Abstract
Objective:
Aurora kinase B (Aurora-B) is a crucial regulator of accurate mitosis. Abnormal Aurora-B expression is associated with aneuploidy and has been implicated in the pathogenesis and drug resistance in a variety of human cancers. However, little evidence is available regarding the role of Aurora-B in regulating drug response in non-small cell lung cancer (NSCLC), which is the most common type of lung cancer, and is characterized with poor prognosis and high mortality.
Method:
In the current study, we investigated the association of Aurora-B with the prognosis of NSCLC patients, and we also used the latest CRISPR/Cas9 system to explore the regulatory role of Aurora-B in NSCLC cells developing resistance to cisplatin (CDDP) and paclitaxel.
Results:
We found that Aurora-B was correlated with significantly reduced overall survival and disease-free survival in NSCLC patients. Aurora-B overexpression was also observed in NSCLC cells developing impaired response to both CDDP and paclitaxel. Moreover, we found, for the first time, that Aurora-B may impair NSCLC drug response by disturbing cell proliferation and inhibiting p53-related DNA damage response and apoptotic pathway, while the knockout of Aurora-B resensitized NSCLC cells to chemo drugs by ensuring correct chromosome segregation and restoring p53 expression.
Conclusions:
Our results demonstrated the association of Aurora-B with chemoresistance in NSCLC, which may finally contribute to the poor prognosis of NSCLC patients. We also suggested Aurora-B as a promising therapeutic target in NSCLC treatment.
Introduction
Lung cancer has emerged as the leading cause of cancer-related mortality worldwide. Every year, more than 25% of cancer patients die from lung cancer (1). Non-small cell lung cancer (NSCLC) is the most common subtype of lung cancer, accounting for about 85% of cases. Of the NSCLC patients, the majority are diagnosed at an advanced stage, which makes chemotherapy the first therapeutic option (2). Although many targeted therapeutic strategies have been recently developed and approved, most of them are only applicable to a small group of NSCLC patients with specific genetic patterns; therefore chemotherapy remains the mainstay of NSCLC treatment. While chemo drugs are capable of inducing cytotoxicity and causing cancer cell death, NSCLC cells are often intrinsically resistant to chemo drugs, which exacerbates the prognosis of NSCLC patients and finally results in a high mortality rate.
During the past decade, much effort has been made to understand the mechanism underlying chemoresistance in NSCLC. An ATP-binding cassette (ABC) transporter superfamily has been revealed as a major regulator of chemoresistance by enhancing drug efflux and decreasing intracellular anticancer drug accumulation (3). Besides, the activation of DNA-repair pathways can also offset the effect of chemotherapeutic agents causing DNA damage and impairing drug response (4). Moreover, abnormal alterations of the antioxidant (e.g. glutathione-S-transferases, superoxide dismutase), cytoskeletal proteins (e.g. tubulin III), and intracellular apoptotic and antiapoptotic proteins (e.g. Bcl-2, Bax, and SAPK/JNK), are also implicated in developing resistance to various chemo drugs in NSCLC (5).
All of this previous evidence expanded our knowledge on drug resistance in NSCLC and implied the complexity of chemoresistance. Thus, a comprehensive understanding of different mechanisms of chemoresistance is of great importance in developing novel therapeutic strategies to restore drug sensitivity and finally overcome lung cancer. Aurora kinase B (Aurora-B) is a key regulator of mitosis by attaching kinetochores to the walls of microtubules and ensuring correct chromosome segregation and spindle assembly (6-8). Aurora-B overexpression has been frequently identified in lung cancer patients (9), and our previous study has demonstrated the potency of Aurora-B silencing in inhibiting NSCLC cell growth (10). Recently, Aurora-B has also been found to contribute to drug resistance in a variety of malignancies, including breast cancer, colorectal cancer, liver cancer, and mesothelioma (11-13), whereas little evidence is available regarding the association of Aurora-B with drug response in NSCLC. In the current study, we therefore explored the association of Aurora-B with the progression and chemoresistance of NSCLC, and we tested if knockout of Aurora-B through the CRISPR/Cas9 system is capable of resensitizing NSCLC cells to chemo drugs.
Methods
Tissue collection
Lung tissue samples were collected from 192 NSCLC patients undergoing surgical tumor resection at the Department of Respiratory Medicine, the First Affiliated Hospital of Nanchang University between December 2006 and June 2011. The study was performed in accordance with the 1964 Helsinki Declaration of World Medical Association. Informed consent was obtained from all patients prior to the study. All procedures were carried out with the approval of the local Ethics Committee at the First Affiliated Hospital of Nanchang University.
