Abstract
Objective:
The aim of this study is to investigate the effect of colchicine and other medications for gout treatment on serum CA72-4 and other tumor markers including alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), CA19-9, CYFRA21-1, neuron specific enolase (NSE), prostate-specific antigen (PSA).
Methods:
A total of 143 gout patients and 40 healthy individuals were randomly recruited. Of these patients, 38 did not receive any treatment; 42 received colchicines; 62 received other medications for gout treatment (Etoricoxib, glucocorticoid, Benzbromarone, allopurinol, Febuxostat, nonsteroidal antiinflammatory drugs or their combinations). Of 42 gout patients receiving colchicines, 23 were followed up. All tumor markers were detected by electrochemiluminescence immunoassays. Statistical analysis was performed by SPSS 17.0 software.
Results:
The serum CA72-4 level was significantly higher in gout patients receiving colchicine than that in healthy control and gout patients without any treatment or who were receiving other treatments (P<0.001). There was no significant difference regarding other tumor markers among the various groups. All patients receiving colchicine, but no healthy individuals, patients with no treatment, or those receiving other medications had increased serum CA72-4 levels. Additionally, none or only a few of the subjects had increased AFP, CA19-9, CYFRA21-1, NSE, CEA, or PSA, while the proportion was not significantly different among various groups.
Conclusions:
Colchicine can cause abnormal elevation of serum CA72-4, but other medications have no effect on serum CA72-4. Furthermore, serum CA72-4 gradually increased and decreased after the initiation and cessation of colchicine therapy, respectively. Other tumor markers are not influenced by colchicine and other medications.
Keywords
Introduction
Colchicine has been widely used for gout treatment. In a recent report, by chance we found that the use of colchicine can cause an abnormal elevation of serum CA72-4. 1 However, we have been wondering whether the finding is a general phenomenon. Moreover, it has remained unknown whether other tumor markers (TMs)—such as alpha -fetoprotein (AFP), carcinoembryonic antigen (CEA), CA19-9, CYFRA21-1, neuron specific enolase (NSE), prostate specific antigen (PSA)—are also influenced by colchicine. Additionally, some medications other than colchicines have been used for gout treatment, such as Etoricoxib, glucocorticoid, Benzbromarone, allopurinol, Febuxostat, nonsteroidal antiinflammatory drugs (NSAIDs), etc. 2 It has remained unclear whether these medications can also cause abnormal elevation of serum CA72-4, as well as other TMs. Therefore, the present study was designed to investigate the effect of colchicine and other medications for gout treatment on various serum TMs.
Subjects and methods
Subjects
We randomly included 143 gout patients (only 1 female) and 40 healthy males in Taizhou First People’s Hospital between January 2018 and December 2019. All the patients met the 1977 preliminary American Rheumatism Association classification criteria for gout. 3 When serum samples were obtained, 38 patients were newly diagnosed with gout and did not receive any treatment; 42 had been receiving colchicines for over 3 days; and 62 had been receiving other medications for gout treatment (Etoricoxib, glucocorticoid, Benzbromarone, allopurinol, Febuxostat, NSAIDs or their combinations) for over 3 days. Of 42 gout patients receiving colchicines, 23 were followed up. They took colchicine orally at a dose of 1 mg three times daily. Serum samples were repeatedly obtained for detecting CA72-4 before and 1, 2, 3, 4, 5, 6, 7 days after treatment, and 1, 2, 3 days and 1, 2, 3, 4, 5 weeks after discontinuing colchicine.
The exclusion criteria included various carcinomas, abnormal renal function, liver cirrhosis, or receiving any medication unrelated to gout, and mushroom and Ganoderma lucidum spore powder (GLSP).
The institutional ethics committee approved this study (No. 2019-KY004-01) and all of the subjects gave their signed informed consent.
Laboratory methods
All the TMs were detected by electrochemiluminescence immunoassays using the Modular E601 automated analyzer (Roche, Basel, Switzerland). The analyzer was routinely maintained according to the manufacturer’s instruction. Dedicated reagents and standard methodologies were used. Internal quality controls were performed by Westgard alert rules during the entire period of the study.
Statistical analysis
Statistical analysis was performed by SPSS 17.0 software. Continuous variables were presented as mean ± SD for normal distribution and median and interquartile ranges (IQR) for non-normal distribution. Categorical variables were described as frequencies and percentages. Continuous variables were compared using Student t test or Mann-Whitney U test when appropriate. χ2 test was used to compare categorical variables. Results were considered statistically significant when P <0.05.
