Abstract
Background
Serum AFP levels are typically elevated in less than 50% of hepatocellular cancer (HCC) patients. Gamma-glutamyl transpeptidase (GGT) levels have been suggested to be a potentially useful HCC biomarker.
Aims
To assess in a cohort of prospectively evaluated HCC patients who underwent liver transplant and whose survival was known; the occurrence, prognosis, and clinical characteristics of patients with elevated serum GGT levels.
Results
Serum GGT levels were found to be elevated in a higher proportion in patients with either small or large HCC than alpha-fetoprotein (AFP) levels, and were significantly related to prognosis in patients with large size HCCs. There was no clear correlation between GGT and AFP levels, likely reflecting different HCC characteristics or HCC cell lineages associated with these two markers. Furthermore, elevated GGT was found in 24% of low-AFP patients with small tumors and 46% with large tumors. Elevated GGT levels were also significantly associated with microvascular invasion and tumor diameter.
Conclusions
Elevated serum GGT levels were associated with HCC size and worse survival, and were unrelated to AFP levels. GGT may be a useful prognostic tumor marker, especially for low-AFP HCC patients.
Introduction
Hepatoma-associated gamma-glutamyl transpeptidase (GGT) was originally purified from a Morris hepatoma,1–4 with reduced glutathione being its best substrate. GGT isoenzyme II or GGT-II was found to be present in 87% of HCC patients, compared with alpha-fetoprotein (AFP) in only 54.5%. 5 GGT-II was also found to be elevated in the serum of patients with small size hepatocellular carcinomas (HCCs or hepatomas) and was found in some patients undergoing surveillance even before HCC was identified radiologically. 5 It was found to be elevated in over 70% of HCC patients whether they were serum AFP positive or negative. 6 The non-hepatoma-specific GGT enzyme is encoded by multiple mRNAs in different tissues, as well as being different in liver and HCC tissues. 7 Hepatoma-specific GGT appears to have no correlation with AFP 8 and can be detected by both slab gel electrophoresis and by the ELISA technique. 9
Since total serum GGT—as determined in routine clinical liver function tests—has been found to be a useful HCC tumor marker, especially in patients with low serum AFP levels, 10 , 11 we examined in the current work, serum GGT levels using standard clinical assays, in a cohort of HCC patients who were treated by liver transplant and whose post-treatment survival was known and recorded at systematic follow-up. We found that serum GGT levels were elevated in a higher proportion of both small and large HCCs than AFP, and that serum GGT levels were significantly related to prognosis in large size, but not small size, HCCs. Furthermore, a significant proportion of low-AFP patients still had elevated GGT levels, suggesting the usefulness of GGT as a biomarker.
Methods
Patients who underwent liver transplant (LT) for HCC at our Liver Transplantation Institute were the subjects of this study. The data were collected prospectively and were analyzed retrospectively. This study was approved by Inonu University Institutional Review Board (Approval no: 2018/1-9).
The clinical parameters and tumor characteristics of 285 patients were analyzed according to the AFP cutoff of 200 ng/mL and the GGT cut-off of 100 IU/mL. These cutoffs were found by an analysis of the receiver operating characteristic and were significantly associated with survival post liver transplant. 12 Categorical (qualitative) variables were expressed as count and percentage. Comparisons of groups were performed by the Pearson chi-square test, the continuity corrected chi-square test, and Fisher’s exact tests for categorical variables. Categorized variables were compared using univariate analysis methods (Pearson chi-square test, continuity corrected chi-square test, Fisher’s exact test). Continuous variables were compared using the Mann-Whitney U test. The Kaplan–Meier survival estimate was used to determine overall survival (OS) and disease-free survival (DFS) of the patients. The follow-up period was defined as the interval between LT until the date of the last visit to the outpatient department for living patients or until the date of death. Time to disease recurrence was defined as the interval between the LT until the date a lesion that appeared to be a tumor was detected by biochemical (AFP) and radiological examination and/or a lesion diagnosed as HCC in another region of the patient. Statistical tests were considered significant when the corresponding P value was less than 5%. All statistical analyses were performed using IBM SPSS Statistics for Windows version 25.0 (New York, USA).
