Abstract
Background:
Helicobacter pylori-induced DNA damage and impaired homologous recombination repair are vital molecular mechanisms for gastric cancer, which mainly count on its virulence factors cytotoxic-associated gene A (CagA) and vacuolating cytotoxin A (VacA). However, the relationship between H. pylori CagA EPIYA motifs and vacA genotypes with DNA damage and homologous recombination repair markers is still not clear.
Methods:
H. pylori positive and negative gastric biopsies were taken from 165 subjects with different gastric precancerous pathologic stages, and DNA damage marker γH2AX and key homologous recombination repair proteins (CtIP and Rad51) were investigated for their association with H. pylori CagA EPIYA motifs and vacAs-, m-, i-, and d-region genotypes and histology (Sydney classification).
Results:
Out of 165 patients, 78 were identified as H. pylori-positive. CagA EPIYA motifs were identified as AB, ABC, and ABD in 2 (3.3%), 21 (35%), and 37 (61.7%) patients, respectively, while vacA alleles were identified as: s1, s2, m1, m2, i1, i2, d1, and d2 in 50 (89.3%), 6 (10.7%), 24 (42.9%), 32 (57.1%), 45 (80.4%), 11 (19.6%), 40 (71.4%), and 16 (28.6%) patients, respectively. vacAs1m1i1d1, s1m2i1d1, and s1m2i2d2 were the most prevailing genotypes. γH2AX was highly localized in H. pylori-positive tissues with corresponding CagA EPIYA motifs and vacA genotypes, while Rad51 and CtIP signals were weak.
Conclusion:
H. pylori were positively correlated with the DNA damage marker in precancerous lesions, but were negatively correlated with the key homologous recombination repair proteins, which may be due to the specific CagA EPIYA motifs and vacA genotypes.
Introduction
Helicobacter pylori is a gram-negative bacterium that colonizes the surface of the gastric mucosa. Its prevalent infection affects about half of the world’s population and causes gastritis, gastroduodenal ulcers, gastric lymphoma, and gastric adenocarcinoma.1,2 H. pylori is mainly localized in the precancerous stages: chronic superficial gastritis (CSG), chronic atrophic gastritis (CAG), and intestinal metaplasia (IM), which means that H. pylori may initiate the occurrence of gastric cancer, 3 but the exact molecular mechanism is still unknown.
Bacterial infection induces DNA damage can be carcinogenic. 4 DNA double-strand breaks (DSBs) are the most cytotoxic form of DNA damage, which can induce deletions, insertions, translocations, and even chromosomal aberrations. 5 Once DSBs occurs, it will activate γH2AX, a marker of DNA damage, which further recruits related proteins to the DNA damage sites and causes cell cycle arrest, apoptosis, or DNA repair. 6 DNA double-strand breaks (DSBs) are mainly repaired in two ways: (a) inexact non-homologous end joining (NHEJ), and (b) precise homologous recombination (HR) repair. 7 Compared to NHEJ, HR is more error free, since it requires an undamaged homologous sequence as a template for repair. HR repair initiated by the resection of the 5′ ends at the DNA break site, which is mainly promoted by MRE11 and CtIP to form single-stranded DNA. 8 Then, replication protein A is coated with the resected DNA, which is further replaced by Rad51 to form a homology-searching filament to pair with the undamaged homologous duplex to guarantee precise repair. 9 HR repair plays a crucial role in maintaining genomic integrity, so a mutation or abnormal expression of a key HR protein, such as CtIP or Rad51, may increase the incidence of cancer. 10
The major virulence factors of H. pylori are CagA and VacA. CagA is directly secreted into the host epithelial cells through the type IV secretion system. Then it undergoes tyrosine phosphorylation at the EPIYA motifs and activates downstream nuclear factor-κB to induce inflammation of the gastric epithelial cells. 11 The phosphorylation of CagA occurs in the carboxyl terminus at the conserved tyrosine residues that are part of a repeated five-amino-acid sequence (Glu-Pro-Ile-Tyr-Ala) referred to as the EPIYA motif, which can be classified as EPIYA-A, EPIYA-B, EPIYA-C, or EPIYA-D. 12 The CagA EPIYA motif has been found to be associated with the development of gastric cancer, but the exact molecular mechanism is not clear. 13 On the other hand, the vacA gene contains several polymorphic regions tightly related to its vacuolating activity and clinical outcomes, including the signal (s)-, intermediate (i)-, middle (m)-, and deletion (d)-region. Based on the amino acid sequence differences within these regions, two primary variants have been described for each region: s1 and s2 for the signal region; i1 and i2 for the intermediate region; m1 and m2 for the middle region; and d1 and d2 for the deletion region. 14 The combination of the vacA s-, m-, and i-region genotypes provides different vacuolating activity for the various H. pylori strains. Among them, H. pylori strains with the vacA s1m1 allele have been associated with increased toxicity and gastric cancer. 15