Drug resistant assay
1 × 104 NSCLC cells were seeded into 96-well plate and left to adhere for six hours. Chemo drugs, cisplatin (CDDP) and paclitaxel (PTX), were then added to cell culture at serial concentrations (0~100uM) and incubated for 72 hours. Cell proliferation was then determined by the MTT cell proliferation assay kit (Thermo Fisher Scientific). The experiment was performed with five replications.
Induction of chemoresistance in NSCLC cell
A549 cells resistant to CDDP (A549/CDDP) and PTX (A549/PTX) were established, respectively, as previously reported (14). Briefly, the original parental cell line was continuously exposed to chemo drugs at initial IC50 concentrations determined by drug resistance assay. After 72 hours incubation, drugs were withdrawn to allow cells to recover for another 72 hours, and drugs were then supplemented to cell culture again at the same concentration. This process was repeated 20 times, and new IC50 concentrations were determined, which were used to treat cell culture for another 20 cycles.
Real time polymerase chain reaction
Trizol reagent (Invitrogen) was used to extract total RNA from NSCLC tissues and cell lines (A549, A549/CDDP, A549/PTX). Total RNA was further reverse-transcribed to cDNA using first-strand cDNA synthesis system, amplified with SYBR Green PCR kit (Invitrogen) and analyzed by LightCycler 480 instrument (Roche). Polymerase chain reaction (PCR) primers used in the experiments include: Aurora-B: Forward: 5’-AGAAGGAGAACTCCTACCCCT-3’ Reverse: 5’-CGCGTTAAGATGTCGGGTG-3’ GAPDH: Forward: 5’-GTGGACATCCGCAAAGAC-3’ Reverse: 5’-GAAAGGGTGTAACGCAACT-3’
CRISPR/Cas9 gene editing
A549/CDDP and A549/PTX cells were grown in PRMI-1640 (HyClone) supplemented with 10% fetal bovin serum (Gibco) and incubated at 37°C in a humidified atmosphere with 5% CO2. sgRNA (GCGCCCTCCCTGAGGTATGG) targeting Aurora-B was annealed to complementary oligo, cloned into pSpCas9(BB)-2A-GFP (PX458) plasmid (Addgene ID 48138), and transfected into NSCLC cells with Lipofectamine 3000 (Thermo Fisher Scientific). After 24 hours incubation, green fluorescent protein (GFP)-positive single cells were sorted by flow cytometer and grown into small colonies in 96-well plates.
Apoptosis assay
Wild type A549/CDDP and A549/PTX cells as well as corresponding Aurora-B knockout cells were harvested after 72 hours exposure to CDDP and PTX. After trypsinization, the cell population was double stained with Annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI) and were analyzed on BD FACScalibur (BD Biosciences). Each experiment was performed with three replicates.
Cell cycle distribution assay
Wild type A549/CDDP and A549/PTX cells as well as corresponding Aurora-B knockout cells were harvested and trypsinized. After being fixed with in ice-cold 70% ethanol overnight at 4°C, the cells were treated with RNase and stained with solution containing 50 ug/mL PI. Cell population was analyzed on BD FACScalibur (BD Biosciences). The experiment was performed with three replications.
Western blot analysis
Cell lysates were harvest with the RIPA cell lysis buffer. A total of 20 ug of protein extracts from each sample were separated in 10% SDS-PAGE and transferred onto nitrocellulose membrane. The membrane was incubated with antibodies against Aurora-B, p53, p21, BAX, and tubulin. Protein bands were detected with the ECL Western Blotting Detection Kit (Amersham).
Results
Aurora-B overexpression is associated with poor prognosis of NSCLC patients
We used real-time PCR to measure Aurora-B mRNA levels in tumor tissues collected from 192 NSCLC patients undergoing surgical resection (Tab. SI). Patients with Aurora-B levels above average were considered to have high Aurora-B expression, while the rest were low Aurora-B patients. Compared with low Aurora-B group, NSCLC patients with high Aurora-B levels had poorer prognosis with significantly lower overall survival (Fig. 1A, log-rank test: P = 4.26 × 10−4) and disease-free survival (Fig. 1B, log-rank test: P = 1.71 × 10−4).
