Abstract
Kawasaki disease is a common systemic vasculitis in children, and early treatment is critical to prevent coronary artery complications. This study aimed to evaluate the impact of different aspirin dosages on the prognosis of Kawasaki disease. A total of 161 children diagnosed with Kawasaki disease were enrolled and divided into four groups based on the dosage of aspirin: 30–34 mg/kg, 35–39 mg/kg, 40–44 mg/kg, and 45–49 mg/kg. Demographic characteristics, pre-treatment laboratory indicators, post-treatment laboratory indicators, the incidence of coronary artery complications, and the rate of IVIG resistance were compared across the groups. No significant differences were found in baseline characteristics such as age, gender, weight, IVIG treatment choice, IVIG treatment duration, or the proportion of patients diagnosed before the age of 1 year. Laboratory indicators before and after treatment were also similar between groups. However, at 3-4 weeks after the onset of illness, the incidence of coronary artery complications was significantly higher in the high-dose aspirin groups compared to the low-dose aspirin group. Additionally, the high-dose aspirin group had a longer duration of fever and a higher proportion of recurrent fever compared to the low-dose group. The results suggest that although aspirin dosage did not appear to have a clear impact on the overall prognosis of Kawasaki disease, high-dose aspirin administered in the 3-4 weeks following disease onset may increase the risk of coronary artery complications and is associated with a higher incidence of recurrent fever. Caution is therefore advised in selecting aspirin dosages to minimize unnecessary risks.
Keywords
Introduction
Kawasaki disease (KD) is a prevalent systemic vasculitis condition predominantly affecting children, with an escalating incidence observed in recent years.1,2 Based on the latest epidemiological data, the global prevalence of KD has exceeded approximately 30-40 cases per 100,000 children. 3 KD primarily affects children under the age of 5, and if left untreated, it can give rise to various complications including coronary artery aneurysms (CAA), thrombosis, and even sudden death.4,5 KD is recognized as the leading cause of acquired heart disease.6,7 The incidence of coronary artery lesions (CAL) in untreated KD children worldwide is approximately 15%-20%, whereas in KD children receiving standardized treatment, the incidence is reduced to about 3%-5%.8,9 Consequently, it is of paramount importance to explore alternative therapeutic approaches to enhance the prognosis of affected children.
Although clear guidelines for KD treatment exist, certain issues pertaining to drug therapy, such as dosage uncertainty, necessitate further research to improve treatment outcomes. The treatment objective for KD is to “terminate the acute phase inflammation as early as possible and minimize the occurrence of coronary artery lesions.” Among these, aspirin has been established as the first-line treatment for KD.10–13Aspirin, also known as acetylsalicylic acid, is a nonsteroidal anti-inflammatory drug (NSAID) with a multifaceted mechanism of action.14,15 It exerts its pharmacological effects by inhibiting the conversion of arachidonic acid, suppressing the release of inflammatory mediators such as histamine, and exhibiting analgesic, anti-inflammatory, and antipyretic properties.16,17 Additionally, aspirin inhibits platelet aggregation by targeting the binding site of platelets and cyclooxygenase, thereby preventing the formation of blood clots. 18 In the context of Kawasaki disease (KD), aspirin primarily demonstrates anti-inflammatory effects during the acute phase and antiplatelet effects during the subacute phase. 19 However, the optimal initial dosage of aspirin for KD treatment remains a subject of debate. Accurate determination of the aspirin dosage during the acute phase is crucial for developing more tailored treatment strategies to enhance KD outcomes.
Presently, there are conflicting recommendations regarding the use and dosage of aspirin across different countries. In North America, the American Heart Association (AHA) guidelines advocate for high-dose aspirin administration (80-100 mg/kg/day) during the acute phase of KD. 10 Conversely, Asian countries, following the Japanese guidelines, widely employ moderate-dose aspirin (30-50 mg/kg/day). 20 Although the clinical practice typically calculates the initial dosage of aspirin within the range of 30–50 mg/kg/day, controversy still surrounds this range. Consequently, the initial aspirin dosage for KD children remains in a state of confusion, lacking standardized regulation.
