Abstract
Objective:
The aim of the study was to evaluate the diagnostic value of soluble fragment of cytokeratin 19 (CYFRA21-1) tests in detecting non-small cell lung cancer (NSCLC), including squamous cell carcinoma, lung adenocarcinoma, and large cell carcinoma.
Methods:
The relevant studies were identified from PubMed, Embase and the Cochrane Library before November 2018. Summary estimates for sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, and diagnostic odds ratio of CYFRA21-1 tests for the diagnosis of NSCLC were calculated using the random effects model. A summary receiver operating characteristic (SROC) curve was used to assess the overall effectiveness of the test. Meta-DiSc 1.4 and Stata11.0 were applied to the statistical analysis. Publication bias was detected using Egger’s test.
Results:
A total of 22 studies consisting of 7910 NSCLC patients (squamous cell carcinoma/lung adenocarcinoma/large cell carcinoma) and 2630 benign lesions patients that met the inclusion criteria were included. The meta-analysis showed that CYFRA21-1 tests had a relatively high accuracy for squamous cell carcinoma detection and a lower accuracy for lung adenocarcinoma detection. The overall sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, and diagnostic odds ratio of CYFRA21-1 tests for squamous cell carcinoma detection were 0.72 (95% confidence interval (CI) 0.70, 0.74), 0.94 (95% CI 0.92, 0.95), 9.73 (95% CI 7.06, 13.40), 0.37 (95% CI 0.29, 0.47), and 27.30 (95% CI 17.68, 42.16), respectively. The area under the SROC curve was 0.9171 (Q* = 0.8500). No publication bias was tested in the squamous cell carcinoma (P = 0.567) and lung adenocarcinoma (P = 0.378) groups.
Conclusions:
CYFRA21-1 tests might be appropriate for detecting squamous cell carcinoma.
Keywords
Introduction
Lung cancer is one of the leading causes of death throughout the world 1 and has the highest mortality of all cancers. 2 The prognosis of lung cancer is poor and less than 15% of patients can survive 5 years after diagnosis. The lack of efficient diagnostic methods and successful treatment for early detection and metastatic disease have caused the poor prognosis. 3 Approximately 85% of all lung cancers are classified as non-small-cell lung cancer (NSCLC), including squamous cell carcinoma (SCC), lung adenocarcinoma (LAD), and large cell carcinoma (LCC), and 10% are small cell lung cancer and 5% other histological variants with distinct biological behavior and genetic alterations. 4 The NSCLC is potentially curable by surgical resection if it is discovered at an early stage. 5 However, patients with resected NSCLC are at risk for recurrence of disease, since there are no effective tools to predict which patients are at highest risk. 6 Early detection and diagnosis is urgent for the sake of an effective treatment strategy for lung cancer. 7
Currently, there are few validated biomarkers that can predict survival or treatment response for NSCLC and most are based on tumor markers. 8 A newly established monoclonal antibody, soluble fragment of cytokeratin 19 (CYFRA 21-1), which is a serum cytokeratin 19 fragment, has been shown to react exclusively with cytokeratin 19. 9 In addition, CYFRA 21-1 has been well documented as an excellent tumor marker for NSCLC. However, a few studies on CYFRA 21-1 for NSCLC reported a limited clinical diagnostic value of CYFRA 21-1 for NSCLC because of low sensitivity. 10 Therefore, whether the diagnostic value of a CYFRA21-1 test may be appropriate for detecting NSCLC is controversial.
In order to achieve a comprehensive assessment of diagnosis for NSCLC (SCC, LAD, and LCC) with CYFRA21-1 tests, it is necessary to consider the findings as a whole, giving attention to methodological characteristics of the studies. Accordingly, we conducted a systematic review of published achievements using meta-analysis techniques to quantitatively combine the results.
Material and methods
Data sources and search strategy
Systematic document retrieval was performed up to November 21st, 2018 from PubMed, Embase, and the Cochrane Library. The search strategy for this meta-analysis was ((soluble fragment of cytokeratin 19) or CYFRA21-1 or (CYFRA 21-1)) and ((non-small cell lung cancer) or NSCLC or (squamous cell lung cancer) or (lung adenocarcinoma)) and (diagnose or diagnosis or diagnostic). Meanwhile, the bibliographies of all selected articles were reviewed to identify additional relevant studies. Only data presented as full-text articles were eligible for inclusion.
