Abstract
Background:
Non-small cell lung cancer (NSCLC) is difficult to treat when metastasis has occurred. This study explores the use of cell-free DNA in the clinical management of NSCLC patients who have Kirsten rat sarcoma viral oncogene homolog (KRAS)-positive mutations and as a marker for prognosis.
Methods:
Peripheral blood collected from advanced NSCLC patients was examined with digital droplet polymerase chain reaction and ultraviolet spectrometry. KRAS mutations were analyzed and quantitated. The specificity and sensitivity of the proposed assay was computed by associating the results with tumor tissue specimens. Comparison against different sub-groups of patients with different metastatic sites and healthy volunteers were made. Patients were subsequently followed up and survival analysis was conducted.
Results:
Among the 186 patients recruited, 150 had concordant KRAS mutational profiles using cell-free DNA with tumor tissues. The assay sensitivity and specificity were 80.6% and 100%, respectively. For the 150 patients with concordant results, the range of cell-free DNA quantities in peripheral blood was 5.3 to 115 ng. Among the patient groups with different metastatic sites, we observed that patients with bone metastasis had higher concentrations of cell-free DNA. Survival analysis showed that these patients had worse survival outcome. Patients with higher KRAS counts in peripheral blood also had worse outcome.
Conclusion:
The use of cell-free DNA presents opportunities for risk stratification of patients and possibly aids in the clinical management of the disease. In the current study for NSCLC, patients with bone metastases showed higher cell-free DNA concentrations. Quantitating the concentrations of cell-free DNA presents a noninvasive biomarker capable of prognostic utility.
Introduction
Non-small cell lung cancer (NSCLC) is a complex disease with increasing global incidence.1, 2 Currently, it is a leading cause of mortality among other diseases with several associated risk factors.3, 4 The main challenge is early detection where symptoms are generally asymptomatic. Many patients are diagnosed when the disease is in an advanced stage. In many instances, dissemination and growth of cancer cells in distant metastatic sites are present at first diagnosis, 5 which leads to various difficulties in treatment. Patients tend to have poor prognosis and will benefit from close monitoring during disease progression. Oncogenic abnormalities associated with the Kirsten rat sarcoma viral oncogene homolog (KRAS) are fairly prevalent gene mutations 6 identified in NSCLC. The mutational rate is as high as 30%7, 8 among other common genetic mutations such as epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase.
In certain western regions, the most frequent oncogenic driver is KRAS, 9 and understanding this mutational impact to treatment is critical. Studies have demonstrated that KRAS-positive patients do not respond well to first-line tyrosine kinase inhibitors. 10 Immunotherapy is a new promising approach9, 11 and better tracking of disease progression will allow timely clinical intervention. For NSCLC, detecting the presence of KRAS is important and often challenging. Molecular profiling is commonly performed on tumor tissue biopsies, which has its difficulties. 12 Intra-tumoral or inter-tumoral heterogeneity may hinder the detection of the mutation if the sampling is not performed at the right site.13, 14
The use of cell-free DNA (cfDNA) is gaining popularity as studies have shown that this can capture the heterogeneous nature of different metastatic sites. 15 This makes it useful in advanced-stage patients where the tumor has metastasized and there is a need for multiple site investigations. Furthermore, the procedure is relatively less invasive than surgical removal of tumor tissues and thus more easily accepted by patients. Although imaging methods have allowed better interrogation of cancerous sites than tumor biopsies, obtaining physical samples is still required for molecular profiling. cfDNA presents a promising route to access molecular signatures associated with the disease. In the National Comprehensive Cancer Network guidelines for NSCLC, 16 the use of cfDNA has been shown to be useful in clinical management of the disease. For NSCLC, numerous studies17, 18 have shown that this disease is highly associated to primary tumor tissues and prove that it originates from these cancerous tumors. Studies have also managed to uncover numerous low frequency mutations associated with disease aggressiveness and proliferation, which would otherwise have been masked in tumor tissue analysis. 19 The use of cfDNA is expanding and its use will greatly aid in offering better clinical management of patients.
While extensive data are available on the predictive and prognostic significance of cfDNA in NSCLC, specific critical groups of patients have not been extensively examined. The current study targets advanced stage NSCLC patients with positive KRAS mutation and its association to different metastatic sites. The aim is to explore the use of cfDNA for the detection and prognostic utility that will aid in better risk stratification of advanced NSCLC patients.
