Abstract
Purpose:
Immunoscore was established to evaluate the prognosis of cancer patients. However, the feasibility of Immunoscore for the prognosis of cervical cancer remains unknown. To find other prognostic markers that contribute to immunological importance, immune checkpoint inhibitors targeting programmed cell death protein (PD-1), or its ligand, PD-L1, are of enormous interest. Our purpose is to investigate the expression of CD8 and PD-1/PD-L1 and their potential role in Immunoscore, supplementing the tumor/node/metastasis (TNM) classification of cervical cancer.
Methods:
Immunoscore was assessed according to the density of PD-1, PD-L1, and CD8 by immunohistochemistry. The association with overall survival and disease-free survival was assessed by the Kaplan–Meier method. To evaluate the effect of Immunoscore, a Cox proportional hazard regression classification was conducted. To compare the prognostic accuracies of Immunoscore and TNM staging, receiver operating characteristic curves were plotted.
Results:
Patients with PD-L1positive and PD-1high in immune cells had poorer overall survival and disease-free survival; however, PD-L1positive in tumor cells that infiltrated more CD8+ T cells were related to better overall survival and disease-free survival. These immune factors can be independent predictors for prognoses. According to these factors, a new Immunoscore system with priority in predicting prognoses was established. In receiver operating characteristic analysis for predictions of overall survival (the area under curve (AUC) = 0.833 vs. 0.766) and disease-free survival (AUC = 0.861 vs. 0.729), Immunoscore is more accurate than TNM staging.
Conclusions:
Thus, this Immunoscore system is an accurate predictive marker, which can be an important supplement to TNM staging for cervical cancer.
Introduction
Cervical cancer is one of the most common gynecological malignancies, which is a severe threat to females’ health in China. 1 Currently, most of methods used to predict clinical prognoses are based on the International Federation of Gynecology and Obstetrics (FIGO)—tumor/node/metastasis (TNM) staging. However, patients’ prognoses may vary with similar staging and pathological characteristics. 2 The reason may be that traditional classifications merely focus on the tumor itself, but not on the molecular signatures of patients. 3 Scholars have proposed that immune status may play significant roles in affecting tumor progression and prognosis. 4 As a result, researchers worldwide have proposed using Immunoscore to predict clinical outcomes of cancer patients during the last decade.5-7 The Immunoscore assessing the condition of intratumor T cells has been defined in various cancers, including liver, 5 melanoma, 6 and colon, 7 which is a prognostic factor superior to TNM classification. However, the prognostic significance and clinical pathological role of Immunoscore in cervical cancer remain unclear so far.
The Immunoscore system for cancer is based on density and location rather than on the function of the immune microenvironment. The immune microenvironment has an important impact on the progression and the prognosis of cervical cancer. The negative regulation of the immune signaling pathway is one of the most important ways in mitigating and terminating immune response, which is also known as immune checkpoint. 8 Immune checkpoints play a significant role in immune suppression and immune escape. Tumor-induced immune suppression and immune escape, which are mediated by the programmed cell death receptor 1 (PD-1) and its ligand (PD-L1), make tumor cells evade the host immunity. 9 Topalion and colleagues 10 treated tumors with PD-L1 and PD-1 monoclonal antibodies (mAbs), which brought a new wave of immunotherapy. Due to the fact that various tumors are likely to be immunosuppressed by the above mechanisms, immunological checkpoint inhibitors may become an “ideal panacea” against tumors. The efficacy of PD-1 and PD-L1 antibodies has been confirmed in current clinical trials. According to a completed phase Ib trial (Keynote-028), pembrolizumab achieved a general response rate of 14.3%, and 91% of patients had a response time of more than 6 months with recurrent or metastatic cervical cancer. 11 Because blockade of PD-L1/PD-1 can be effective in cervical cancer treatment, the expression of PD-L1 and PD-1 was thought to have prognostic significance, and high-risk human papillomavirus (HPV) is a major etiological factor of cervical cancer. 12 Because large numbers of immune cells are found at tumor sites, these complex components of the tumor microenvironment may be attributed to complicated characteristics of tumor immune responses. 7 Therefore, a comprehensive and reliable system of multiple immune effectors is needed and may provide more accurate prognoses for cervical cancer patients when compared to that of single-factor predictors.
Our study has not only evaluated the infiltration of CD8+ T cells, but also included the expression of PD-L1 and PD-1 in immune cells and tumor cells to make the Immunoscore targeting the immune response status of patients. We set up a multi-factor Immunoscore system—PD-L1 in tumor cell (TC-PD-L1), PD-L1 in immune cell (IC-PD-L1), PD-1 in immune cell (IC-PD-1), and CD8—to make a more accurate prognosis prediction for cervical cancer treated by surgery compared to TNM staging. Furthermore, the system may provide more precise prognoses and abundant information for the choices of immunotherapy in patients with cervical cancer.