Selection of studies
The observers independently checked all retrieved articles according to the inclusion criteria. Disagreements were resolved by consensus. The inclusion criteria were as follows: (a) published literature mainly focused on the clinical value of serum CYFRA21-1 for diagnosis of different subtypes of NSCLC patients, including SCC, LAD, and LCC; (b) pathological analysis or the World Health Organization (WHO) classification was considered in the diagnosis of lung cancer, and subjects adults (⩾18 years old) were eligible for inclusion; (c) patients with benign lung lesions treated in the same hospital and at the same time were selected as the control group; (d) the effect sizes were sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR) and diagnostic odds ratio (DOR); (e) Studies providing data regarding the diagnosis results of CYFRA21-1 test for different subtypes of NSCLC were included, including the number of true positives (TP), false positives (FP), false negatives (FN), and true negatives (TN). However, studies were excluded with the following characteristics: (a) healthy subjects as the control group; (b) incomplete data that are difficult to perform statistical analysis; (c) the pleural fluid or pleural effusion CYFRA 21-1 could be used for the diagnosis of NSCLC; (d) non-original articles: including abstracts, reviews, letters, and comments; and (e) duplicate publications from the same cohort.
Data extraction and study quality assessment
Data from the published studies were extracted independently by two reviewers. The following data were extracted for each study: study details (e.g., the first author’s name, year of publication, design of studies, region and duration of the study, etc.), characteristics of participants (e.g., age, sex and sample size, etc.). After the completion of data extraction, the investigators exchanged the data sheets, and any inconsistencies were resolved by discussion.
To assess study quality characteristics, Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2, a tool used in the quality assessment of studies of diagnostic accuracy included in systematic reviews 11 ), was used as a guideline. QUADAS is structured as a list of 11 items, which should be answered with “yes”, “no” or “unclear”. When a specific item was achieved, a score of “1” was given, “0” if this item was “unclear” and “−1” if the item was not fulfilled. 12 The total scores were calculated by counting up the score of each item. The same two reviewers independently assessed the study quality, and disagreements were solved by consensus.
Statistical methods
A summary receiver operating characteristic (SROC) curve, based on the sensitivity and specificity of each selected study, was created in this study. The area under the curve (AUC) was employed as potentially available summaries of the curve. Concerning the publication bias for meta-analyses of diagnostic studies, we explored the potential publication bias through “midas”—Meta-DiSc statistical software version 1.4 (http://www.hrc.es/investigacion/metadisc_en.htm)—and the STATA software package v.11.0 (Stata Corporation, College Station, TX, USA) were used for all the above statistical analyses. All statistical tests were two sided, and P < 0.05 was considered to be statistically significant. The heterogeneity test was performed through Cochran’s Q statistics and I 2 test. If there was significant heterogeneity with P < 0.05 or I 2 > 50%, the random effects model was chosen to obtain the combined effect value; otherwise, the fixed effects model was adopted (P ⩾ 0.05 or I 2 ⩽ 50%). Also, a subgroup analysis was performed based on the regional differences (Asian area vs. Western area), study types (prospective vs. retrospective study) and the detection methods of serum CYFRA21-1 (ECLIA vs. EIA, ELISA, and IRMA), investigating the effect of the above factors on the results of sensitivity, specificity, PLR, NLR, and DOR in different subtypes of NSCLC.
Results
Characteristics of eligible studies
There were 976 papers potentially relevant to the search terms (PubMed = 423; Embase = 527; Cochrane Library = 26). First, 267 duplicates were excluded. Then, 668 studies that were clearly not relevant were excluded. Subsequently, 19 studies (9 articles with healthy control, 7 articles without interested outcomes and 3 only with abstracts) were removed by reviewing the full-text based on the inclusion and exclusion criteria. Finally, 22 studies13-34 were included in this meta-analysis. The flow chart of study selection and literature search is shown in Supplementary Figure 1.
The characteristics of included studies are presented in Table 1. In total, 22 studies including 7910 NSCLC patients and 2630 benign lesions patients were included in this study. The publication year of the included studies ranged from 1993 to 2018. These studies were conducted in China, France, Sweden, Turkey, Israel, and Japan. The diagnostic results of CYFRA21-1 test for NSCLC (SCC/LAD/LCC) with the number of TP, FP, FN, and TN detections are shown in Supplementary Table 1. In addition, QUADAS-2 was used for the quality assessment for the included studies, and the results showed that the quality of the studies was relatively high (Supplementary Table 2).
Characteristics of studies included in the meta-analysis.