Results
The serum CA72-4 level was significantly higher in gout patients receiving colchicine than that in healthy control and gout patients without any treatment or receiving other treatments related to gout (P<0.001). There was no significant difference regarding other TMs including AFP, CEA, CA19-9, CYFRA21-1, NSE, and PSA among various groups (Table 1). All (100%) of patients receiving colchicine for more than 3 days had increased serum CA72-4 levels beyond the upper limit of normal (ULN), while none of the healthy individuals, the gout patients without any treatment and those receiving other medications had increased CA72-4 beyond ULN. Also, none of the subjects had increased AFP, CA19-9, CYFRA21-1, and NSE over ULNs. Increased CEA or PSA over but near the ULNs was detected for a few subjects, while the proportion was not significantly different among the various groups (Table 2).
Serum TM levels in various groups.
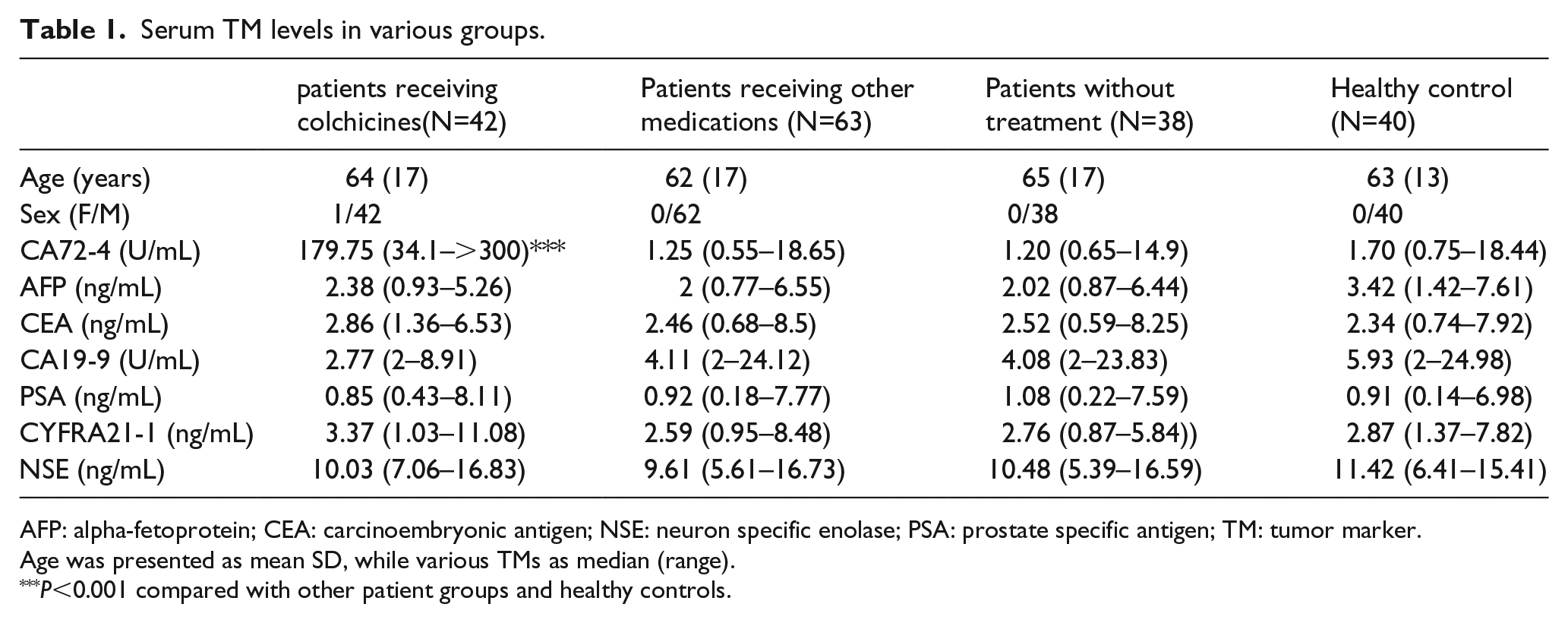
AFP: alpha-fetoprotein; CEA: carcinoembryonic antigen; NSE: neuron specific enolase; PSA: prostate specific antigen; TM: tumor marker.
