in serum peptide levels can occur at the early stage of a tumor, and MALDI-TOF-MS technology is highly sensitive to detecting small amounts of peptides. Therefore, the applications of MALDI-TOF-MS technology to detect serum peptides and establish a diagnostic model are of great clinical value for the early diagnosis of tumors.
Two of the five peptides in the model were identified: AHSG (m/z 2081.0) and serotransferrin (m/z 1954.9). AHSG is a multifunctional protein involved in calcium homeostasis, bone development, and insulin sensitivity. 24 AHSG has been reported to be related to a variety of tumor diseases and has different expression levels in breast cancer, lung cancer, and liver cancer.25-27 A previous study suggested that by blocking the binding of transforming growth factor β (TGF-β) to its receptor, AHSG could regulate the TGF-β signaling pathway and promote or inhibit the development of tumors. 28 Therefore, we speculated that AHSG may be related to the growth and proliferation of BC through TGF-β signaling pathway, which needs to be verified in future studies.
Transferrin is a major iron-binding protein that is involved in metabolic processes that require iron, such as DNA synthesis, electron transport, proliferation, and cell survival. 29 Transferrin is of great importance for rapidly growing cells, such as tumor cells, which may be due to the increased demand for iron as a cofactor for ribonucleotide reductase. 30 It was reported that neutrophil-derived transferrin is a key regulator of the growth of metastatic tumor cells and a therapeutic target for anti-metastasis therapy, indicating that transferrin is very important for the proliferation of cancer cells. 31 A new study showed that transferrin was highly expressed in the plasma of BC patients. 32 Therefore, we speculated that the high expression of serum transferrin may promote the proliferation of BC.
There were some limitations in this study. Because the current protein database is incomplete, some peptides were not identified; we will explore new methods to identify these peptides in the future. Also, in order to fully explore the clinical value of the diagnostic model, further studies are needed in a larger number of BC patients and healthy controls.
Conclusion
The five-peptide diagnostic model established in this study had high sensitivity and specificity, especially in the diagnosis of early BC, and could differentiate between healthy volunteers and hematuria patients. This was of great significance for the early screening, early detection, and early treatment of BC, which may make up for the lack of biomarkers for BC.
Supplemental Material
Supplementary_figure1 – Supplemental material for Novel serum peptide model revealed by MALDI-TOF-MS and its diagnostic value in early bladder cancer
Supplemental material, Supplementary_figure1 for Novel serum peptide model revealed by MALDI-TOF-MS and its diagnostic value in early bladder cancer by Dapeng Ding, Mingying Chen, Xiaoguang Xiao, Penglong Cao and Shijun Li in The International Journal of Biological Markers
Supplemental Material
Supplementary_material – Supplemental material for Novel serum peptide model revealed by MALDI-TOF-MS and its diagnostic value in early bladder cancer
Supplemental material, Supplementary_material for Novel serum peptide model revealed by MALDI-TOF-MS and its diagnostic value in early bladder cancer by Dapeng Ding, Mingying Chen, Xiaoguang Xiao, Penglong Cao and Shijun Li in The International Journal of Biological Markers
Footnotes
Abbreviations
WCX-MB: Weak cation exchange magnetic beads
MALDI-TOF-MS: Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry
KNN: k-nearest neighbor algorithm
LC-MS/MS: Liquid chromatography-tandem mass spectrometry
Ethics approval and consent to participate
Informed consent was signed by all subjects before the serum samples were collected, and the ethics committee of the First Affiliated Hospital of Dalian Medical University approved the study (PJ-KS-KY-2019-71). The study was conducted according to the Declaration of Helsinki.
Author contributions
Dapeng Ding and Mingying Chen contributed equally to this work and should be considered co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant (no.20180550088) from Natural Science Foundation of Liaoning Province of China.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