Materials and methods
Patient population
We conducted an observational study initially with 186 recruited patients and 30 healthy individuals. All participants provided informed consent to be part of the study and the institutional review board approved all study protocols. The study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. The clinico-pathological background of NSCLC patients is shown in Table 1. All patients had advanced NSCLC and were mostly at stage IV. KRAS mutations were confirmed using molecular profiling of the tumor tissues as per routine assays. NSCLC patients were subsequently categorized based on the metastatic patterns and levels of their cfDNA detected. Patients with cancer of unknown origin were excluded from this study. Healthy volunteers with no family history of lung cancer were enlisted to provide a direct comparison to cancer patients tested using cfDNA.
Clinico-pathological characteristics of KRAS-positive NSCLC patients and healthy donors.
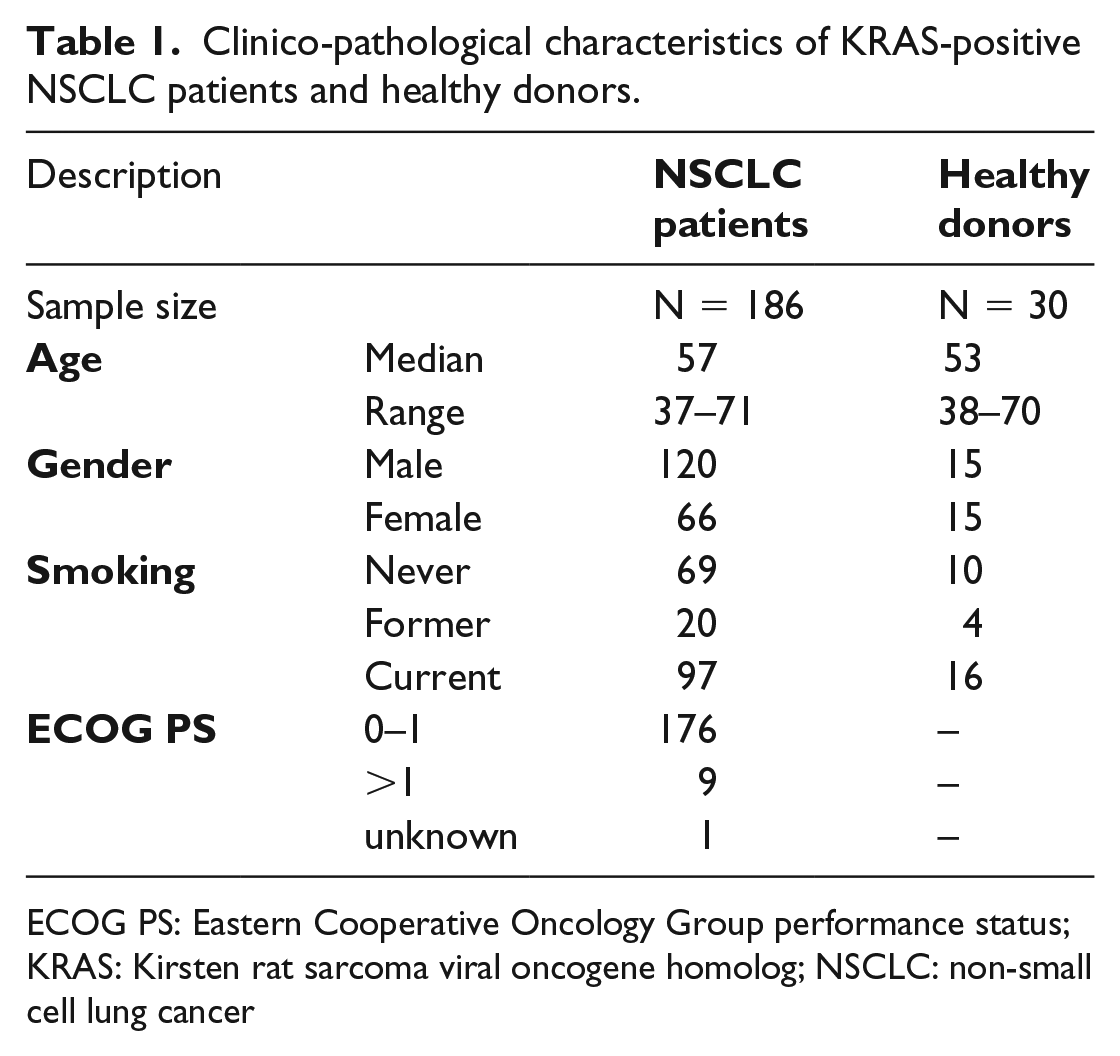
ECOG PS: Eastern Cooperative Oncology Group performance status; KRAS: Kirsten rat sarcoma viral oncogene homolog; NSCLC: non-small cell lung cancer
Sample collection and blood processing procedures
Blood specimens were drawn using EDTA vacutainer tubes by a trained phlebotomist. Specimens were collected within a week of tumor tissue extraction and processed immediately. Whole venous blood of 5 mL was taken from each participant and spun down to separate out the plasma. This was accomplished using a two-step centrifugation process at 3000 xg in a refrigerated condition. The first step involved removing blood cells and platelets, and fresh blood samples were spun at 3000 xg for 10 minutes at 4°C. The separated plasma was carefully removed to a new centrifuge tube and spun at the same condition again. This ensured that all cellular components and debris were entirely removed. Separated plasma was aliquoted into two 1.5 mL conical tubes and stored frozen or purified immediately to recover cfDNA. For cfDNA purification, this was achieved using spin columns from the Qiagen’s QIAamp circulating nuclei acid kit (Qiagen Inc., USA). The procedures strictly followed the manufacturer’s instructions, and cfDNA from each reaction tube was eluted in 10 µL nuclease-free water. Purified cfDNA was then profiled using the ultraviolet (UV) spectrometer (Nanodrop 2000, Thermo Fisher, USA) and stored frozen prior to molecular profiling. For healthy volunteers, the process followed the same sample processing pipeline. The reported concentration of cfDNA was derived from the average of the readings from each of the aliquots separated during plasma extraction.
Molecular processing of cfDNA to confirm KRAS mutations
