Abstract
Background:
The PreImplantation Factor (PIF)—a peptide secreted by viable embryos—exerts autotrophic protective effects, promotes endometrial receptivity and controls trophoblast invasion. Synthetic PIF (sPIF) has both immune-protective and regenerative properties, and reduces oxidative stress and protein misfolding. PIF is detected by immunohistochemistry (IHC) in hyperplastic endometriotic lesions and advanced uterine cancer. sPIF reduces graft-versus-host disease while maintaining a graft-versus-leukemia effect.
Methods:
PIF detection in prostate cancer was assessed in 50 human prostate samples following radical prostatectomy using tumor-microarray-based IHC correlating PIF immune staining with Gleason score (GS) and cancer aggressiveness.
Results:
PIF was detected in moderate-to-high risk prostate cancer (GS 4+3 and beyond, prognostic groups 3 to 5). In prostate cancer (GS (WHO Grade Group (GG)5), PIF was detected in 50% of cases; in prostate cancer (GS 4+4 GG4), PIF was observed in 62.5% of cases; in prostate cancer (GS 4+3 GG3), PIF immunostaining was observed in 57.1% of cases. In prostate cancer, (GS 3+4 GG2) and (GS 3+3 GG1) cases where PIF staining was negative to weak, membranous staining was observed in 20% of cases (staining pattern considered negative). High-grade prostate intraepithelial neoplasia PIF positive stain in 28.57% of cases (6 of 21) was observed. In contrast, PIF was not detected in normal prostate glands.
Importantly, sPIF added to the PC3 cell line alone or combined with prostate cancer fibroblast feeder-cells did not affect proliferation. Only when peripheral blood mononuclear cells were added to the culture, a minor increase in cell proliferation was noted, reflecting local proliferation control.
Conclusions:
Collectively, PIF assessment could be a valuable, simple-to-use immunohistochemical biomarker to evaluate aggressiveness/prognosis in specimens from prostate cancer patients.
Keywords
Introduction
Cancer is characterized by sustained proliferation, activation of invasion, and evasion of cell death. 1 As cells become abnormal—usually deriving from one stem cell clone—they gain growth advantage and take over adjacent normal cells. The well-differentiated tumor becomes progressively less differentiated, loses cell-to-cell contact, and becomes prone to metastasis.2-4 Once cells become anaplastic, they may metastasize, become of unknown origin, and ultimately will closely resemble embryonic cells. Consequently, they are expected to express compounds such as those present in the fetal period. Indeed, the alpha-fetoprotein and the carcino-embryonal antigen are two known markers of tumorigenesis.5-8 However, whether advanced tumors can also express the earliest markers of embryogenesis has not been investigated so far. In that respect, preimplantation factor (PIF), a 15-amino acid peptide secreted by viable embryos from the zygote stage onwards, presents the necessary features to become a cancer biomarker candidate. The seminal role of PIF in pregnancy biology has been reported.9-14 Increased PIF levels in culture correlate with development to the blastocyst stage. PIF is present in viable embryos, and is absent in non-viable ones. Synthetic PIF (sPIF), due to its immune regulatory and regenerative effects, was shown to be effective in clinically relevant models in the treatment of diverse immune and transplantation disorders.15-23 These studies paved the way to a successfully completed FAST-TRACK Phase I trial in patients with an autoimmune condition and an award of orphan drug designation status (ODD) by the US Food and Drug Administration (FDA). The Phase I study showed that PIF is safe and devoid of toxicity or deleterious side effects, enabling progression to Phase II clinical trials (NCT02239562).