Age was presented as mean SD, while various TMs as median (range).
P<0.001 compared with other patient groups and healthy controls.
The frequencies and percentages of subjects with TMs over upper limits of normal values.
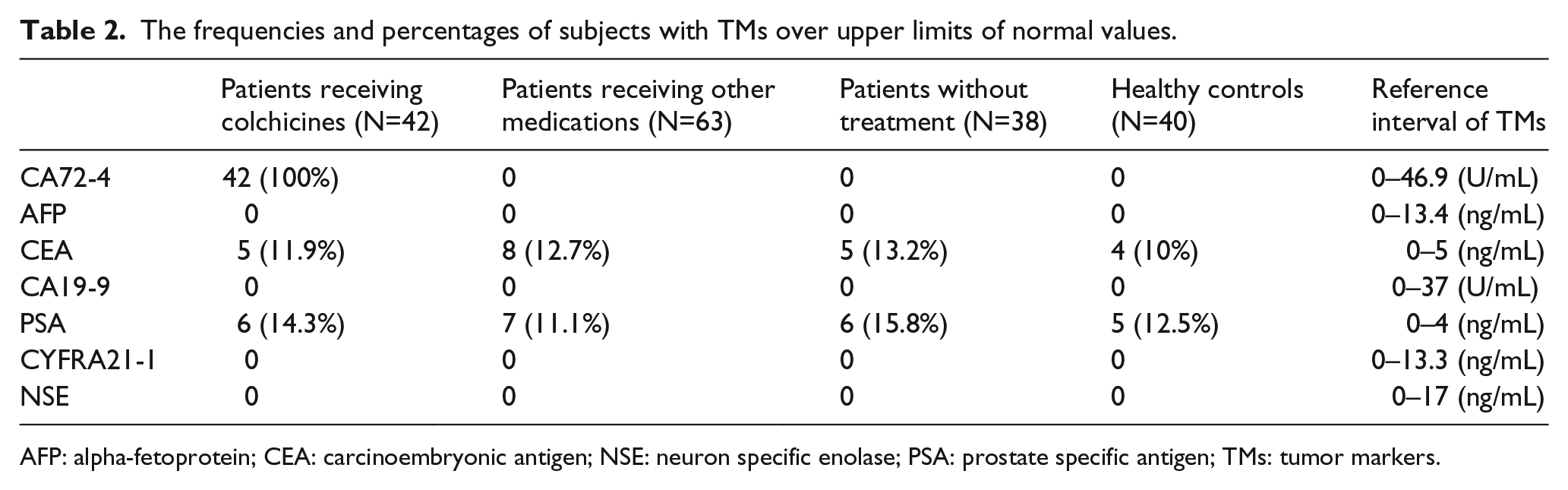
AFP: alpha-fetoprotein; CEA: carcinoembryonic antigen; NSE: neuron specific enolase; PSA: prostate specific antigen; TMs: tumor markers.
None of the patients had increased CA72-4 over ULN before and on day 1 after colchicine therapy. Serum CA72-4 levels showed a gradually increased trend during the course of colchicine therapy and a decreased trend after discontinuing colchicine. In more detail, on day 2 after colchicine therapy, increased serum CA72-4 over ULN was detected in 47.8% of patients, and the proportion reached 95.7% on day 3. From day 4, all of the patients had increased CA72-4 >ULN. Two days after discontinuing colchicines, none of the patients had serum CA72-4 in the normal range. Serum CA72-4 fell to the normal range in 17.4%, 47.8%, and 73.9% of patients at 3 days, 1 week, and 2 weeks, respectively, after withdrawal. All of the patients had decreased serum CA72-4 into the normal range 4 weeks after withdrawal (Figure 1).
