Abstract
Purpose:
Hepatitis B virus infection is a well-known risk factor for intrahepatic cholangiocarcinoma. However, its prognostic impact has rarely been investigated in advanced intrahepatic cholangiocarcinoma.
Methods:
Between April 2010 and May 2015, 296 patients with unresectable or metastatic intrahepatic cholangiocarcinoma who received gemcitabine plus cisplatin (GemCis) were categorized into a hepatitis B virus group (n=62; 21%) and a non-hepatitis B virus group (n=234; 79%). Clinicopathological features and survival outcomes were retrospectively reviewed and analyzed.
Results:
The median age of patients was 59 years (range, 27–78). The median overall survival with first-line GemCis was 9.4 months (95% CI 8.4, 10.4). Compared to the non-hepatitis B virus group, the hepatitis B virus group was younger (median age, 57 vs. 61 years, P = 0.001), mainly male (74% vs. 57%, P = 0.02), and had lower frequency of elevated cancer antigen (CA) 19-9 (34% vs. 59%, P = 0.001) and alkaline phosphatase (43% vs. 61%, P = 0.01). In a univariate analysis, the hepatitis B virus infection showed a marginal relationship with poor overall survival compared to the non-hepatitis B virus infection (median, 8.3 vs. 10.0 months; P=0.13). A multivariate analysis of potential prognostic factors revealed a significant association with poor overall survival in the hepatitis B virus group (hazard ratio (HR) =1.50, P = 0.02). Initial metastatic disease (vs. recurrent/unresectable disease; HR=1.50), metastatic sites ⩾ 2 (vs. 0–1; HR=1.51), Eastern Cooperative Oncology Group performance status ⩾ 2 (vs. 0–1; HR=1.93), elevated total bilirubin (vs. normal; HR=1.83), and low albumin (vs. normal; HR=1.52) were significantly related to an unfavorable overall survival.
Conclusions:
This study suggests that the hepatitis B virus infection may be associated with distinctive clinicopathological characteristics and poor outcome in advanced intrahepatic cholangiocarcinoma treated with GemCis.
Keywords
Introduction
Intrahepatic cholangiocarcinoma (ICC) is the second most common hepatic malignancy following hepatocellular carcinoma (HCC), and accounts for approximately 10% of all liver cancers. 1 Surgical resection remains the only curative treatment option for ICC. Most patients are unresectable at the time of diagnosis and tumor recurrence is common even following curative resection, resulting in a dismal prognosis with a median survival of less than 2 years. 2
To date, various risk factors of ICC have been identified, including hepatolithiasis, primary sclerosing cholangitis, primary biliary cirrhosis, choledochal cysts, and hepatic parasite infections. 3 Recently, hepatitis B virus (HBV)—a widely known cause of liver cirrhosis and HCC—has been recently found to be associated with the development of ICC in HBV epidemic areas.4,5 Although the mechanism of the development of ICC by chronic hepatitis B infection is unclear, patients with HBV-associated ICC have been reported to have different clinicopathological characteristics and survival outcomes compared to patients without HBV infection. However, such previous studies were mainly performed in patients with early resectable ICC. Because many ICC patients present with advanced disease at the time of initial diagnosis, and have a high recurrence rate even after curative resection, evaluating the impact of HBV infection on the clinical outcomes of patients with gemcitabine plus cisplatin (GemCis), the standard first-line chemotherapy for unresectable or metastatic cholangiocarcinoma, is important.6,7
In the present study, we retrospectively analyzed patients with advanced ICC who received first-line GemCis to compare clinicopathological features and clinical outcomes according to HBV infection.