The relationship and parallel between pregnancy and cancer is rather unique. Pregnancy actually protects against cancer, including breast, brain, colon, ovary, and endometrium. 24 On the other hand, the presence of cancer during pregnancy, paradoxically, despite the intense proliferation the prognosis, frequently does not worsen the cancer. Finally, metastasis of cancer from the mother to the fetus is extremely rare. 25 This indicates that pregnancy is actually “cancer protective,” and the identification of specific compounds that are involved could be utilitarian. 26 This premise has led to studies to examine the sPIF effect in cancer using clinically relevant models. sPIF also reduced semi and fully/allogeneic bone-marrow-transplant-induced graft-versus-host development all while preserving the beneficial graft-versus-leukemia (BCL1) effect, thereby reducing spleen metastasis and mortality. 27
sPIF administration reduced spleen metastasis in the melanoma model, which was amplified by low-grade magnetic resonance imaging exposure. In human metastatic melanoma cell cultures, sPIF administration led to cancer autophagy, while it did not affect melanoma and other cancer cell lines. 28
PIF levels in maternal circulation correlate with a favorable pregnancy outcome. 29 Placental PIF expression (monoclonal antibody based) is highest in the first trimester and declines toward term. 13 PIF is also expressed in selective immune fetal organs including the liver, spleen, and thymus. 9 PIF is not detected in non-pregnant subjects, indicating that circulating levels are pregnancy specific (NCT02239562). Placental PIF expression is modulable and is upregulated following lipopolysaccharide (LPS) injection in the murine model at term. 30 In contrast, in preeclampsia and intrauterine growth retardation a premature decline in PIF expression is found. 31
Outside pregnancy, PIF re-expression was examined to determine whether cellular “de-differentiation” can lead to PIF expression. This is based on the concept that embryo development reflects increased complexity where from a zygote of over 250 cell types will develop and morph into regional complexity as different organs develop to form an integrated fetus, from structure to function. On the other hand, a breakdown in complexity is observed when the cancer becomes highly anaplastic where the metastatic cells spread to various regions of the body, meanwhile the original tumor site remains unknown. In contrast to normal endometrium, in the ectopic hyperplastic endometrium (i.e. endometriosis caused by chronic inflammation) PIF was detected, perhaps reflecting local immune privilege. 32 Importantly, sPIF blocked endometriosis cell culture proliferation and regulated local immune response, thus supporting clinical translation to therapy, which is being pursued. High PIF expression in advanced uterine cancer was also recently shown, and was associated with worse prognosis. 33
Specific to this study, in the prostate gland and even in a higher number in prostatic ducts, the presence of local stem cells was demonstrated. These primordial cells remain dormant when the cancer is androgen sensitive. However, as the prostate cancer (PC) progresses to an androgen-resistant phase, these cells are activated and start to proliferate. 34 This aberrant proliferation leads to the re-expression of several embryonic markers.35-37 PIF starts being expressed by the earliest and most proliferative stage embryo. Whether this will also lead to PIF re-expression in advanced PC had not been previously investigated. Such information could be complementary or even pivotal to currently used PC markers. The most commonly used PC markers are still circulating PSA levels, the Gleason score (GS), and the WHO Grade Group (GG).
The current study aimed to determine the correlation between PIF (monoclonal antibody based on immunohistochemistry (IHC) detection) and the GS and GG in patients with PC underwent radical prostatectomy. PIF detection was also examined in normal prostate tissue and in high-grade prostate intraepithelial neoplasia (HG-PIN). Further, to provide possible insight to local PIF effect, sPIF was added to the PC tumor cell line and microenvironment in order to assess its effect on proliferation. Data generated indicate that PIF detection by IHC in PC closely correlates with high GS and GG. Thus, PIF has the necessary characteristics to become an independent marker for diagnosing aggressive PC, thereby improving both the timing and the patients’ treatment choices.
Overall, PIF expression has the features to become a useful, simple-to-use marker for tumor clinical management, correlating with its aggressiveness.
Material and methods
Prostate cancer patient samples and tissue microarray construction
The study series included 50 laparoscopic radical prostatectomy specimens.
Representative tissue areas were used to build tissue microarrays (TMAs), which included benign and pre-neoplastic lesions in addition to PC. No other information than the GS and GG were accessed, maintaining strict patient confidentiality.
The surgical specimens were fixed in 10% formalin, were step sectioned and embedded in paraffin. Hematoxylin-eosin–stained sections from each histologic tissue block were reviewed to confirm the presence of PC and to document the presence of HG-PIN and benign prostatic tissue. A total of 149 different cores taken for the 50 prostatectomy samples were selected for TMA construction.
IHC was performed on 3μ-thick tissue sections obtained from the resulting TMA blocks. All sections were deparaffinized in Bio-Clear (Bio-Optica) and hydrated through graded ethanol. Antigen retrieval was performed by a calibrated water bath capable of maintaining the Epitope Retrieval Solution EDTA (pH 9.0) at 97°C for 15 minutes. The sections then were allowed to cool down to room temperature (RT) for 20 minutes. To block endogenous peroxidase activity, slides were treated with EnVision FLEX™ Peroxidase-Blocking Reagent (Dako Agilent). Anti-PIF mouse monoclonal antibody (BioIncept LLC) was conjugated with biotin (Biosynthesis) and was then applied to TMA sections at 1:25 dilution, which was followed by 1-hour incubation at room temperature. The immunohistochemical reaction was visualized by using the streptavidin-peroxidase ready to use (Thermo Fisher Scientific) with 3,3′ diaminobenzidine as chromogen. The positive control included in every run was placental tissue. The negative control was performed by substituting the primary antibody with Mouse IgG1 Isotype Control (Thermo Fisher Scientific).
Control sections were run in parallel with the study samples. The sections were lightly counterstained with Mayer’s hematoxylin.