The identification of positive KRAS mutations was achieved by comparing healthy samples and patients’ assay results. Digital droplet polymerase chain reaction (ddPCR) was used as this assay could detect low mutational frequencies within high background noise specimens. The primers and probes used were procured from a commercial company that validated their primer design (Bio-Rad PrimePCR ddPCR mutational assay, Bio-Rad, USA) for KRAS mutations. For validation, we performed a serial dilution of KRAS-positive plasmid samples prior to testing on clinical specimens. The procedure for processing is fairly straightforward and samples were batched processed to minimize experimental bias. Briefly, each PCR reaction consisted of 20 µL containing master mixes, DNA template, primers/probes, and nuclease-free water. The master mixes were prepared fresh as recommended by the manufacturer, and samples were mixed thoroughly prior to droplet generation in the QX200 (Bio-Rad, USA). The droplets were then transferred to a 96-well PCR plate for PCR amplification. The thermocycling conditions was set at 95°C, 10 minutes (1 cycle); 94°C, 30s, and 55°C, 1 minute (40 cycles); 98°C, 10 minutes (1 cycle); held at 4°C. The analysis of positive sample concentrations was collated on the QuantaSoft software (Bio-Rad, USA).
Statistical analysis
The diagnostic accuracy of the ddPCR assay was examined using cfDNA and tumor tissue in NSCLC patients, in addition to blood samples from healthy volunteers. Analysis of variance (ANOVA) was performed for concentrations of cfDNA from different patient groups. Receiver operating characteristics (ROC) curves were used to analyze the cutoff for optimal positive discrimination of NSCLC from healthy controls. The Kaplan–Meier (KM) estimator was used in the survival analysis of patients in longitudinal follow-ups. Hazard ratios (HR) for each category were calculated using the COX regression model. All variables are represented as mean and standard deviation. All statistical analyses were performed with the aid of PRISM software (GraphPad Inc., USA).
Results
Clinical study design and establishing the optimal cfDNA cutoff
Given that KRAS mutations may have a direct impact on NSCLC prognosis, the current study investigated this patient cohort with positive mutations using cfDNA. The clinic-pathological characteristics of patients are shown in Table 1 together with their different metastatic pattern of the cohort. Accordingly, the most frequent sites of metastases for NSCLC patients were lung (45.3%), bones (28.0%), brain (17.3%), adrenal glands (16.4%), and liver (11.3%). Most patients had metastases affecting multiple organs. The median age of NSCLC patients was 57 years, and the gender ratio (male:female) was 1:1.82. For the healthy cases, median age and gender ratio was 53 years and 1.00:1.00 (male:female), respectively. A total of 186 patients and 30 healthy participants were enrolled after giving informed consent. The study design is shown in Figure 1(a), which addresses the diagnostic accuracy using cfDNA compared with tumor tissue and its prognostic utility for NSCLC patients.