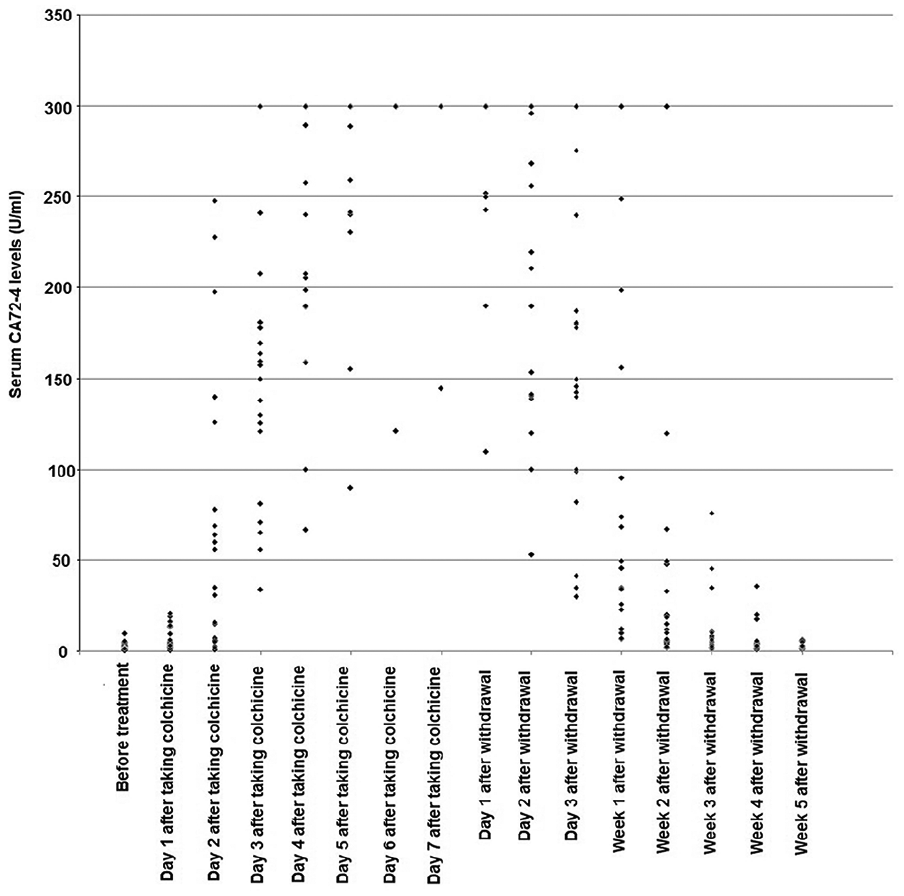
Dynamic changes of serum CA72-4 levels in 23 gout patients during colchicine therapy.
Discussion
It has been known that mushroom and GLSP can cause abnormal elevation of serum CA72-4.4-6 Our previous 1 and current studies confirmed that use of colchicine can also cause abnormal elevation of CA72-4. Meanwhile, we found that gout itself and the uses of other medications related to gout treatment—including Etoricoxib, glucocorticoid, benzbromarone, allopurinol, febuxostat, and NSAIDs—had no effect on serum CA72-4. Additionally, our study indicated that gout itself and all the medications had no effect on the levels of other serum TMs, including AFP, CEA, CA19-9, PSA, CYFRA21-1, and NSE.
Furthermore, we found that serum CA72-4 is gradually increased and decreased after initiation and cessation of colchicine therapy, respectively. We had not detected increased CA72-4 over ULN in 100% of patients until the fourth day after the use of colchicine, and until the 4th week after cessation all patients had decreased CA72-4 falling into the normal range. We cannot explain the reason why colchicine only causes abnormal elevation of serum CA72-4, but has no effect on other serum TMs. Nor do we know the mechanisms by which colchicine causes CA72-4 increase in vivo. However, in the previous study, we verified that colchicine may not directly interfere with the CA72-4 assay. Moreover, the dynamic change of CA72-4 in this study suggests that the biological mechanisms may be complex.
The important implications of our findings lie in three points. First, the clinician can explain to the condition to the patient with abnormal elevation of serum CA72-4, who has no carcinoma and is receiving colchicine therapy. Second, the feature of dynamic change of CA72-4 should be considered during the course of therapy. Since the effect of colchicine on serum CA72-4 may be a chronic course in most patients, an acute elevation of serum CA72-4 might have clinical implications on patients’ general health concerns. Third, other TMs, such as AFP, CEA, CA19-9, PSA, CYFRA21-1, and NSE, may not be influenced by colchicine and other medications related to gout treatment.
In conclusion, it is necessary to communicate with patients about the effect of colchicine on serum CA72-4 in order to alleviate an unnecessary economical and mental burden.
Footnotes
Author contributions
Bing Zhao and Miaomiao Zhang contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Taizhou Science and Technology office program (1801ky62).
Informed consent
Informed consent was obtained from all participants.