PIF immunostaining evaluation
PIF immunostaining (IS) was semi-quantitatively assessed and considered positive if any staining was seen in the cytoplasm of the cells. PIF IS shows granular stain in the cytoplasm of the tumor cells. Samples were graded according to the intensity of staining; for example, negative, weak, or strong. The weak intensity showed finely granular or incomplete stain with often prevalent apical enhancement. Samples were considered strong positive when there was a diffuse and intense cytoplasmic granular stain easily visible at low magnification (5×). In positive cases, a marked powdery granular stain in whole cytosol with different-sized colorful spots was observed. Only samples with strong staining were considered positive; meanwhile samples with weak or absent IS, were considered negative (Figure 1).
(a) Negative PIF staining in prostate cancer. (b) Positive PIF staining in prostate cancer.
Cell cultures
Human prostate cancer cell lines (PC3) were purchased from European Collection of Cell Culture (ECACC). Prostate fibroblasts were obtained from prostate cancer specimens (CAFs) from patients undergoing radical prostatectomy. Written informed consent was obtained by all patients in accordance with the Ethics Committee of Azienda Ospedaliera Universitaria Careggi (Firenze, Italy). PC3 cells and fibroblasts were routinely cultured in DMEM (Sigma/Aldrich) supplemented with 2 mM glutamine, with penicillin (100 U/mL, Sigma/Aldrich), streptomycin (100 μg/mL, Sigma/Aldrich), and 10% fetal bovine serum (FBS, Euroclone), and incubated at 37°C in a humidified atmosphere of 5% CO2. Peripheral blood mononuclear cells (PBMC) were isolated from human peripheral blood using Ficoll-Paque. 38
Proliferation assay
Synthetic PIF (MVRIKPGSANKPSDD) >95% purity (PPL (Proprietary)). The effect on sPIF on PC3 cell proliferation was determined in sequential experiments, cells alone, and combined with CAFs, and finally with PBMC, as a three-partite assay. Cell proliferation was evaluated using a CFDA-SE fluorescent probe. PC3 cells were labeled with the 2.5 μM CFDA-SE in a PBS buffer for 15 minutes, detached, then plated alone or in co-culture with fibroblasts (CAFs) and/or PBMC. After 24 hours, cells were detached, fixed in 3% paraformaldehyde, and analyzed by flow cytometry. The fluorescence values were analyzed by ModFit software to measure the cell proliferation index.
Cell proliferation was evaluated using carboxyfluorescein diacetate succinimidyl ester (CFDA-SE, Invitrogen™) fluorescent probe. PC3 cells were labeled with the 2.5 μM CFDA-SE in PBS buffer for 15 minutes, detached, then plated alone or in co-culture with fibroblasts (CAFs). After 24 hours, cells were detached, fixed in 3% paraformaldehyde and analyzed by flow cytometry. The fluorescence values were analyzed by ModFit software to measure cell proliferation index.
Statistical analysis
Immunohistology analysis: Univariable linear regression analysis was applied to assess the relationship between PIF expression and other variables. All analyses were performed with Microsoft Excel version 2016, and a value of P < 0.05 was considered to indicate a statistically significant difference.
Cell proliferation analysis: For comparison between two groups, significance was determined by using the Student’s test; P<0.05 was considered significant.
Results
Pathologic diagnosis at radical prostatectomy included GS 3+3= 6 (GG 1) PC; GS 3+4= 7 (GG 2) PC; GS 4+3= 7 (GG 3) PC; GS 4+4 = 8 (GG 4) PC; and GG 5 PC in 4, 20, 14, 8, and 4 cases, respectively. Among the 4 GG 5 PCs, 3 cases were GS 4+5= 9 PC and 1 case was GS 5+4=9 PC.
Among the 149 prostatic tissue samples evaluated, 105 showed PC, while 32 showed benign prostatic tissue, and 12 showed HG-PIN. Among the 105 TMAs showing PC in 9 cases, there was a coexistence of focal HG-PIN in the same sample.