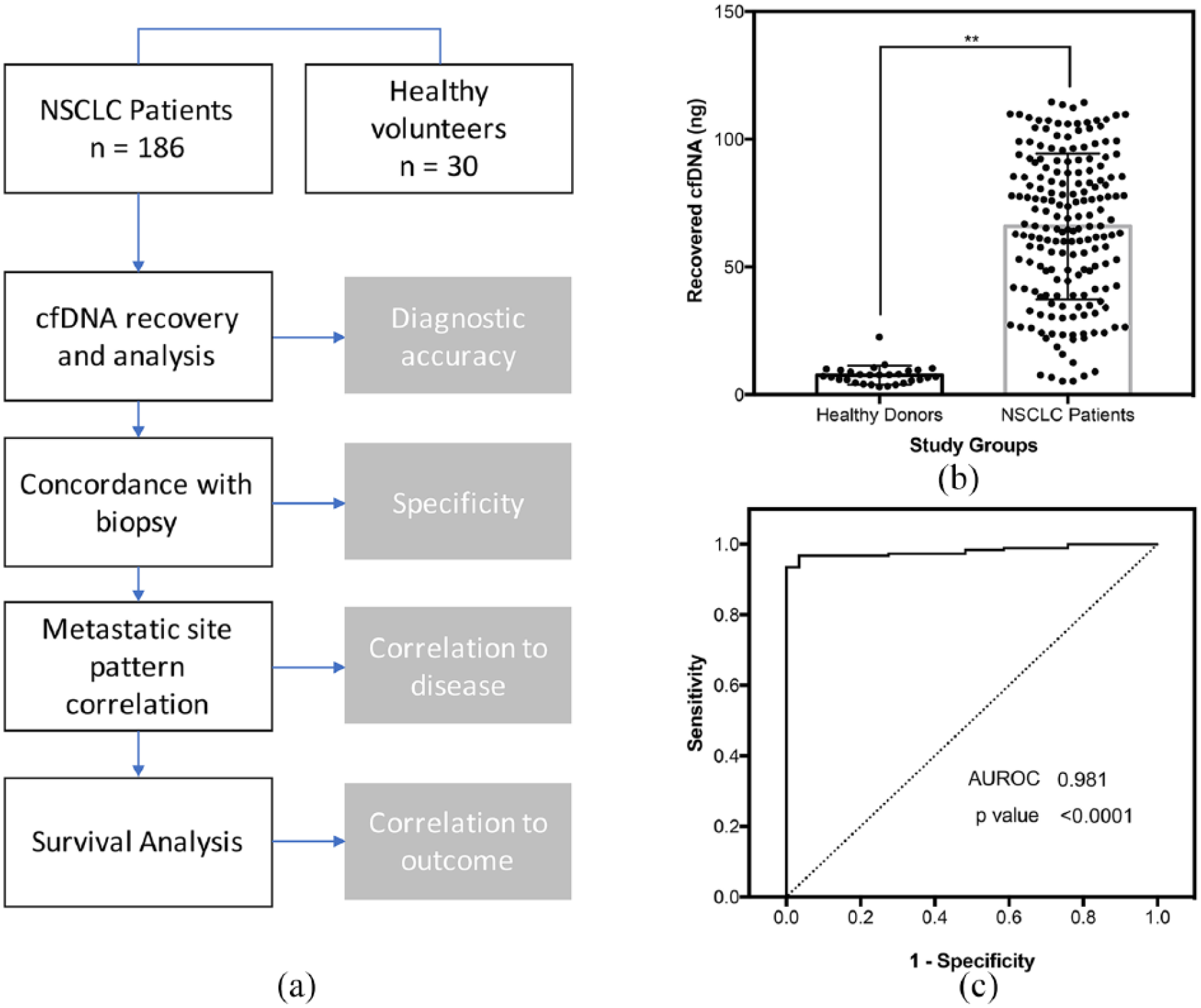
Study design and baselines cfDNA measurements. (a) Study characteristics for diagnostic accuracy analysis and correlation to survival outcome. (b) Comparison of recovered cfDNA from healthy donors and NSCLC patients show markedly higher mean values associated with disease. (c) ROC curve of healthy against NSCLC patients showing good clinical relevance in cfDNA measurements.
We examined the quantity of cfDNA extracted from NSCLC and healthy participants to ascertain the relevance of the potential marker. Mean cfDNA concentrations obtained from all 186 patients were markedly higher than the healthy controls (Figure 1(b), P < 0.001). The mean quantity recovered from NSCLC patients and healthy volunteers were 65.8 ng and 7.6 ng, respectively. This was 8.7-fold higher for cancer patients. We performed the ROC curve analysis using cfDNA concentrations derived from the trial participants as shown in Figure 1(c) (AUROC = 0.98). The optimal cutoff was 22.9 ng for cfDNA derived from plasma (sensitivity: 93.5%; specificity: 100%).