Materials and methods
Patients
This study enrolled 222 cervical carcinoma patients who underwent radical hysterectomy with pelvic lymphadenectomy and were followed-up in the Harbin Medical University Cancer Hospital. Patients with 5 years of follow-up data (between October 2005 and October 2007) were selected. The analysis of survival, death, or completion was followed up to October 31, 2014. All the patients gave oral informed consent before enrollment in the study. The ethical approval was acquired from the Medical Ethics Committee of the Harbin Medical University Cancer Hospital, and all procedures were conducted in accordance with ethical principles. The tumor stage was determined according to FIGO staging. 13 The histological grade was classified based on the World Health Organization classification standards. 14 Patients were enrolled with the following criteria: stage I-IIA1; radical standardized hysterectomy; lymphadenectomy; none of these patients were given preoperative radiotherapy or chemotherapy before primary treatment of surgery; patients with high risks factors received a standardized combination chemotherapy regimen consisting of paclitaxel/paclitaxel liposome 135–175mg/m2 and cisplatin/nedaplatin 75mg/m2.
The main clinical and pathological characteristics of the patients, including age at diagnosis, cigarette smoking, weight loss, infection of HPV, FIGO staging, histologic type, histologic grade and lymph node metastasis, are described in the Supplementary Table 1.
Immunohistochemistry and evaluation of staining
All sections were examined by pathologists for the evaluation of TNM staging, differentiation, positive lymph nodes, and so on. Samples were harvested from paraffin-embedded specimens. Briefly, embedded tumor tissues were sectioned to 4μm thickness, dewaxed, and rehydrated. Paraffin-embedded slides were stained using an immunohistochemical technique that labeled the primary antibodies CD8 (MAB-0021, Maicin Biotech), PD-L1 (#13684, clone E1L3N, CST), PD-1 (ZM-0381, clone MRQ-22, OriGene). All the specimens were examined by two independent pathologists in a blind manner based on staining percentage and intensity of positive cells. The staining pattern of PD-L1 was defined as negative or positive if < 1% or ⩾ 1% in TCs, 11 or if < 5% or ⩾ 5% in ICs, respectively. 15 Staining of CD8 cells was defined as less or more if infiltration was < 20% or ⩾ 20% at the tumor site. 7 PD-1 was quantified by staining intensity: PD-1low if intensity was negative or weak, and PD-1high if intensity was intermediate or strong. 16
Statistical analysis
Statistical analyses were performed by SPSS 21.0 software (Chicago, IL, USA). The characteristics of patient tumors were compared using a t test for continuous variables or a Spearman test for categorical variables. Overall survival (OS) was defined as the time from surgery until death or the last follow-up and disease-free survival (DFS) was defined as the date from surgery to the first time of relapse or death. Survival analysis was performed using the Kaplan–Meier method. To evaluate the effect of the expression of CD8, PD-1, and PD-L1 on the OS and DFS, univariate and multivariate analyses using a Cox proportional hazard regression classification were carried out, and hazard ratios (HRs) were estimated with 95% confidence interval (CI) limits. P < 0.05 was considered as statistically significant. The prognostic accuracy of the model compared with that of the TNM staging was conducted by receiver operating characteristic (ROC) analysis.
Establishment of the Immunoscore system
We enrolled TC-PD-L1, IC-PD-L1, IC-PD-1, and numbers of CD8+ with independent prognostic significance in Immunoscore system. We assigned TC-PD-L1positive =0; TC-PD-L1negative =1; IC-PD-L1negative =0; IC-PD-L1positive =1; IC-PD-1low =0; IC-PD-1high =1; CD8more =0; and CD8less =1. These score indexes were then added together.
Results
Patients characteristics
The clinical characteristics of cervical cancer patients with stage I-IIA1 are summarized in Supplementary Table 1. The study included 222 patients, with the median age at the diagnosis being 49 years (range, 21–75 years). Of these patients, 95.05% tested positive for an HPV infection and only 4.95% were HPV negative. Together, 50 out of 222 patients had lymph node metastasis, and 80.63% patients belonged to stage I, according to the criteria released by the TNM staging. The characteristics of patients in current studies were consistent with those of Chinese cervical cancer patients. 17
Univariable and multivariable analyses of independent prognostic factors
Clinical characteristics and immune effectors were included and subjected to univariable analysis (Supplementary Table 2). FIGO staging and immune variables TC-PD-L1, IC-PD-L1, IC-PD-1, and CD8 had independent prognostic values for OS and DFS, according to the results of multivariable analyses (Table 1).
Multivariate Cox proportional hazard analyses for OS and DFS among patients with cervical cancer.