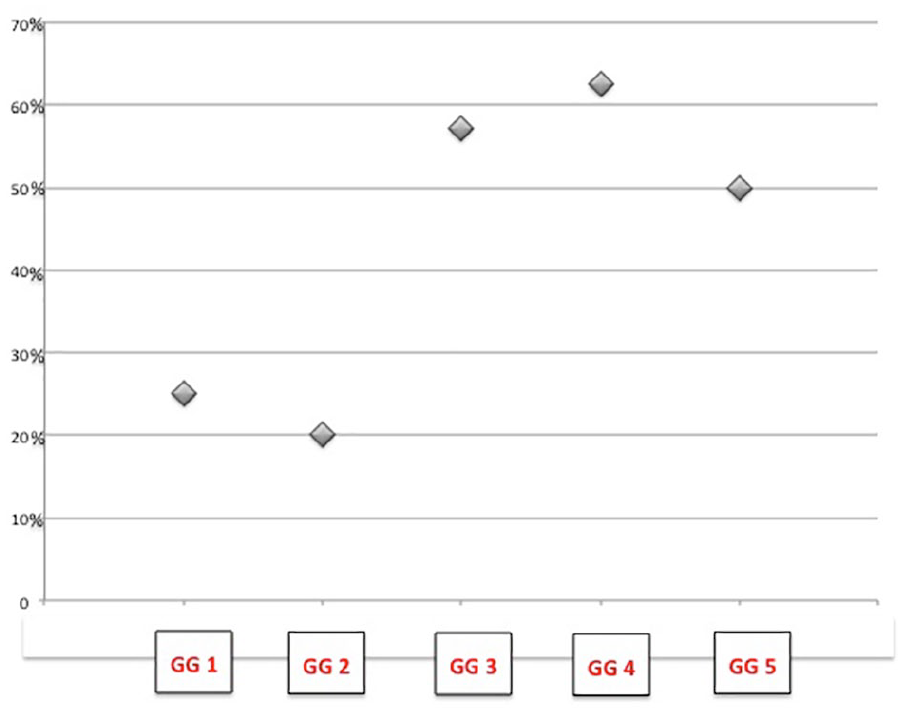
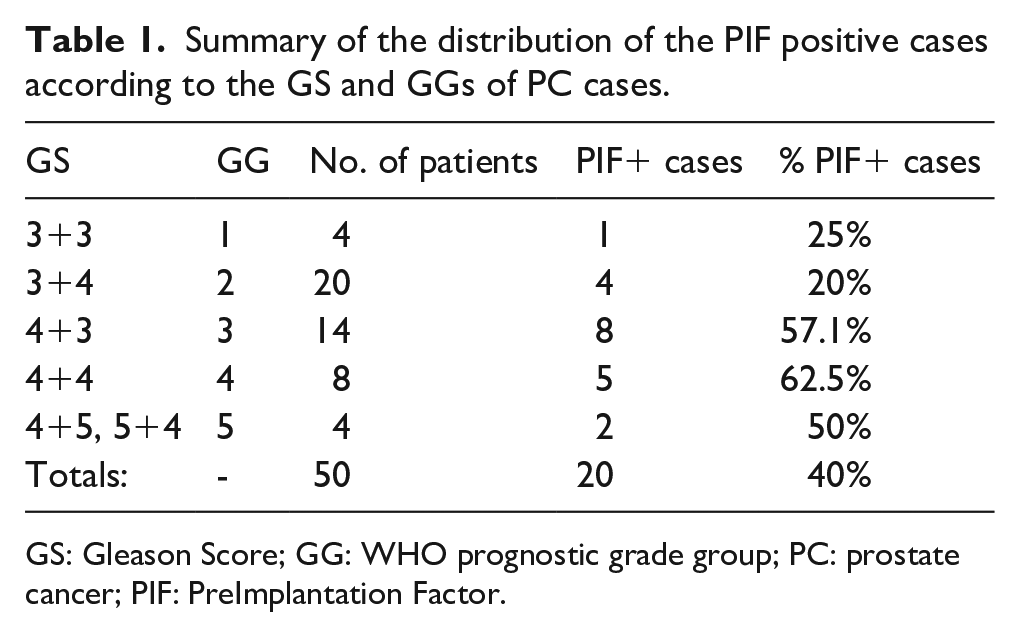
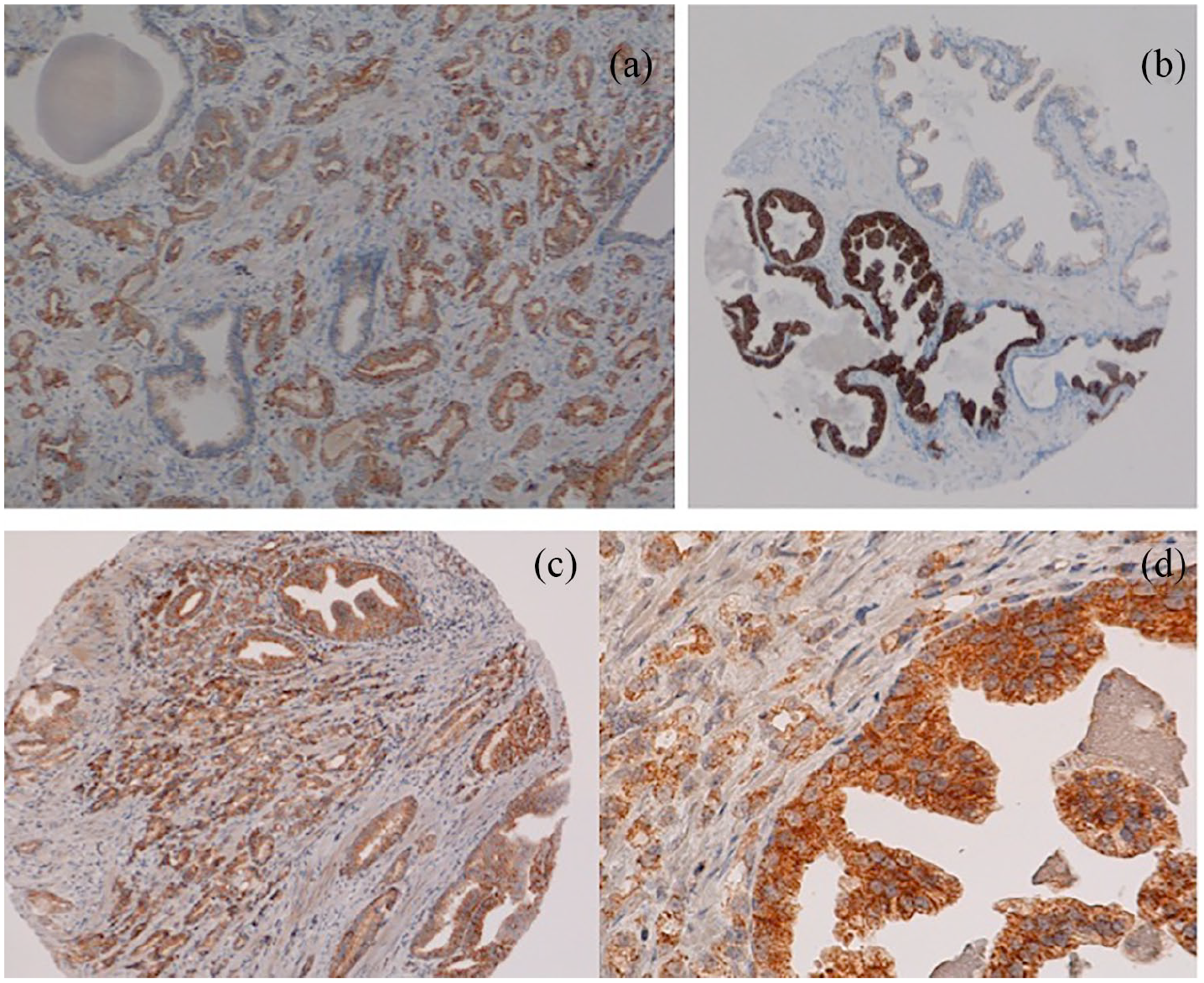
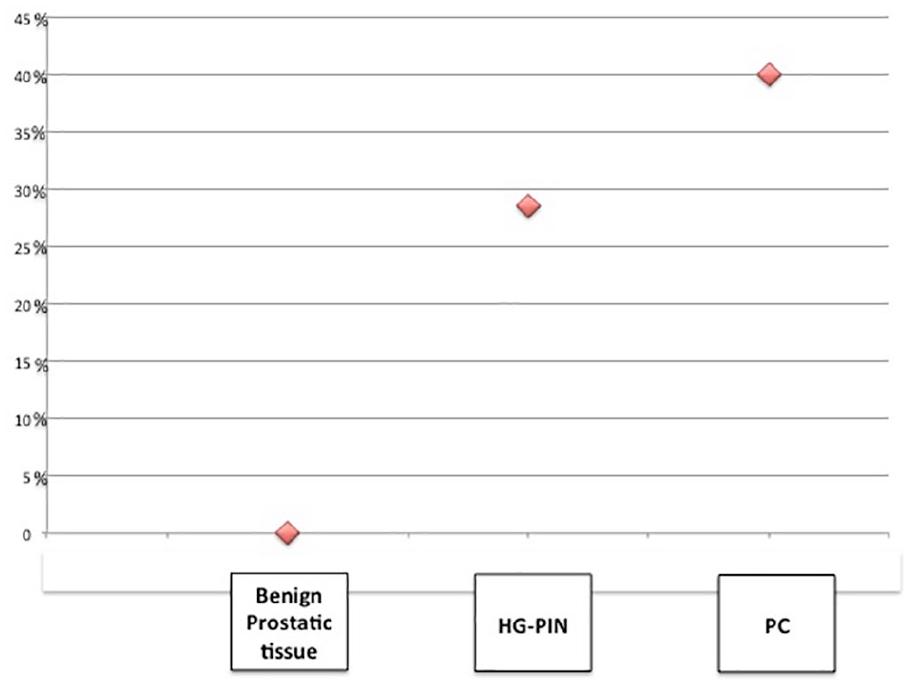
In the PC samples, a strong PIF-positive IHC-based staining in 40% of the cases was observed; in the PC GS 3+3 GG1 cases, the PIF IS was observed in 25%; in the PC GS 3+4 GG2 cases, the PIF IS was observed in 20%; in the PC GS 4+3 GG3 cases, the PIF IS was observed in 57.1%; in the PA GS 4+4 GG4 cases, the PIF IS was observed in 62.5%; and in the PC GS GG5 cases the PIF IS was observed in two (50%) of the four analyzed cases (Figure 2). Table 1 summarizes the distribution of the PIF positive cases according to the GGs of PA cases. Figure 2 shows the distribution of the percentage of the cases with PIF IS according the stratification in GGs of PC and the PIF IS, which is statistically related to the PC cases with an extensive presence of pattern 4 (P = 0.01). Figure 3 shows that the negative IS in benign prostatic tissue (100% of the cases) and the different PIF detection in HG-PIN. PIF IS was not present in any of the 32 benign prostatic tissue samples, while in the HG-PIN we observed PIF-positive IS in 28.57% of the cases (6 of 21; Figure 4). In the 9 cases where there was coexistence in the TMA of HG-PIN adjacent to PC, the IS of PIF was the same in HG-PIN and PC. In the nine TMAs with PC and HG-PIN, HG-PIN and the PC adjacent showed positive stain in four cases (44.4%). While in the other HG-PIN samples, non-PC-associated, we observed the PIF IS in 2 of the 12 cases (16.7%).