Concordance rates of cfDNA and tumor tissue, and links to NSCLC metastatic pattern
We next examined the cfDNA concordance rates using the KRAS mutation assay that utilized ddPCR. This technique has been previously reported to be sensitive for detecting these low frequency mutations in cfDNA. 20 Collectively, from the results of healthy volunteers, we examined the diagnostic accuracy for cfDNA in KRAS detection. Table 2 summarizes the results. A total of 150 patients of the 186 NSCLC cases examined had concordant tumor tissue and cfDNA. All 30 healthy volunteers were detected with wild type KRAS. The sensitivity and specificity of the test was 80.6% and 100%, respectively. Similarly, the positive predictive value was 100%. Overall, the results showed that cfDNA may be a good potential source of tumor tissue for the molecular detection of KRAS (Figure 2(a)).
Concordance measure of healthy donors and NSCLC patients for KRAS mutation.
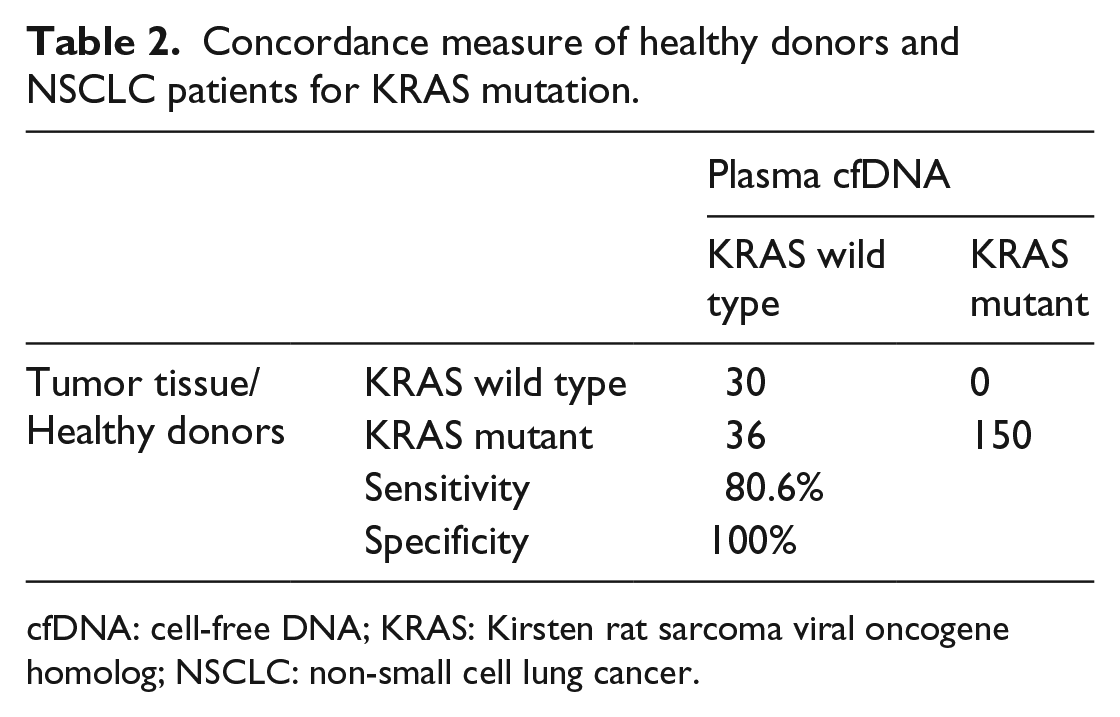
cfDNA: cell-free DNA; KRAS: Kirsten rat sarcoma viral oncogene homolog; NSCLC: non-small cell lung cancer.
