Abstract
Purpose:
With the recent developments in novel molecular targeted therapy such as immune-checkpoint blockades, serine/threonine-protein kinase B-Raf, and mitogen-activated protein kinase kinase inhibitors, the prognosis of advanced malignant melanoma has been improving. 5-S-cysteinyl-dopa (5-S-CD), a precursor of pheomelanin, has been previously revealed to be a useful biomarker for advanced-stage malignant melanoma, especially in patients with distant metastases. Here, we aimed to assess and compare the utility of serum 5-S-CD and lactate dehydrogenase levels as markers for predicting the effects of nivolumab in advanced malignant melanoma.
Methods:
Baseline serum 5-S-CD and lactate dehydrogenase levels in patients with unresectable stage IIIC and IV malignant melanoma treated with nivolumab (n = 21) were analyzed to determine their utility as predictive markers for survival. We also analyzed the prognostic value of these markers among patients with only stage IV malignant melanoma (n = 17).
Results:
Our analysis showed that patients with baseline serum 5-S-CD levels >25.0 nmol/L had significantly poor prognosis. In contrast, serum lactate dehydrogenase levels at the upper limit of the normal range did not exhibit such changes.
Conclusions:
Serum 5-S-CD levels have the potential to be an excellent predictive marker for the efficacy of nivolumab therapy in patients with advanced malignant melanoma.
Introduction
Malignant melanoma (MM) is the most aggressive form of skin cancer that exhibits dismal prognosis especially with distant metastasis. 1 Nonetheless, recently developed serine/threonine-protein kinase B-Raf (BRAF), mitogen-activated protein kinase kinase (MEK) inhibitors, and immune-checkpoint blockades have improved the prognosis of advanced-stage MM.2,3 Therefore, identifying cancer biomarkers that can precisely reflect the progression of advanced-stage MM may help improve prognosis. 4 In addition, recognizing predictive markers for the efficacy of immune-checkpoint blockades will maximize the effects of therapy and minimize the cost and risk of side effects.
Black eumelanin and yellow to reddish-brown pheomelanin are two types of melanin pigments that are produced in the melanocytes and melanoma cells. 5-S-cysteinyl-dopa (5-S-CD), which belongs to the catecholamine family, is a precursor of pheomelanin.5,6 It has been reported that 5-S-CD is released into the blood stream, and several studies have shown that the serum 5-S-CD level is a useful biomarker for advanced-stage MM, especially in patients with distant metastases.7–11 We had also previously reported that serum 5-S-CD is more efficient in detecting advanced-stage MM and its recurrence compared to serum lactate dehydrogenase (LD). Moreover, among patients with stage III and IV MM, serum 5-S-CD levels greater than 15.0 nmol/L at the initial hospital visit correlated with poor prognosis. We thus indicated the possibility that serum 5-S-CD levels could predict the prognosis of advanced MM. 12
In Japan, 5-S-CD and LD are the only commercially available biomarkers for MM. Elevation of serum LD levels has been included under the M category in both the seventh and eighth edition of the American Joint Committee on Cancer (AJCC) melanoma tumor node metastasis staging manual.1,13 Some reports have also shown that baseline serum LD levels were effective in predicting the prognosis of patients with MM receiving anti-PD-1 antibody, including nivolumab.14,15 Therefore, we hypothesized that serum 5-S-CD levels also may be useful in predicting the efficacy of nivolumab therapy.
The present study assessed and compared the utility of baseline serum 5-S-CD and LD levels as predictive markers for the efficacy of nivolumab therapy in advanced-stage MM.