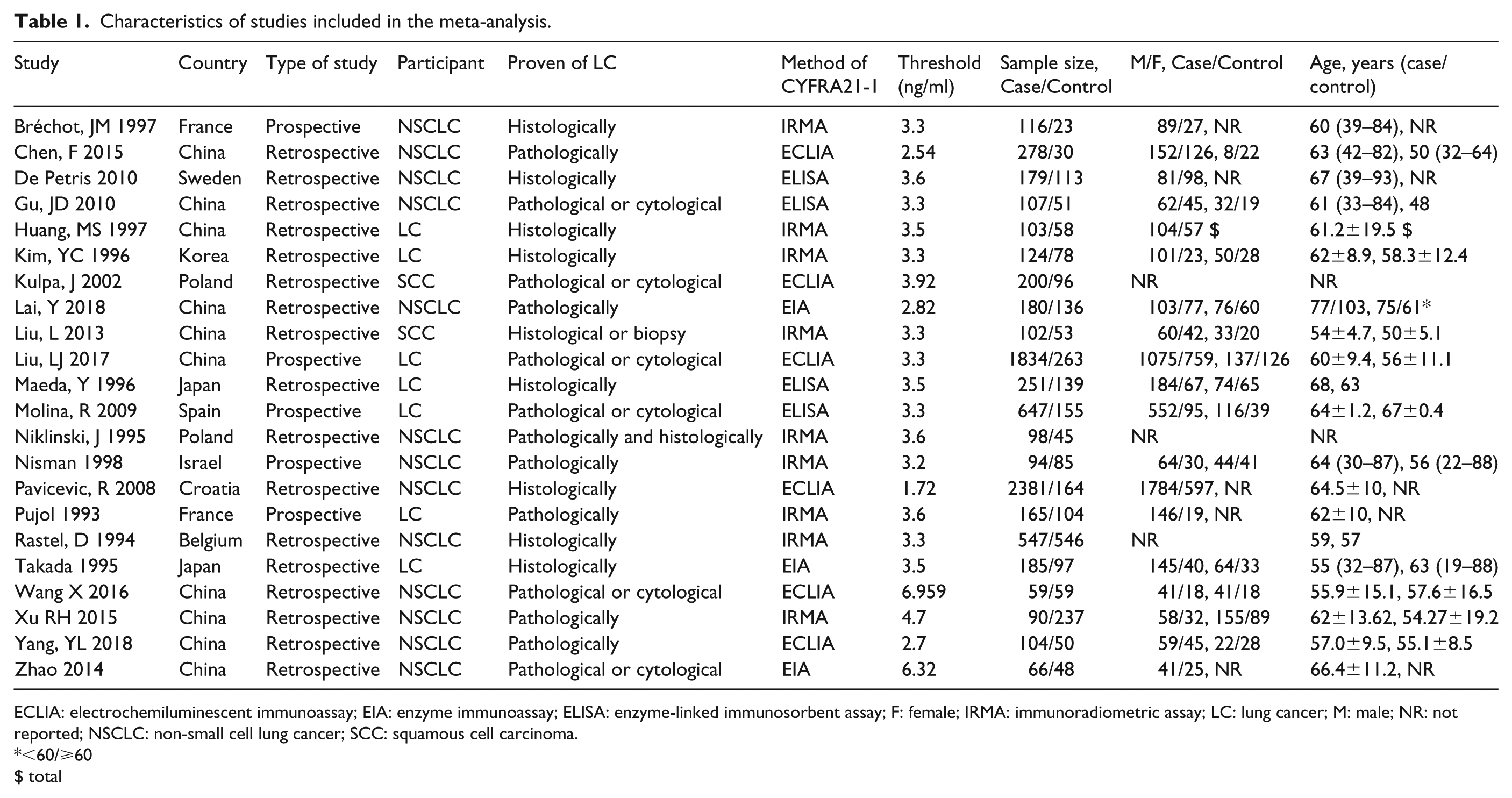
ECLIA: electrochemiluminescent immunoassay; EIA: enzyme immunoassay; ELISA: enzyme-linked immunosorbent assay; F: female; IRMA: immunoradiometric assay; LC: lung cancer; M: male; NR: not reported; NSCLC: non-small cell lung cancer; SCC: squamous cell carcinoma.
<60/⩾60
$ total
Diagnostic accuracy
The random effects model was performed for the meta-analysis of pooled quantitative data due to statistically significant heterogeneity across the included studies (P < 0.05 or I 2 > 50%). In this pooled analysis of serum CYFRA 21-1 level in all NSCLC patients showed that the pooled sensitivity, specificity, PLR, NLR, and DOR was 0.60 (95% confidence interval (CI) 0.58, 0.61), 0.90 (95% CI 0.89, 0.91), 5.93 (95% CI 4.10, 8.57), 0.40 (95% CI 0.32, 0.50), and 16.17 (95% CI 10.04, 26.04), respectively, and the area under the SROC curve was 0.8676 (Q*=0.7981) (Figure 1). Then, the diagnostic value of CYFRA 21-1 tests in different subtypes of NSCLC was also evaluated separately. As shown in Figure 2, the pooled sensitivity (0.72, 95% CI 0.70, 0.74), specificity (0.94, 95% CI 0.92, 0.95), PLR (9.73, 95% CI 7.06, 13.40), NLR (0.37, 95% CI 0.29, 0.47), DOR (27.30, 95% CI 17.68, 42.16), and the area under the SROC curve (AUC = 0.9171, Q*=0.8500) were displayed in the pooled analysis encompassing SCC patients. In addition, in LAD patients, the meta-analysis results showed that the pooled sensitivity was 0.48 (95% CI 0.46, 0.50), specificity was 0.94 (0.93, 0.95), PLR was 8.32 (4.60, 15.05), NLR was 0.52 (0.38, 0.72), DOR was 16.25 (7.00, 37.71), and the area under the SROC curve was 0.9714 (Q*=0.9221) (Figure 3). Furthermore, in LCC patients, the pooled sensitivity, specificity, PLR, NLR, and DOR was 0.67 (95% CI 0.61, 0.72), 0.93 (95% CI 0.91, 0.95), 8.48 (95% CI 4.75, 15.14), 0.53 (95% CI 0.35, 0.81), and 17.16 (95% CI 7.61, 38.68), respectively, and the area under the SROC curve was 0.9584 (Q*=0.9022) (Figure 4). These results showed that the diagnostic value of the CYFRA21-1 test for SCC patients was higher than that for the LAD and LCC patients, and the LAD patients had the lowest diagnostic accuracy.
