Abstract
Introduction
Severe acute respiratory syndrome-corona virus-2 (SARS-CoV-2) reportedly surfaced in the Chinese city of Wuhan in late 2019.1,2 The virus spread rapidly throughout the globe, and in just couple of months, the total reported cases of infection crossed 20 million. The World Health Organization (WHO) declared it a pandemic on 11 March 2020. Until 11 January 2022, more than 308 million individuals were reportedly infected with SARS-CoV-2 infection. Additionally, 5.4 million SARS-CoV-2-associated deaths were reported across the globe. 3
Reverse transcriptase-polymerase chain reaction (RT-PCR) is currently used as the gold standard for acute COVID-19 diagnosis. 4 In the first 7 days after the onset of infection, the sensitivity for detection of virus in RT-PCR is 66.7% which decreases afterward. 5 Alternatively, serological tests which are based on the humoral responses of the human body become positive after 1–3 weeks of the infection.5,6 Antibodies are produced against different epitopes of SARS-CoV-2 including receptor-binding domain (RBD), spike glycoprotein, and nucleocapsid protein. 7 Among these, the nucleocapsid is more immunogenic, highly expressed, and more sensitive to detection than spike protein.7,8 Nucleocapsid antigen plays an important role in viral pathogenesis, it has numerous epitomes that stimulate T and B cell responses, and is suitable for vaccine formulation. 9 It has been reported that COVID-19 patients become seronegative gradually with some studies suggesting rapid decline within few months post-COVID-19 infection.10,11 However, there is limited research available regarding the post-infection persistence and response to SARS-CoV-2 virus up to 12 months period.
This study collected periodic samples (quarterly) from 120 HCWs previously infected with SARS-CoV-2 infection up to 12 months from their enrolment into the study to understand the magnitude and durability of the anti-nucleocapsid IgG antibodies titer in the long run.
Material and methods
Participant cohort
This was a single-arm follow-up study. Healthcare workers who participated in this study were employees of Prime Foundation (PF) group of teaching hospitals, Peshawar city, Pakistan. Healthcare workers including doctors, nurses, and paramedical staff were invited to participate. A total number of 134 HCWs previously infected with COVID-19 infection (confirmed by RT-PCR) were included in the study while severely ill participants were excluded. Nine HCWs dropped out after first follow-up and five after second follow-up (Figure 1: flow chart). All participants provided written informed consent for study participation. The study was approved by Institutional Review Board (IRB) of Prime Foundation. Blood samples were collected periodically (quarterly) from the study cohort between July, 2020 and April 2021 at four time points (July 2020, Oct 2020, Jan 2021, and April 2021). Flow chart.
Blood sampling and serological testing
SARS-CoV-2 recombinant (N) protein (IgG)