Methods
Cohort of nivolumab-treated patients
From February 2012, 28 patients were treated with nivolumab at the Department of Dermatology, Okayama University Hospital. Baseline serum 5-S-CD and LD levels were measured in 21 of these patients prior to the first administration of nivolumab. Among them, 17 and 4 were stage IV and IIIC MM, respectively, according to the eighth edition of the AJCC melanoma staging manual. 1 Five patients were also receiving BRAF/MEK inhibitors. Considering the remarkable effects these inhibitors have on the treatment of metastatic MM, we were concerned about the positive bias on the prognosis of such patients. We thus excluded four patients receiving BRAF/MEK inhibitors subsequent to nivolumab, whereas the remaining one patient who showed progressive disease despite vemurafenib treatment followed by nivolumab treatment was included in the analysis (Table 1). Among these 21 patients, 11 received only nivolumab, 2 received nivolumab subsequent to ipilimumab (patient nos. 1 and 5), 4 received ipilimumab subsequent to nivolumab (patient nos. 9, 15, 16, and 17), and 1 received nivolumab followed by ipilimumab and then nivolumab (patient no. 10). Except for patient nos. 15 and 18, all others received 2 mg/kg of nivolumab every 3 weeks. Patient no. 15 received 2 mg/kg of nivolumab biweekly followed by ipilimumab, while patient no. 18 received 3 mg/kg of nivolumab biweekly followed by 2 mg/kg of nivolumab every 3 weeks. Although serum 5-S-CD levels were previously shown to spuriously increase upon chronic renal failure 16 and Agaricus extract intake, 17 none of the included patients exhibited severe renal failure or had ingested Agaricus extract.
Clinical characteristics of the 21 cases receiving nivolumab.
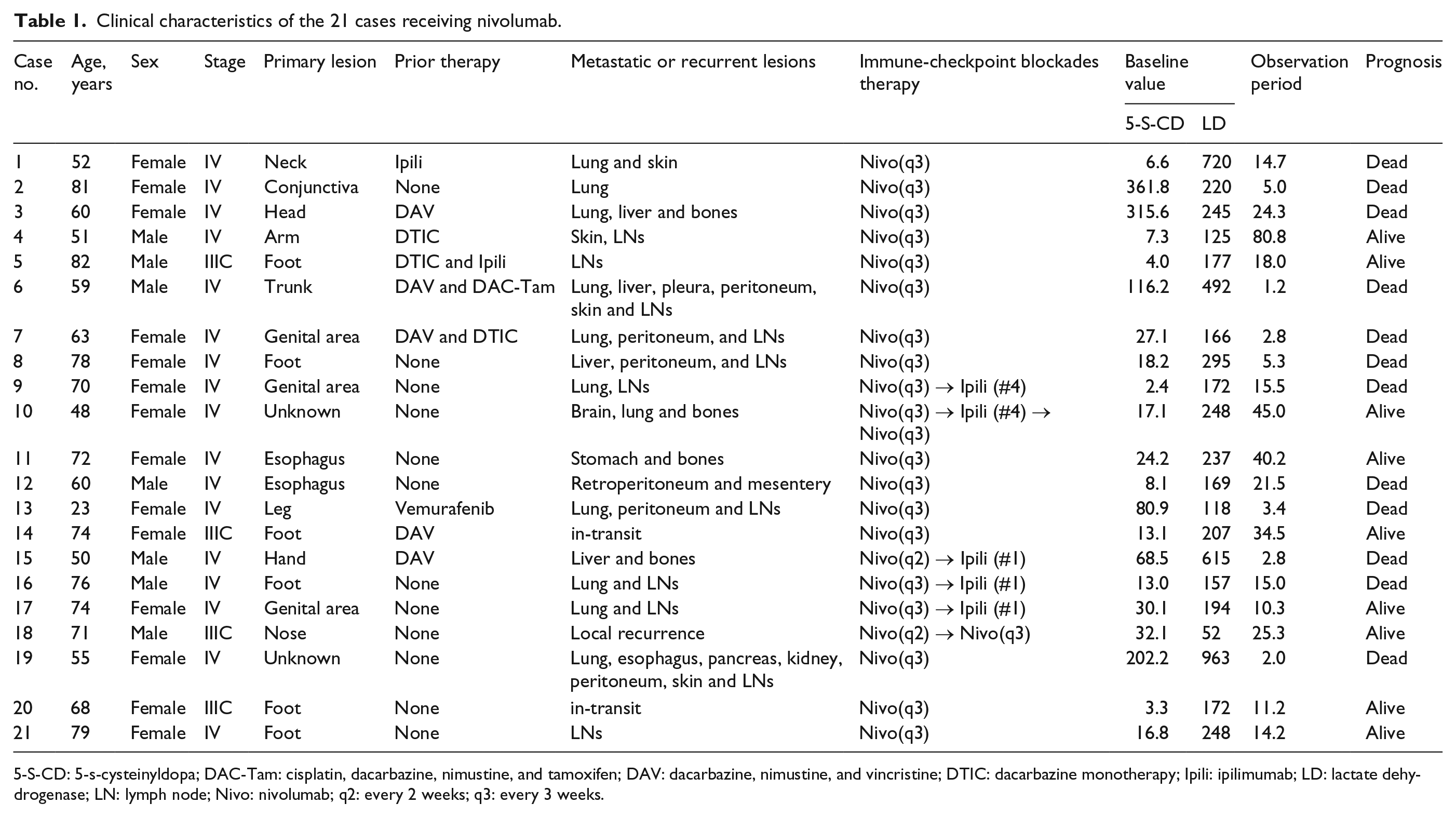
5-S-CD: 5-s-cysteinyldopa; DAC-Tam: cisplatin, dacarbazine, nimustine, and tamoxifen; DAV: dacarbazine, nimustine, and vincristine; DTIC: dacarbazine monotherapy; Ipili: ipilimumab; LD: lactate dehydrogenase; LN: lymph node; Nivo: nivolumab; q2: every 2 weeks; q3: every 3 weeks.