Methods
Patients and clinical information
A total of 305 patients with histologically documented advanced ICC between April 2010 and May 2015 received GemCis as a first-line systemic treatment at the Asan Medical Center, Seoul, Korea. Among these, nine patients were excluded because of incomplete data for baseline liver function or HBV infection, and the remaining 296 (97%) were included in this study. The following patient data were retrospectively obtained from the medical records: patient demographics; tumor characteristics; Eastern Cooperative Oncology Group (ECOG) performance status; results of blood laboratory tests within 2 weeks prior to the initiation of chemotherapy, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), total bilirubin, albumin, prothrombin time and cancer antigen (CA) 19-9; HBV and HCV infection status; date of disease progression; and survival status at last follow-up. Tumor responses were assessed every 6 weeks using computed tomography or magnetic resonance imaging, and were graded according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The Institutional Review Board of the Asan Medical Center waived informed consent for this study because of its nature of retrospective analysis.
Evaluation of HBV and HCV infection
Hepatitis B serologic tests including hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (anti-HBs) were performed on all included patients before chemotherapy. Patients that were HBsAg positive were considered to have chronic HBV infection and had additional diagnostic tests to determine HBV DNA levels, hepatitis B e antigen (HBeAg), and hepatitis B e antibody (anti-HBe). Hepatitis B core immunoglobulin (Ig)M antibody levels to exclude acute hepatitis B infection were only measured if there was evidence of acute hepatitis. Liver cirrhosis was diagnosed and graded using abdominal imaging, laboratory test results, and clinical findings. As a baseline test, all patients were initially screened for hepatitis C virus (HCV) antibody test. To confirm, an HCV RNA quantification assay was followed in the patients who had the reactive HCV antibody.
Statistical analysis
To compare the differences in clinicopathologic factors between the HBV and the non-HBV groups, categorical variables were analyzed using the chi-square test or Fisher’s exact tests, as appropriate. Survival curves were estimated using the Kaplan–Meier method and compared by log-rank tests. Overall survival (OS) was defined as the length of time from the start of GemCis treatment to the date of death from any cause. Progression-free survival (PFS) was defined as the length of time from the start of GemCis treatment