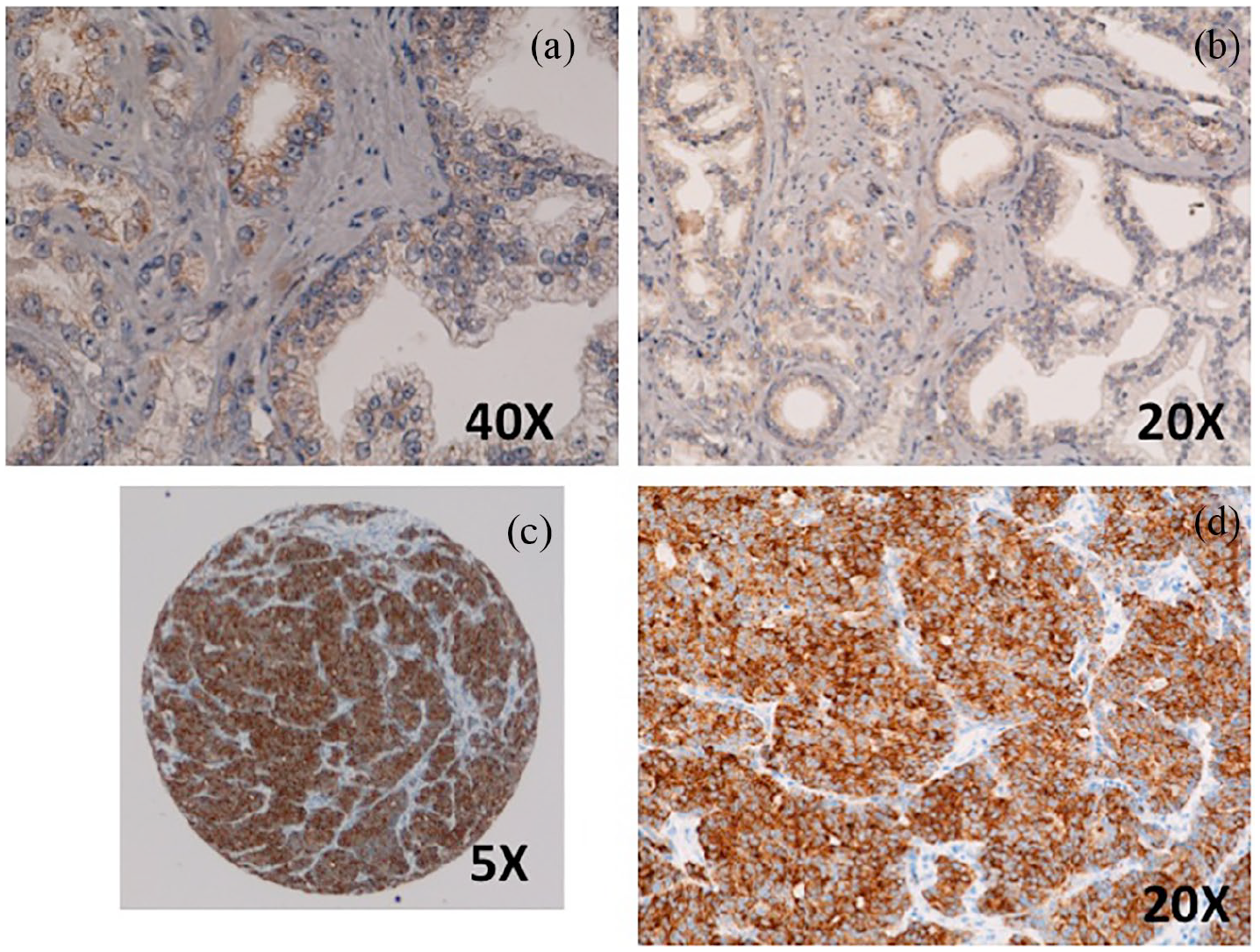
Positive PIF stain in Gleason pattern 3 prostate cancer (PC) (a) and (b). Positive PIF stain in pattern 4 PC (not well-formed glands (c); fused glands (d)). Positive PIF stain in Gleason Pattern 4, glomeruloid-type PC (e). PC with single cell Gleason pattern, 5 (f).