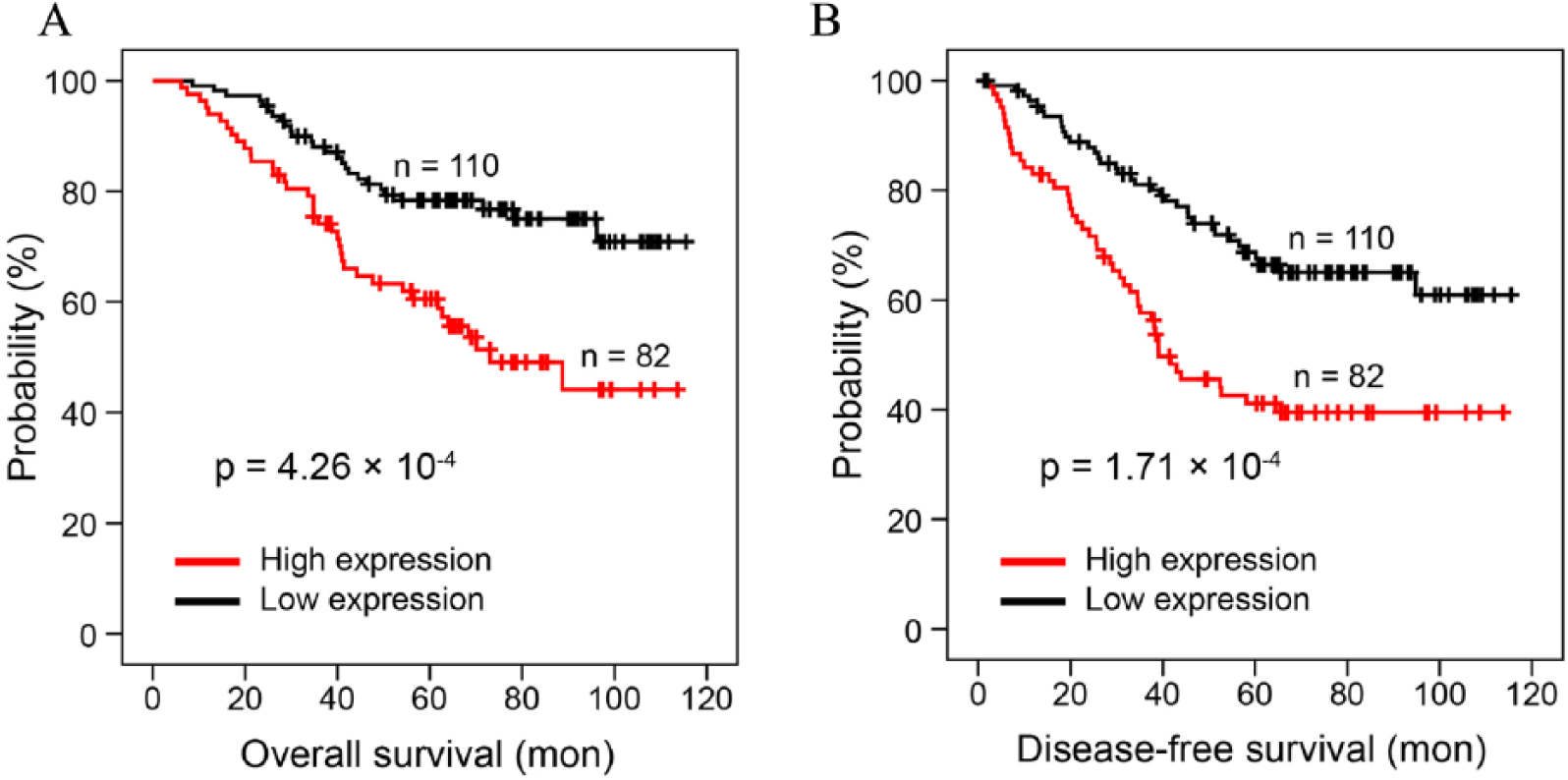
Kaplan–Meier analysis of the overall survival A) and disease-free survival B) between NSCLC patients with high and low Aurora-B expression.
Aurora-B is involved in regulating the cell cycle process, and increased cell proliferation has been frequently implicated in NSCLC chemoresistance and relapse. It is possible that Aurora-B overexpression might be caused by increased proliferation in cancer tissue. We therefore adjusted the influence of the cell cycle with the Cox regression model. As shown in Table I, in a univariate analysis, Aurora-B as well as cell cycle regulators (Cyclin A and Cyclin E) were all significantly associated with NSCLC prognosis. However, after controlling the confounding effects, increased Aurora-B expression (Tab. I, P = 0.018; relative risk (RR): 1.951; 95% confidence interval (CI) 1.120, 3.399) and higher disease stage (Tab. II, P = 0.001; RR: 1.558; 95% CI 1.193, 2.034) remained as the major determinants of decreased disease-free survival. Decreased disease-free survival implied increased resistance to chemo drugs and the failure of chemotherapy. Our results suggested the association of Aurora-B with chemoresistance in NSCLC patients.