Results
Patients were analyzed according to the presence of small or large tumors (maximum tumor size or maximum tumor diameter (MTD) <5 cm or ≥5 cm). For patients with small (<5 cm) tumors, 25% had high GGT levels; whereas for patients with larger tumors, 48.2% had high GGT levels (Table 1). The corresponding numbers for serum AFP levels were: 15.4% had high AFP for small tumors, but 32.2% had high AFP for large tumors. Thus, a greater percentage of patients had elevated GGT levels than AFP levels, for either small or large tumors.
GGT or AFP distribution in small or large MTD groups.
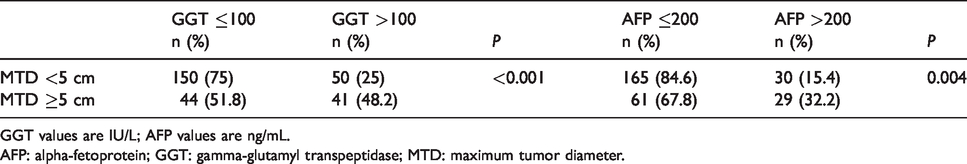
GGT values are IU/L; AFP values are ng/mL.
AFP: alpha-fetoprotein; GGT: gamma-glutamyl transpeptidase; MTD: maximum tumor diameter.
OS was then examined, according to the Kaplan–Meier method. Survival was significantly worse in patients having large tumors and higher versus lower serum GGT levels (P = 0.008; Table 2), but not in patients having smaller tumors with high versus low GGT levels. By contrast, survival was not significantly different in the high versus low AFP groups, for either small or large tumors (p = 0.161).
Survival in small or large MTD groups according to serum GGT or AFP levels.

GGT values are IU/L; AFP values are ng/mL.
AFP: alpha-fetoprotein; GGT: gamma-glutamyl transpeptidase; MTD: maximum tumor diameter; OS: overall survival.
To examine whether the significant survival differences in patients with large tumors was attributable simply to the patients with elevated GGT also having elevated AFP levels, the distribution of AFP levels according to the GGT group was examined (Table 3). We found that the percentage of high or low AFP patients showed no difference between high and low GGT groups, whether or not patients had small or large tumors. Thus the survival differences between patients having higher versus lower serum GGT levels was not attributable to whether they had either higher or lower serum AFP levels.
AFP distribution according to GGT level, in small or large MTD groups.
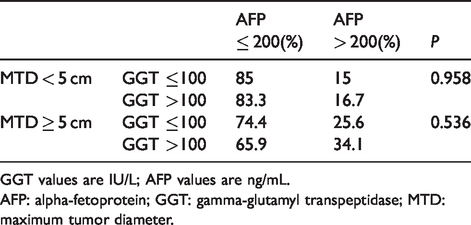
GGT values are IU/L; AFP values are ng/mL.
AFP: alpha-fetoprotein; GGT: gamma-glutamyl transpeptidase; MTD: maximum tumor diameter.
The clinical characteristics between patients having either high or low serum GGT levels were then compared (Supplementary Table 1; clinical characteristics of high and low GGT patient groups). The elevated GGT group had significantly higher levels of serum aspartate amino transferase, alanine amino transferase, alkaline phosphatase, and platelet values than the low GGT level group. They also had more aggressive tumor characteristics, with significantly higher AFP levels, larger tumors, and a higher percentage of patients with microscopic portal vein invasion by tumor.
We were interested to know whether serum GGT levels increased with increasing MTD. We found that the proportion of patients with elevated GGT increased with an increase in MTD, but was significant only when the largest MTD group was compared to the others (Table 4(a)).
GGT levels in MTD groups and survival in low AFP patients.