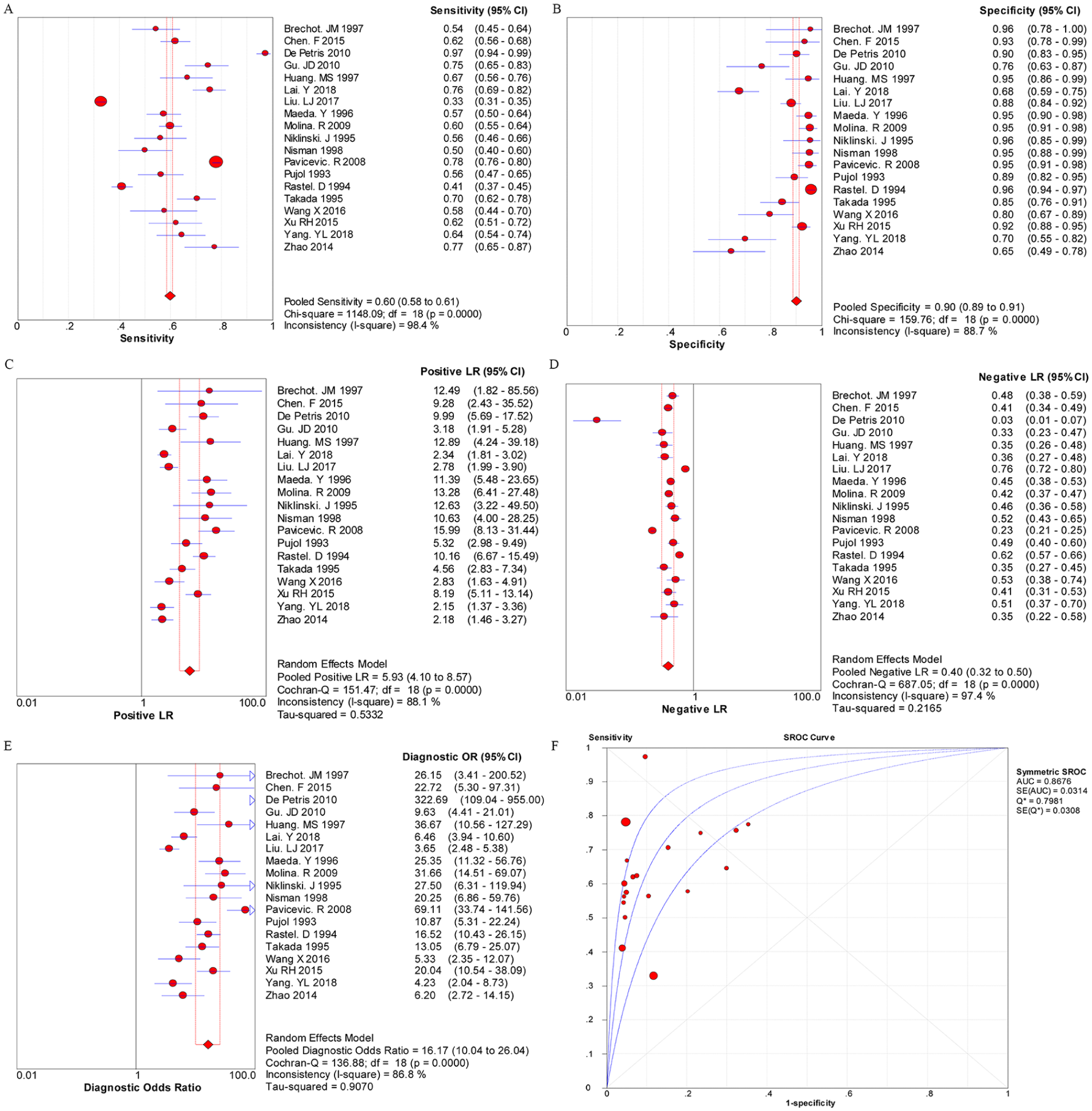
The pooled results for the diagnostic accuracy of soluble fragment of cytokeratin 19 (CYFRA 21-1) tests for all non-small cell lung cancer (NSCLC) patients.