Previously, in-vitro studies have shown that H. pylori induce DNA DSBs and inhibit the function of many DNA repair pathways’ genes in gastric epithelial cells, including the HR repair genes mre11 and rad51, which triggers the risk of gastric cancer. 16 However, less supporting data are available regarding studies on clinical tissues. Moreover, it is not clear if the H. pylori virulence factors regulate HR repair in the gastric precancerous lesions. In this study, we aimed to find the association of H. pylori with γH2AX and key HR proteins (Rad51 and CtIP) in gastric precancerous lesions. We also aimed to study whether CagA EPIYA motifs and vacA polymorphisms correlate with the DNA damage and HR repair systems in gastric precancerous lesions. Findings of the current study will provide novel insights for a molecular mechanism for H. pylori-induced gastric adenocarcinoma.
Materials and methods
Samples collection
Gastric tissues were collected from 165 patients undergoing gastroscopy in the 5th Affiliated Hospital of Zhengzhou University. Sampling was performed from March 2017 to May 2019. Patients were investigated for symptoms of digestive tract discomfort, acute bleeding, use of oral anticoagulants, medical history for H. pylori eradication, use of medicines in the last 2 weeks (use of antibiotics, proton pump inhibitors, or antitumor drugs, etc.). All the sampling and experimental procedures were approved by the ethical review board of Zhengzhou University. Informed written consent was obtained from all patients before sampling. All sampling and experimental procedures were performed by strictly adhering to guidelines of the Helsinki declaration 1964 and its latest amendments.
Histological examination
Endoscopic gastric tissues were taken from the gastric corpus and antrum, fixed in 10% formaldehyde overnight at 4˚C and embedded in paraffin for microtome sectioning. Hematoxylin-eosin staining was performed to determine the histopathological types, including 80 cases of CSG, 32 cases of CAG, 35 cases of IM, and 18 cases of dysplasia (Dys). Pathological diagnosis and classification were performed by two experienced pathologists strictly following the criteria of the World Health Organization and the updated Sydney system. 17
Reagents and antibodies
Rabbit anti-H. pylori polyclonal antibody (ZA-0127) (Beijing Zhongshan Jinqiao Biology Corporation, P.R. China), γH2AX antibody (#2595) (Cell Signaling Technology, Danvers, MA, USA), mouse anti-RAD51 polyclonal antibody (MA5-14419) and rabbit anti-CTIP polyclonal antibody (PA5-20963) (Thermo Fisher Scientific, Waltham, MA, USA), a rabbit secondary antibody kit (SP-9001), a mouse secondary antibody kit (SP-9002), goat serum, horseradish peroxidase labeled streptavidin, hematoxylin and DAB color reagents (Beijing Zhongshan Jinqiao Biology Corporation, P.R. China), and PBS powder and sodium citrate antigen recovery solution (Beijing Solarbio Biology Corporation, P.R. China) were used in this study.
Immunohistochemistry
To determine the expression and localization of H. pylori, γH2AX, Rad51, and CtIP immunohistochemical analysis was performed. Paraffin sections were mounted on slides and dewaxed in dimethylbenzene, followed by rehydration through 100%, 95%, and 85% ethanol, and incubated with fresh 3% H2O2 for 10 min to block endogenous peroxidase activity. Goat serum was used for blocking for 15 min. Primary antibodies were incubated overnight with the tissue at 4°C. The next day, the biotinylated secondary antibody was added drop-wise and incubated at 37°C for 20 min. The sections were washed with PBS and incubated with horseradish peroxidase labeled streptavidin for 20 min then washed with PBS and incubated with DAB for 5 min. A blank control group was incubated overnight with PBS instead of a primary antibody. All sections were counterstained with hematoxylin for histopathological examination.