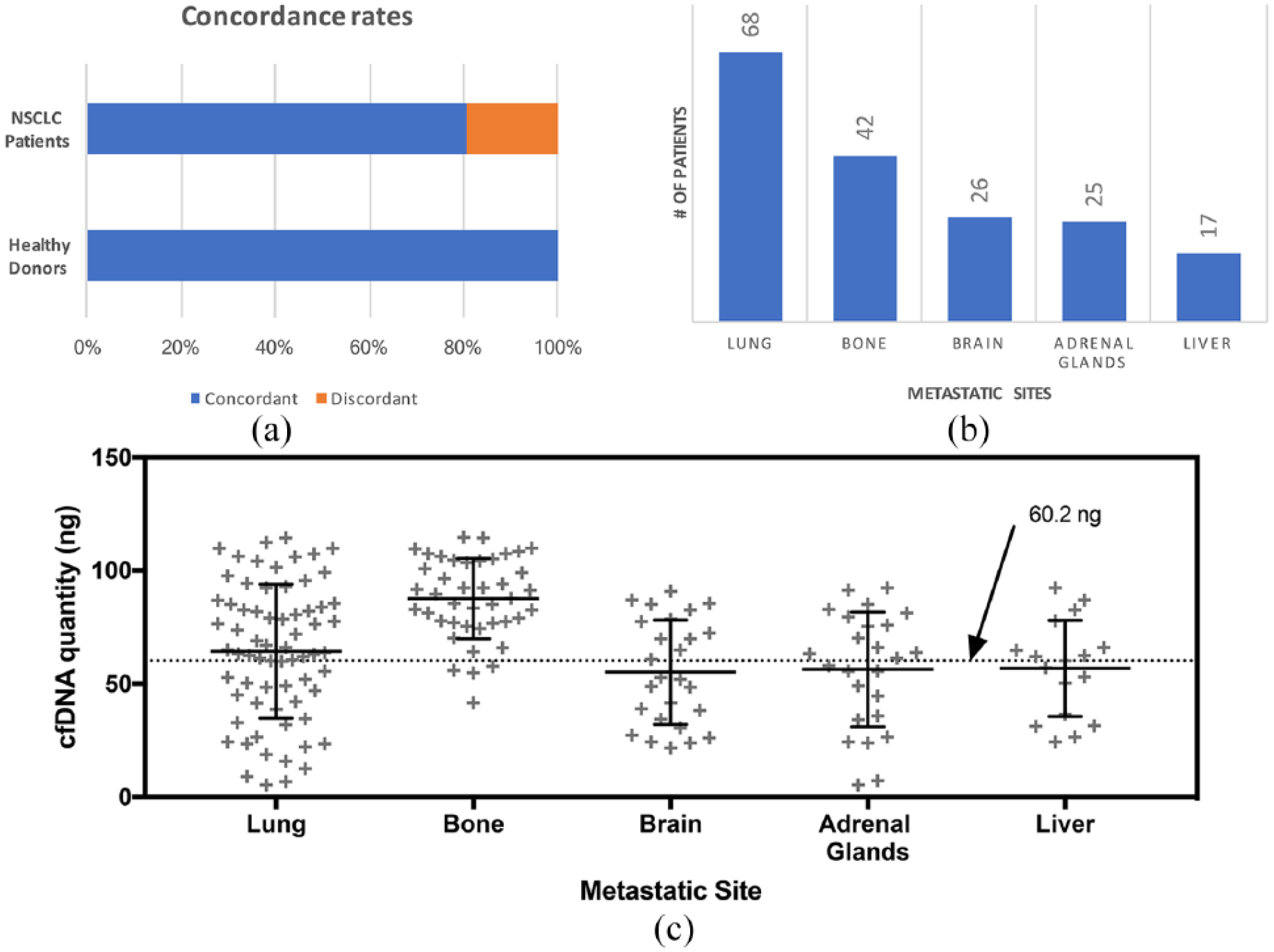
Direct correlation of cfDNA molecular profiling among different study groups. (a) KRAS mutation detection. NSCLC patients were compared with tumor tissue biopsies, and healthy volunteers were assumed to have wild type KRAS. (b) Metastatic pattern distribution among NSCLC patients that had concordant cfDNA results. (c) Distribution of cfDNA levels among NSCLC patients with different secondary metastasis.
Within the NSCLC patient cohort, we investigated the metastatic pattern and its link to cfDNA concentrations. The results were from the collective analysis of the 150 patients with concordant KRAS results (shown in Table 2). In the following investigation, we did not observe any clear major trends between the metastatic pattern of NSCLC patients and their corresponding recovered cfDNA. Interestingly, NSCLC patients that had bone metastases had higher mean recovered cfDNA concentrations compared with others (Figure 2(b)). This was similar true for KRAS mutation concentrations detected in these samples. An ANOVA among cases other than bone metastasis patients showed no statistical significance (P = 0.339). A direct comparison between patients with and without bone metastases showed a significant difference (P < 0.001) using a Student t test. Mean cfDNA quantities for patient groups with and without bone metastases were 87.6 ng and 60.2 ng, respectively, as shown in Figure 2(c).