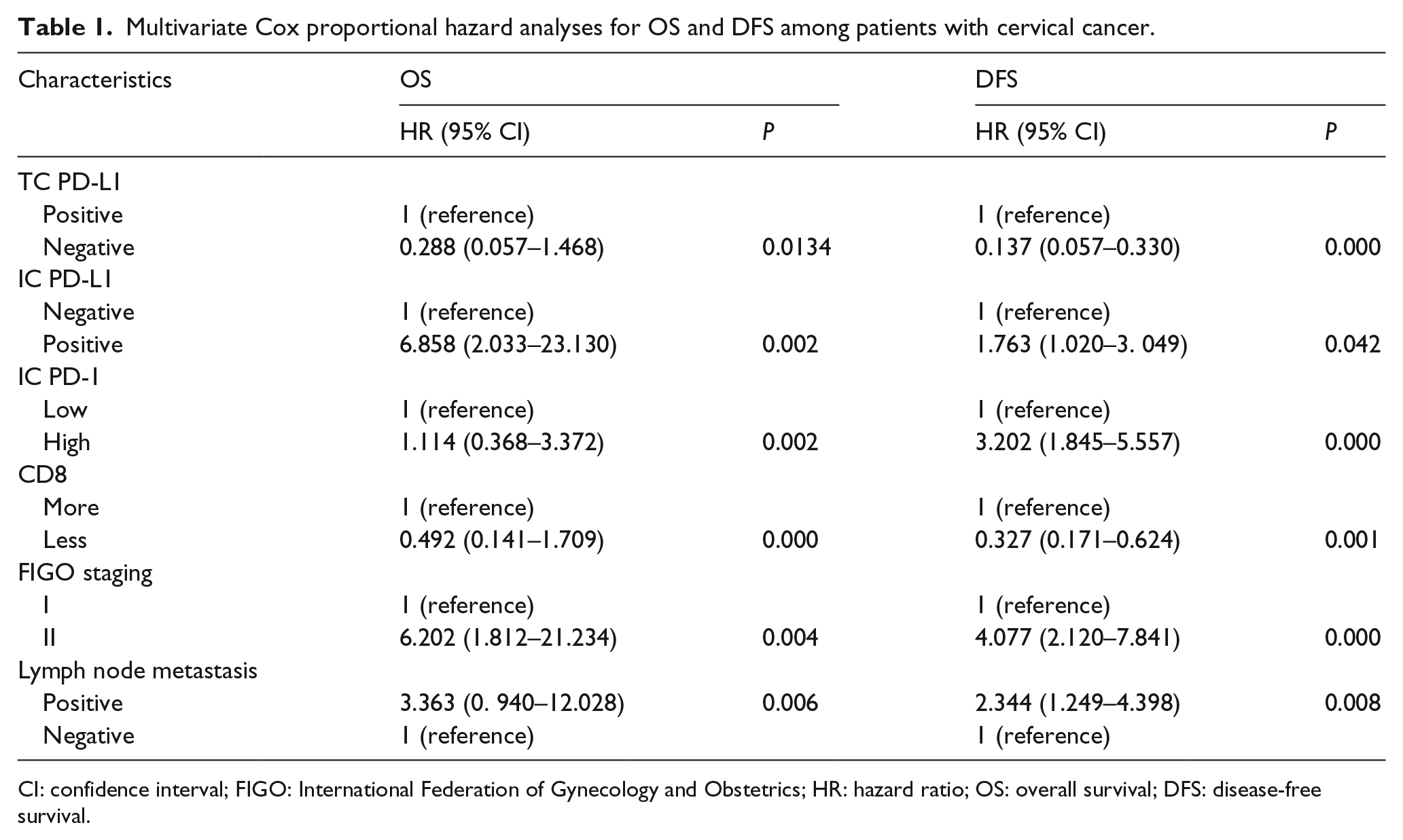
CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; HR: hazard ratio; OS: overall survival; DFS: disease-free survival.
IC-PD-L1positiveor IC-PD-1high in tumor-infiltrating immune cells predicted poor prognoses
We detected the expression of IC-PD-L1 infiltrating the tumor site (Figure 1(a) and (b)). IC-PD-L1positive was observed in 22.07% of the samples. Patients with IC-PD-L1positive had shorter OS and DFS compared with those with IC-PD-L1negative (P < 0.0001) (Figure 2(a) and (e)). The 5-year survival rate was 79.59% in the IC-PD-L1positive group versus 96.65% in the IC-PD-L1negative group, and the DFS rate was 40.82% versus 82.66%, respectively.