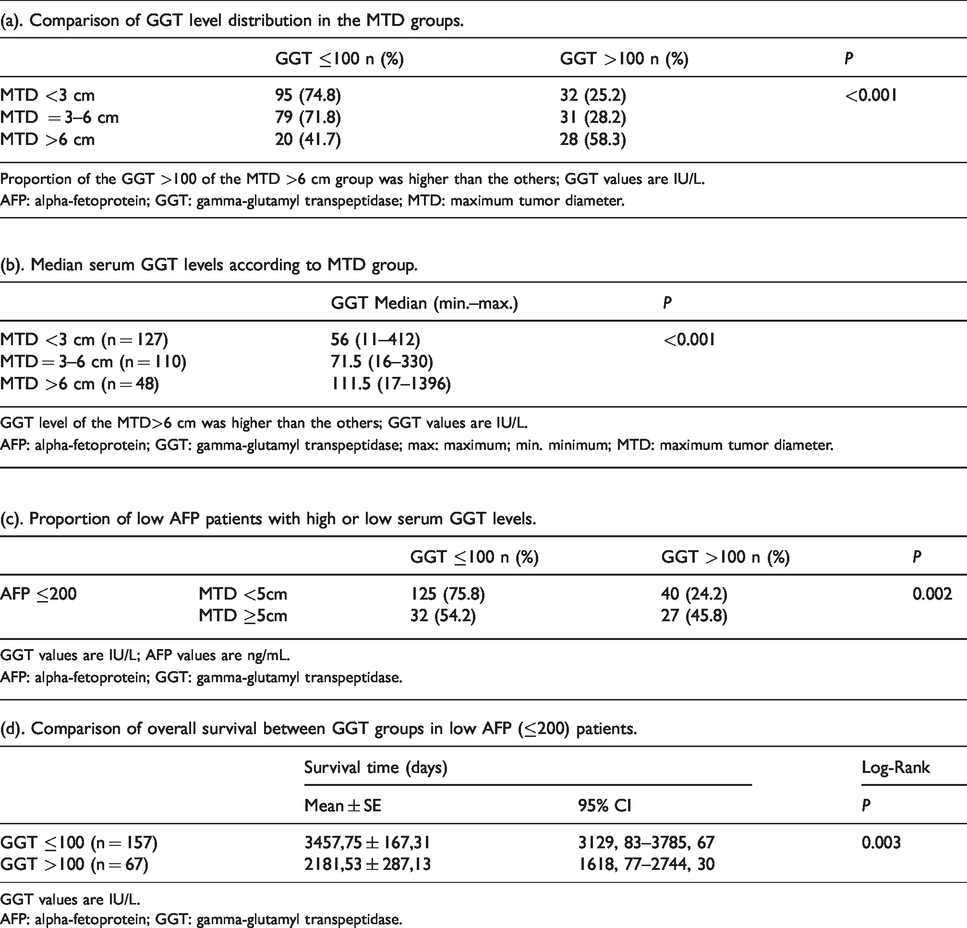
GGT values are IU/L.
AFP: alpha-fetoprotein; GGT: gamma-glutamyl transpeptidase.
The serum GGT levels were also increased with an increase in MTD (Table 4(b)), but the differences were also only significant for the largest tumor size band versus the others.
We then examined low-AFP patients (the majority, or 84.6% of patients with small tumors and 67.8% of patients with large tumors; Table 1), and found that the proportion of patients with high serum GGT was 24.2% in those with low-AFP and small tumors, and increased to 45.8% in those with low-AFP and larger size tumors (Table 4(c)). Survival was then analyzed for these two GGT groups of patients with low-AFP values and was found to be significantly shorter for patients with high GGT levels compared with those having low GGT levels (2181 versus 3457 days; P = 0.003; Table 4(d)). Thus, elevated serum GGT could be prognostically useful in low AFP level patients.