Review and scoring for tissue sections
Slides were examined under a light microscope and the results were organized according to the Sydney criteria. Cells stained with yellow or brown color in the nucleus and/or cytoplasm were defined as positive in the gastric mucosa epithelial or glandular tube tissues. Immuno-histochemical scoring was performed using a semi-quantitative method. 18 Positive cell number scoring criteria: I. the percentage of positive glandular epithelial cells in total glandular epithelial cells; that is, 0: <5%; 1: 5%–25%; 2: 26%–50%; 3:51%–75%; 4: ⩾75%. II. The staining intensity: 0: no staining, 1: weak staining, 2: moderate staining, and 3: strong staining. Then, the “area × intensity” was calculated, by which the overall expression levels of the proteins in the section were defined, as follows: score 0 to 2 for negative (−); score 3 to 5 for weak positive (+); score 6 to 8 for moderate positive (++); and score 9 to 12 for strong positive (+++).
Genomic DNA extraction of H. pylori and PCR methods
DNA extraction of H. pylori was performed using a DNA Mini Kit (Qiagen, Duesseldorf, Germany, #51304) following the manufacturer’s instructions. Amplification of DNA was performed in a PowerCycle SL96 Gradient machine (Biometra GmbH, Goettingen, Germany) with 15 µL 2×Taq Master Mix (Solarbio, Beijing, China, #PC1120), 8 µL RNase-free water, 1 µL of each forward and reverse primer (10 µmol/L), and 5 µL H. pylori DNA. Ten primer sets were used for the study: CagA EPIYA motif, vacA s1, vacA s2, vacA m1, vacA m2, vacA d1, vacA d2, vacA i1, and vacA i2 (Supplementary Table 1). Polymerase chain reaction (PCR) was carried out as follows: enzyme activation at 94°C for 2 min, 30 cycles of denaturation at 98°C for 10 s, annealing for 30 s followed by final extension at 68°C for 7 min. PCR products were visualized by agarose gel electrophoresis, ethidium bromide staining, and a DL2000 DNA Marker (Takara, Dalian, China, #3427A) was used as a molecular weight marker.
Statistical analysis
The data obtained were statistically analyzed using SPSS20.0 (SPSS Inc., Chicago, IL, USA) software. The χ2 test and the McNemar test were used for two unpaired groups or multiple groups, respectively. The χ2 test and the Fisher’s exact test were used for contingency tables. P<0.05 was considered as statistically significant.
Results
Expression of γH2AX and key HR repair proteins in different gastric precancerous lesions
DNA damage marker (γH2AX) and the key HR repair proteins (Rad51 and CtIP) in different gastric precancerous lesions were successfully detected by immunohistochemistry in tissues obtained from 165 patients (Figure 1). Furthermore, we analyzed the correlation of γH2AX and HR repair proteins with the gastric precancerous lesions. No significant correlation was seen between γH2AX and gastric precancerous lesions, but high expression of γH2AX was found in all stages (P>0.05, Supplementary Table 2). Comparably, high expression of the key HR proteins Rad51 and CtIP was noted at a high level in CAG, CSG, and IM, but at a lower level in Dys (P<0.05, Supplementary Tables 3 and 4).
