Abstract
The utility of positron emission tomography (PET) for the evaluation of response to immunotherapy has been considered a hot topic, particularly in the last 2 to 3 years. Different experiences have been collected in clinical practice, with 18F-Fluorodeoxyglucose (FDG) PET/computed tomography (CT), particularly in patients affected by lymphoma, malignant melanoma, and lung cancer. It has been tested in different settings of disease, from the prediction to the prognosis relative to the response to immunotherapy. In the present mini-review, some evidence is reported about the role of FDG PET/CT in patient candidates to or treated with immunotherapy.
18F-Fluorodeoxyglucose in patient candidates t(FDG) positron emission tomography (PET) has been extensively used for the evaluation of response to conventional chemotherapy, both in the interim and after the end of all the cycles, demonstrating a significant impact on patient management and prognosis.1-4 However, until now, few preliminary data are available about the utility of PET for the evaluation of response to immunotherapy. 5 Difference experiences have been collected in the recent years in clinical practice with FDG PET/computed tomography (CT), particularly in patients affected by lymphoma,6-7 malignant melanoma,8-12 and lung cancer.13-15 Its role has been tested in different setting of disease, such as the prediction, the assessment, and the prognosis relative to the response to immunotherapy.
In the study by Grizzi et al., 16 FDG PET/CT was able to identify lung cancer patients who will respond to immunotherapy by using the semiquantitative analysis (sensitivity of 88.9% in case of a SUVmax< 17.1 in the index lesion for the identification of the fast progression after 8 weeks of therapy). Similarly, Evangelista et al. 17 demonstrated that the sum of the data obtained by semiquantitative PET analysis was able to predict the responsiveness to immunotherapy in lung cancer, based on the patient’s gender.
For the assessment of response to immunotherapy, the experiences are variable. In 2016, the Lugano criteria for lymphoma were refined as LYRIC (Lymphoma Response to Immunomodulatory Therapy Criteria) introducing the concept of indeterminate response, for those cases with “delayed response” or “pseudo-progression,” which may be related to recruitment of immune cells to disease site. 6 FDG PET/CT has been used for the indeterminate response in different clinical trials, particularly to confirm complete response or progressive disease. 7 However, it also seems to be useful in cases of patients with stable or unclear response to therapy at CT imaging, by using RECIST1.1 (Response Evaluation Criteria in Solid Tumors) or iRECIST (immunoRECIST) criteria for the interpretation of the morphological images. 11 In melanoma patients, it has been demonstrated that the response to immunotherapy can be predicted with a sensitivity of 100% and a specificity of 93%8,10 (Figure 1). Similarly, the inclusion of a third PET scan, after 4 weeks from the second one (usually performed after 2 months from the start of immunotherapy) was able to differentiate between unconfirmed and confirmed progressive disease.