Discussion
AFP has been the most time-tested and useful marker for HCC diagnosis, prognosis, and response to therapy. However, it is typically not increased in more than 50% of HCCs, 11 thus limiting its diagnostic usefulness as well as its applicability in screening. Several other biomarkers have been proposed and become accepted in HCC clinical practice, including lectin-binding AFP (AFP-L3), the US Food and Drug Administration-approved (and vitamin K-associated) des gamma carboxyprothrombin, glypican-3, and several novel biomarkers in development, including circulating microRNAs and circulating tumor cells, among others. 13
Multiple reports have shown the diagnostic and prognostic usefulness of both gamma glutamyl transpeptidase, as well as its hepatoma-specific isoform, GGT-II.1–9 Several studies have also shown the prognostic usefulness of GGT in various treatments of HCC, including chemoembolization, 14 , 15 radiofrequency ablation, 16 resection, 17 , 18 and liver transplantation. 19
Among the main findings of this study was the absence of an association of serum levels of AFP and of GGT (Table 3). Thus, patients with low or high GGT levels had a similar percentage of patients with high AFP levels. Thus, the prognostic significance of elevated GGT could not be explained by concomitant and proportional levels of AFP, suggesting that the two markers were independently regulated or reflected different HCC biological processes. AFP levels were elevated in only 32.2% of our patients with large tumors; by contrast, GGT levels were elevated in 48.2% of patients with large tumors. Thus, GGT may have a wider application than AFP in patients with larger tumors. However, in patients with tumors <5cm, GGT was elevated in only 25% (15.4% for AFP). Thus, other tumor markers are still needed for the surveillance that is necessary to identify smaller tumors (Tables 1 and 4(a)). Thus, Table 1 shows that 25% of patients with <5cm tumors have elevated GGT levels, which is better than 15% that have elevated AFP levels, but still in need of more sensitive markers. Similarly, for patients with 3–6 cm tumors, only 28.2% had elevated GGT levels (Table 4), which is useful, but still in need of improvement in sensitivity. Despite this, GGT also showed its usefulness in patients with low AFP levels, as an additional 25% of them could potentially be flagged on surveillance screening, as well as 45.8% of low AFP patients with tumors ≥5 cm (Table 4(c)). Although patients with elevated GGT levels had lower survival than patients with lower GGT levels, the differences for survival were statistically significant only for the patients with tumors ≥5 cm (Table 2). In order to try to understand the basis for the significant differences in survival between patients with larger tumors who had elevated versus normal GGT levels (Table 2), we compared the clinical characteristics between these two clinical groups (Supplementary Table 1). We found that patients with elevated GGT levels had significantly larger MTD and a significantly lower percent of them had portal vein thrombosis, compared to patients with normal or low GGT levels. Thus, elevated GGT levels appear to be associated with more aggressive HCC characteristics. Exactly what the mechanisms involved might be is not really clear from this or other studies, although we showed an increase in median serum GGT levels with an increase in tumor diameter (Table 4(b)). A useful finding from this study is the relatively high proportion of patients with elevated GGT levels who did not also have elevated AFP levels. Two of the biggest clinical needs in HCC clinical practice is for (a) a suitable tumor marker for the >50% of patients without elevated AFP levels; and (b) a tumor marker for smaller tumors. GGT seems to succeed in the first clinical need, but less so in the second.
This study was performed on the basis of GGT levels that were measured in standard clinical laboratory practice, and not by the use of hepatoma-specific GGT or GGT-II, which was not available to our clinical laboratory. However, this might be a technically feasible approach to take in future studies in order to examine the applications and limitations of serum or possibly tumor GGT-II levels in clinical HCC practice.
Supplemental Material
sj-pdf-1-jbm-10.1177_1724600820921869 - Supplemental material for Gamma glutamyl transpeptidase as a prognostic biomarker in hepatocellular cancer patients especially with >5 cm tumors, treated by liver transplantation
Supplemental material, sj-pdf-1-jbm-10.1177_1724600820921869 for Gamma glutamyl transpeptidase as a prognostic biomarker in hepatocellular cancer patients especially with >5 cm tumors, treated by liver transplantation by Volkan Ince, Brian I. Carr, Harika Gozukara Bag, Cemalettin Koc, Sertac Usta, Veysel Ersan, Adil Baskiran, Tevfik Tolga Sahin and Sezai Yilmaz in The International Journal of Biological Markers
Footnotes
Authors’ contributions
VI and BIC made equal contributions; VI, BIC, SY, VE participated in research design; VI, BIC, CK, SU, SY participated in the writing of the paper; VI, BIC, SY, CK, TTS, VE, AB participated in the performance of the research; and HGB, VI, BIC, TTS, AB participated in data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