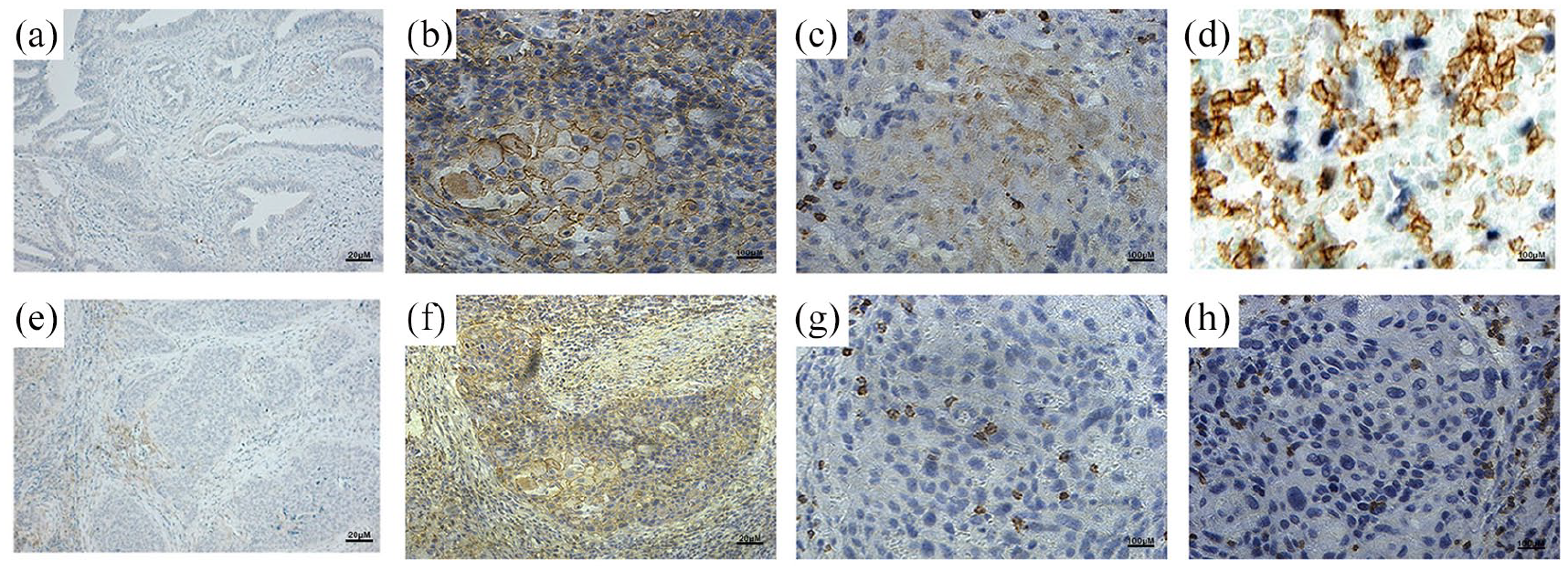
Representative images of IC-PD-L1, IC-PD-1, TC-PD-L1 and CD8. Negative (a) and positive (b) expression of IC-PD-L1. Low (c) and high (d) expression of IC-PD–1. Negative (e) and positive (f) expression of TC-PD-L1. Less (g) and more (h) expression of CD8 in tumor tissue.
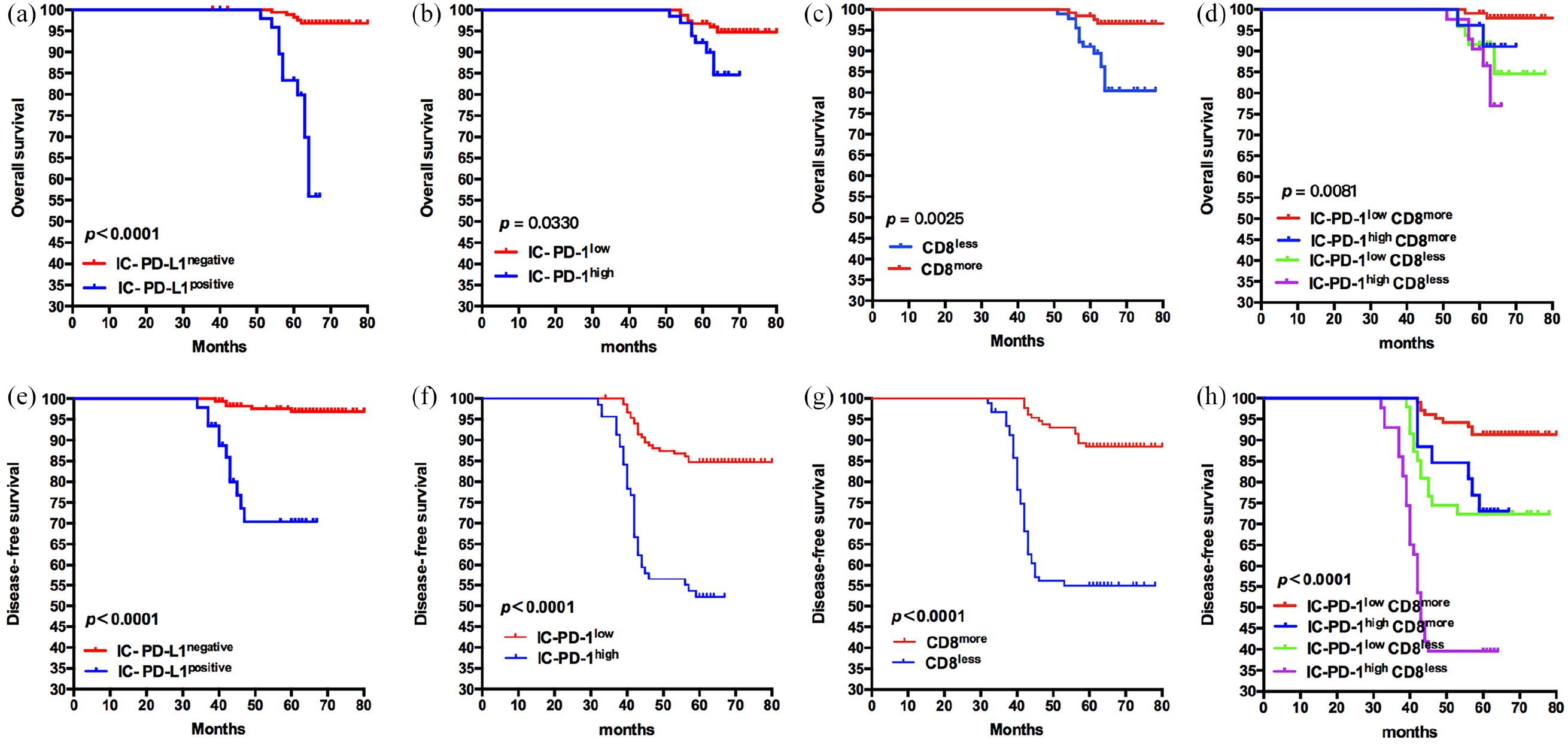
OS and DFS of cervical cancer patients based on the expression of IC-PD-L1, IC-PD-1, and CD8+ T cells. (a) and (e), OS and DFS of patients divided by the expression of IC-PD-L1 (P < 0.0001); (b) and (f), OS and DFS of patients grouped by the intensity of IC-PD-1 (OS: P = 0.0330; DFS: P < 0.0001); (c) and (g), OS and DFS of patients grouped by the infiltration of CD8 (OS: P = 0.0025; DFS: P < 0.0001). (d) and (h), the Kaplan–Meier curves for OS and DFS are shown for cervical cancer patients grouped according to IC-PD-1low CD8more, IC-PD-1high CD8more, IC-PD-1low CD8less, IC-PD-1high CD8less (OS: P = 0.0081; DFS: P < 0.0001). The probability of OS and DFS were estimated by using the Kaplan–Meier method and compared using the log-rank statistic.
We then studied the expression of IC-PD-1 infiltrating the tumor sites (Figure 1(c) and (d)). IC-PD-1high were detected in 31.08% patients. IC-PD-1high had poorer OS compared with IC-PD-1low (P = 0.0330) (Figure 2(b)). The 5-year survival rate was 89.86% in IC-PD-1high group versus 94.12% in IC-PD-1low group. IC-PD-1low had better DFS than IC-PD-1high (P < 0.0001) (Figure 2(f)), and the DFS rate was 50.72% versus 83.66%, respectively. No correlation was found between IC-PD-L1 or IC-PD-1 and the clinical characteristics of the patients.
Prognostic value of CD8+ T cell infiltration combined IC-PD-1 expression