Serum 5-S-CD level measurement
Serum 5-S-CD levels were measured using high-performance liquid chromatography (SRL, Tokyo, Japan) in accordance with a previously published method. 18
Serum LD level measurement
Serum LD levels were measured at the Department of Laboratory Medicine, Okayama University Hospital using a conventional method that quantifies lactate conversion to pyruvate with a reference range of 124–222 IU/L.
Statistical analysis
All statistical analyses were performed using the EZR statistics software (version 1.37). 19 Patient survival was assessed from the date of the first nivolumab administration to either the date of death due to melanoma or the last follow-up. Survival of nivolumab-treated patients was evaluated using the Kaplan–Meier method, and the survival curves generated were compared using the log-rank test. P < 0.05 was considered statistically significant.
Results
Prognostic discriminative performance of serum 5-S-CD levels at baseline in nivolumab-treated patients
In patients with advanced MM receiving nivolumab (n = 21), baseline serum 5-S-CD levels ranged from 2.4 to 361.8 nmol/L (median; 17.1 nmol/L). As previously described, 10.0 nmol/L was considered to be the cut-off value for serum 5-S-CD levels when discriminating patients with MM from the control group. 10 In addition, our previous study revealed that in patients with stage III and IV MM, serum 5-S-CD levels greater than 15.0 nmol/L at the initial hospital visit correlated with poor prognosis. 