Abstract
Background:
Primary pulmonary enteric adenocarcinoma (PEAC) is a rare non-small cell lung cancer subtype sharing morphologic and immunohistochemical features with colorectal adenocarcinoma. Given the frequency of lung metastases in colorectal cancer, the differential diagnosis of PEAC according to routine morphological and immunohistochemical findings may be difficult. Genome sequence by next-generation sequencing has recently introduced new perspectives to better define the diagnosis and tumor sensitivity to treatments, while the rarity of this subtype of cancer still limits the current knowledge of its molecular features and provides no information to address patients to tailored therapies.
Methods:
We diagnosed a rare case of subcutaneous metastasis as a first symptom of a PEAC. Formalin-fixed paraffin-embedded samples of the primary tumor and subcutaneous metastases were examined by immunohistochemistry, and subsequently by targeted next-generation sequencing analysis.
Results:
Morphological and immunohistochemical findings suggested a rare case of metastatic pulmonary adenocarcinoma with enteric aspects. Next-generation sequencing analysis performed on both the primary tumor sample and the cutaneous lesion identified two pathogenic variants on CDKN2A and KRAS in both of them. However, the metastasis showed two additional pathogenic mutations located in SMAD4 and FLT3 genes.
Conclusions:
We describe for the first time an extensive molecular analysis on a rare case of PEAC with an unusual cutaneous metastasis. Our observation suggests that a specific pattern of mutations is harbored in this neoplasm, and that additional molecular studies may provide further information to identify prognostic and hopefully predictive genes of response to treatment.
Keywords
Introduction
Pulmonary enteric adenocarcinoma (PEAC) was described in 1991 for the first time by Tsao and Fraser, 1 and officially included within the non-small cell lung cancer (NSCLC) classification only in 2011, according to the International Association for the Study of Lung Cancer (IASLC), the American Thoracic Society (ATS), and the European Respiratory Society (ERS). This rare tumor is defined by a morphological enteric cell differentiation involving more than 50% of tumor cells, and by the positive immunohistochemistry (IHC) staining for at least one marker of intestinal differentiation, including cytokeratin 20 (CK20), CDX-2, and Mucin 2 (MUC2). The eventual IHC positivity of cytokeratin 7 (CK7) and thyroid transcription factor-1 (TTF-1) in approximately 50% of the cases may be useful in the differential diagnosis with the metastatic colorectal adenocarcinoma. In 2015, the World Health Organization (WHO) included this type of adenocarcinoma in the classification of lung malignancies as “pulmonary adenocarcinoma with enteric differentiation.” 2
To date, the high histological similarity between PEAC and colorectal cancer and the absence of non-traditional pathological tools for a certain diagnosis, imposes the execution of colonoscopy as well as computed tomography (CT) scan or positron emission tomography (PET-CT) during the staging process to provide auxiliary information to the histologic description. Also, considering the small number of cases observed, no differences in onset, lung location type, clinical course, and radiographic features between PEAC and conventional lung adenocarcinoma are reported. 3 In contrast to lung cancer, in several cases PEAC shows an increase in circulating tumor markers CA19-9 and carcinoembryonic antigen (CEA) with an absence of CYFRA 21.1 or Neuron Specific Enolase (NSE). 3
With the advance of nucleic acid sequencing technologies over the recent years, new perspectives are emerging in the field of cancer characterization, but the rarity of PEAC hindered the identification of specific molecular signatures to apply in differential diagnosis, prognostic stratification, and prediction of response to therapy. 4 Contrasting data suggest the presence of typical NSCLC driver mutations (EGFR, ALK, and ERBB2) with variable percentages, 5 while more recent (but small) studies indicate that the methylation or microRNA (miRNA) patterns can be used as molecular indicators of differentiation from colon carcinoma.4,6
Here, we describe the clinical case of a patient admitted to our clinical evaluation after diagnosis of metastatic enteric lung adenocarcinoma. Formalin-fixed paraffin-embedded (FFPE) samples from the primary tumor and one cutaneous metastasis were examined by IHC, and subsequent molecular investigation by next-generation sequencing (NGS) to investigate the genomics of this peculiar cancer subtype.