Cox regression model
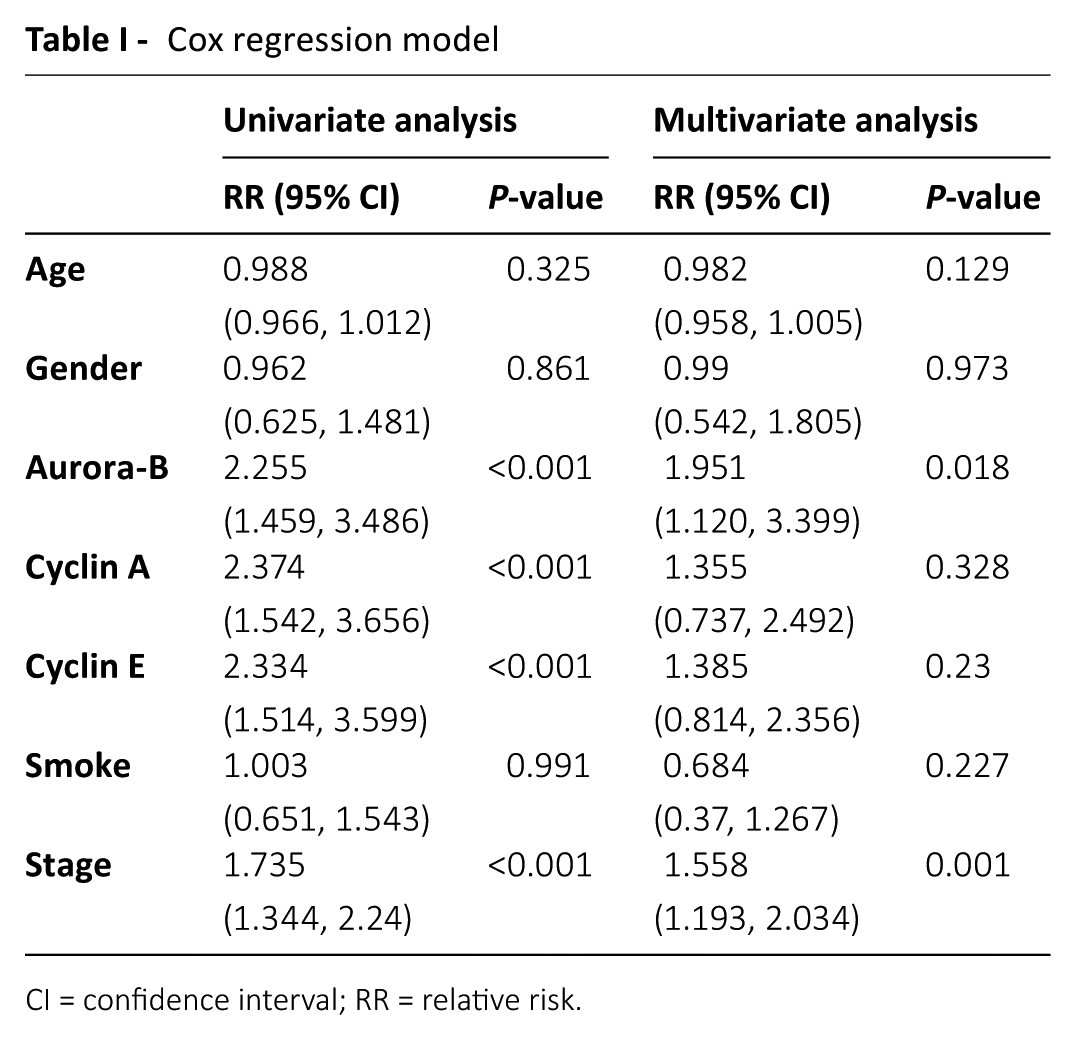
CI = confidence interval; RR = relative risk.
Aurora-B overexpression is associated with increased drug resistance in NSCLC cells
To understand the role of Aurora-B in chemoresistance, we generated chemoresistant NSCLC cell line by continuously exposing A549 to CDDP and PTX, which are widely used chemotherapeutic agents in NSCLC treatment (15). As shown in Figure 2A, CDDP- and PTX-resistant NSCLC cell (A549/CDDP, A549/PTX) displayed significantly higher IC50 concentrations than parental cells after 72 hours’ incubation. (Fig. 2A: CDDP: 48.44 ± 5.05 uM vs. 18.33 ± 2.71 uM, P = 2.52 × 10−6; PTX: 63.57 ± 7.64 uM vs. 46.56 ± 5.34 uM, P = 0.0035). Notably, the expression level of Aurora-B mRNA was also significantly increased in both CDDP- and PTX-resistant cells (Fig. 2B: CDDP: P = 6.58 × 10−5; PTX: P = 0.0001), indicating the association of Aurora-B with NSCLC chemoresistance. In addition, cyclin A and cyclin E were also overexpressed in both A549/CDDP and A549/PTX cells, suggesting the activation of cell proliferation during the development of drug resistance in NSCLC.