12 Considering these values of 5-S-CD, we evaluated the significance of baseline serum 5-S-CD levels in the prognosis of nivolumab-treated patients according to four different 5-S-CD threshold levels, 10.0, 15.0, 20.0, and 25.0 nmol/L, using the Kaplan–Meier method (Figure 1(a) to (d)). Accordingly, at a threshold level of 25.0 nmol/L, baseline 5-S-CD was able to significantly distinguish between patients with good and poor prognoses (Figure 1(d)).
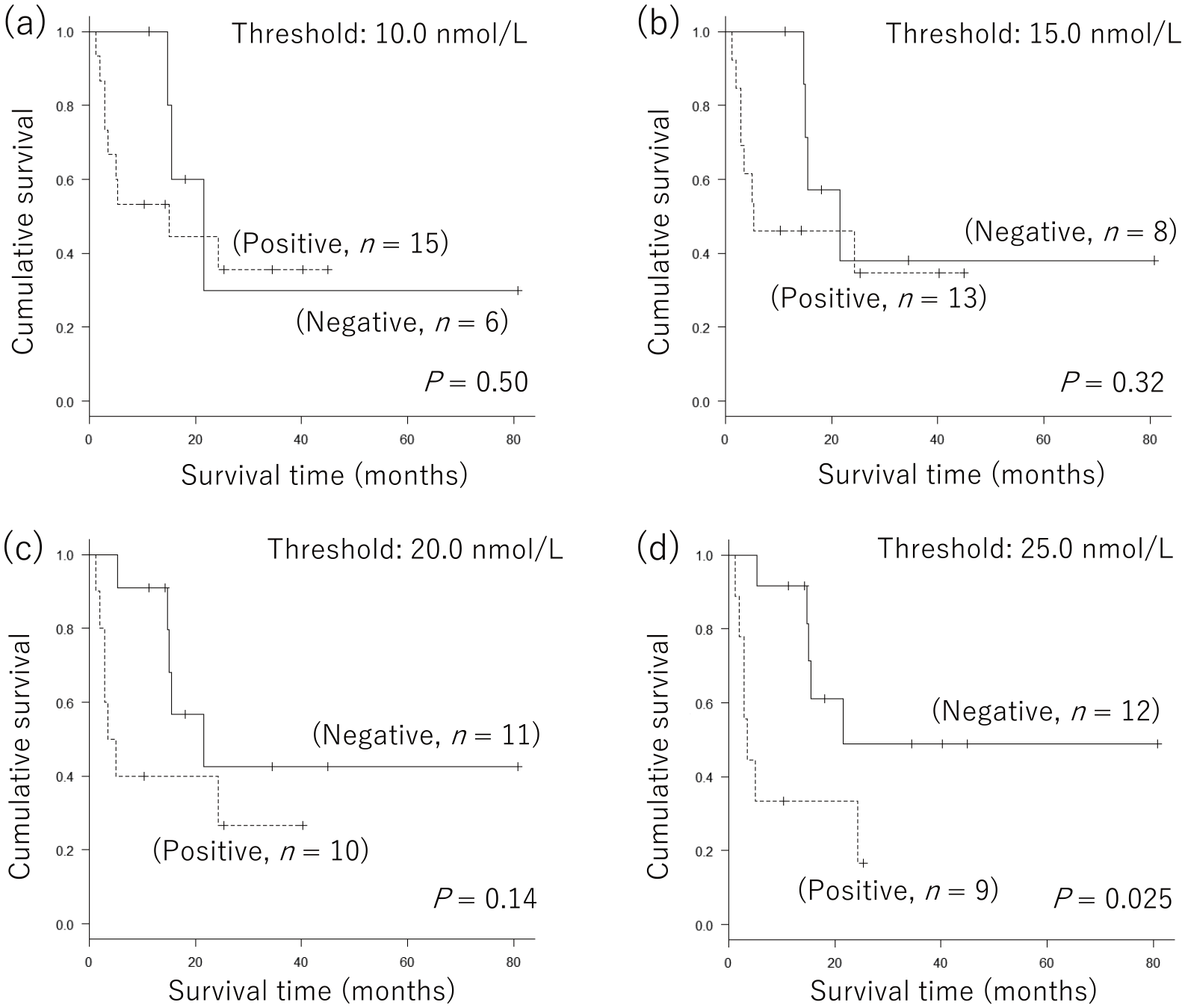
Significance of baseline serum 5-S-cysteinyl-dopa levels in the prognosis of all nivolumab-treated patients. For patients with advanced-stage malignant melanoma (n = 21), Kaplan–Meier curves at threshold levels of (a) 10.0, (b) 15.0, (c) 20.0, and (d) 25.0 nmol/L were created. Log-rank test showed significant differences at a threshold of 25.0 nmol/L (P = 0.025).
In our previous analysis, the cohort included four stage IIIC patients. To exclude the confounding factors, such as staging differences, we again evaluated the significance of baseline serum 5-S-CD levels on prognosis focusing on stage IV patients (n = 17). Among the 17 patients with stage IV MM, baseline serum 5-S-CD levels ranged from 2.4 to 361.8 nmol/L (median; 24.2 nmol/L). Similar analysis using the four threshold levels were also performed (Figure 2(a) to (d)). Among the four threshold levels, 25.0 nmol/L was again determined to be the level at which baseline 5-S-CD could significantly distinguish between patients with good and poor prognoses (Figure 2(d)).