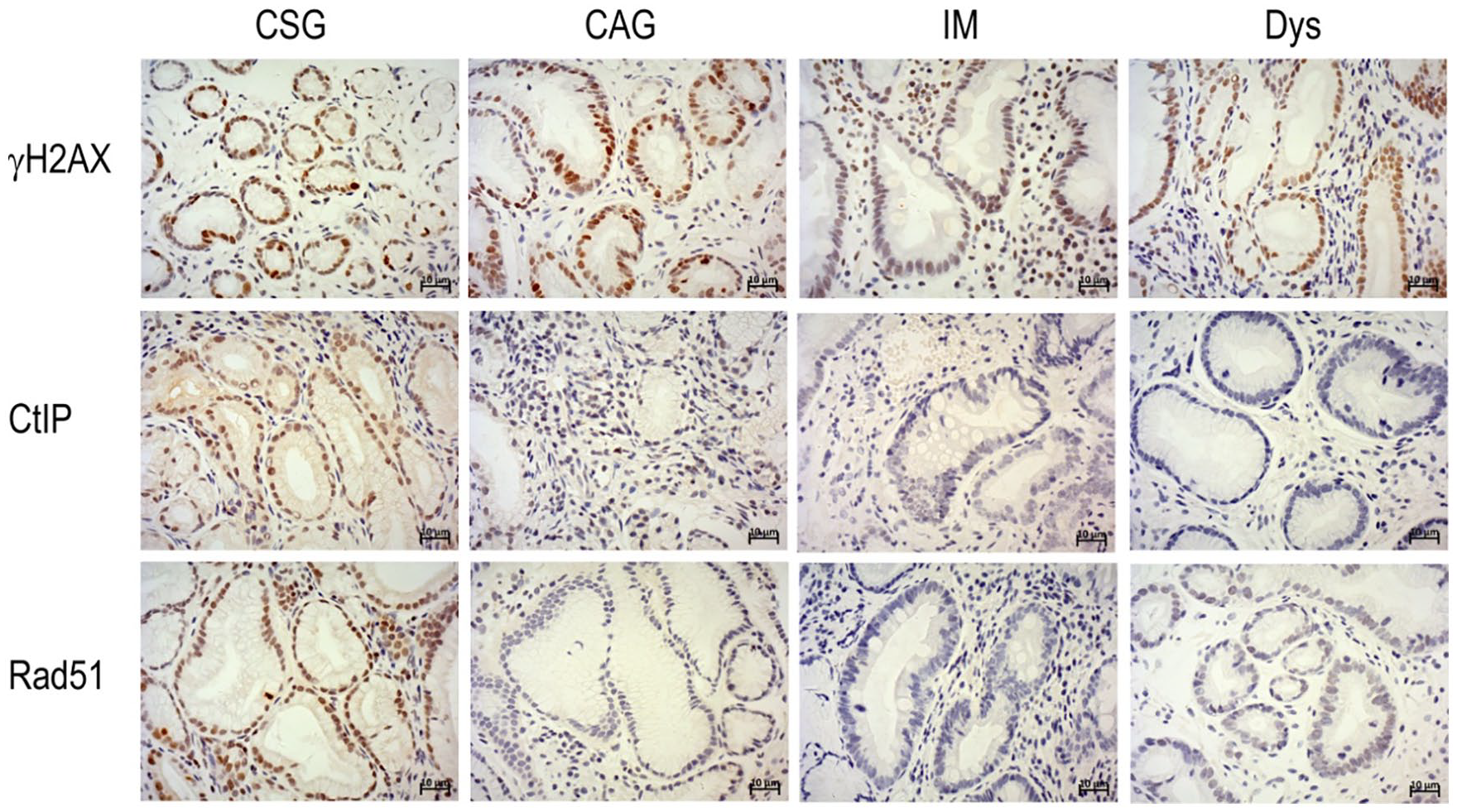
Immunohistochemically staining of γH2AX, Rad51 and CtIP protein in chronic superficial gastritis (CSG), chronic atrophic gastritis (CAG), intestinal metaplasia (IM) and dysplasia (Dys) (magnification, ×400; scale bar, 10µm).
Effect of H. pylori on the expression of the γH2AX and key HR repair proteins in different gastric precancerous lesions
We further investigated the correlation of H. pylori with γH2AX and HR proteins during the gastric precancerous cascade. H. pylori infection was measured by IHC in all patients, and γH2AX and HR protein levels were analyzed in the H. pylori-negative and H. pylori-positive groups. Higher expression of γH2AX was noted in the H. pylori-positive than in the H. pylori-negative group in CSG, CAG, and IM (P<0.05, Table 1), but for Dys, no significant difference was found between the H. pylori-positive and the H. pylori-negative subgroups. However, for the HR proteins, the expression of Rad51 was significantly lower in the H. pylori-positive subgroup than in the H. pylori-negative group in CSG, but was significantly higher in CAG (P<0.05, Supplementary Table 5). For the IM and Dys, no statistically significant difference was found between the H. pylori-positive and H. pylori-negative subgroups. Furthermore, Rad51 was maintained at a low level in IM and Dys compared with CAG and CSG in the H. pylori-positive group. Similarly, the expression of CtIP was significantly lower in the H. pylori-positive subgroup than the H. pylori-negative group in CSG and CAG (P<0.05, Supplementary Table 6); however, in IM and Dys, no significant difference in expression was found between the H. pylori-positive and H. pylori-negative groups. Similar to Rad51, the CtIP was maintained at a low level in IM and Dys compared with CAG and CSG in the H. pylori-positive group.
Expression of γH2AX in patients with various histological observations in relation to H. pylori infection.