to the date of disease progression or death from any cause, whichever occurred first. Univariate and multivariate analyses for OS and PFS were analyzed using the Cox proportional hazards model. In the multivariate analysis, variables showing potential association with survival outcomes (P < 0.20) in the univariate analyses were included, and backward selection was used to eliminate non-significant variables based on the probability of the Wald statistic. A two-sided P value less than 0.05 was considered statistically significant. All statistical analyses were performed using the Statistical Package for the Social Sciences (IBM, Armonk, NY, USA) version 22.0.
Results
Patient characteristics
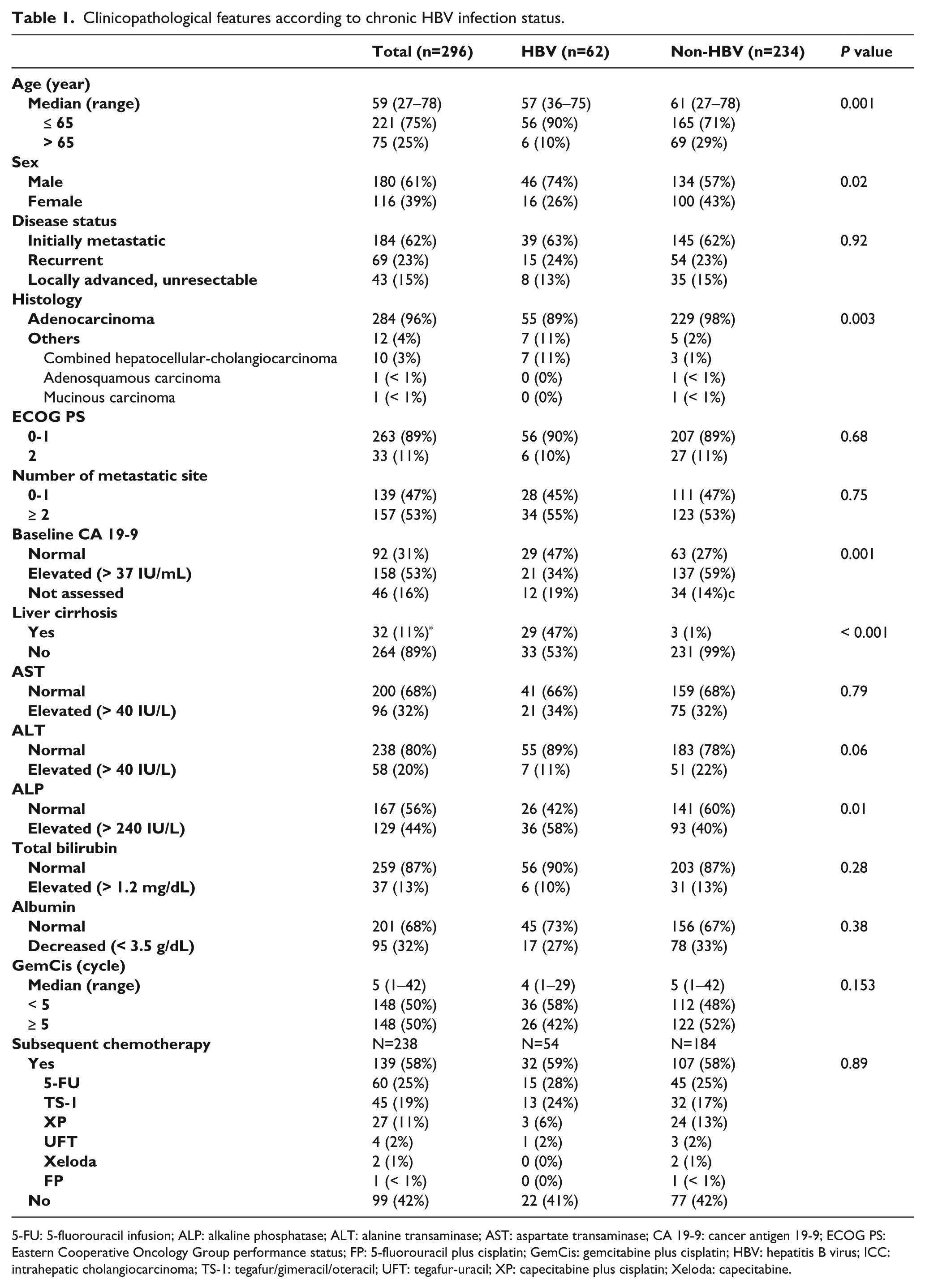
Baseline characteristics and clinical information of the study cohort (n = 296) are presented in Table 1. The median age was 59 years (range, 27–78), with males comprising 61% (n=180) of study participants. A total of 62 patients (21%) had chronic HBV infection. Of these, 27 had been already treated with antiviral agents; 15 patients recently commenced treatment with antiviral agents for high HBV DNA levels (> 2,000 IU/mL) indicating chronic active hepatitis B in an immune clearance phase or HBV reactivation; and 11 patients commenced treatment with antiviral agents as a prophylaxis. Liver cirrhosis was found in 47% of patients with HBV infection (n=29). Three patients had liver cirrhosis without HBV infection, with gall-stone-related cirrhosis in one patient and no identifiable cause in the remaining two patients. Among the 32 patients with liver cirrhosis, no patient was classified into Child–Pugh class C (class A, n=28; class B, n=4). Only one patient had HCV infection without accompanying HCV-related liver cirrhosis. Metastatic disease at presentation (n=184, 62%) was the most common disease status, followed by recurrent (69, 23%) and locally advanced unresectable disease (43, 15%). The most common histological type was adenocarcinoma (n=284, 96%) followed by combined hepatocellular-cholangiocarcinoma (10, 3%), adenosquamous carcinoma (1, < 1%), and mucinous adenocarcinoma (1, < 1%).
Clinicopathological features according to chronic HBV infection status.
5-FU: 5-fluorouracil infusion; ALP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate transaminase; CA 19-9: cancer antigen 19-9; ECOG PS: Eastern Cooperative Oncology Group performance status; FP: 5-fluorouracil plus cisplatin; GemCis: gemcitabine plus cisplatin; HBV: hepatitis B virus; ICC: intrahepatic cholangiocarcinoma; TS-1: tegafur/gimeracil/oteracil; UFT: tegafur-uracil; XP: capecitabine plus cisplatin; Xeloda: capecitabine.