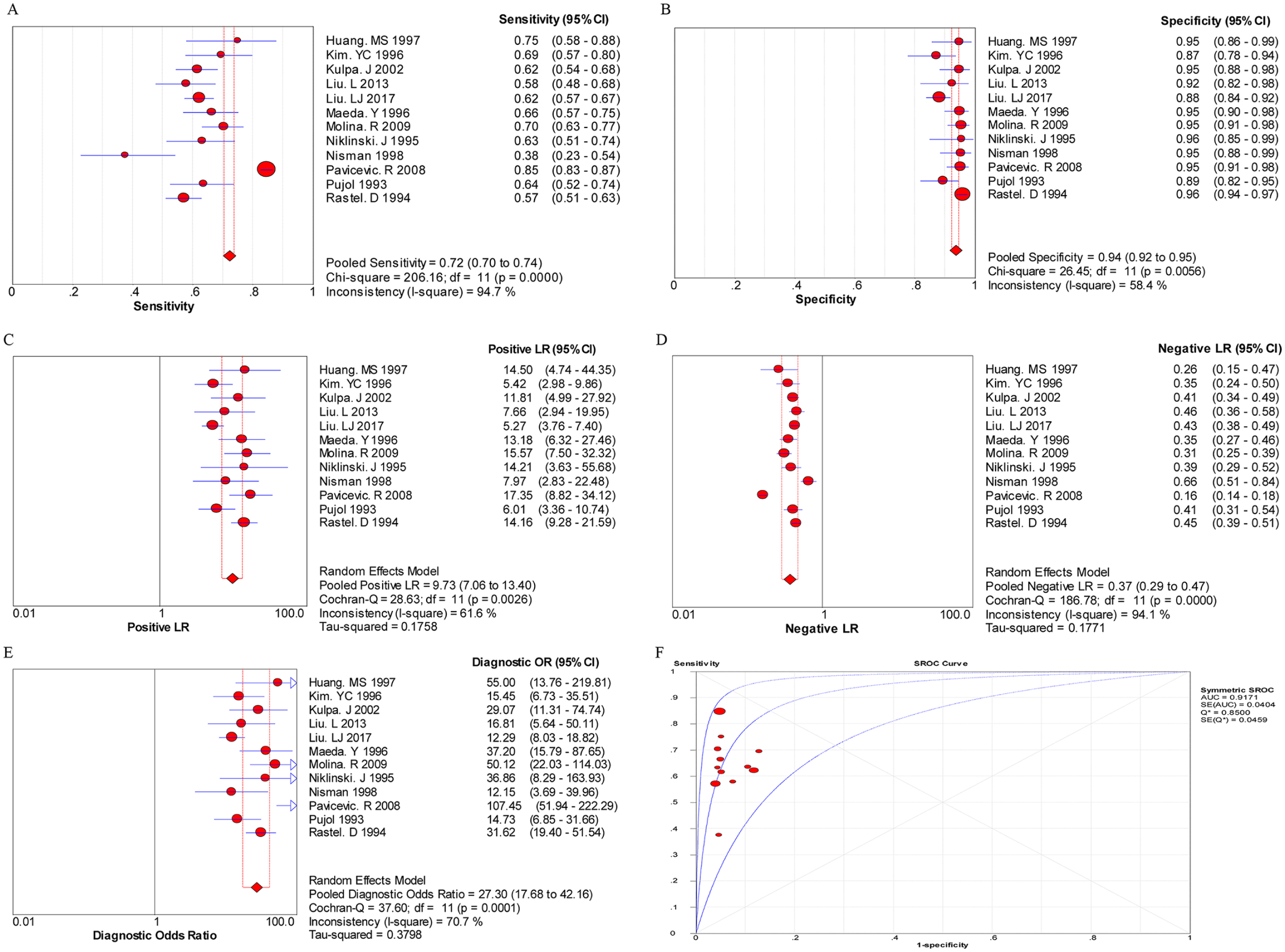
The pooled results for the diagnostic accuracy of CYFRA 21-1 tests for SCC patients.