Case description
A 63-year-old man was admitted in November 2017 to the Medical Oncology Unit of the University Hospital “Policlinico of Bari” with lumbar bone pain and a painful left-sided thoracic soft tissue mass, which had progressively grown over the previous 2 months. The patient had been a heavy smoker since the age of 30 and had a personal medical history of ischemic heart disease, arterial hypertension, type 2 diabetes mellitus, and lumbar bone pain. An ultrasound scan of the chest mass was immediately performed, and showed a nodule of 4 cm in its major diameter with regular shape, altered ultrasound structure, and with no intra-lesion vascularization. A similar lesion with scarce peri-nodular vascularization was also found on the volar surface of the right arm, approximately 15 × 23 mm in size.
The patient underwent whole body CT scan, which showed a 35 mm irregular nodule of the right pulmonary hilum associated with metastases involving mediastinal lymph nodes, lungs, liver, multiple skeletal sites, subcutaneous tissue of the chest, and the right gluteal muscle. A 18F-labeled fluoro-2-deoxyglucose (18F-FDG)-PET-CT confirmed the above-mentioned areas (Figure 1). Hence, fibrobronchoscopy was performed, revealing the presence of obstructing vegetation in the right superior lobar bronchus, which underwent bioptic sampling.
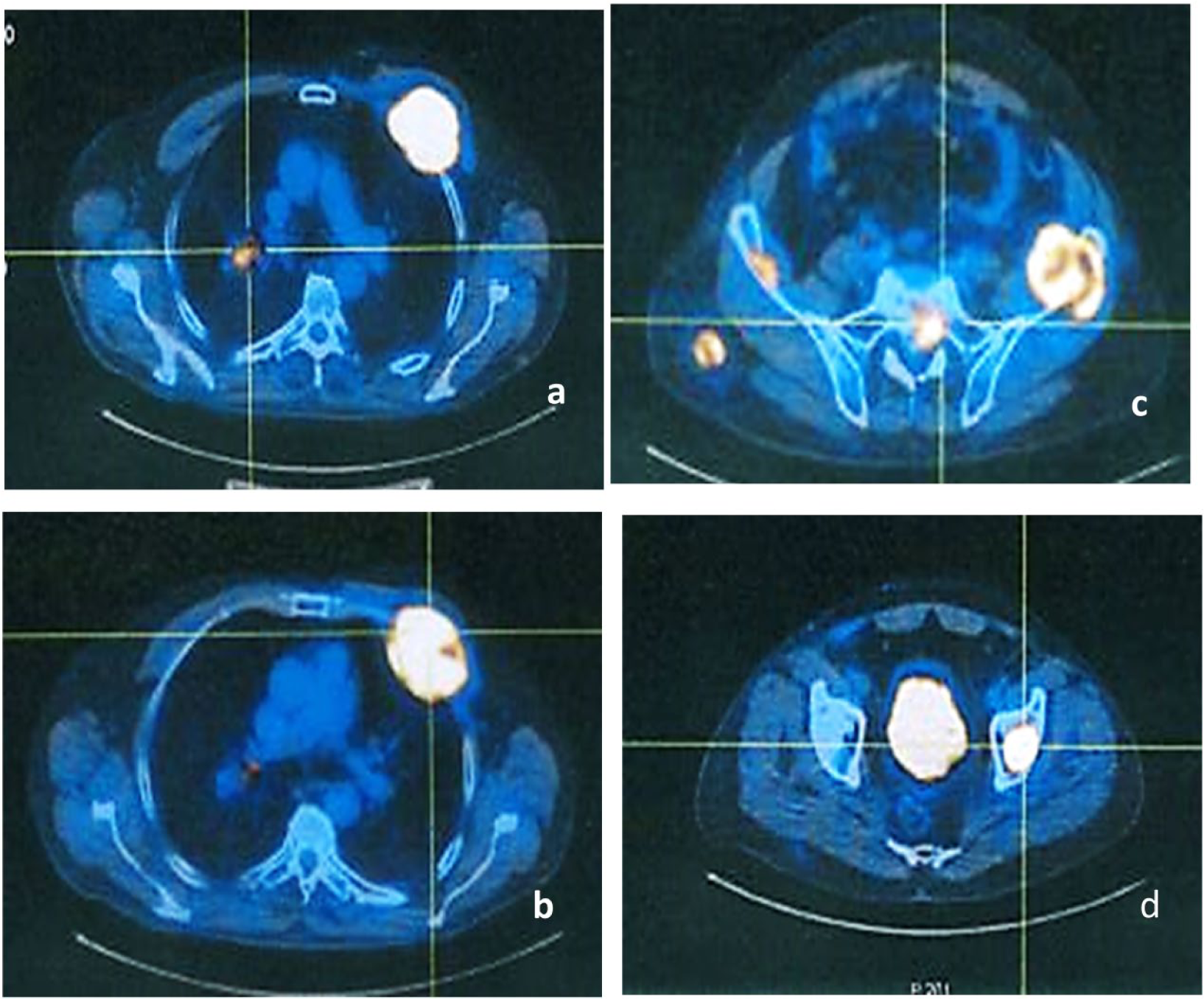
Images of most representative areas of pathologic 18F-FDG uptake detected by PET-CT scan. PET scan showed pathological uptake of 18F-FDG in: (a) right pulmonary hilum (SUV 10.4); (b) anterior left hemithorax with partial involvement of some costal arches (SUV 21.1); (c) left iliac ala (SUV 11.0); (d) left femur (SUV 18).