Clinical outcomes of overall patients
Patients were treated with a median of 5 cycles (range, 1–42 cycles) of GemCis. With the median follow-up duration was 23.9 months, the median OS and PFS after first-line GemCis therapy were 9.4 months (95% CI 8.4, 10.4) and 5.1 months (95% CI 4.4, 5.8), respectively. Complete and partial responses were achieved in 1 (< 1%) and 35 (12%) patients, respectively, with an overall response rate (ORR) of 12%. The disease control rate, the proportion of patients with at least stable disease, was 68%. No patient experienced Child–Pugh class C hepatic impairment while receiving first-line GemCis chemotherapy. Subsequent second-line treatment was administered to 58% (139 of 238) of patients who progressed after GemCis therapy. The regimens are listed in Table 1.
Baseline characteristics according to HBV infection status
Table 1 also shows the differences in baseline clinicopathological characteristics of patients with ICC in the HBV and non-HBV groups. Compared to the non-HBV group, the HBV group was significantly younger (median age, 57 vs. 61 years, P = 0.001), predominantly male (74% vs. 57%, P = 0.02), had higher rates of HCC-combined or adenosquamous histology (11% vs. 2%, P = 0.003), and lower frequency of elevated CA 19-9 levels (34% vs. 59%, P = 0.001) and alkaline phosphatase (43% vs. 61%, P = 0.01). A significant association was found between the HBV group and liver cirrhosis (53% vs. 1%, P < 0.001); however, AST, ALT, total bilirubin, albumin, and