Several studies have investigated the optimal dosage of aspirin (ASP) in the management of Kawasaki disease (KD). However, the findings from these studies have been inconsistent in recent years. A multicenter, randomized, single-blind trial conducted in Taiwan demonstrated no significant difference in short-term coronary artery lesions between high-dose and low-dose aspirin during the acute phase of KD. 21 Similarly, Huang et al. reported that both low-dose and moderate-dose aspirin had no impact on coronary artery damage in KD. 22 Furthermore, Migally et al. observed that the duration of high-dose aspirin administration did not correlate with the long-term prognosis of coronary arteries. 23 These studies collectively suggest that the prognosis of KD may not significantly differ with the use of high, moderate, or low-dose aspirin. However, contrasting conclusions have also been reported. Dhanrajani et al. found that the administration of low-dose aspirin was associated with an increased risk of intravenous immunoglobulin (IVIG) resistance. 24 Kim et al reported that the administration of moderate to high-dose aspirin, after adjusting for various confounding factors, was associated with a higher incidence of coronary artery aneurysms compared to low-dose aspirin. 25 These findings suggest that the dosage of aspirin plays a crucial role in the prevention of coronary artery lesions. However, the optimal dosage of aspirin remains a subject of debate, and the potential side effects associated with changes in aspirin dosage have not been thoroughly investigated in previous studies. Therefore, further investigation is warranted to determine the optimal initial dosage of aspirin.
Methods and materials
Study population and inclusion/exclusion criteria
This retrospective study included 161 children diagnosed with Kawasaki disease who received different dosages of aspirin treatment at a single medical center from January 2020 to December 2021. Inclusion criteria were as follows 10 : (1) age less than 12 years, with all children admitted to our hospital for Kawasaki disease treatment; (2) fulfillment of the diagnostic criteria for the disease as outlined in “Practical Pediatrics” by Chu Futang; (3) initial treatment with intravenous immunoglobulin (IVIG) (2 g/kg/d) and oral aspirin (ASP); (4) voluntary participation of parents after being informed about the study's purpose; (5) availability of complete data and successful completion of the study. Exclusion criteria were as follows: (1) children with known drug allergies; (2) children with organ dysfunction or other severe diseases identified during pre-study examination; (3) children who had previously received similar drug treatments; (4) children who withdrew from the study midway.
Study methods
A comprehensive assessment of the baseline condition and physical status of all study subjects was conducted. Targeted anti-infective treatment, maintenance of electrolyte balance, and provision of nutritional support were implemented. Intravenous administration of IVIG (2 g/kg/d) from Chengdu Rongsheng Pharmaceutical Co., Ltd was performed. ASP (Bayer HealthCare Manufacturing S.r.l) was administered at initial doses of 30–35 mg/kg/d, 35–40 mg/kg/d, 40–45 mg/kg/d, and 45–49 mg/kg/d in three divided doses. The dose was gradually reduced by 5 mg/kg every 2 days, starting 3–5 days after defervescence, until a maintenance dose of 3-5 mg/kg/d in three divided doses was reached. All four groups received continuous treatment for 6 weeks.
Collection of patient study parameters
Pre- and post-treatment, various laboratory parameters were collected for all four patient groups, including white blood cell count (WBC), platelet count (PLT), C-reactive protein (CRP), D-dimer, prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT), hemoglobin (HB), neutrophil count (ANC), erythrocyte sedimentation rate (ESR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, fibrinogen (FIB), neutrophil percentage (Neutrophils), and sodium ion concentration (Na+).
Criteria for intravenous immunoglobulin (IVIG) resistance
IVIG is a primary treatment method for Kawasaki disease. However, some patients exhibit poor response to IVIG treatment, leading to IVIG resistance. The criteria for determining IVIG resistance are as follows 26 : 1. Persistent fever: Patients continue to experience fever of 38°C or higher within 48 h after receiving IVIG treatment. 2. Persistent or recurrent rash: Patients develop persistent or recurrent rash within 48 h after receiving IVIG treatment. 3. Persistent or recurrent conjunctivitis: Patients experience persistent or recurrent conjunctivitis within 48 h after receiving IVIG treatment. 4. Cardiac involvement: Patients develop new coronary artery lesions, myocarditis, pericarditis, or other cardiac abnormalities within 48 h after receiving IVIG treatment. Based on these criteria, patients meeting one or more of them are classified as IVIG resistant.