The histological examination of the primary lesion described fragments of the bronchial mucosa in a pattern of moderately differentiated infiltrating adenocarcinoma. Acinar glandular structures were present, with a visible lumen delimited by cylindrical epithelia with dysmetric and hyperchromic nuclei positioned mainly at the base of the cell. Some glandular structures had a cribriform appearance, others a solid aspect, but more than 50% of the neoplastic cells were recognized as showing intestinal-like glandular structures. Furthermore, a surrounding stromal desmoplastic reaction was noted. IHC investigations revealed positivity for CDX2 and CK7 staining, while TTF1, Napsin A, CK20, and p40 markers were not detectable (Figure 2).
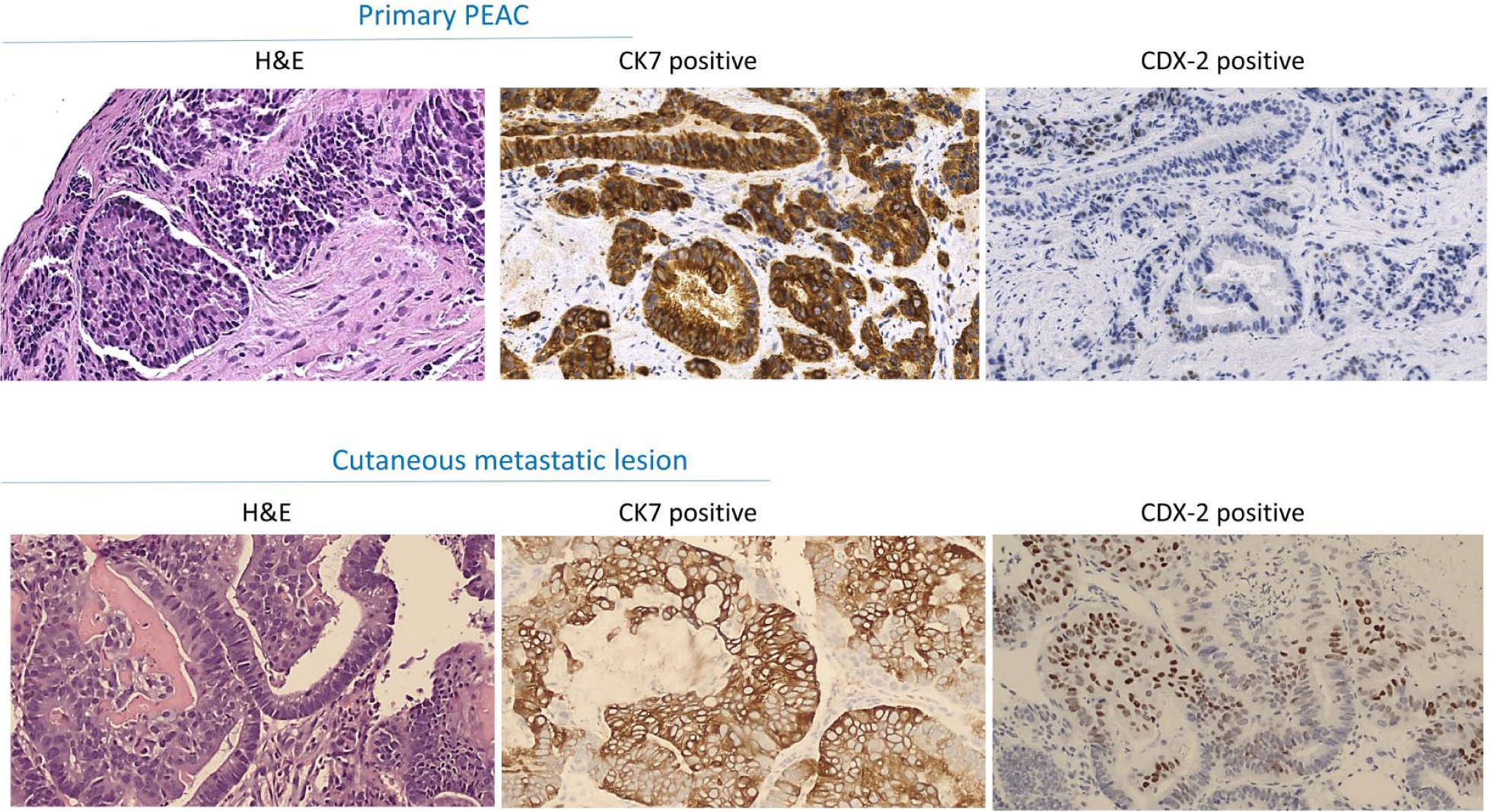
Hematoxylin and eosin staining and immunohistochemical findings of the primary pulmonary enteric adenocarcinoma (PEAC) and the cutaneous metastatic lesion both positive for cytokeratin 7 (CK7) and caudal type homeobox (CDX2) (original magnification × 40).