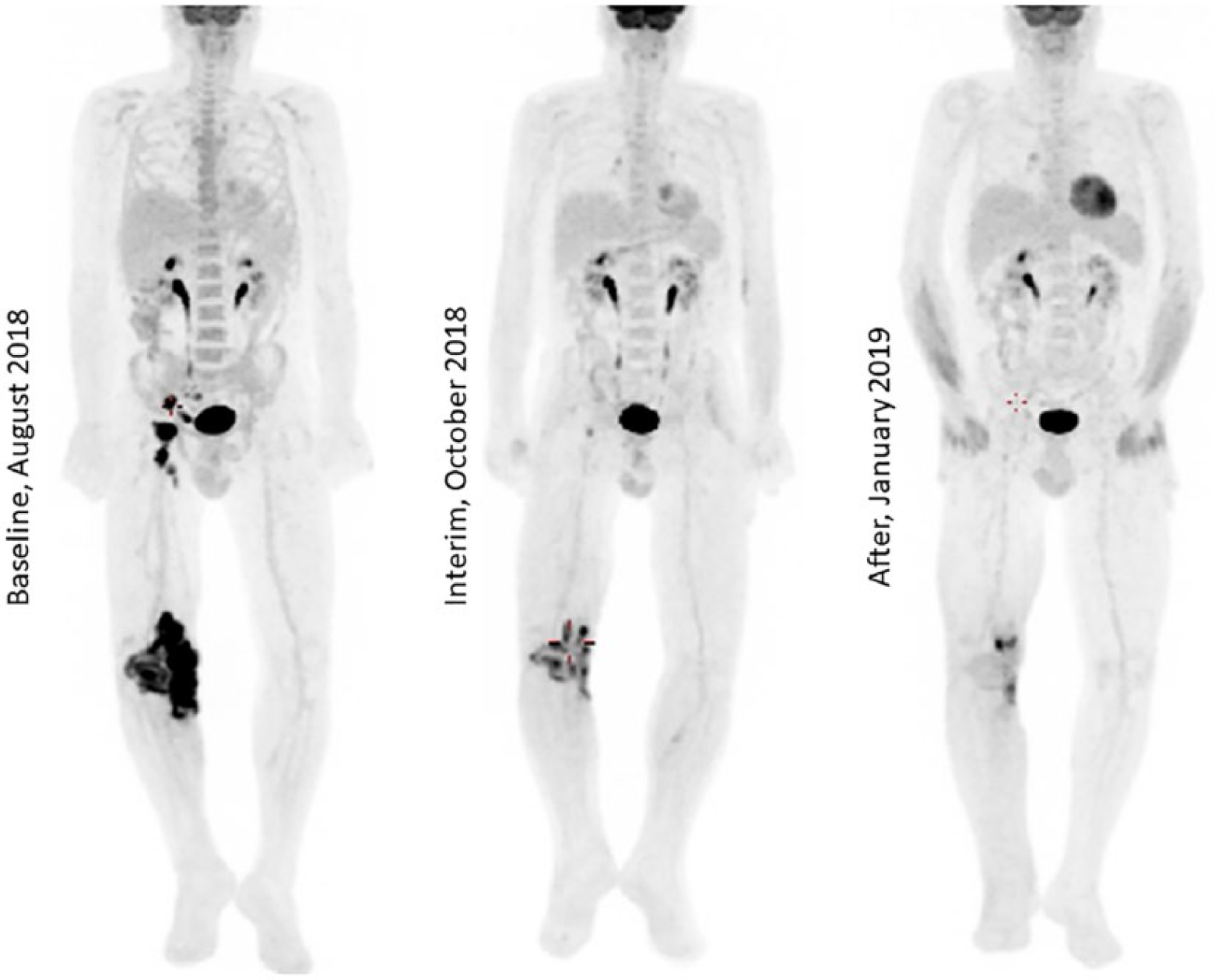
A 69-year-old man affected by a T/NK nasal type lymphoma treated with immunotherapy (Pembrolizumab). PET/CT scan showed a partial response to immunotherapy. The patient was later submitted to radiotherapy.
Some efforts have been made in recent years to identify specific criteria for the definition of response to therapy with FDG PET/CT (Table 1). The PERCIMT (PET Response Evaluation Criteria for Immunotherapy) criteria (by considering both morphological and functional size of the lesions) were tested at interim evaluation, after two cycles of immunotherapy in patients with melanoma, and were compared with the EORTC (European Organisation for Research and Treatment of Cancer) criteria. The PERCIMT showed a significantly higher sensitivity than the EORTC in predicting the clinical benefit (93.4% vs. 64.5%, respectively); however, it showed a lower specificity in predicting no clinical benefit. 10 The PECRIT, a mix of RECIST 1.1 and PERCIST criteria, predicted the response to immunotherapy at 21 to 28 days, with 100% sensitivity, 93% specificity, and 95% accuracy. 8 Finally, the above mentioned iPERCIST criteria were assessed in 28 patients affected by lung cancer, and showed a significant reclassification than iRECIST in 39% of cases. 15
Suggested criteria for the interpretation of images.