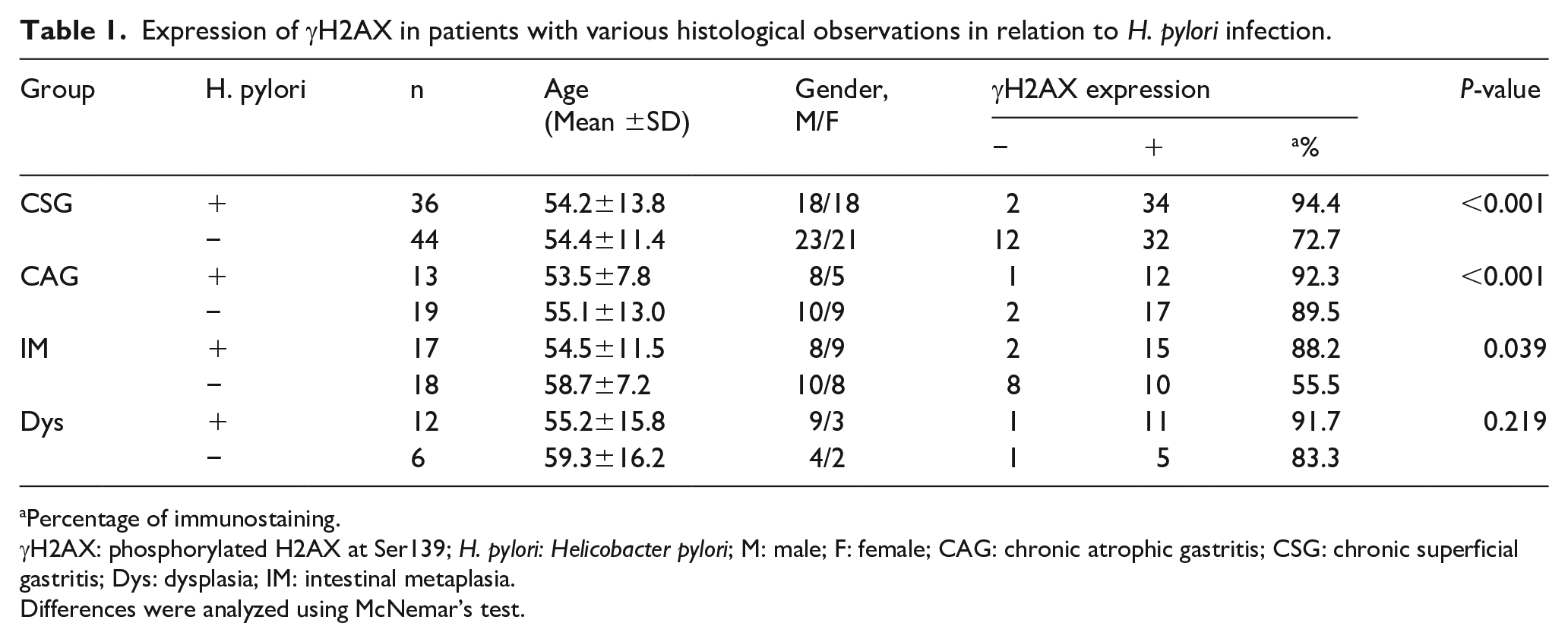
Percentage of immunostaining.
γH2AX: phosphorylated H2AX at Ser139; H. pylori: Helicobacter pylori; M: male; F: female; CAG: chronic atrophic gastritis; CSG: chronic superficial gastritis; Dys: dysplasia; IM: intestinal metaplasia.
Differences were analyzed using McNemar’s test.
Association between the CagA EPIYA motif, vacA genotypes, and gastric precancerous lesions
We evaluated the correlation between CagA EPIYA motifs and vacA genotype with gastric precancerous lesions. According to our results, 60 H. pylori strains were successfully identified with their corresponding EPIYA motif, which were further classified as AB, ABC, and ABD in 2 (3.3%), 21 (35%), and 37 (61.7%), respectively. Interestingly, both the eastern type ABD and western type ABC formed the main proportion (Table 2). The vacA alleles were evaluated based on four polymorphic regions: s-, m-, i-, and d-regions. A total of 56 H. pylori strains were successfully identified by their vacA alleles, which were classified as s1, s2, m1, m2, i1, i2, d1, and d2 in 50 (89.3%), 6 (10.7%), 24 (42.9%), 32 (57.1%), 45 (80.4%), 11 (19.6%), 40 (71.4%), and 16 (28.6%), respectively (Table 2). Further analysis showed that the three most highly detected vacA genotypes were s1m1i1d1, s1m2i1d1, and s1m2i2d2, with detection rates of 37.5%, 26.8%, and 17.9%, respectively. The genotype with the highest frequency in the CSG and CAG groups was s1m1i1d1, accounting for 38.5% and 72.7% of the detected expression, respectively. The genotype with the highest frequency of detection in the IM and Dys groups was s1m2i1d1, accounting for 36.4% and 25.0% of the detected expression, respectively (Table 2).