In consideration of the clinical and immuno-morphological features, the histological specimen was suggestive for a “pulmonary adenocarcinoma with enteric aspects” but the metastatic nature of the lesion could not be excluded. Blood tests revealed increased levels of Ca 19.9 (1571 IU/mL, normal range 0–37 IU/mL), CEA (483 ng/mL, normal range 0–5 ng/mL), lactate dehydrogenase (397 IU/L, normal range 87–241 IU/L), NSE 29,8 mcg/L (normal range <18.3) and CYFRA 14.1 ng/mL (normal range <3.3). In order to exclude the diagnosis of a primary gastrointestinal malignancy, the patient underwent esophago-gastroduodenoscopy and colonoscopy, which were both negative. A biopsy of the left-sided thoracic subcutaneous lesion was addressed and the histological examination confirmed the diagnosis of enteric adenocarcinoma, with focal areas of necrosis with positive CDX2 and CK7, negative TTF1, Napsin A and CK20 staining by IHC. Mutational analysis performed on the primary tumor sample and the metastatic lesions did not identify mutations involving EGFR gene, nor ALK gene translocations.
To explore the potential mechanisms underlying the onset of subcutaneous metastases, FFPE samples of the primary tumor and the subcutaneous thoracic metastasis were recruited from the Pathology Department of the University of Bari, after receiving the patient’s written informed consent.
The DNA was extracted using the QIAamp DNA FFPE Tissue Kit (Qiagen) and quantified by Qubit® fluorimeter (Life Technologies™ Carlsbad, CA, USA). Molecular screening was performed through the Ion AmpliSeq™ Cancer Hotspot Panel v2 (Life Technologies), which includes 2800 somatic mutations in 50 genes involved in cancer initiation and progression. Briefly, 10 DNA ng were employed and barcoded the libraries using the Ion AmpliSeq™ Library kit 2.0 and the Ion Xpress™ barcode adapters (Life Technologies). After purification with Agent court AMPure XP (Beckman Coulter, Indianapolis, USA), the libraries were quantitated with the Ion Library Quantitation Kit (Life Technologies) on the StepOne Plus system (Applied Biosystem, Foster City, CA 94404, USA) and utilized for the preparation of templates with the Ion OneTouch™ 2 System and Ion One Touch ES (Life Technologies). NGS reactions were performed on the NGS Ion Torrent PGM™ system by using Ion Torrent™ 316 chips (Life Technologies).
Mutational results were analyzed with the Torrent Suite Software 5.0.4 and variant calling was performed running the Torrent Variant Caller plugin version 5.0.4.0. NGS analysis.
All sequence variants were interpreted and verified using the visualization by IGV (Integrative Genomics Viewer) browser (Broad Institute, Cambridge, Massachusetts, United States).
Each variant was investigated in its potential pathogenic role consulting COSMIC (http://cancer.sanger.ac.uk/cosmic), dbSNP (https://www.ncbi.nlm.nih.gov/snp/), ExAC (http://exac.broadinstitute.org/), and HGMD (http://www.hgmd.cf.ac.uk/ac/index.php) databases.