Summary of the distribution of the PIF positive cases according to the GS and GGs of PC cases.
GS: Gleason Score; GG: WHO prognostic grade group; PC: prostate cancer; PIF: PreImplantation Factor.
The distribution of the percentage of the cases with positive PIF immunostaining according the stratification in prognostic grade groups of prostate cancer (PC), and that the PIF detection is statistically related to PC cases with the presence of extensive prognostic grade group 4 (GG 4) (P = 0.01).
Negative PIF stain in a benign prostatic tissue (a). Negative PIF stain in HG-PIN (b). Positive PIF stain in a HG-PIN, while the benign prostatic tissue shows PIF negative stain (c). Positive PIF stain in HG-PIN and high-grade prostatic carcinoma (d).
Effect of sPIF on PC cell line proliferation
To evaluate the local role of PIF on the tumor microenvironment, the effect of sPIF on cultured tumor cells proliferation using a prostatic tumor cell line (PC3) as a model was tested. Initially, the sPIF effect was tested on cultured PC3 cells, and a mild a mild inhibitory effect was observed. Subsequently, in independent experiments, activated fibroblasts derived from patients with PC (CAFs)—as the support matrix promoted tumor cells proliferation—were added together with 400nM sPIF. Similarly to that observed using PC3 cells alone, sPIF had only a mild inhibitory effect following this combined co-stimulatory culture. Finally, when a large number of circulating PBMCs were added, the sPIF effect was tested in this three-partite culture, where a modest 15% was noted, but a significant increase in PC3 proliferation was also noted (P<0.05). Labeling PC3 cells with the CFDA-SE probe enabled us to determine the proliferation rate despite the presence of other cell types in this complex culture system (Figure 1S, supplementary material).
Discussion
The pivotal finding of the current study is that PIF IS, as analyzed by IHC, correlates with high-grade GS and GG in patients with PC. Treatment planning in patients with PC is based on the pathological findings obtained from the prostate by needle biopsy and/or following radical prostatectomy. Prior to the current study, the GS system represented the single most powerful prognostic test in PC, permitting the selection of the most appropriate patient treatment. 39 Nevertheless, despite recent revisions of the GS and the introduction of the GGs40,41 there is an expanded diagnostic utility to use biomarkers and molecular testing for selecting tailored therapies. Recent research and literature have focused on the identification of new biomarkers that are useful in predicting disease progression, specifically Ki-67, BCl-2, CD147, Cox2, and ALDH1A1.42-47
Frequently, PC presents as an indolent progressive disease, and pathologists experience difficulties in identifying aggressive cases that necessitate definitive therapy compared with a truly indolent cases, which would warrant only active surveillance. The search for a reliable biomarker for PC that enables the detection of aggressive cases is an active field of research.
Ideally, a biomarker that rapidly and directly correlates with the cancer’s aggressiveness would provide the physician with a simple to use, reliable, utilitarian tool to choose both the type and the timing of treatment accordingly tailored to each patient.
The present study offers important elements of such a solution. We observed that benign prostatic tissue does not stain with PIF under any circumstances; however, conversely, in a large number of patients, intense PIF staining was observed in malignant prostatic glands. PIF immune staining correlated with higher GS PC: 50%–62% of the PC GS ⩾ 4+3 patients strongly stained with PIF; conversely, in the cases with a GS ⩽ 3+4 score PC, PIF was detected in only 20%–25% of cases. Importantly, PIF staining was statistically related to PC cases with an extensive presence of pattern: GS 4 (P = 0.01). Furthermore, HG-PIN adjacent to PC showed PIF positivity in 44.4% of cases compared to the 16.7% of PIF positive immune stain in the case of isolated PIN-HG. These data suggest that positive PIF expression in PIN-HG may be a marker for an invasive (or even aggressive) neoplasia in the core biopsy.
Regarding staining specificity, in benign prostate samples PIF is not detected, and similarly, even in non-malignant tissue adjacent to PC. Of note, despite the high GS, only in a certain percentage of patients PIF staining was present, which may reflect a different prognostic outcome in this subgroup. This may also be the case in those with HG-PIN, which may herald future invasiveness. Thus, PIF could serve as a potential predictive biomarker that complements both the GS and the GG scoring systems.
Solid tumors are composed of heterogeneous subpopulations of cancer cells, and by tumor stroma mainly CAFs, cells that promote tumor growth, neo-angiogenesis, and metastasis. Inflammation is instrumental in local immune effects and drives the access of immune cells to the tumor microenvironment. Transformed and non-transformed cells within tumors have an ongoing complex multidirectional crosstalk, which is essential for tumor progression. This intimate interaction is difficult to replicate in vitro using cell lines that have lost their primary tumor properties and the critical interaction among the cell types that is present in the tumor microenvironment.