Relationships between CagA EPYIA motif, vacA polymorphism, and gastric precancerous lesions.
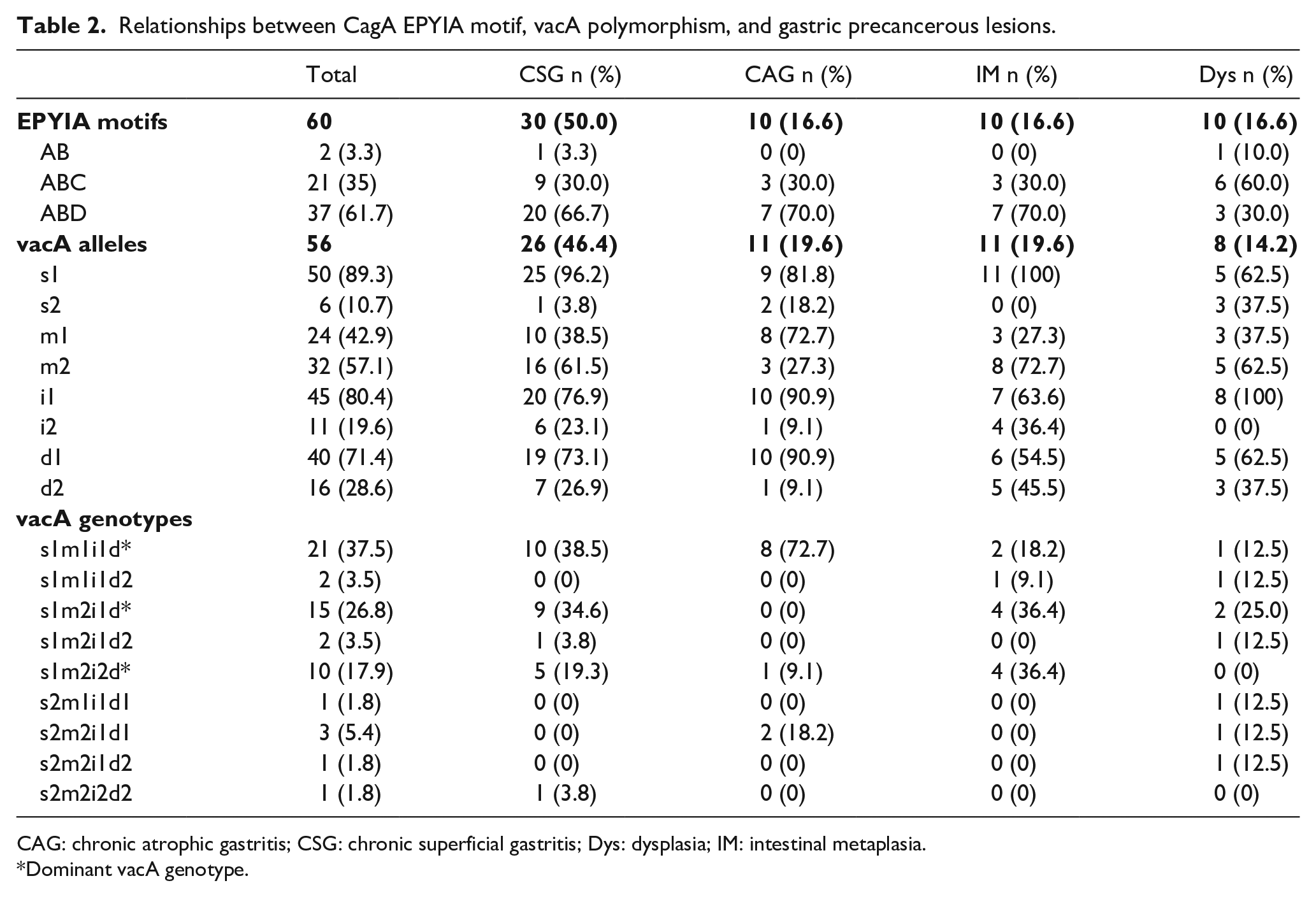
CAG: chronic atrophic gastritis; CSG: chronic superficial gastritis; Dys: dysplasia; IM: intestinal metaplasia.