NGS results from both primary tumor and skin metastasis were compared, as shown in Table 1. In the primary tumor sample, we identified 13 sequence variants, 2 of which have already been reported as pathogenic variants according to COSMIC algorithms, namely the CDKN2A c.237_247del11; p.Pro81fs*35 and the KRAS c.34G>T; p.Gly12Cys. However, all the gene variants identified in the primary tumor were also found in the subcutaneous metastasis. Moreover, the metastatic lesion showed two additional pathogenic mutations located in the SMAD4 (c.767A>T; p.Gln256Leu) and the FLT3 genes (c.1999delC; p.Gln667fs). The latter is responsible for the production of a truncated protein and is interpreted as deleterious by predictive algorithms, although it has not been reported until today in the mutational databases (Table 1).
Comparison of sequence variants between the pulmonary enteric adenocarcinoma and subcutaneous metastasis.
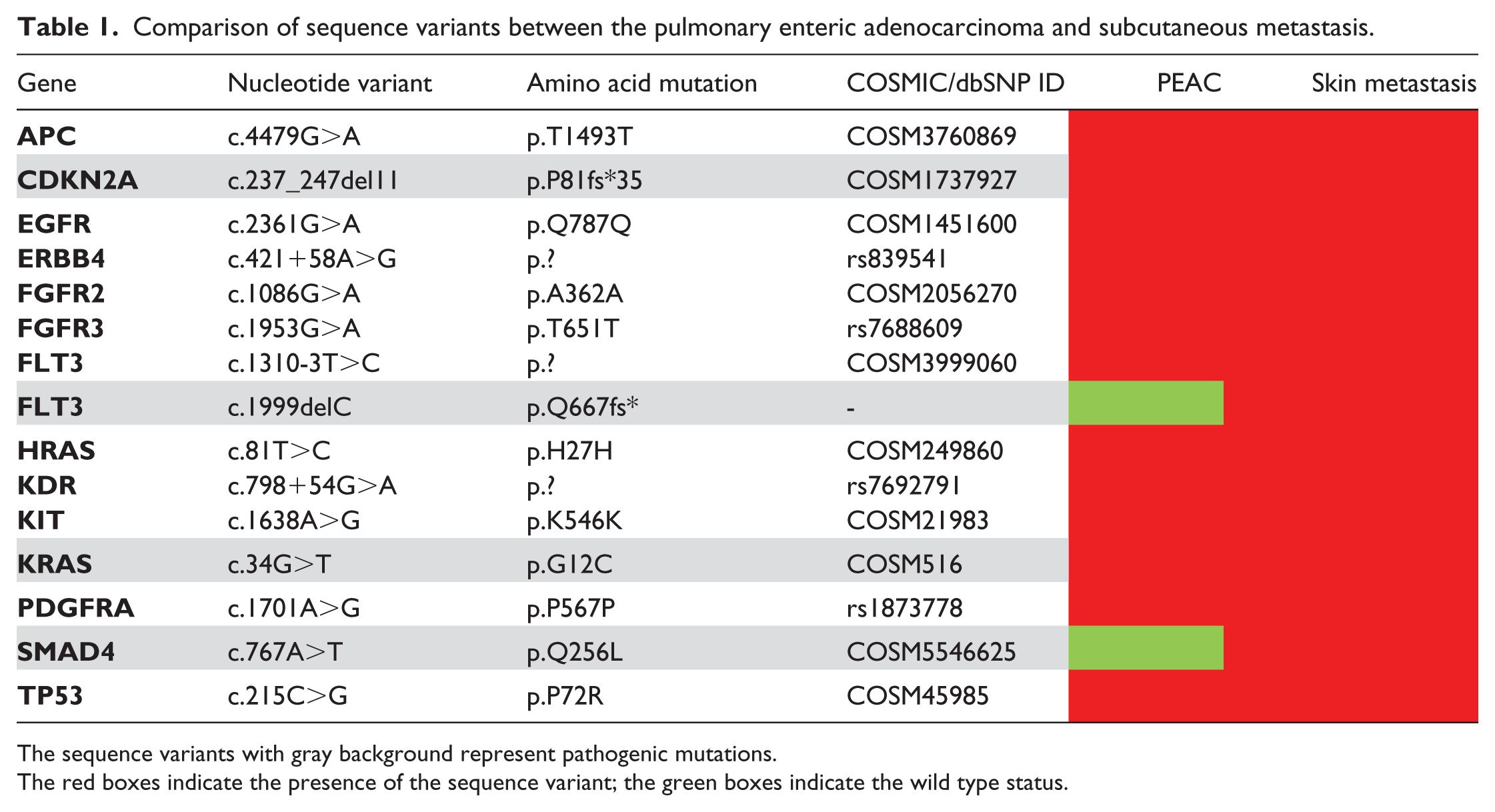
The sequence variants with gray background represent pathogenic mutations.