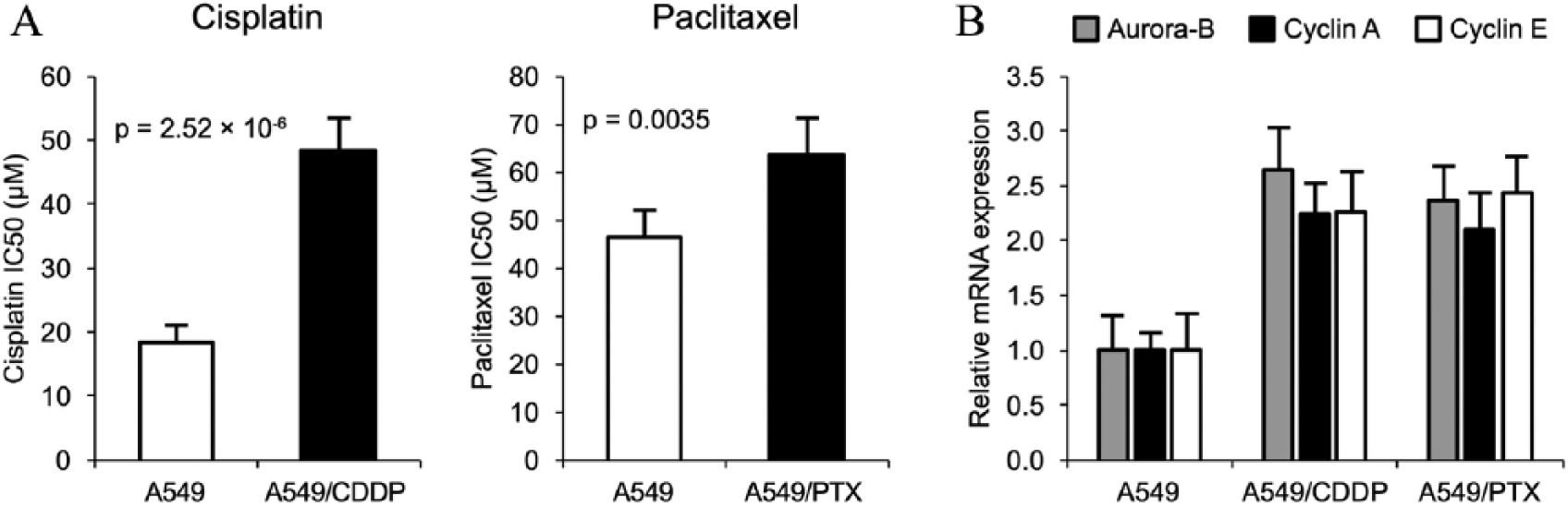
A) IC50 concentration for cisplatin (left) and paclitaxel (right) on NSCLC A549 cells with and without resistance to the corresponding drugs. B) Aurora-B, cyclin A, and cyclin E expression in parental A549, A549/CDDP, and A549/PTX cells.
Aurora-B knockout resensitizes NSCLC cells to chemo drugs
Since increased Aurora-B was associated with impaired CDDP and PTX response, we therefore asked if inhibiting Aurora-B expression could resensitize NSCLC to chemo drugs. To this end, we used CRISPR/Cas9 system to knockout Aurora-B from A549/CDDP and A549/PTX. As shown in Fig. 3A and 3B, Aurora-B expression was successfully depleted from both A549/CDDP and A549/PTX cells. More importantly, as revealed by MTT cell proliferation assay, the proliferation of Aurora-B knockout A549/CDDP and A549/PTX cells was significantly reduced by increasing the drug concentrations, while wide type A549/CDDP and A549/PTX remained resistant to chemotherapy (Fig. 3C and 3D). Moreover, fluorescent activated cell sorting (FACS) analysis demonstrated that, on the treatment of chemo drugs at IC50 concentration determined from Figure 2A (CDDP: 48.44 uM; PTX: 63.57 uM), Aurora-B knockout resulted in significant apoptosis in both A549/CDDP and A549/PTX cells. (Fig. 3E, apoptotic A549/CDDP cells: 1.24 ± 0.37% vs. 12.26 ± 2.54%, P = 0.0017; Fig. 3F, apoptotic A549/PTX cells: 1.29 ± 0.79% vs. 12.01 ± 2.22%, P = 0.0014). These results suggest the potency of Aurora-B as a promising target to overcome chemoresistance in NSCLC therapy.