prothrombin time (PT) levels did not differ between the HBV and the non-HBV groups. In addition, there was no difference in the proportion of patients who received subsequent second-line chemotherapy following disease progression in either group (59% vs. 58%, P = 0.89).
Univariate and multivariate analysis for survival outcomes
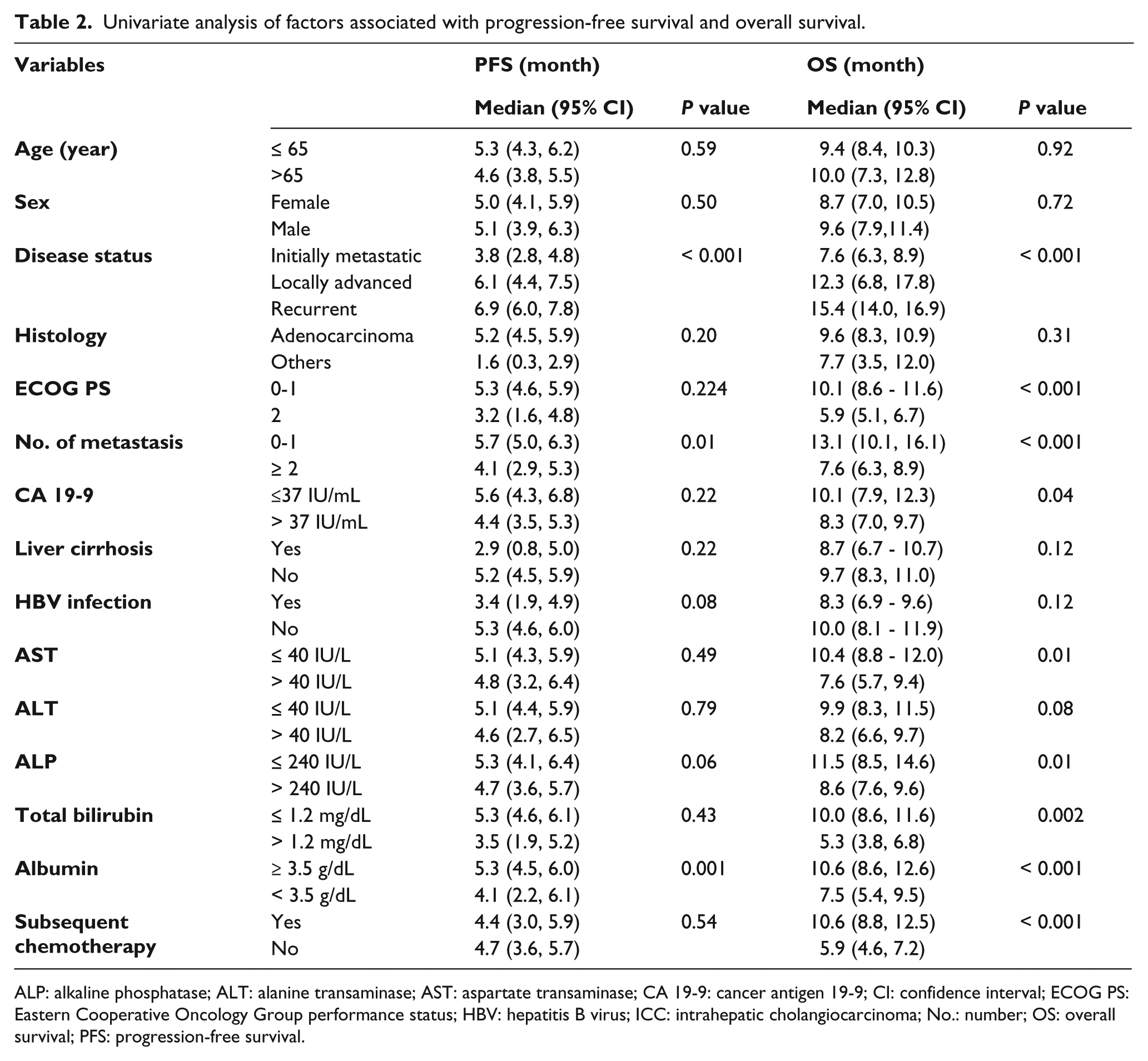
The results of univariate analyses for PFS and OS including potential prognostic factors are shown in Table 2. The HBV group showed a trend toward poorer survival outcomes compared to the non-HBV group (OS, median 8.3 (95% CI 6.9, 9.6) vs. 10.0 (95% CI 8.1, 11.9) months, P = 0.12; and PFS, 3.4 (95% CI 1.9, 4.9) vs. 5.3 (95% CI 4.6, 6.0) months, P = 0.08) (Figure 1). Within the HBV group, whether patients accompanied liver cirrhosis or not was not significantly related to PFS (with vs. without liver cirrhosis, median 2.9 (95% CI 0.8, 4.9) vs. 4.3 (95% CI 1.5, 7.1) months, P = 0.55) and OS (with vs. without liver cirrhosis, median 8.2 (95% CI 6.0, 10.5) vs. 8.7 (95% CI 6.6, 10.8) months, P = 0.48).
Univariate analysis of factors associated with progression-free survival and overall survival.
ALP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate transaminase; CA 19-9: cancer antigen 19-9; CI: confidence interval; ECOG PS: Eastern Cooperative Oncology Group performance status; HBV: hepatitis B virus; ICC: intrahepatic cholangiocarcinoma; No.: number; OS: overall survival; PFS: progression-free survival.