The red boxes indicate the presence of the sequence variant; the green boxes indicate the wild type status.
The patient received systemic treatment with Pemetrexed-Cisplatin and bisphosphonates associated with radiotherapy on the pelvis (20 Gy divided into five fractions), with significant improvement of bone pain. However, after two cycles there was a worsening of performance status leading to death.
Conclusions
The frequency of skin metastases in lung cancer is relatively low, ranging between 1.7% and 3.1%, and occurring more frequently in males, where it is the second cause of cutaneous metastasis after melanoma, while in women it is much rarer. 7 The most common sites of involvement are the anterior chest wall, the dorsal trunk, the abdomen, and the scalp. In literature, evidence of cutaneous metastases at the time of diagnosis of lung cancer is rarely described. 7 To date, very few case reports and small cohorts of the PEAC subtype have been described, and only one case of subcutaneous metastasis of the left upper chest wall in a 62-year-old man with PEAC is currently reported. After comparing cytological features of the primary lung neoplasm and subcutaneous metastasis, these two pathological tissues showed similar morphological and IHC characteristics. 8
With the advent of NGS technologies in recent years, new perspectives have emerged in delineating new molecular profiles of PEACs, but at present only a limited number of studies have applied these methodologies. 4 Once any gastrointestinal lesion was excluded in our case at the staging diagnostic examination, the identification of the same IHC features and pathogenic gene variants confirmed the common origin of the cutaneous lesion with the primary pulmonary nodule. In particular, both in primary and subcutaneous metastasis, we found the pathogenic variants of CDKN2A and KRAS genes whose mutations in lung tumors are widely described in the literature and are already identified in PEAC. 4
Interestingly, we also demonstrated two additional pathogenic mutations in the metastatic lesion, located in the SMAD4 and the FLT3 genes, which suggests a possible genetic evolution from the primary lesion.
The tumor suppressor gene SMAD 4 (OMIM # 600993), also known as DPC4, encodes for a fundamental protein that mediates the signal transduction of TGF-β and BMP superfamilies, acting on the transcriptional activation of target genes and pathways, such as WNT/β-catenin, MAPK, and PI3K/AKT. 9 In particular, the TGF-β/SMAD4 signaling pathway controls numerous cellular responses such as proliferation, expression of extracellular matrix proteins, differentiation, apoptosis, cell adhesion, migration, and cancer initiation and progression processes. SMAD4 gene mutations are often found in pancreas, colorectal, esophagus, and stomach cancers, and less frequently in other neoplasms such as prostate and lung tumors. 9 Further evidence indicates that the SMAD4 loss of function is correlated with poor prognosis and tumor metastasis. 10 In this regard, the SMAD4 mutation (c.767A> T; p.Gln256Leu, Q256L) discovered by ourselves in cutaneous metastasis has been described in very few cases, including a renal metastasis from primitive thyroid cancer, whose interpretation may appear intriguing. 11
The FLT3 gene (OMIM # 136351) encodes for a protein that belongs to the growth factor receptor tyrosine kinase family. This specific receptor is mainly expressed in the hematopoietic compartment, and after activation by its ligand (FLT3 ligand, FL) recruits a series of proteins involved in signal transduction pathways that regulate crucial cellular processes such as proliferation, survival, and differentiation. Activating mutations of the FLT3 gene occur in acute myeloid leukemia and apparently correlate with a poor prognosis. 12
Oncogenic somatic mutations are also described in lung cancer, and both experimental and computational evidence suggest that FLT3 may be implicated in the metastatic processes of this neoplasm. 13
In conclusion, we are the first to describe the molecular characterization of a lung enteric adenocarcinoma with a rare synchronous cutaneous metastasis. Our study emphasizes the need for focusing studies by NGS analysis, as well as extended multicenter collaborations given the rarity of this neoplasm. Therefore, we hope that this case report may increase the knowledge on this rare type of tumor and suggest novel molecular indications for diagnosis and personalized therapy.
Footnotes
Acknowledgements
The authors wish to thank Dr. Gerardo Cazzato, Division of Pathology University of Bari Aldo Moro, for his excellent assistance and Associazione per la Ricerca Biomolecolare Organizzazione Non Lucrativa di Utilità Sociale (A.R.B. ONLUS).
Author contributions
AT and VI contributed equally to this work.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by a grant from the Italian Association for Cancer Research (AIRC, grant number 17536) and from the Apulia Region (Oncogenomic Project Jonico-Salentino Project) (to F.S.).