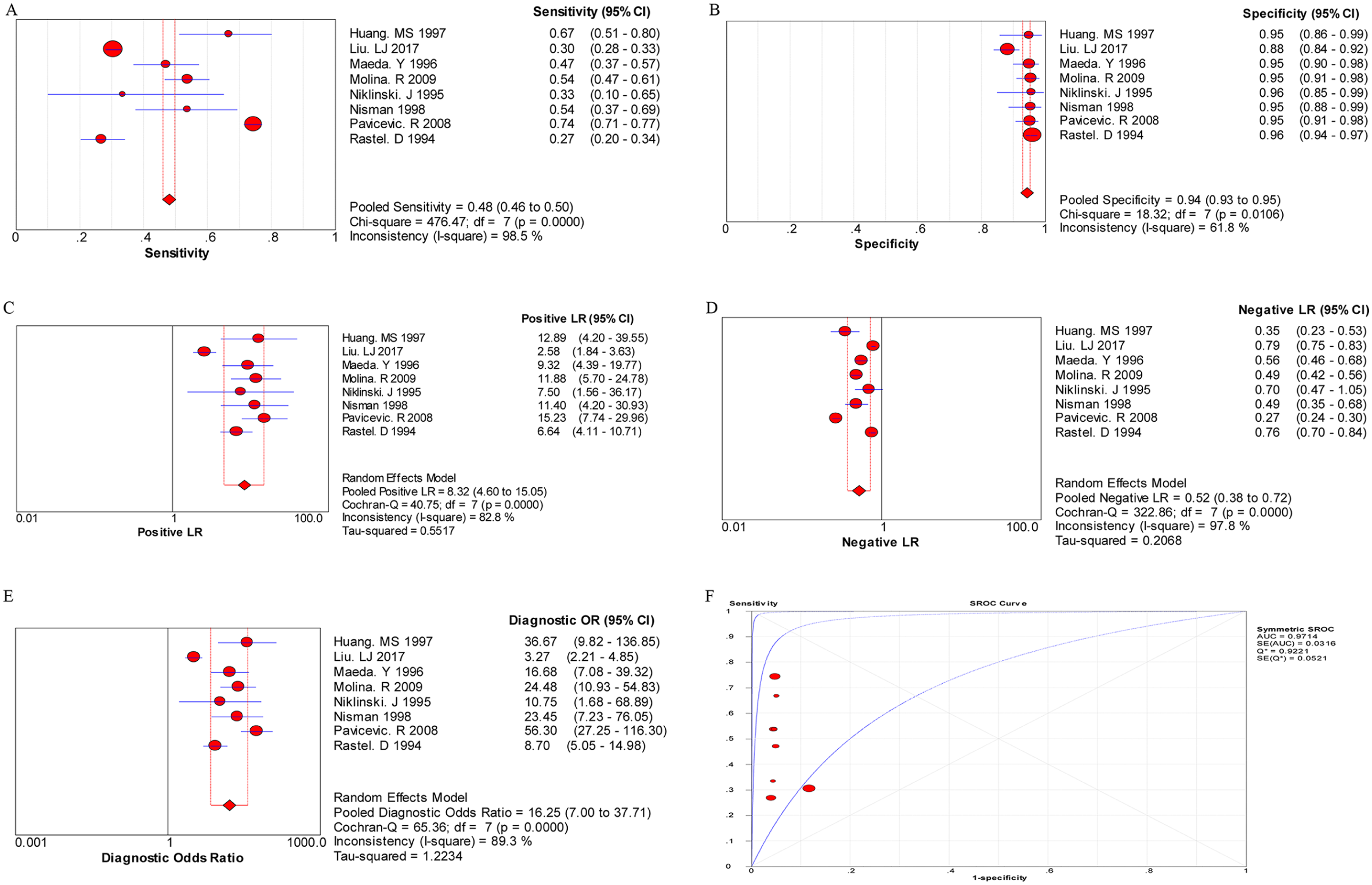
The pooled results for the diagnostic accuracy of CYFRA 21-1 tests for LAD patients.