Criteria for coronary artery damage
Coronary artery damage is a significant complication of Kawasaki disease, often resulting in the development of coronary artery aneurysms, thrombosis, and life-threatening situations. The criteria used to assess coronary artery damage include the following 27 : 1. Coronary angiography: Coronary angiography is employed to evaluate the presence of abnormal conditions such as stenosis, dilation, or aneurysm formation in the coronary arteries. 2. Echocardiography: Echocardiography is utilized to examine coronary artery blood flow and identify any abnormalities, such as coronary artery aneurysms. 3. Electrocardiogram: Electrocardiogram is employed to detect any abnormal changes, such as ST segment alterations or T wave inversion. By employing these criteria, clinicians can comprehensively evaluate the extent of coronary artery damage in patients and determine appropriate treatment strategies.
Statistical analysis
Statistical analysis was conducted using SPSS software (version 23). Descriptive statistics were employed to analyze laboratory parameter data before and after treatment. Intergroup comparisons were performed using t-tests or analysis of variance (ANOVA). Chi-square tests were utilized to compare the incidence of coronary artery complications and IVIG resistance. Logistic regression analysis was employed to predict the relationship between the incidence of coronary artery complications, IVIG resistance, and the proportion of recurrent fever with aspirin dosage. The analysis was adjusted for factors such as age, weight, gender, IVIG treatment regimen, IVIG treatment duration, hemoglobin level, albumin level, and fibrinogen level, among other relevant clinical information.
Results
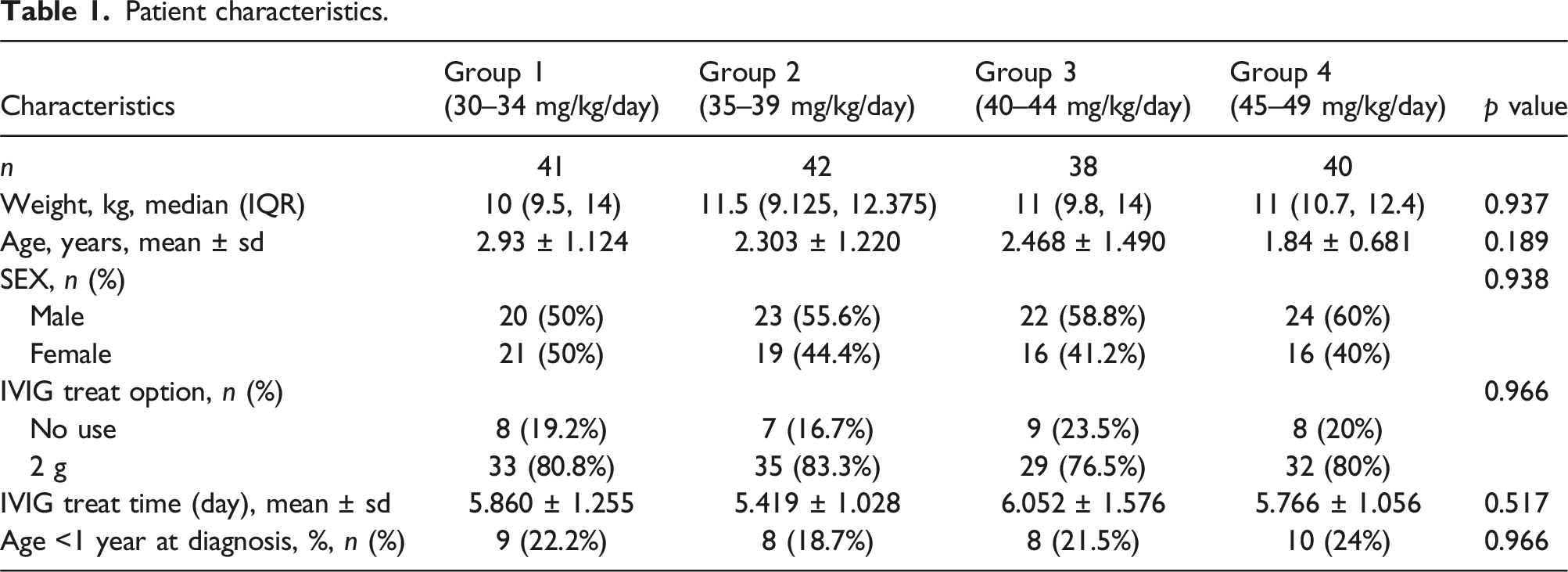
Demographic characteristics of the study population
Patient characteristics.