CD8+ T cells play a pivotal role in anti-tumor immunity. 18 We detected infiltrations of CD8+ T cells by immunohistochemistry (IHC) staining (Figure 1(g) and (h)). CD8less was observed in 41.44% patients. Patients with CD8less tended to have poorer OS compared to those with CD8more (P = 0.0025) (Figure 2(c)), and the 5-year survival rate was 86.96% versus 96.92%, respectively. CD8less had poorer DFS compared to those with CD8more (P < 0.0001) (Figure 2(g)), and DFS rate was 54.35% versus 86.92%, respectively. We found that patients with CD8more/PD-1low had significantly better OS and DFS than those with CD8less/PD-1high (P = 0.0081 and P < 0.0001, respectively; Figure 2(d) and (h)). No correlation was observed between the expression of CD8 and other clinical characteristics of patients. The infiltration of CD8more T cells with PD-1low was associated with better prognoses.
The expression of TC-PD-L1positive correlated with CD8+ T cells infiltration and better prognoses
We detected expression of PD-L1 in tumor tissue by IHC staining (Figure 1(e) and (f)). PD-L1positve was observed in 61.71% patients. Patients with TC-PD-L1positive had better OS and DFS compared with those with TC-PD-L1negative (P < 0.0001) (Figure 3(a) and (b)). The 5-year survival rate was 97.81% versus 84.71%, and the DFS rate was 94.89% versus 38.82%, respectively. We confirmed that patients with TC-PD-L1positive and CD8more had the best OS and DFS (P < 0.0001) (Figure 3(c) and (d)). The association between TC-PD-L1positive and CD8more was also confirmed in the Spearman test (Supplementary Table 3), which may represent an adaptive immune status in cervical cancer.