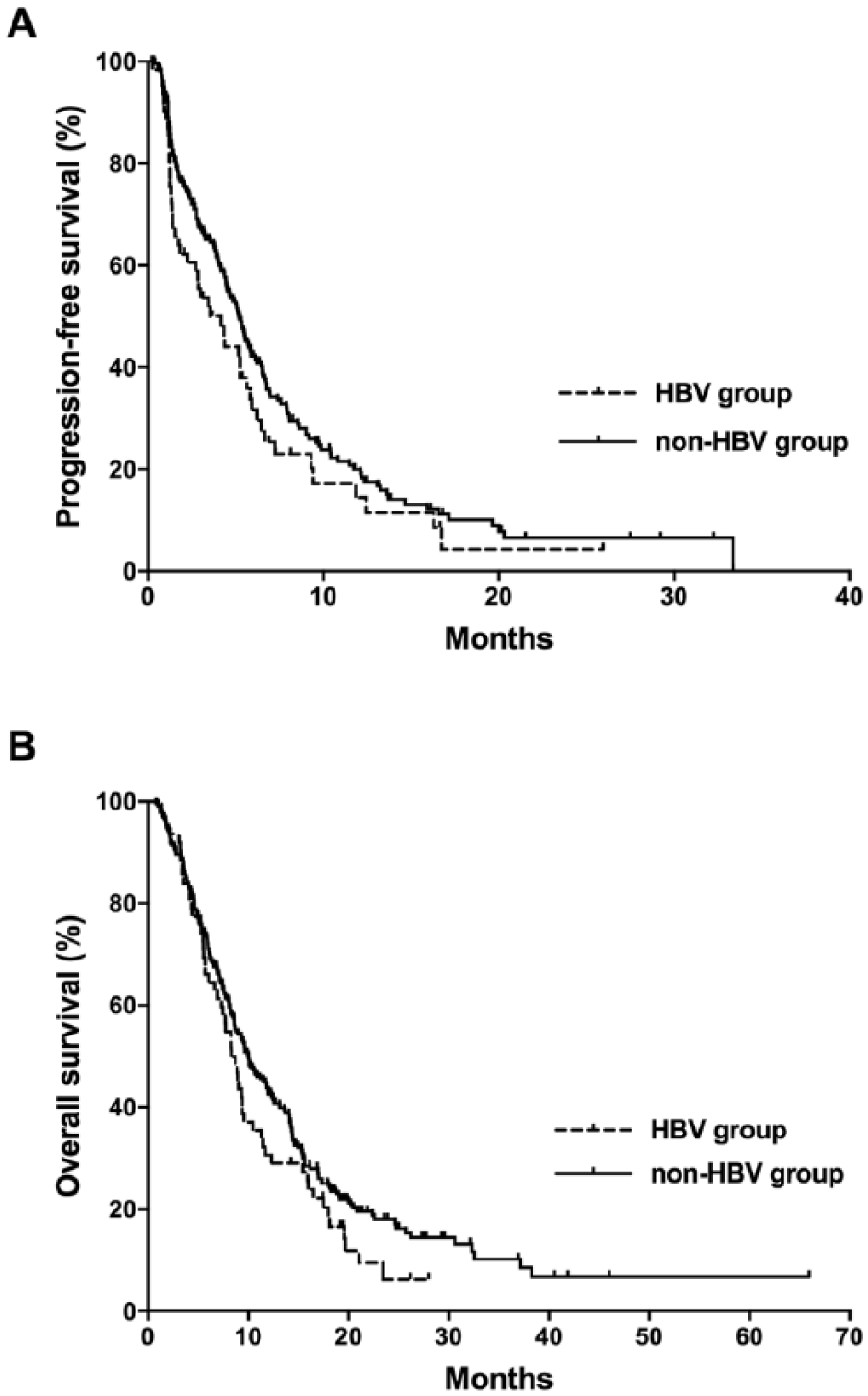
Comparison of progression-free survival outcomes between the HBV and non-HBV groups.