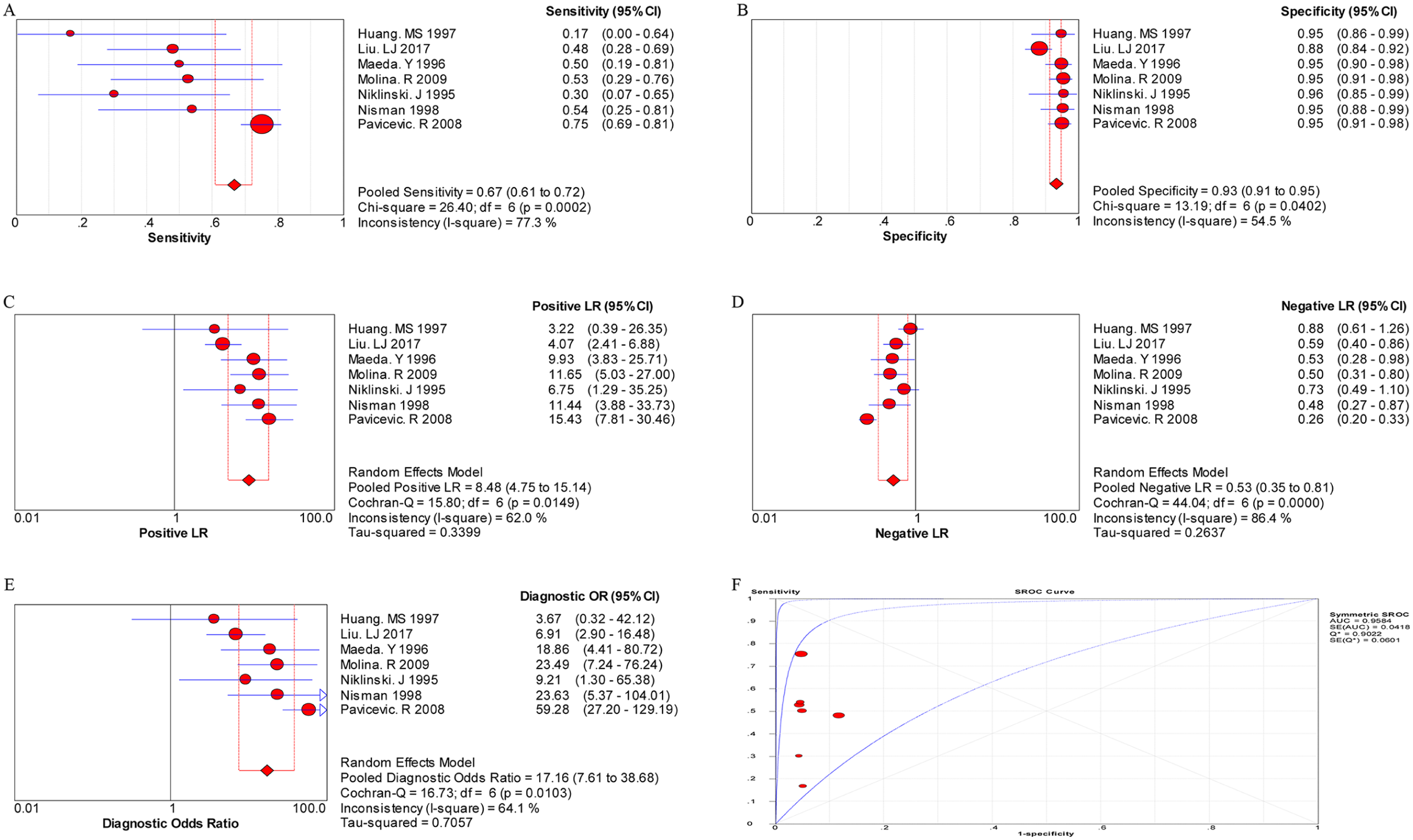
The pooled results for the diagnostic accuracy of CYFRA 21-1 tests for LCC patients.
Subgroup analysis
Subgroup analysis of the sensitivity, specificity, PLR, NLR, and DOR among different regions, types of study, and detection methods of CYFRA21-1 in different subtypes of NSCLC patients is listed in Supplementary Table 3. In SCC patients, subgroup analysis by regional difference showed that the PLR of the CYFRA21-1 test in patients from both the Asian and the Western areas was 7.24 (95% CI 5.07, 10.34) and 12.20 (95% CI 8.51, 17.49), respectively, with no significant heterogeneity. Similar findings regarding the PLR of the CYFRA21-1 test were observed in the LCC patients from different areas, suggesting that regional difference might be an influential factor for the diagnostic value of CYFRA21-1 tests and CYFRA21-1 values pooling. Furthermore, there was no significant heterogeneity between the studies that applied different methods for the detection of serum CYFRA21-1; thus, technical methodologies used for CYFRA21-1 detection were not responsible for the differences merging from the performed meta-analysis.
Publication bias
No publication bias was detected in NSCLC (P = 0.773), SCC (P = 0.567), and LAD (P = 0.378) using Egger’s test, while publication bias was found in the LCC group (P = 0.030).
Discussion
NSCLC is a major cause of death in both developed and developing countries and remains a major challenge to medical oncologists. 35 Lack of sensitivity and specificity has been one of the problems in the use of the carcinoembryonic antigen (CEA) and the SCC antigen (SCC-Ag) in NSCLC. 36 CYFRA21-1, a fragment of cytokeratin 19, is a biomarker of NSCLC. 37 Recently, many studies have reported the diagnostic value of CYFRA21-1 tests in detecting NSCLC.38, 39 However, these studies have shown controversial and inconclusive results regarding the diagnostic value of this method in different subtypes of NSCLC due to small sample sizes or low statistical power. Therefore, the present meta-analysis evaluated the diagnostic value of CYFRA21-1 tests for different subtypes of NSCLC (SCC, LAD, LCC), suggesting that CYFRA21-1 tests had a relatively high accuracy for SCC detection and a lower accuracy for LAD detection.