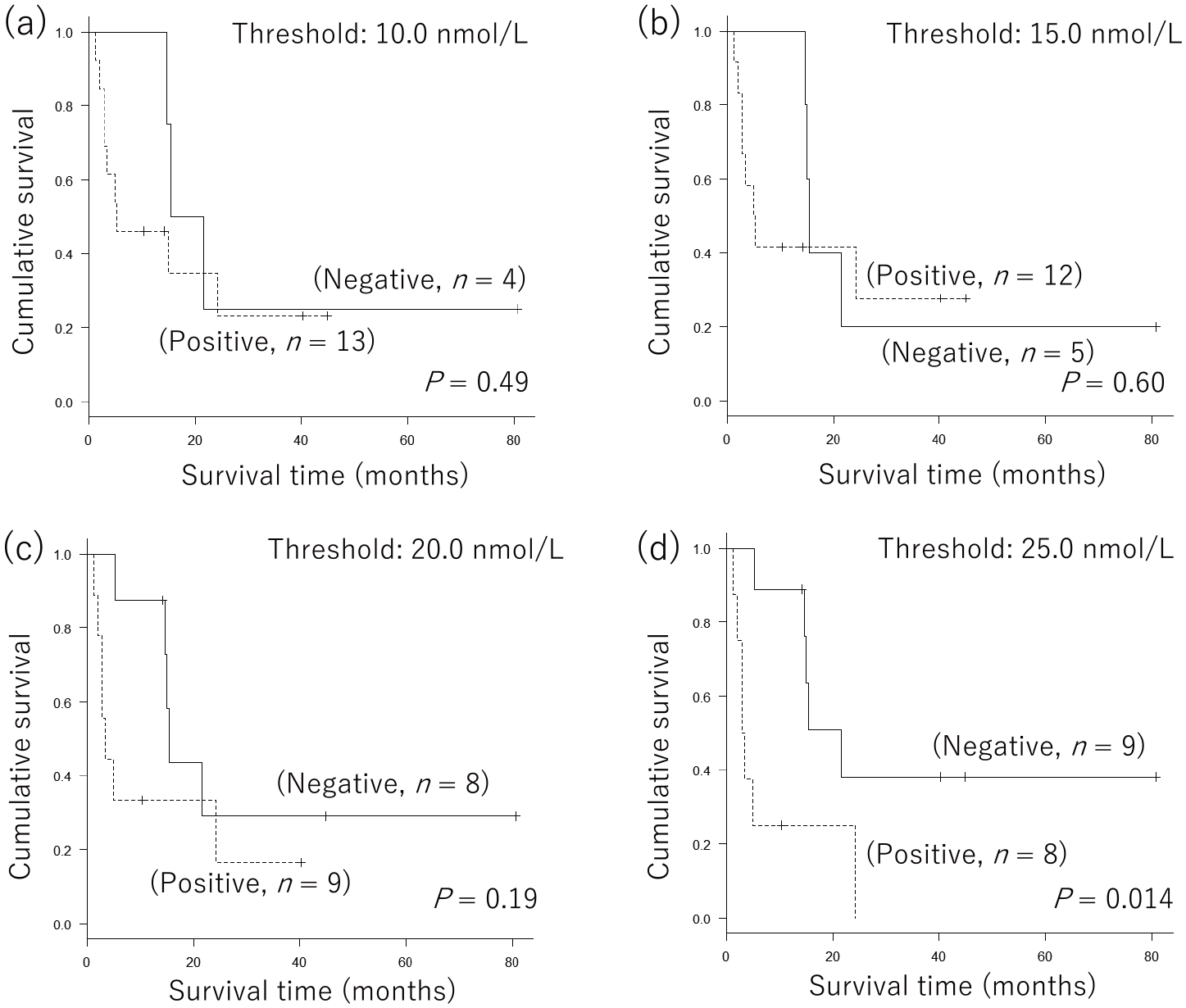
Significance of baseline serum 5-S-cysteinyl-dopa levels in the prognosis of nivolumab-treated patients with stage IV malignant melanoma. For patients with stage IV malignant melanoma (n = 17), Kaplan–Meier curves at threshold levels of (a) 10.0, (b) 15.0, (c) 20.0, (d) 25.0 nmol/L were also created. Log-rank tests showed significant differences at a threshold of 25.0 nmol/L (P = 0.014).
Summarizing both analyses, we speculated that baseline serum 5-S-CD levels >25.0 nmol/L have the potential to be a predictor of poor prognosis among patients with advanced MM receiving nivolumab therapy.
Prognostic discriminative performance of baseline serum LD levels in nivolumab-treated patients
We evaluated the significance of baseline serum LD levels on the prognosis of nivolumab-treated patients. At the 222 IU/L threshold; that is, the upper limit of the normal range (ULN), baseline LD levels failed to distinguish between good and poor prognoses (Figure 3). We also analyzed the significance of serum LD levels exclusively among patients with stage IV MM. Similarly, baseline serum LD levels at the 222 IU/L threshold did not show a significant difference in the prognosis of patients with stage IV (Figure 4).