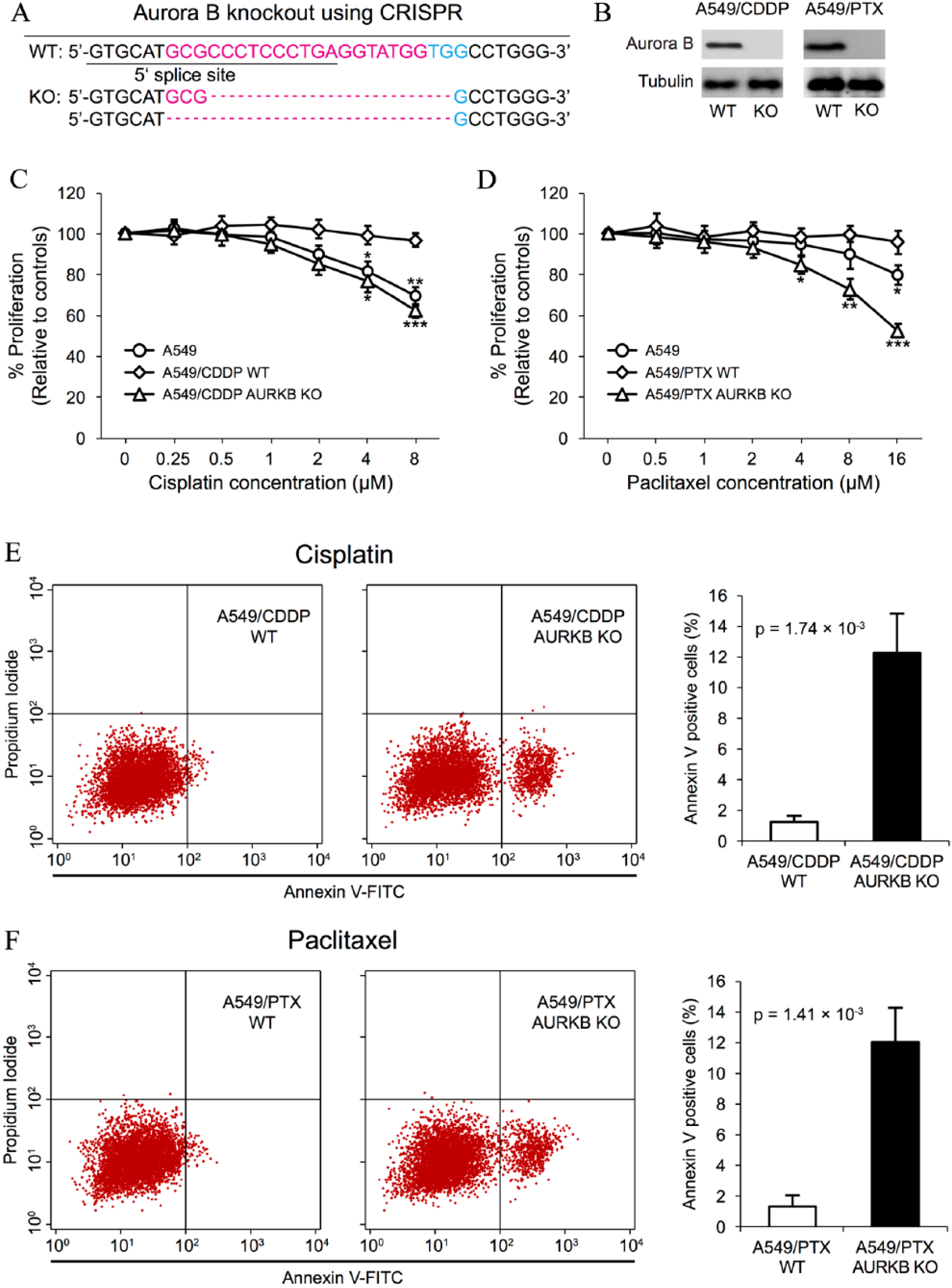
A) Aurora-B was knockout from A549/CDDP and A549/PTX cells using CRISPR/Cas9 system and B) Aurora-B expression was assessed in wide type (WT) and knockout (KO) cells using Western blot. Cisplatin C) and paclitaxel D) at different dilutions were used to treat parental A549 cells, WT and Aurora-B KO A549 cells with resistance to corresponding drugs. Cell proliferation was assessed using MTT method. * P < 0.05, ** P < 0.01, *** P < 0.001. Cisplatin E) and paclitaxel F) at IC50 concentration were used, respectively, to treat WT and Aurora-B KO A549 cells with resistance to corresponding drugs. Apoptotic cells were detected by flow cytometer.