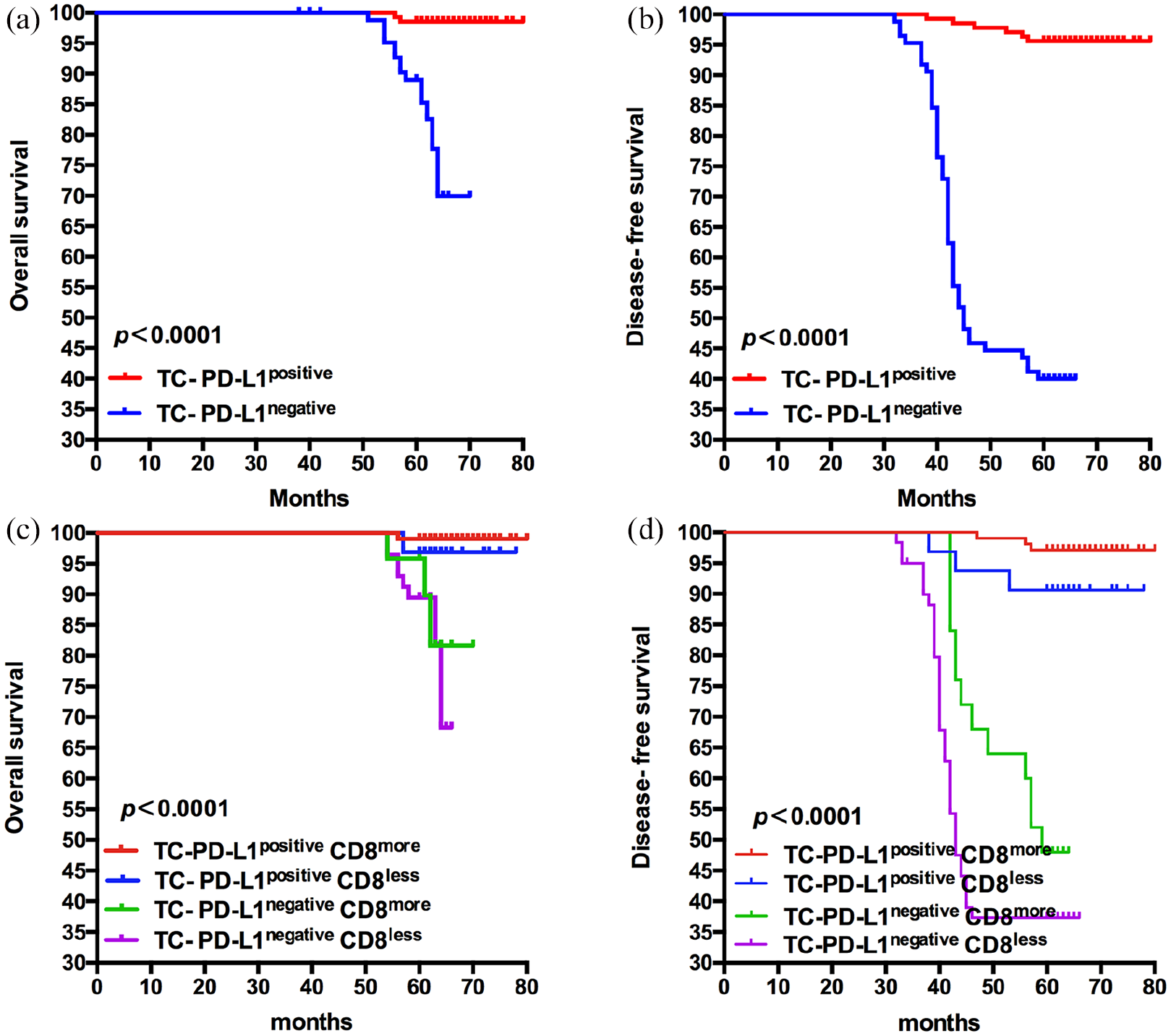
The expression of TC-PD-L1positive correlated with infiltration of CD8+ T cells predict better OS and DFS. (a) and (b), the OS and DFS of patients grouped by the expression of TC-PD-L1 (P < 0.0001). (c) and (d), the OS and DFS of patients grouped by TC-PD-L1positive CD8more, TC-PD-L1positive CD8less, TC-PD-L1negative CD8more, TC-PD-L1negative CD8less (P < 0.0001). The probability of OS and DFS were estimated by using the Kaplan–Meier method and compared using the log-rank statistic.
Establishment and validation of the Immunoscore system
Although the four-factor expression of TC-PD-L1, IC-PD-L1, IC-PD-1, and numbers of CD8+ had independent prognostic significance, their complicated interaction during anti-tumor immune responses prevented these factors from providing accurate prognosis predictions. Therefore, we proposed and established a comprehensive Immunoscore system for predicting patients’ survival. As shown in the Kaplan–Meier survival analysis, patients scored as 0 tended to have the best OS and DFS, and those with a higher index had poorer prognoses than those with lower (Figure 4(a) and (b)). For the convenience of classification, we combined Immunoscores 1 and 2 into one group and Immunoscores 3 and 4 into another group with no significant differences. In staging I, we found that patients with a lower Immunoscore had significantly better OS and DFS than those with higher (P = 0.0308 and P = 0.0074, respectively) (Figure 4(c) and (d)). The tendency in staging II is consistent with I (P = 0.0055 and P = 0.0002, respectively) (Figure 4(e) and (f)). To verify the accuracy of Immunoscore compared with TNM staging, ROC analysis was performed. Our system yielded an area under the ROC analysis for predictions of OS (the area under curve (AUC) = 0.833 vs. 0.766) and DFS (AUC = 0.861 vs. 0.729), and the Immunoscore is more accurate than for TNM staging (Figure 4(g) and (h)). The sensitivity values for TNM staging and Immunoscore were as follows: 69% and 75% in OS; 54% and 80% in DFS, respectively. The specificity results were as follows: 85% and 83% in OS; and 92% and 74% in DFS, respectively.