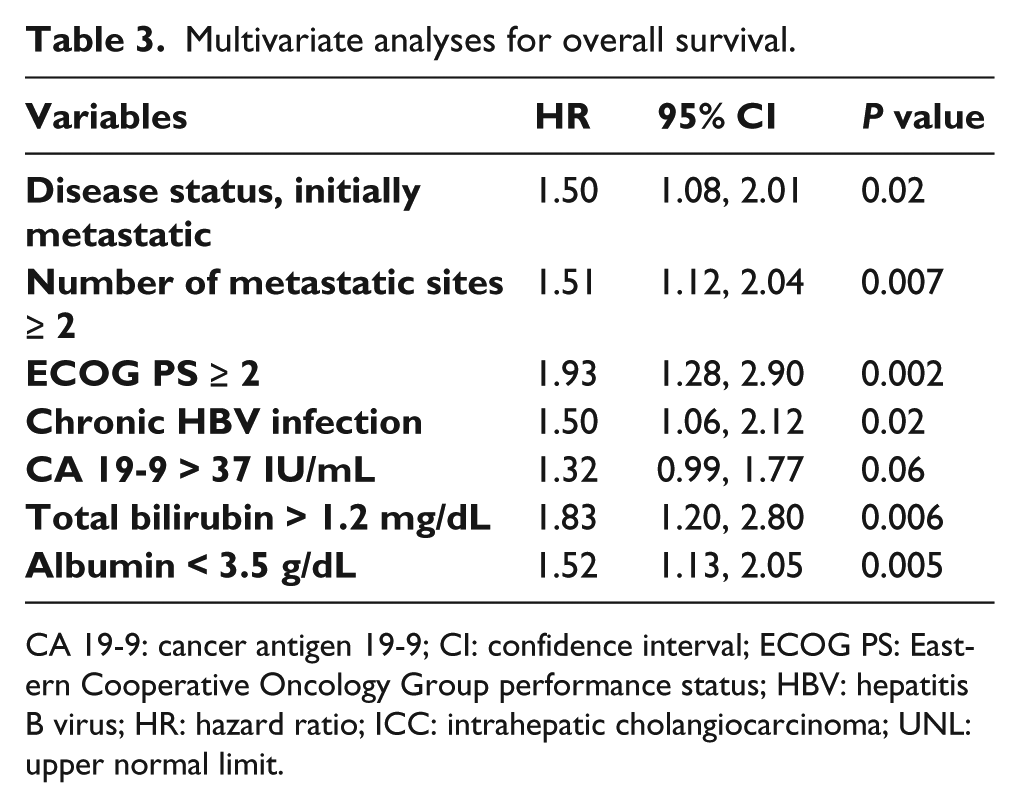
All predictors that were potentially significant in a univariate model with a P value less than 0.20 were included in the multivariate analysis. The multivariate analysis for OS was performed with covariates of disease status, ECOG performance status, number of metastasis, CA 19-9, liver cirrhosis, HBV infection, AST, ALT, ALP, total bilirubin, and albumin. Chronic HBV infection was significantly associated with poorer OS after the multivariate analysis (HR=1.50 (95% CI 1.08, 2.12), P =0.02; Table 3). In addition, metastatic disease at presentation, the number of metastatic sites ⩾ 2, poor ECOG performance status, high CA 19-9 levels, elevated total bilirubin levels, and low albumin levels were significantly associated with poorer OS. The other variables, such as liver cirrhosis, AST, ALT, and ALP, were eliminated after backward selection.
Multivariate analyses for overall survival.
CA 19-9: cancer antigen 19-9; CI: confidence interval; ECOG PS: Eastern Cooperative Oncology Group performance status; HBV: hepatitis B virus; HR: hazard ratio; ICC: intrahepatic cholangiocarcinoma; UNL: upper normal limit.
The multivariate analysis for PFS was carried out with covariates of disease status, histology, number of metastasis, HBV infection, ALP, and albumin. Patients with metastatic disease at presentation (vs. recurrent or locally advanced disease; HR 1.49; (95% CI 1.13, 1.96), P = 0.004) and low albumin levels (vs. normal; HR 1.46 (95% CI 1.10, 1.95), P = 0.01) showed significantly poorer outcomes. Chronic HBV infection did not remain significant in the multivariate model for PFS.
Discussion
Our results demonstrate that chronic HBV infection is an adverse prognostic factor for OS in patients with advanced ICC treated with first-line GemCis. In addition, patients with HBV-related ICC were found to have different clinicopathological features compared to patients with non-HBV-related ICC.
In the present study of 296 patients with metastatic or locally advanced unresectable ICCs who received the first-line GemCis chemotherapy, patients with chronic HBV infection were an independent prognostic factor for poor OS in the multivariate analysis, which adjusted the potential confounding effects of other clinicopathological factors. The HR for the non-HBV group was 1.50. Despite the marginal relationship in univariate analysis, this was not a significant factor in the multivariate analysis for PFS.