Dominant vacA genotype.
Effect of CagA EPIYA motifs and vacA genotype on expression of γH2AX and key HR proteins
In the final step, we analyzed whether CagA EPIYA motifs and the three dominant vacA genotypes affected the expression of γH2AX and key HR proteins (CtIP and Rad51). No significant correlation was found between the expression of DNA damage proteins and CagA EPIYA motifs and vacA genotypes (Tables 3 and 4), which may be due to the low sample size. However, it was shown that γH2AX maintained at a higher level in the tissue with the corresponding CagA EPIYA motifs at 100%, 90%, and 94.6%, respectively. However, the HR protein Rad51 expressed comparably lower levels in the corresponding CagA EPIYA motifs at 50%, 52.4%, and 73%, respectively. CtIP showed comparably lower expression levels in the tissue with corresponding CagA EPIYA motifs at 50%, 71.4%, and 73%, respectively (Table 3). Similarly, DNA damage marker γH2AX showed higher levels in the tissues with the levels of the three dominant vacA genotypes (s1m1i1d1, s1m2i1d1, and s1m2i2d2) at 95.2%, 86.7%, and 100%, respectively (Table 4). However, the HR protein Rad51 showed comparably lower levels in the tissues with the corresponding levels of the three dominant vacA genotypes at 76.2%, 66.7%, and 70.0%, respectively. CtIP also had lower levels in tissues with the corresponding levels of the three dominant vacA genotypes at 71.4%, 60.0%, and 50.0%, respectively (Table 4).
Relationships between CagA EPYIA motif and DNA damage marker and key HR repair protein.
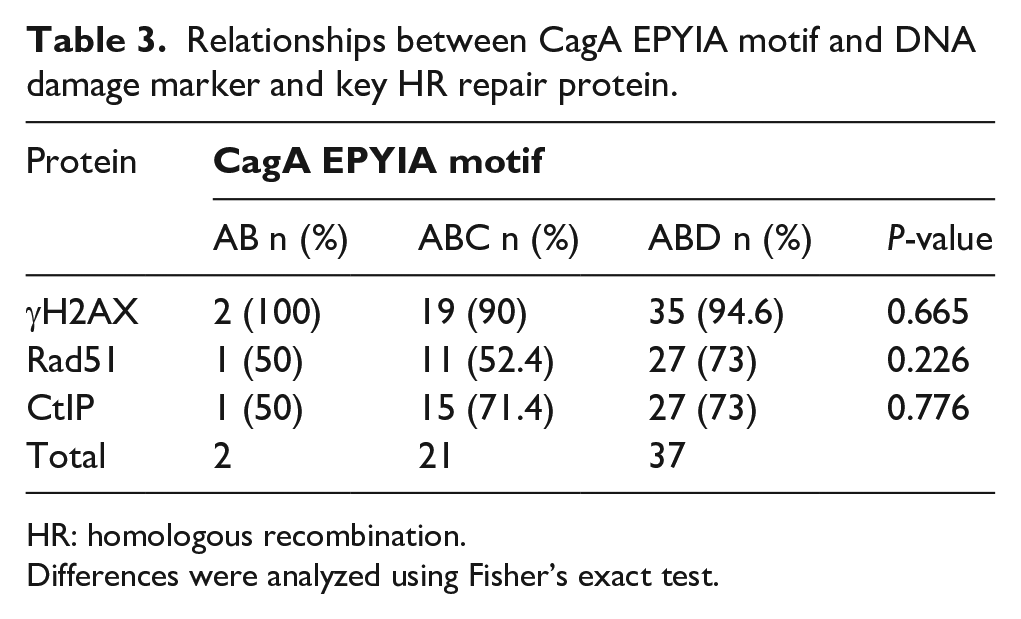
HR: homologous recombination.
Differences were analyzed using Fisher’s exact test.
Relationships between vacA polymorphism and DNA damage marker and key HR repair protein.

HR: homologous recombination.