Five milliliter of blood sample was collected and kept in lithium heparin bottles. Samples were centrifuged and serum was isolated. SARS-CoV-2 antibodies in the serum sample were identified using the Roche assay (Roche, Basel, Switzerland): an electro-chemiluminescence immunoassay that uses a protein expressing the nucleocapsid antigen (N) binding (sensitivity of 100% and specificity of 99.8%). The manufacturer’s instructions were followed for the result interpretation. (reactive: antibody titer cutoff index 1.0).
Statistical analysis
Statistical analyses were performed using SPSS software Mac version 25.0 (IBM, New York USA). Graphs were plotted using Prism software Mac version 9.0 (Graph Pad, CA, USA). Counts/percentages were calculated for categorical variables and descriptive statistics for continuous variables. The median COI and 95% CI were plotted against the different time points. Pearson’s correlation was drawn to evaluate correlation between anti-N-IgG and time interval/age. A
Results
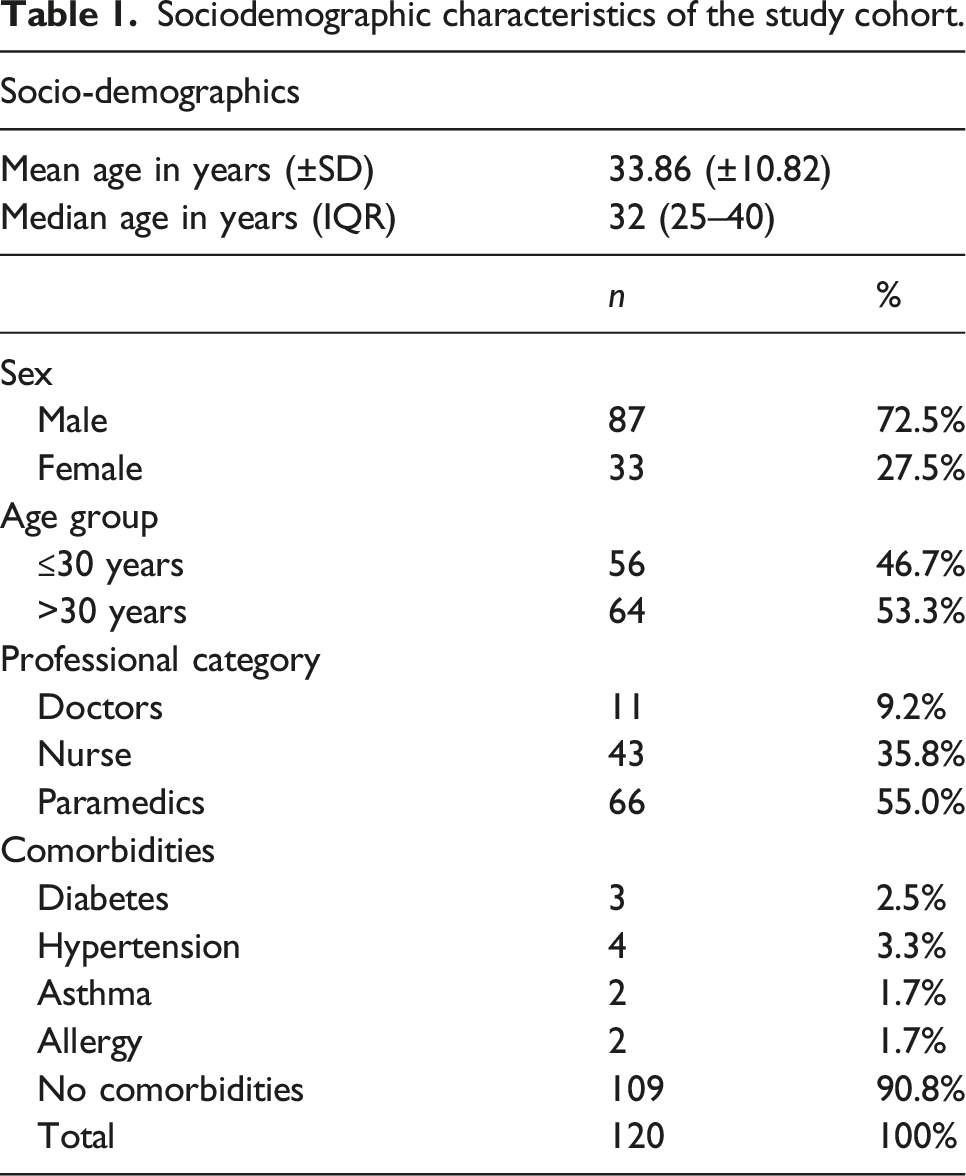
Sociodemographic characteristics of the study cohort
Sociodemographic characteristics of the study cohort.
Twelve months anti-N-IgG seropositivity rates
Serum samples were periodically tested for SARS-CoV-2 antibody titers in HCWs. We compared the anti-N-IgG at 3, 6, 9, and 12 months interval. The first serum sample provided by the healthcare workers within 12 weeks after the onset of symptoms showed detectable SARS-CoV-2 anti-N-IgG antibodies in all individuals. During the follow-up period, three (2.5%) individuals at 6 months, 11 (9.2%) individuals at 9 months, and 12 (10%) individuals at 12 months were found seronegative (Figure 2). Majority of the seronegative individuals (66.6%) at month 12 were aged 30 years or less. The relationship between anti-N-IgG index and time interval was plotted using the median and IQR (Figure 3). The median anti-N-IgG antibody level identified at 3 months was 23.7 CO-index (IQR: 9.13–50.27) and increased to 32.9 CO-index (IQR: 11.8–84.4) at 6 months. At 9 months, the median anti-N-IgG antibody level started to wane in the subsequent time and was dropped to 14 CO-index (IQR: 3.4–37.6) and declined further to 9.8 at 12 months (IQR: 2.8–27.8) (Figure 3). When classified by gender, there was no statistically significant difference in anti-N-IgG between male and female at all time points examined (3 months, Positive and negative tests over time. Legend: Number of positive and negative tests over 12 month post-infection. IgG antibody response against nucleocapsid. Periodic changes of anti-SARS-CoV-2 IgG antibodies over 12 month period. Blue scatter dot-plot with median and IQR showing the distribution of SARS-CoV-2-anti-nucleocapsid IgG with C/O index in serum samples of HCWs at July 2020, Oct 2020, Jan 2021, and April, 2021 post-infection. Anti-SARS-CoV-2 IgG antibodies (median, IQR) over 12 month post-infection. Median and interquartile (IQR) SARS-CoV-2-anti-nucleocapsid IgG of HCWs at 3, 6, 9, and 12 months post-infection confirmed by RT-PCR. *Refer to significance with Mann–Whitney U test.