Survival analysis links to bone metastases in NSCLC
We investigated the survival outcomes of these patients via a long-term follow-up based on the analysis using cfDNA. In the comparison with healthy volunteers, we noticed that the significantly higher cfDNA concentrations recovered were associated with NSCLC patients. As a result, we hypothesize that levels of NSCLC may be related to the disease severity. In the patient cohort, an equal split of the NSCLC patient group by ranking the recovered cfDNA quantities was conducted and analyzed as shown in Figure 3(a). Results indicated that patients with higher detected cfDNA had worse outcomes (HR 1.58; 95% confidence interval (CI) 1.13, 2.20). Median survival for this patient group was 2 months less. In a direct comparison with other risk factors, we observed that cfDNA counts had statistical significance in determining this patient group’s overall survival (supplementary Table 1). In the metastatic pattern analysis for cfDNA, patients with bone metastases were also observed with a higher quantity of recovered cfDNA. We investigated whether this affected the survival outcome for this subset of patients (Figure 3(b)). HR was determined at 1.71 (95% CI 1.13, 2.58) and median survival for patients with bone metastases was 6 months. The results indicated statistical significance over the non-bone metastases patient cohort (P < 0.001). As a comparison, we performed the same analysis for patients with lung metastases (Figure 3(c)), which has the highest incidence rates but lower mean cfDNA counts. The results showed a lowered HR of 0.81 (95% CI 0.59, 1.12; P = 0.14) compared with patients stratified by having bone metastases.
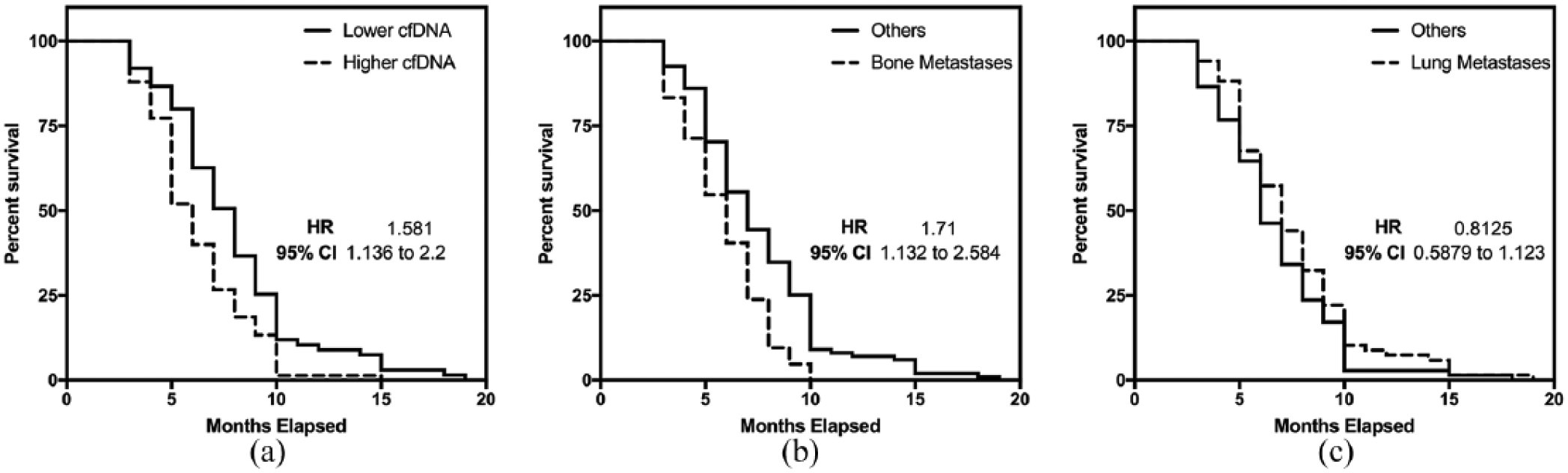
Survival analysis of different patient cohorts. (a) Comparison of survival outcomes of patients stratified using recovered cfDNA quantity (median split). (b) Comparison of survival outcomes with patients of bone metastases having worse outcome. (c) Comparison of patient outcomes with NSCLC patients with lung metastases.
Discussion
Clinical management and treatment of NSCLC patients with positive KRAS mutations remains challenging. 6 Immunotherapies present new options for these patients but remain critical in disease monitoring. 4 In this controlled population of 186 patient samples, we addressed the use of cfDNA to detect this molecular aberration and to provide prognostic utility. The technique provided ease for tumor material access compared with traditional invasive surgery and allowed early risk stratifications for patients with potentially worse outcomes. This enables us to probe the dynamics of the disease, which will hopefully lead to better clinical interventions in the future.