In terms of its prognostic relevance, the result of our analysis on patients with unresectable or metastatic ICC is in contrast with those of previous studies, which evaluated the prognostic significance of HBV infection in early resectable ICC.8-11 Although consistent results have not been demonstrated in all of these studies, a recent meta-analysis demonstrated that chronic HBV infection appears to be associated with better clinical outcomes after surgery in early resectable ICC. 12 These previous studies have suggested that regular imaging surveillance for the detection of HCC in patients with chronic HBV infection may help detect early ICC, which may be associated with favorable post-resection outcomes. These studies also showed that tumors with less frequent periductal invasion and lymph node metastasis in patients with HBV infection may result in better survival outcomes in resectable ICC.
In contrast to the results observed in resectable ICC, our current study demonstrated that HBV-related ICC in patients treated with GemCis with a locally advanced or metastatic setting was associated with poor survival. This demonstrates that HBV-related ICC may have distinctive clinicopathological and molecular characteristics, which may impact sensitivity to chemotherapy. This is supported by results from a previous study in patients with ICC using high throughput molecular profiling, with different mutation patterns according to the presence of chronic liver disease. 13 In this study, which included 38 ICC patients with chronic advanced liver diseases (29 with HBV infection) and 43 with normal livers, the patients with ICC and chronic advanced liver disease showed higher EGFR mutation rates (13.2% vs. 2.3%) and lower mutation rates for KRAS (7.9% vs. 18.6%), MLH1 (5.3% vs. 11.6%), and GNAS (2.6% vs. 11.6%) compared to patients with ICC that had a normal liver. In our study, patients with advanced HBV-related ICC were younger, more often male, and had a higher prevalence of liver cirrhosis and a lower frequency of elevated serum CA 19-9, which is consistent with results from previous studies on early resectable ICC.10-14 The gross morphology of ICC has been reported to differ according to predisposing factors; viral hepatitis-associated ICCs seemed to have mass-forming type arising from smaller bile ducts or liver progenitor cells rather than the periductal infiltrating or intraductal growth type originating from the epithelial cells of larger bile ducts.15,16 These clinical and genetic findings suggest that HBV-related ICCs are distinguishable from ICCs with other etiologies. However, there is currently insufficient biological evidence to draw a conclusion that such differences make result disparities in tumor responses to GemCis. Liver decompensation associated with chronic HBV infection may also potentially contribute to a poorer outcome. However, baseline liver function did not differ between the HBV and the non-HBV groups, and no patient developed liver decompensation of Child–Pugh class C during the first chemotherapy treatment. Also, whether a patient accompanied liver cirrhosis or not did not affect both PFS and OS within the HBV group, which indicates that liver dysfunction is unlikely to be the main factor in survival difference.
The strength of the present study is the evaluation of the prognostic role of chronic HBV infection among ICC patients with metastatic or locally advanced unresectable disease, which has been rarely investigated in contrast to early ICCs. However, our study had several limitations. First, it was performed retrospectively and there may be an inherent selection bias in the study population. Second, several factors related to OS, including chronic HBV infection, were not significantly associated with PFS, which is partially the result of the small number of patients with HBV infection to attain statistical significance.
In conclusion, our study indicates that chronic HBV infection is associated with distinctive clinical features and an unfavorable OS in patients with advanced ICCs treated with first-line GemCis. ICC is a heterogeneous disease and further translational research should aim to determine genetic, molecular, and phenotypical differences between HBV-associated and non-HBV-associated ICC and how these differences influence the response to anti-tumor treatment.
Footnotes
Authors’ note
These data were presented at the annual meeting of the Korean Association for Clinical Oncology held in Seoul on 19 May 2017.
Author contributions
Heejung Chae and Hyungwoo Cho contributed equally to this work and should be considered co-first authors.
Changhoon Yoo and Baek-Yeol Ryoo contributed equally to this work and should be considered co-corresponding authors.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Bio and Medical Technology Development Program of the National Research Foundation funded by the Korean government, MSIP (NRF-2016M3A9E8941331) and a grant from the Asan Institute for Life Sciences at the Asan Medical Center in Seoul, Korea (2017-728).
Informed consent
The Institutional Review Board of the Asan Medical Center waived consent for this study because of its nature of retrospective analysis.