Increased PIF staining was also found in endometriosis, while in culture of endometriosis cells sPIF had an inhibitory effect on proliferation. 32 Following semi-allogeneic bone marrow transplantation, PIF prevented graft-versus-host disease while reducing mortality and spleen metastasis. Similarly, in human metastatic melanoma, sPIF increased immune recognition (increased footprint) and reduced spleen metastasis. Moreover, sPIF led to autophagy of cultured human melanoma cells derived from metastatic lymph nodes while it did not affect melanoma or other cell lines. Hence, we also investigated whether PIF, beyond being an effective biomarker, has a regulatory role in PC, promoting autophagy or immune privilege. We attempted to recreate a tumor microenvironment in PC. Data generated on the sPIF effect on PC3 prostate cancer cell line proliferation showed that sPIF did not affect PC cells, either when cultured alone (minimal decrease), or when CAFs, fibroblasts from PC patients were present, which recreated a favorable tumor microenvironment. Only when systemic immune cells were added to the culture a modest increase in proliferation was noted. Therefore, PIF does not influence a single cell type, but acts by interfering with the crosstalk between cells belonging to the tumor microenvironment, although the mechanisms behind this are not fully elucidated. This evidence indicates that sPIF acts on primary tumor cells where specific signaling and receptors are still preserved. Mechanistically, data have also emerged revealing that in primary human glioma cells sPIF prevents stemness through reduced SOX2 expression (Brodie, under submission), which is also highly relevant for PC.12,47 In contrast, in vivo sPIF promotes normal neural cell differentiation by reduced let-7 levels, and through the PKC/PKA phosphorylation pathway.18,48,49
Of note, PIF was not detected in all high GS PCs; this may help to better categorize the tumor’s aggressiveness and long-term prognosis.
Even within the same patient when PIF staining in the cancer was intense, in the adjacent normal gland it was negative. The ability to carry out this assay rapidly in any laboratory with minimal tools and no difficulties could help to implement the patient’s clinical management. This will overcome the current limitation of the study where a high number of cases will be needed to establish the assay’s clinical value. Recent data published on uterine cancer support that PIF detection in advanced uterine cancer is associated with worse prognosis. 33 Whether this is also the case in this study remains to be shown. An additional weakness is that PIF identification is based on an antibody, although monoclonal, and therefore tissue extraction and proteome analysis is needed to confirm whether the entire PIF sequence is intact, truncated, or is present in altered form.
Importantly, the method used for diagnosis by PIF was a simple IHC method, which was already validated in the human placenta and served as a positive control. The large number of normal prostate and benign prostatic hyperplasia samples where expression was low to negative represent a strength of this research.
Currently used PC tumor markers appear to proceed in a reverse fashion, first associated with late fetal development proteins and, as it becomes anaplastic, their embryonic origin becomes more prominent. Current evidence indicates that the prostate stem cells are the most aggressive since they survive castration in PC patients. 50 Adult cell expressions of PIF based on IHC are mostly minimal and remain dormant. However, as seen in chronic inflammation due to endometriosis (known to trigger propensity to malignancy), specifically where cellular hyperplasia is present, PIF re-expression is prominent. This may be analogous to what is found with PIF detection in PIN samples.
In conclusion, PIF immune staining in PC is associated with a high GS. This is observed in up to 60% of cases and with a subset of HG-PIN. The increase in the score directly correlates with increasing PIF detection, while in a normal prostate gland PIF is not detected. PIF detection thus may be of value for clinical decisions in patients with PC. Additional studies are ongoing to assess the impact of PIF IHC in the risk stratification of patients with high-grade GS PC. Data herein strongly support the use of PIF as a biomarker for PC. Overall, PIF expression has the features to become a useful marker for tumor clinical management, correlating with its aggressiveness.
Supplemental Material
Figure_1S – Supplemental material for PreImplantation Factor immunohistochemical expression correlates with prostate cancer aggressiveness
Supplemental material, Figure_1S for PreImplantation Factor immunohistochemical expression correlates with prostate cancer aggressiveness by Maria Rosaria Raspollini, Ilaria Montagnani, Paolo Cirri, Gianna Baroni, Alessia Cimadamore, Marina Scarpelli, Liang Cheng, Antonio Lopez-Beltran, Rodolfo Montironi and Eytan R. Barnea in The International Journal of Biological Markers
Footnotes
Acknowledgements
We thank Amy Carter and Stephanie Zinn for their valuable editorial assistance.
Author contributions
Antonio Lopez-Beltran and Eyton R. Barnea contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Lopez-Beltran is supported in part by the Grant PI17/01981 FIS (Ministry of Health), Madrid, Spain.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