In line with previous research findings, we observed good concordance rates using cfDNA to detect KRAS mutations in these patients. Assay specificity with normal wild type volunteers showed 100% agreement, and diagnostic accuracy with positively identified patients via tumor biopsy samples was at 80.6%. In a similar study, Wang et al. 21 demonstrated 64% concordance with EGFR mutations for NSCLC patients using plasma cfDNA. Our study results showed significantly higher agreement, which was likely a result of different patient cohorts and monitored mutations. An interesting aspect demonstrated by Wang et al. 21 showed clinical significance in detecting the EGFR T790M mutation, a well-characterized drug-resistant mutation. This impact therapy selection for NSCLC patients and cfDNA allowed the constant monitoring of disease progression. 21 In our study of advanced stage NSCLC, we analyzed the metastatic patterns and their association with plasma cfDNA. It is interesting that patients linked with bone metastases had significantly higher cfDNA levels than other common metastatic sites, as shown in Figure 2. Patients with KRAS mutation and bone metastases are likely to have poor prognosis, 22 and our results showed that cfDNA potentially can have prognostic capabilities. The use of cfDNA in the clinical management of NSCLC is potentially advantageous. The specimen collection is non-invasive, and this is better received by patients compared with other invasive means such as tumor tissue biopsy. Computed tomography (CT) and magnetic resonance imaging (MRI), which are used to address disease progression are potentially useful as well, but the high cost and long waiting time prohibits their routine clinical use. In addition, the constant exposure to CT and MRI radiation can be potentially harmful for patients. We observed that cfDNA has good diagnostic accuracy for NSCLC patients given the relatively high sensitivity in a ROC analysis. Levels of cfDNA are also strongly elevated in cancer patients compared to controls that demonstrated the clinical significance of the potential marker.
Compared with imaging techniques, cfDNA offers capabilities for precise molecular profiling. We observed good concordance with KRAS mutation detection against the gold standard tested by tissue biopsy and alongside the metastatic patterns of advanced stage NSCLC. In addressing the prognostic potential with the marker, we performed a KM analysis on different population groups. We hypothesized that the level of cfDNA could be a good indicator, given its close association to the disease. Indeed, in a median split, we observed worse outcomes with the cohort of higher cfDNA levels, and median survival was 2 months less. This is consistent with existing literature that has examined cfDNA in NSCLC. Several studies23, 24 noted that high levels of cfDNA were associated with overall survival; Hu et al. 25 demonstrated that these can potentially correlate in relapse-free survival for patients on therapy. An interesting observation from our study showed that NSCLC with secondary bone metastases had higher cfDNA at the reference measurement. This was found to be 8.7-fold higher than other cohorts. The survival confirmed that patients in this category do have significantly poorer outcomes compared with their peers. Our study adds to the existing understanding of the use of cfDNA in clinical disease management, and this may allow quicker profiling of patients to better understand their risk and condition.
We envision that cfDNA will aid in current NSCLC clinical testing, given the ease of sample collection, and its use may expand in routine clinical monitoring. The main limitation of the study is that clinical interventional data and correlation are limited, which makes it difficult to understand the disease dynamics. Nonetheless, this study serves to realize the potential for better scrutiny of KRAS-mutation-positive patients, and, in our case, the variations in metastatic patterns for NSCLC.
Conclusion
The use of cfDNA in the clinical testing of NSCLC to understand variations across different metastatic profiles shows interesting trends in the current study. This method offers a relatively straightforward means for patient risk stratefication. Our study indicated that the patient group with bone metastasis has higher cfDNA levels, which are linked to poorer prognosis. The ability to risk stratify NSCLC patients early can potentially lead to better tailored therapy regimes. Further studies will be required to understand the cfDNA changes in the long term for these high-risk patients and the effects due to different treatments. Potentially, cfDNA offers an aid to complement current clinical management routines and has utility in the detection and prognosis of KRAS-positive NSCLC patients.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Use of cell free DNA as a prognostic biomarker in non-small cell lung cancer patients with bone metastasis
Supplemental material, Supplementary_Table_1 for Use of cell free DNA as a prognostic biomarker in non-small cell lung cancer patients with bone metastasis by Yongjian Ye, Zhihang Luo and Dejun Shi in The International Journal of Biological Markers
Footnotes
Author contributions
YY and ZL contributed equally and are co-first authors.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/ or publication of this article: This study was supported by a research grant provided by Yinzhou Second General Hospital (201700116).
Ethical approval
All human and animal studies have been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