Comparison of pre-treatment laboratory parameters
Pre-treatment laboratory investigations.

Comparison of laboratory parameters after treatment
Laboratory examinations after treatment.

Comparison of the incidence of coronary artery complications and IVIG resistance among four groups of patients
Comparison of treatment and outcomes.

Logistic regression analysis for predicting the incidence of coronary artery complications and IVIG resistance
Multiple regression analyses.

Interestingly, we did not observe a significant correlation between aspirin dosage and the incidence of coronary artery complications 2 weeks after onset, even after adjusting for factors such as age, weight, gender, IVIG treatment selection, and IVIG treatment duration. However, between three to 4 weeks after onset, we found a significant correlation between aspirin dosage and the incidence of coronary artery complications. We conducted analyses with and without adjusting for the same relevant factors, and the results showed no correlation between the risk of IVIG resistance and the dosage of aspirin used in combination therapy. However, multivariate logistic regression analysis revealed that receiving a high dosage of aspirin had a predictive effect on the proportion of recurrent fever.
Discussion
Kawasaki disease (KD) is a prevalent systemic vasculitis primarily affecting pediatric patients. 28 Timely diagnosis and appropriate treatment are paramount in effectively managing this condition. Currently, the internationally recognized treatment approach involves the combined administration of intravenous immunoglobulin (IVIG) and aspirin. 29 IVIG treatment has been extensively studied and proven to effectively alleviate symptoms and reduce the incidence of coronary artery damage, thus playing a pivotal role in KD management. 30 The significance of IVIG treatment lies in its multifaceted effects, including its anti-inflammatory, anti-platelet aggregation, and immunomodulatory actions. 31 By suppressing inflammatory reactions, IVIG can ameliorate systemic symptoms and cardiac damage in affected children. Moreover, IVIG can inhibit platelet aggregation, mitigating the risk of thrombosis and subsequently reducing the occurrence of coronary artery damage. 32 Therefore, IVIG treatment holds immense importance, particularly in the early stages of KD. Coronary artery damage represents a major complication of KD and can lead to severe consequences such as coronary artery aneurysm and thrombosis. Consequently, the prevention and treatment of coronary artery damage are pivotal in the management of KD. It is worth noting that the use of aspirin in KD treatment predates the adoption of IVIG. 33 Previous studies have demonstrated the beneficial effects of high-dose aspirin in combination with intravenous immunoglobulin (IVIG) in preventing coronary artery damage in Kawasaki disease. 27 However, recent research has suggested that the extent of coronary artery damage is only negatively correlated with the dosage of IVIG and is unrelated to aspirin treatment. 34 The relationship between aspirin dosage and coronary artery damage in Kawasaki disease remains uncertain. The American Heart Association (AHA) guidelines recommend the use of high-dose aspirin (80-100 mg/kg/day) during the acute phase of Kawasaki disease in North America. 10 In contrast, the Japanese guidelines advocate for the widespread use of moderate-dose aspirin (30-50 mg/kg/day) in Asian regions. 20 Therefore, this study aims to investigate the impact of different initial aspirin dosages, specifically within the range of 30-50 mg/kg, as determined by consensus, on children with Kawasaki disease. To ensure comparability among the groups, no significant differences were observed in terms of gender, age, and duration of IVIG treatment in this study.
In recent years, numerous studies have been conducted to investigate the optimal dosage of aspirin for treating Kawasaki disease (KD). These investigations have challenged the current recommended dosage as being unreasonable. Notably, it has been demonstrated that there is no discernible difference in the effectiveness of moderate and high-dose aspirin in the treatment of KD. 35 Moreover, the incidence of coronary artery damage among KD patients remains unaffected by varying aspirin dosages.22,36 Similarly, the duration of fever experienced by KD patients shows no correlation with different aspirin dosages.37,38 Furthermore, the long-term prognosis of coronary arteries in KD patients is not influenced by the dosage of aspirin administered. 23 Collectively, these findings suggest that high, moderate, and low dosages of aspirin do not impact the overall prognosis of KD.