There are several tumor markers that have been studied in lung cancer, including CYFRA21-1, CEA, neuron-specific enolase (NSE), cancer antigen 125 (CA-125), and SCC-Ag).40,41 CYFRA 21-1 is responsible for NSCLC diagnosis. 42 CYFRA 21-1, a fragment of cytokeratin 19 which is responsible for the structural integrity of epithelial cells, 43 was investigated in NSCLC with reasonable diagnostic performances, 44 which had also been verified by the present meta-analysis. Cytokeratins are the main components of cellular microfilaments. Every epithelial cell contains a differentiation-dependent combination of two or more cytokeratin proteins. 45 Cytokeratins 8, 18, and 19 are expressed in simple epithelium, including the bronchial epithelium, and in malignant tumors derived from these cells. Insoluble cytokeratin filaments are released into the bloodstream in the form of soluble fragments from dead or dying cells after proteolytic degradation. Therefore, they could be used as serum tumor markers. 38
In the present meta-analysis, studies were enrolled according to the including criteria to evaluate the diagnostic value of CYFRA21-1 tests in detecting different subtypes of NSCLC, including SCC, LAD and LCC. The average QUADAS scores of the included studies were relatively high. As a whole, the pooled sensitivity and specificity of CYFRA21-1 tests for all NSCLC patients calculated by the bivariate random effects model were 0.60 (95% CI 0.58, 0.61) and 0.90 (95% CI 0.89, 0.91), respectively, which was higher than other tumor markers, such as NSE and CA-125. 46 It has been reported that CYFRA 21-1 is recognized as a prognostic and predictive marker in NSCLC. 47 Furthermore, CYFRA 21-1 is positively related to disease stage, performance status, different pathological types of lung cancer, and a high CYFRA 21-1 level indicates worse prognosis. 42 Similarly, our data also found that the diagnostic value of CYFRA 21-1 tests for SCC patients was relatively higher than that for LAD and LCC detection. This results was similar to Liu et al.’s 48 study that the SCC cases had the highest level of CYFRA 21-1; the LAD group was the second; and the lowest CYFRA 21-1 was detected in the LCC group. In addition, DOR is a single overall indicator of diagnostic accuracy, which represents the frequency of positive test results among patients with or without the condition of interest. The value of a DOR ranges from 0 to infinity; the higher values indicate the better diagnostic performances. A DOR of 1.0 indicates that a test does not discriminate between patients with the disorder and those without it. 49 The present meta-analysis found that the mean DOR for the diagnosis of SCC was 27.30 (95% CI 17.68, 42.16), which indicated a relatively high level of overall accuracy since a previous study had achieved a DOR of 20.33 in the diagnosis of lung cancer. 49 Besides, our data also achieved an AUC of 0.9171 (Q*=0.8500) by a meta-analysis. Overall the pooled results above exhibited a relatively high level of overall accuracy of CYFRA21-1 tests in patients with SCC.
There were several drawbacks and limitations within this study that must be acknowledged. First, although articles focused on the diagnostic value of the detection of CYFRA21-1 in NSCLC were included in this meta-analysis, relatively few studies were available for meta-analysis on the diagnostic value of the CYFRA21-1 test for LAD or LCC patients, indicating that more updated literature is needed to make the results more reliable. Second, only published studies were included in this meta-analysis, which might introduce bias. Third, it was a retrospective review based on studies, and the selection bias could not be completely avoided. We minimized the likelihood of bias by developing a detailed protocol before initiating this study, performing an elaborative search for published studies and employing explicit methods for study selection, data extraction, and data analysis. Meanwhile, high heterogeneity was found in this meta-analysis, which might be due to the regional difference according to the results of the subgroup analysis. Therefore, continuous updating and further studies should be conducted to confirm these results in our future work.
In summary, the present meta-analysis suggests a relatively high diagnostic value in CYFRA21-1 tests for the detection of NSCLC, especially for patients with SCC. Considering the limitations and the heterogeneity among the selected studies, large and well-designed prospective studies will be needed to evaluate the diagnostic value of CYFRA21-1 tests. In addition, in order to accurately understand the diagnostic value of CYFRA21-1 tests for NSCLC, further studies should be performed to study the stage and the earliest time that NSCLC can be identified by CYFRA21-1.
Supplemental Material
Supplementary_material – Supplemental material for CYFRA21-1 tests in the diagnosis of non-small cell lung cancer: A meta-analysis
Supplemental material, Supplementary_material for CYFRA21-1 tests in the diagnosis of non-small cell lung cancer: A meta-analysis by Lei Fu, Rong Wang, Ling Yin, Xiaopu Shang, Runtong Zhang and Pengjun Zhang in The International Journal of Biological Markers
Footnotes
Authors’ Note
Lei Fu currently is also affiliated with Joint Logistics College, National Defence University of PLA, Beijing, China
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Key Program; project number 71532002); and Science and Technology Basic Work of Key Projects of the Ministry of Science and Technology of China (project number 2013FY110900).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