Dominant vacA genotype.
Differences were analyzed using Fisher’s exact test.
Discussion
H. pylori mainly colonized the gastric mucosa in precancerous lesions, including CSG, CAG, and IM, which implies that H. pylori initiated the progression towards gastric cancer. 19 An in vitro study showed that H. pylorus inhibits most of the DNA repair genes and some HR repair proteins. 16 Clinical studies confirmed that the expression of the DNA damage marker γH2AX gradually increased in precancerous stages of gastric adenocarcinoma but showed a comparable lower level in the terminal adenocarcinoma stage, 20 which is consistent with our study.
However, there is not enough literature available on DNA repair proteins in precancerous gastric tissues—especially the HR repair proteins, which are essential for maintaining genome stability. In our study, we evaluated the expression of the key HR proteins CtIP and Rad51 in gastric precancerous lesions. In contrast to the DNA damage marker γH2AX, HR proteins CtIP and Rad51 had lower expression in IM and Dys. This implies that HR repair was dysfunctional or insignificant, which contributes to malignant progression. CtIP and Rad51 also showed lower expression at CSG stages in the H. pylori-positive group; low expression of the HR is reasonable as it was already known that DNA damage repair occurs by NHEJ repair instead of HR repair in H. pylori-infected gastric epithelial cells. 21
Here, we report for the first time that the tumor suppressor CtIP expresses in gastric precancerous lesions, predicting the role of CtIP in the initiation of the HR repair pathway and the central role of Rad51 in HR repair. Their decreased expression proves that H. pylori may inhibit HR repair in the gastric precancerous stage, which increases the genome instability and initiates the malignant process.
The CagA EPIYA motif correlated with the development of gastric cancer, especially the eastern CagA EPIYA-D motif with a higher affinity to SHP-2, which could be a clue to explain its toxicity. 22 Interestingly, we found a high proportion of western EPIYA-ABC motifs in the Henan province in China. Furthermore, the DNA damage marker γH2AX had a higher expression while the HR repair proteins Rad51 and CtIP showed comparably lower levels in tissues with the CagA EPIYA motif, which implies that H. pylori may inhibit HR repair proteins through the CagA EPIYA motif.
VacA s1m1 genotype is more toxic in inducing DNA damage, but vacA i and d alleles have not been well studied. 23 We discovered three dominant vacA genotypes s1m1i1d1, s1m2i1d1, and s1m2i2d2 for the first time in the Chinese population. Furthermore, we found that s1m1i1d1 plays a predominant role in CAG stage; the s1m1i1d1 and s1m2i1d1 genotypes take up the main proportion in the CSG stage; and s1m2i2d2 and s1m2i1d1 play a predominant role in the IM stage. DNA damage marker γH2AX is highly expressed in patients with all three dominant vacA genotypes while HR repair proteins Rad51 and CtIP have low expression, which implies that H. pylori may inhibit HR repair though the specific vacA polymorphisms.
In summary, we conclude that the decreased expression of HR proteins in gastric precancerous lesions may be associated with the CagA EPIYA motif and specific vacA genotypes. These results further confirmed the role of HR proteins during the progression of gastric precancerous lesions and supplied new insights to explain the molecular mechanisms of H. pylori virulence proteins to initiate gastric cancer.
Supplemental Material
supplemental_material – Supplemental material for The association of Helicobacter pylori CagA EPIYA motifs and vacA genotypes with homologous recombination repair markers during the gastric precancerous cascade
Supplemental material, supplemental_material for The association of Helicobacter pylori CagA EPIYA motifs and vacA genotypes with homologous recombination repair markers during the gastric precancerous cascade by Yang Mi, Haibin Dong, Xiangdong Sun, Feifei Ren, Youcai Tang and Pengyuan Zheng in The International Journal of Biological Markers
Footnotes
Author contributions
Yang Mi and Haibin Dong contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All human studies were approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Zhengzhou Major Collaborative Innovation Project (No.18XTZX12003); Medical service capacity improvement project of Henan Province in China (grant number Yu Wei Medicine [2017] No.66); International Science and Technology Cooperation Project of Henan Province in China (grant number 182102410018); Medical Science and Technology Project of Henan Province in China (grant number 2018020224); The Foundation of Henan Science and Technology Department (grant number 172102410014); The key scientific research project of the Foundation of He’nan Educational Committee (FHEC, #18A320011).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