Aurora-B overexpression disturbs cell proliferation and inhibits p53-related pathway
Aurora-B is a crucial cell cycle regulator involved in chromosome condensation, chromosome alignment, and spindle assembly checkpoint (6). Aurora-B overexpression has been reported to cause abnormal cell cycle progression and the generation of aneuploidy (7). As shown in Figure 4A-D, a large 4N peak and more cells with DNA contents > 4N were observed in both A549/CDDP and A549/PTX cells, while Aurora-B depletion brought the cell cycle back to normal profiles, implying a regulatory role of Aurora-B in the proliferation of NSCLC cells. Notably, we also found that Aurora-B knockout was accompanied with increased expression of p53 and its downstream transcriptional targets p21 and BAX, suggesting that Aurora-B may disturb cell proliferation through p53-related pathway (Fig. 4E).
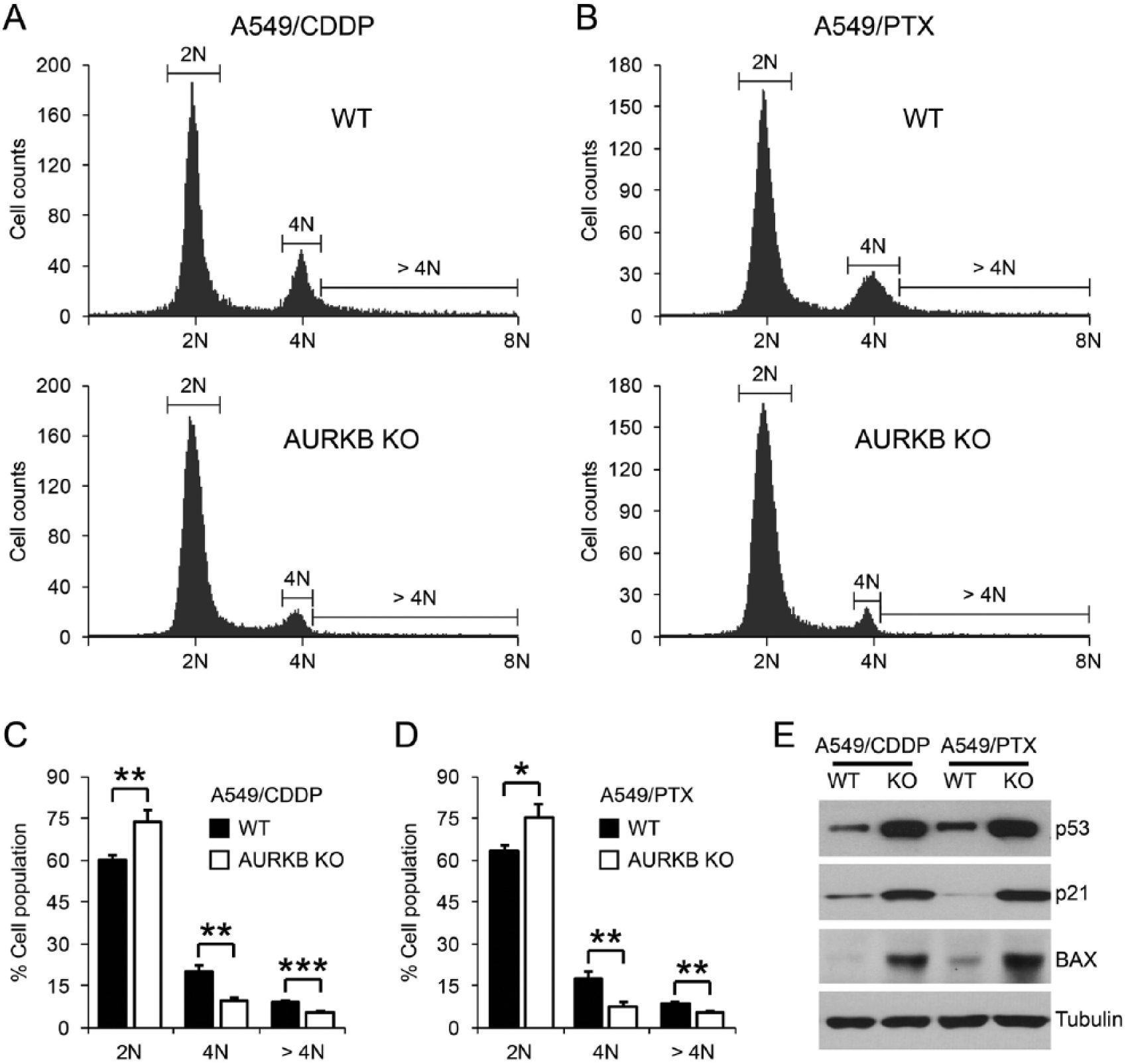
Accumulation of aneuploidy cell in both A549/CDDP A,B) and A549/PTX (B,D), while normal cell cycle distribution was observed in Aurora-B knockout (KO) cells. * P < 0.05, ** P < 0.01, *** P < 0.001. E) Western blotting analysis revealed that Aurora-B overexpression is correlated with reduced expression of p53, p21, and BAX.