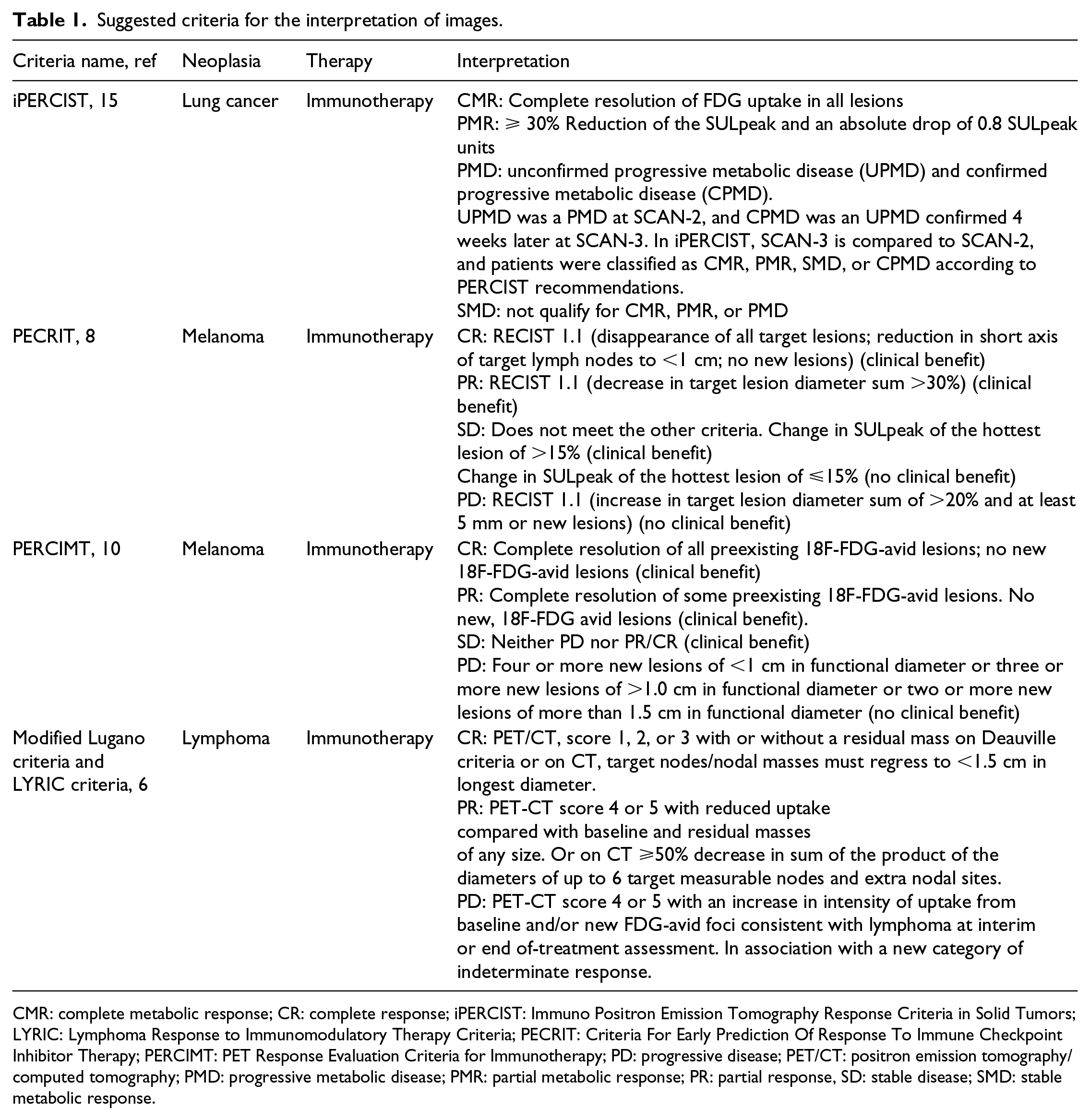
CMR: complete metabolic response; CR: complete response; iPERCIST: Immuno Positron Emission Tomography Response Criteria in Solid Tumors; LYRIC: Lymphoma Response to Immunomodulatory Therapy Criteria; PECRIT: Criteria For Early Prediction Of Response To Immune Checkpoint Inhibitor Therapy; PERCIMT: PET Response Evaluation Criteria for Immunotherapy; PD: progressive disease; PET/CT: positron emission tomography/computed tomography; PMD: progressive metabolic disease; PMR: partial metabolic response; PR: partial response, SD: stable disease; SMD: stable metabolic response.