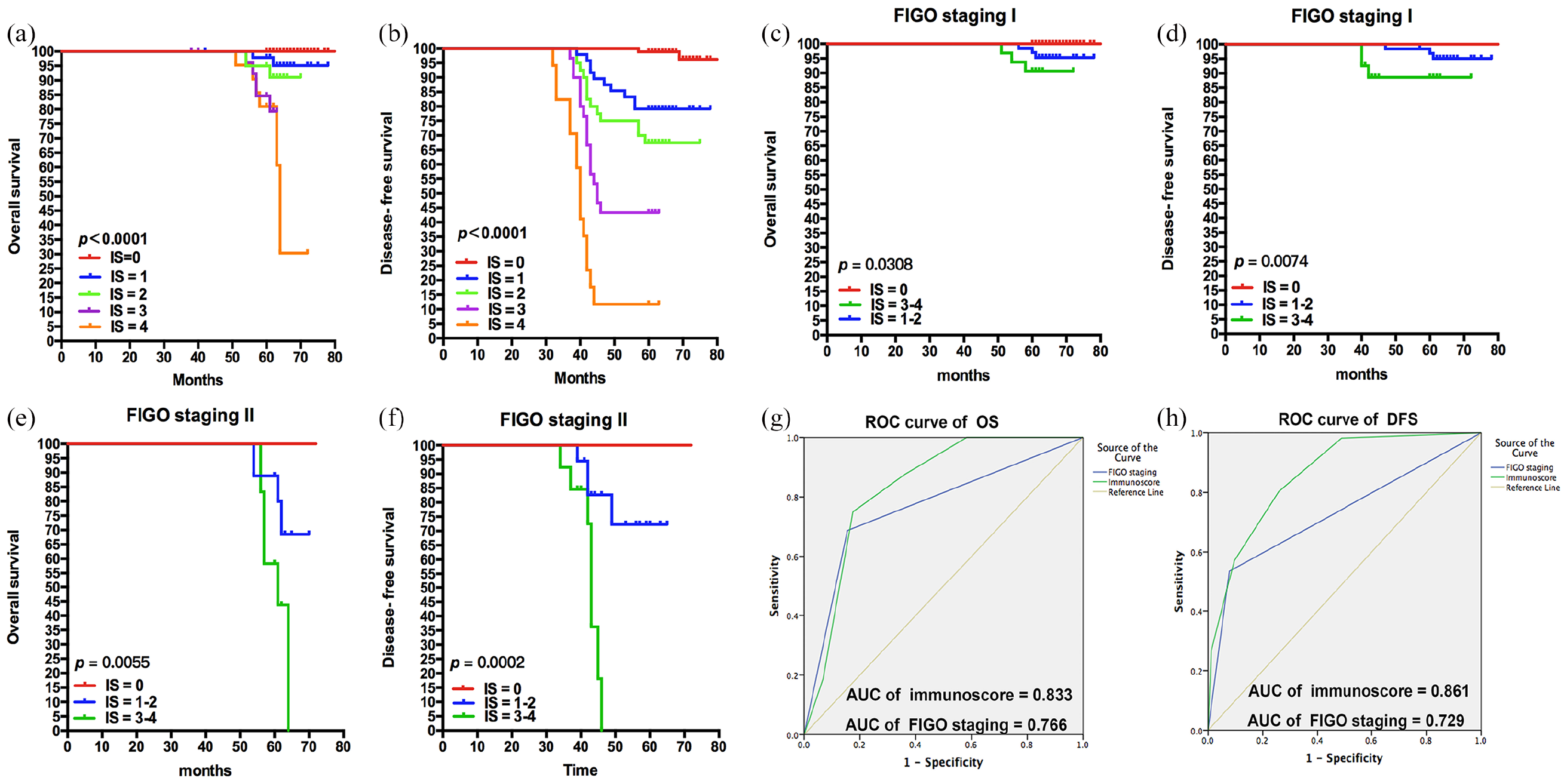
Kaplan–Meier curves of OS and DFS based on the Immunoscore system. According to the expression of TC-PD-L1, IC-PD-L1, IC-PD-1, and the infiltration of CD8, we set up a comprehensive Immunoscore system to predict the survival accurately. (a) and (b), OS and DFS of patients divided by the Immunoscore (P < 0.0001). We combined Immunoscore = 1 and 2, 3, and 4 into one group with no significance. (c) and (d), condition of Immunoscore with FIGO staging I (OS: P = 0.0308; DFS: P = 0.0074). (e) and (f), condition of Immunoscore with FIGO staging II (OS: P = 0.0055; DFS: P = 0.0002). (g) and (h), ROC analysis of OS and DFS based on the Immunoscore system, AUC of Immunoscore (OS: 0.833; DFS: 0.861) is prior to FIGO-TNM staging (OS: 0.766; DFS: 0.729).