Correlation analysis for anti-N-IgG
Spearman’s correlation coefficient was computed to assess the strength of linear association between time interval (interval between symptom onset and blood sampling) and anti-N-IgG index. The correlation coefficient was negatively associated between anti-N-IgG and time interval (
Discussion
Infection with SARS-CoV-2 produces a complex immune response that changes over time and varies between individuals. 12 Due to this diversity, different studies have suggested a variable range of time for antibody persistence in COVID-19 infection. Initial studies demonstrated that antibodies against SARS-CoV-2 decrease rapidly in the first 3 months.10,13 While others reported that antibodies persist in the majority of the patients for more than 9 months.14,15 This study investigated the antibody levels for 1 year in HCWs who identified positive for SARS-CoV-2 antibody (anti-N-IgG) in the first wave of COVID-19 outbreak in Peshawar, Pakistan.
In our study, we observed persistence of anti-N-IgG antibodies against SARS-CoV-2 in 97% of the cases at 6 months, in 88% of the patients at 9 months, and 89% of the patients at 12 months. (Graph A). In accordance with our observations, a study from the USA found detectable anti-N-IgG in 92% of patients within 3–6 months after the onset of symptoms. 16 Jose F. Verona reported seropositivity in 75% of HCWs at 9 months. 14 Zhao Y et al. from China also observed antibody persistence in 88.06% of the participants at 12 months. 15 In contrast to our study, Jan Van Elslande found persistence of antibodies in only 38% of the participants within the first 6 months in mildly infected individuals 17 while Floriane Gallais et al. and Stephane Pelleau et al. observed seropositivity in 20% and 7% of participants, respectively, after 1 year of onset of symptoms.18,19
Longitudinal studies on antibody levels reported an initial peak followed by a decline.20–22 The peak time for antibodies in our study was 3–6 months which is in accordance with a study by Arnaud et al. who observed antibody peak at 3 months. 22 While Sheila F Lumley et al. found peak levels at 24th-day post-infection. 21 Similarly, a systematic review by Post et al. observed peaked IgG around weeks 3–7 post-symptom onset, persisting for at least 8 weeks. 23 We noticed a gradual decline in antibody levels with time and the decline appeared to slow down as time progressed (Graph B), which is in accordance with the study by Floraine Gallais et al. 18 and Maurice Steenhuis et al. 24
We observed inter-individual variability in our project which was consistent with previously reported studies.14,21 However, a clear difference was found in below and above 30 years of age at the end of 12 months. In accordance with our study, Jose F. Verona et al. also reported a significant difference but for the age group below and above 45 years. 14 Sheila F Lumley and Meddalena Peghin also reported faster decay of antibodies in young adult than older participants.21–25
This study adds to the existing literature about the anti-nucleocapsid-IgG antibody immune response after natural infection up to 12 month period. Moreover, the coronavirus vaccines are based on spike protein, and the detection of response against nucleocapsid antigen may help to distinguish between serological response to vaccination and infection.
Our study has some limitations. First, we did not use and study anti-S antibody levels, which might have diverse antibody kinetics. Second, due to mild symptoms and asymptomatic cases, we did not determine the association between antibody level and clinical course; third, the study participants were from a single center with small sample and the results cannot be generalized.
Conclusion
In conclusion, our study demonstrates a persistent immunological response to COVID-19 infection. SARS-CoV-2 antibody levels started steadily declining after 6 months but remained detectable in the majority of patients up to 12 month period. Additionally, the decrease over time was more in young individuals (≤30 years) than older individuals after 6 months. Long-term monitoring is recommended to reveal whether this immunologic memory provides protection against current or new variant of SARS-CoV-2 re-infection.
Footnotes
Author’s note
The cost of the study is beared by the principal and the co-authors of this study.
Acknowledgments
The authors acknowledge all the healthcare workers for voluntarily participating in this study.
Authors’ contributions
1Concept, study design, data collection, introduction, discussion, and data entry. 2Introduction, discussion, and references. 3Results, methodology, data-analysis, and abstract. 4 Concept, discussion, conclusion, and proof reading.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was granted from institutional review board (IRB) of Prime Foundation, Peshawar Medical College, Pakistan. IRB approval number: Prime/IRB/2020-279.
Written informed consent
Written informed consent was obtained from all subjects before the study.
Availability of data and material
The datasets used during the current study are available from the corresponding author on reasonable request.