FDG PET/CT has always been considered an optimal imaging instrument for the prognostic assessment. Patients with well-differentiated cancer has a better prognosis than those with scarcely differentiated tumors. Also, in patients undergoing immunotherapy, FDG PET/CT has reported some advantages in this setting. In the recent FIR-trial, lung cancer patients with a stable disease at CT RECIST analysis showed an early progression of disease at FDG PET/CT with a strong impact on the final prognosis. 14 Therefore, in patients with a stable disease by CT-based RECIST criteria at week 6, metabolic response with PET further informs the outcome. In the study by Goldfarb et al., 15 FDG PET monitoring with iPERCIST was an effective tool for discerning lung cancer patients who could benefit from treatment with nivolumab. The authors found that the 1-year survival rate was greater than 90% in responders and 11% for non-responders. A later FDG PET/CT (after 1 year from the end of immunotherapy), also may be considered a useful prognostic instrument in patients with malignant melanoma. 12
FDG is trapped in tissues with high glycolytic activity and thus it is not specific. Therefore, a high rate of false positive findings would be found, especially in patients undergoing immunotherapy, due to the activation of the immune system. Patient preparation and the physician’s interpretation of FDG PET/CT images during immunotherapy is essential for defining the response to therapy. First, the differential diagnosis between pseudo-progression 18 and hyper-progression 5 would be difficult to assess by using only two PET/CT scans; therefore, a third scan is warranted, as reported by Goldfarb et al. 15 and Cho et al. 8 Second, immune-related side effects (colitis, thyroiditis, others) should be taken into account for the interpretation of FDG PET/CT images, as they are associated with a high rate of false positive findings in the colon, thyroid, lung, and others. 5 However, Eshghi et al. 19 reported that the appearance of FDG uptake in the thyroid gland during immunotherapy was associated with the development of immune-related adverse events and therefore has a potential response to immunotherapy, due to the activation of the immune system (Figure 2). In order to overpass the limitations of FDG, alternative radiopharmaceuticals have been tested in preclinical studies, such as anti-PD-L1, anti-CTL-4, and similar, radiolabeled with radioisotope for single-photon emission computed tomography (SPECT; i.e. 111In) or PET imaging (i.e. 64Cu or 18F). The preliminary data are encouraging for the in vivo evaluation of PD-L1 expression in the lesions (both primary and metastasized). 20 To date, only one study is available regarding the role of 99mTc-labeled anti-PD-L1 sdAb SPECT/CT in patients affected by lung cancer. 21 The results demonstrated the feasibility and safety of this new agent, which would be used for the selection of patients who will respond to immunotherapy, and for monitoring the response to this treatment.