However, it is worth noting that contradictory conclusions have also been reported by certain clinical researchers. Some studies have indicated an association between different aspirin dosages and the prevention of coronary artery aneurysms, with low-dose aspirin being prone to inducing IVIG resistance. 24 Additionally, moderate to high-dose aspirin has been identified as a risk factor for coronary artery aneurysms. 25 Remarkably, these findings are consistent with the results of our own research. Through logical analysis and adjustment for essential clinical factors such as age, weight, gender, IVIG treatment selection and duration, as well as hemoglobin levels, our study reveals significant differences in the risk of coronary artery aneurysms and recurrence of fever among the four groups. This indicates a positive correlation between aspirin dosage and the incidence of coronary artery aneurysms and recurrence of fever. Furthermore, our investigation finds no significant variation in the proportion of IVIG resistance among KD children receiving different aspirin dosages.
Aspirin, a nonsteroidal anti-inflammatory drug (NSAID), functions primarily by inhibiting prostaglandin synthesis through the suppression of prostaglandin H synthase (PGHS). Kawasaki disease, a systemic vasculitis inflammatory condition, is mitigated by the inhibition of prostaglandin synthesis by aspirin, thus reducing inflammatory response and platelet aggregation, thereby exhibiting anti-inflammatory and anti-platelet properties. Furthermore, aspirin ameliorates symptoms such as fever and joint pain in Kawasaki disease patients, ultimately enhancing their quality of life. The potential mechanism underlying the prolonged fever duration in Kawasaki disease patients receiving high-dose aspirin may be attributed to its impact on the thermoregulatory center. 39 By inhibiting prostaglandin synthesis, aspirin may disrupt the function of the thermoregulatory center, resulting in thermoregulatory abnormalities and prolonged fever in Kawasaki disease patients. The heightened recurrence of fever in Kawasaki disease patients associated with high-dose aspirin could stem from its inadequate therapeutic efficacy. High-dose aspirin might not adequately suppress inflammatory response and platelet aggregation, leading to uncontrolled disease progression and recurrent fever. The elevated incidence of coronary artery damage in Kawasaki disease patients administered high-dose aspirin may be linked to its suboptimal inhibition of platelet aggregation. Kawasaki disease, being a vasculitis inflammatory ailment, encompasses platelet aggregation as one of its pathogenic mechanisms. Ineffectual inhibition of platelet aggregation by high-dose aspirin could exacerbate coronary artery damage in Kawasaki disease patients. Our research findings suggest that the lack of effect of high-dose aspirin on white blood cell count (WBC), platelet count (PLT), C-reactive protein (CRP), D-dimers, prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT), hemoglobin (HB), neutrophil count (ANC), erythrocyte sedimentation rate (ESR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, fibrinogen (FIB), neutrophil percentage (Neutrophils), and sodium ion concentration (Na+) may be attributed to the minimal impact of aspirin on these parameters or the insufficiency of aspirin dosage to elicit significant effects.
This study has several limitations, including a relatively small sample size, a retrospective study design, and a lack of long-term follow-up. Furthermore, our findings may be influenced by unconsidered confounding factors. Nevertheless, our study results present preliminary evidence regarding the impact of aspirin dosage on the prognosis of Kawasaki disease. In summary, Kawasaki disease is a prevalent systemic vasculitis in pediatric patients, with the prevention of coronary artery damage and other complications being the key to its treatment. Intravenous immunoglobulin (IVIG) and aspirin are the mainstay drugs for Kawasaki disease treatment, with the dosage of aspirin playing a crucial role in preventing coronary artery damage.
Conclusion
The findings of this study indicate that aspirin dosage significantly impacts the prognosis of Kawasaki disease. Specifically, the administration of high-dose aspirin (45–49 mg/kg per day) during the 3- to 4-week period following onset is linked to an elevated incidence of coronary artery damage and extended duration of fever. Consequently, we advise exercising caution in the clinical application of high-dose aspirin and advocate for further research to identify optimal therapeutic strategies aimed at reducing the risk of coronary complications.
Footnotes
Author contributions
ZAB and PLY conceived and designed the study, analyzed and interpreted the data, and wrote the manuscript. LF, LHZ, ZB, ZYY and GYJ collected clinical samples and completed the related experiments. All authors approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was financially supported by Changzhou Science and Technology Plan Project (CJ20239037).
Trial registration
This retrospective study was not registered because retrospective studies do not require registration prior to initiation as they analyze existing data collected for clinical, not research, purposes.