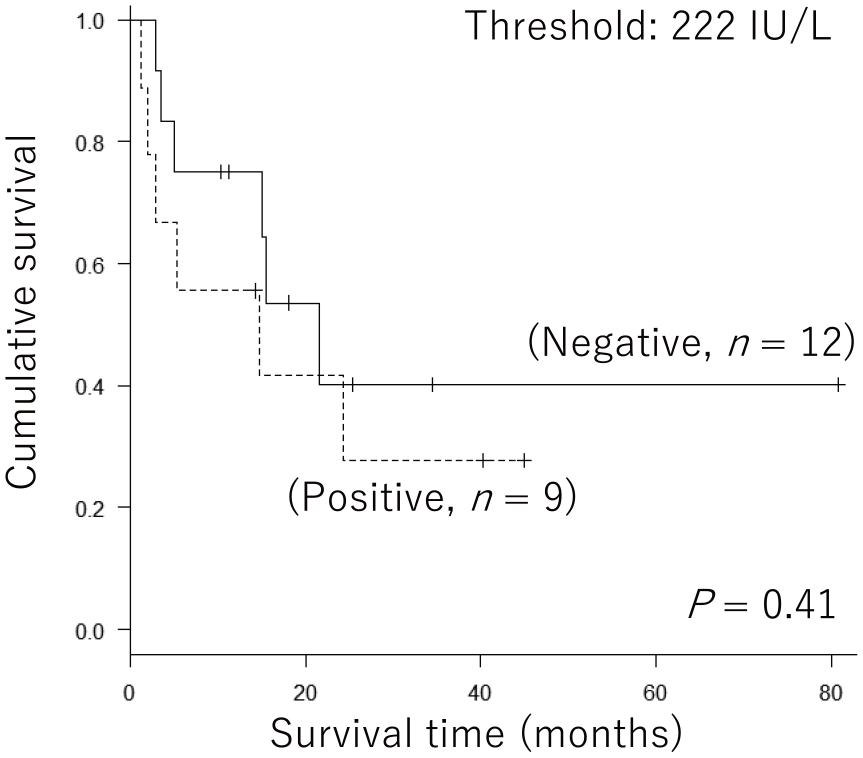
Significance of baseline serum lactate dehydrogenase levels in the prognosis of all nivolumab-treated patients. For patients with advanced-stage malignant melanoma (n = 21), Kaplan–Meier curves at a threshold of 222 IU/L (upper limit of the normal range) were created. Log-rank tests showed no significant difference in prognosis (P = 0.41).
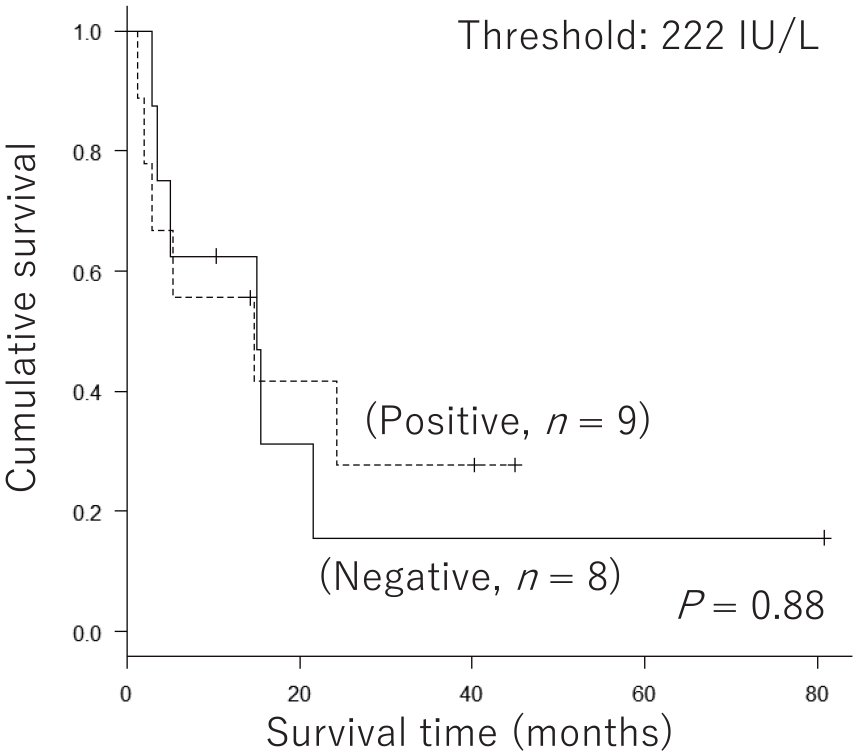
Significance of baseline serum lactate dehydrogenase levels in the prognosis of nivolumab-treated patients with stage IV malignant melanoma. For patients with stage IV malignant melanoma (n = 17), Kaplan–Meier curves at a threshold of 222 IU/L (upper limit of the normal range). Log-rank tests showed no significant difference in prognosis (P = 0.88).