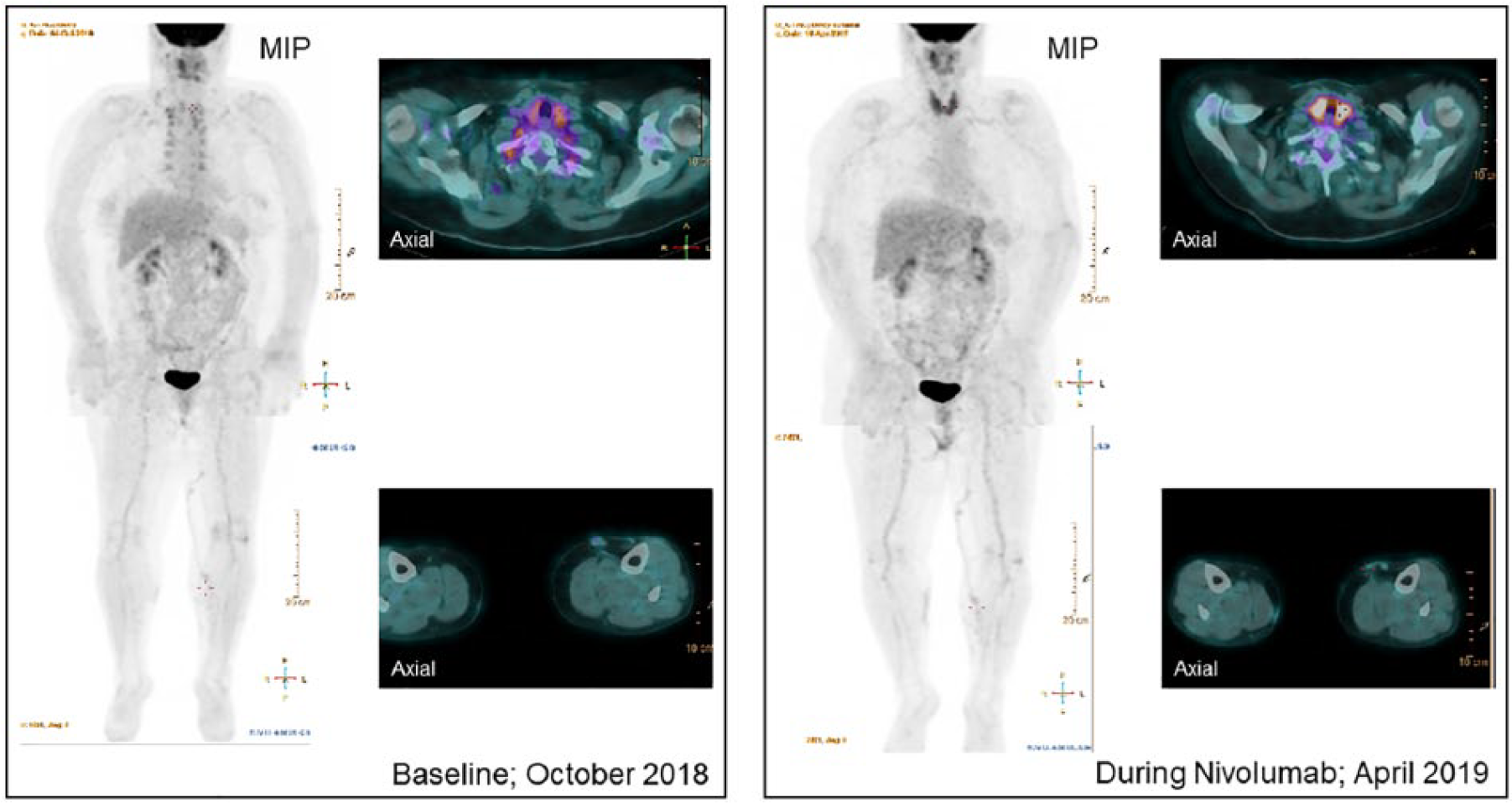
A 65-year-old woman with a cutaneous malignant melanoma treated with nivolumab. A partial response was noted after 4 months of treatment. The patient developed a thyroiditis relative to the immunotherapy.
In conclusion, first, more clinical trials are necessary in order to identify the role of FDG PET/CT in patient candidates to or undergoing immunotherapy; second, the standardization of criteria both for the definition of the optimal time between the immunotherapy cycle and the FDG PET/CT images, and for the definition of the response to therapy is necessary; and finally the potential benefit of PD-L1 theranostic imaging for the selection of patients should be tested in large cohorts of patients.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