Discussion
Cervical cancer is a kind of cancer with high heterogeneity. 19 Classically, TNM staging is the most widely used method to predict the clinical outcomes of cancer patients. However, clinical outcomes may vary significantly in patients with the same TNM staging. The reason may be that this classification method merely focuses on the tumor characteristics but not on the immune function status of patients. As a result, the relationship between tumor cells and infiltrating immune cells was neglected. 20 Thus, it is not enough to obtain an accurate outcome prediction by TNM staging alone. Galon and colleagues5,7 suggest that Immunoscore can provide a more accurate clinical prognosis compared to that of TNM staging. However, they were not taken the function and status of immune microenvironment into the system. Increasing evidence has suggested that the prognoses are associated with immune effectors. 7 Numerous studies have indicated the prognostic importance of immune cells and immune checkpoint molecules, such as PD-L1 and PD-1. 21 However, the function of immune factors has not been validated in cervical cancer. 11 Since no single factor was sufficient for the prognoses of cancer patients, an improved, comprehensive system with multi-factors is needed. Based on the considerations above, we have suggested a multi-factor Immunoscore, which has combined the function and the status of immune cells infiltrating tumor sites with the previous system. Our study not only analyzed the infiltration of CD8+ T cells and the expression of PD-L1 and PD-1, but also validated the relationship between these immune effectors and the patients’ survival. We found that TC-PD-L1, IC-PD-L1, IC-PD-1, and CD8 can be independent prognostic biomarkers. Patients with PD-L1positive and PD-1high in immune cells had poorer OS and DFS; however, PD-L1positive in tumor cells, which infiltrated more CD8+ T cells, related to better outcomes. According to the factors above, we set up a comprehensive Immunoscore system with immune effectors, which is superior to TNM in predicting clinical prognoses. In the ROC analysis for assessing the accuracies of the two systems, the Immunoscore is more accurate than TNM staging. All these findings suggest the prognostic significance and clinical pathological role of Immunoscore in cervical cancer.
According to Smyth and colleagues, 20 the tumor microenvironment can be divided into four types according to the expression of tumor infiltrating lymphocytes (TILs) and PD-L1. TILs are the main anti-tumor immunity for killing tumor cells. 4 The main expressions of PD-l and PD-L1 are expressed on TILs and tumor cells, which reflect the universality of immune escape in tumor cells. 22 In general, the tumor microenvironment is immunosuppressive. 7 Tumor cells may reduce the immune killing effect of TILs by expressing PD-1 binding on PD-L1 and TILs. Expression of PD-L1 in tumor cells was regulated mainly by mechanisms of extrinsic immune-induced factors. 22 As high-risk HPV infection is the major etiological factor of cervical cancer, which leads to an inflammatory reaction, 12 the expression of PD-L1 could be upregulated. Consistently, these results were also confirmed by the Spearman test to explore correlations between TC-PD-L1 and CD8+ T cells in our study. Patients with TC-PD-L1positive and CD8more T cells had the best prognoses compared to other groups, indicating an adaptive immune status existing in cervical cancer patients.
Tumor response to therapies are also based on the tumor microenvironment, in which various cells, especially immune cells, interact with tumor cells. 7 PD-L1 expressed in TILs reflected good responses to PD-L1 mAb treatment in a series of cancers. 21 In our study, TC-PD-L1positive, rather than IC-PD-L1positive, correlated with better prognoses, which could be associated with the response to immune checkpoint inhibitors. In melanoma, high expression of PD-1 of CD8+ T cells is associated with the response to PD-1 mAb therapy. 23 In our results, patients with CD8more PD-1low had significantly better OS compared with those with CD8less PD-1high. This may provide clues that the intensity of PD-1 is bound to give the number and function of intratumor CD8+ T cells with impact. The facts given above could be used to predict the cervical cancer patients’ response to immunotherapy.
We set up a comprehensive and multi-factor Immunoscore system according to the immune status of patients, which not only predicts prognoses accurately and is an essential complement to the TNM staging system, but also helps oncologists to evaluate the effectiveness of immunotherapy and carry out more effective options of cervical cancer treatment. In further studies, other critical immune markers can be included in the system, which may assist the doctors to choose more effective immunotherapy options for cervical cancer patients.
Supplemental Material
Supplementary_materials – Supplemental material for Immunoscore system combining CD8 and PD-1/PD-L1: A novel approach that predicts the clinical outcomes for cervical cancer
Supplemental material, Supplementary_materials for Immunoscore system combining CD8 and PD-1/PD-L1: A novel approach that predicts the clinical outcomes for cervical cancer by Hong Chen, Bairong Xia, Tongsen Zheng and Ge Lou in The International Journal of Biological Markers
Footnotes
Abbreviations
AUC: area under the curve; DFS: disease-free survival; FIGO: International Federation of Gynecology and Obstetrics; HPV: human papillomavirus; HRs: hazard ratios; IC: immune cell; IC-PD-1: PD-1 in immune cells; IC-PD-L1: PD-L1 in immune cells; mAb: monoclonal antibody; OS: overall survival; PD-1: programmed cell death receptor 1; PD-L1: programmed death receptor ligand 1; ROC: receiver operating characteristic; TC: tumor cell; TC-PD-L1: PD-L1 in tumor cells; TILs: tumor infiltrating lymphocytes; TNM: tumor/node/metastasis.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81872507); the 2017 Harbin Medical University Ph.D. Innovation Fund Project; and the 2017 Harbin Medical University Ph.D. Academic Exchange Program.
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Harbin Medical University Cancer Hospital and all patients were provided informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