Discussion
The results of the present study showed that among patients with advanced MM who underwent nivolumab therapy, baseline serum 5-S-CD levels >25.0 nmol/L were able to significantly predict poor prognosis. Recently, Omodaka et al. 20 also described that serum 5-S-CD levels in the early treatment phase (3–6 weeks after initial administration of nivolumab) could possibly predict therapeutic responses in metastatic MM. Analyzing 12 metastatic MM patients under nivolumab therapy, they showed that serum 5-S-CD levels decreased in 2 of 3 patients having a partial response and increased in 4 of 8 progressive cases. 20 Their results suggest the possible monitoring efficacy of 5-S-CD in the early phase of nivolumab therapy. On the other hand, the present study showed the utility of 5-S-CD as a predictive marker for the efficacy of long-term nivolumab therapy. Taking the aforementioned results together, serum 5-S-CD levels may be considered useful in predicting the prognosis and monitoring the effects of the nivolumab therapy in advanced MM.
Serum LD, which is also included in the staging criteria for MM, has been widely used as a marker for various malignancies. 1 Increased baseline LD levels reflect the prognosis of patients receiving nivolumab or pembrolizumab therapy.14,15 However, in the present study, serum LD levels >ULN did not show worse prognosis. Thus, baseline serum 5-S-CD levels may be potentially superior to LD in predicting the efficacy of nivolumab therapy.
As shown in Table 1, four patients with baseline LD levels >ULN also presented high 5-S-CD levels >25.0 nmol/L (patient nos. 3, 6, 15, and 19) and poor prognosis. In addition, two of the four patients with LD levels <ULN and 5-S-CD levels >25.0 nmol/L (patient nos. 2 and 13), as well as two of the five patients with LD levels >ULN and 5-S-CD levels <25.0 nmol/L (patient nos. 1 and 8), demonstrated poor prognosis. The combined use of baseline LD and 5-S-CD may be beneficial in detecting patients with poor prognosis prior to nivolumab therapy. However, two patients with LD levels >ULN and 5-S-CD levels <25.0 nmol/L (patient nos. 10 and 11) had relatively good prognosis over 3 years of survival. Hence, to further confirm the practical use of these markers in predicting the efficacy of nivolumab therapy, future studies with larger sample sizes are warranted.
Owing to its retrospective nature, the present study carries some limitations. Among the 21 patients included, 5 also had been treated with ipilimumab subsequent to nivolumab. To completely exclude of the effects of ipilimumab, multivariate analysis should have been performed. Unfortunately, multivariate analysis was not possible in the present study given the small number of patients. Considering that three of the five patients (patient nos. 15, 16, and 17) received ipilimumab only once, its effects may have been negligible. Additionally, the therapeutic effects of ipilimumab on the prognosis of advanced MM has been revealed to be relatively small in comparison to nivolumab.21,22 Thus, in the present study, we considered the effects of ipilimumab to be minimal.
Conclusion
The serum 5-S-CD level has the potential to be a good predictive marker for the efficacy of nivolumab therapy in patients with advanced MM.
Footnotes
Acknowledgements
We are grateful to Dr. Shigeki Umemura (Department of Thoracic Oncology, National Cancer Center Hospital East, Kashiwa, Japan) for giving us insightful comments and suggestions on this work.
Ethical approval
This study was approved by the Ethics Committee of the Okayama University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences and the Okayama University Hospital.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