Discussion
Aurora-B has been accepted as an oncogene by inducing abnormal cell division and the subsequent aneuploidization (6). Elevated Aurora-B expression is frequently observed in various cancer types and has been characterized as a regulator of chemoresistance (7, 9-13), which is a common and major problem in cancer therapy. In the current study, we found that Aurora-B overexpression is also involved in drug resistance in NSCLC and is associated with the poor prognosis of NSCLC patients. We also provide evidence that elevated Aurora-B expression may disturb the cell proliferation process (Fig. 4A-D). In normal conditions, the deregulated cell cycle process triggers the activation of p53 as well as the downstream repair and apoptotic pathways to clear the cells in abnormal proliferative status (16). However, in the context of Aurora-B overexpression, a large part of p53 is subjected to ubiquitin-mediated protein degradation, which further decreases the expression of p21 and BAX (17). BAX is a proapoptotic factor and p21 is a responder of DNA damage by arresting the cell cycle at the G1/S phase and activating apoptotic cascades (18, 19). The absence of p21 and BAX enables the cells with DNA damage and abnormal chromosome number to bypass cell cycle checkpoints and escape damage repair, which finally impairs the efficacy of the drugs targeting cancer cell DNA and the cell cycle, and leads to drug resistance. Moreover, the accumulation of cells with DNA defects further aggravate the disease and finally contribute to treatment failure.
With the advances of cancer research, many regulatory pathways have been revealed to modulate NSCLC drug response, and numerous candidates have been proposed as predictive markers and promising therapeutic targets (3-5). However, we are still far from fully understanding the complicated drug resistant mechanism, and it is still essential to developing more therapeutic strategies to finally overcome NSCLC. In this study, we proposed for the first time that Aurora-B-related p53 signaling pathway is involved in drug response in NSCLC. We also provided evidence that the knockout of Aurora-B expression with the latest CRISPR gene-editing technology is capable of restoring p53-related DNA damage response and apoptotic pathway, and helps to resensitize NSCLC cells to chemo drugs (Figs. 3 and 4) (20).
Recently, several inhibitors targeting Aurora-B have been developed and some of them have already been approved for clinical usage. In contrast to CRISPR gene knockout, these inhibitors aim at reducing Aurora-B expression and deactivating Aurora-B activity (21). A comparison between CRISPR technology and Aurora-B inhibitors is still being undertaken to evaluate the effectiveness of different strategies. If it is necessary, we will modify our current CRISPR strategy to establish a more robust system targeting Aurora-B. Moreover, in the current study, we only explored the role of Aurora-B in regulating resistance to a small group of drugs: cisplatin and paclitaxel. Considering the many chemo drugs on the market and the fact that more drugs are under investigation, we still need to do more work to understand the interaction between Aurora-B and many other agents. Also, as mentioned above, chemo drug response involves a complex regulatory network and the p53 pathway is insufficient to fully understand Aurora-B-related regulatory network. Also, in addition to Aurora-B, many other candidates remain to be characterized to generate a comprehensive view of NSCLC and to develop novel strategies to finally cure lung cancer.
Supplemental Material
Table_S1 – Supplemental material for High expression of Aurora-B is correlated with poor prognosis and drug resistance in non-small cell lung cancer
Supplemental material, Table_S1 for High expression of Aurora-B is correlated with poor prognosis and drug resistance in non-small cell lung cancer by JingJing Yu, Jing Zhou, Fei Xu, Wei Bai and Wei Zhang in The International Journal of Biological Markers
Footnotes
Disclosures
Financial support: No financial support.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
