Abstract
Investigating the role of surface-modified 3D printed titanium implants in modulating oral soft tissue closure and antimicrobial properties. Eighteen 6-month-old healthy adult New Zealand White rabbits were selected as experimental animals, the left and right mandibular teeth of each experimental animal were randomly divided into two groups, and the bilateral first premolar teeth were extracted after general anesthesia, and one implant was implanted into each of the magnesium/zinc ionized surface-treated group (experimental group) and the surface-untreated group (control group), with a total of 36 implants, i.e., 18 implants for the experimental group and 18 implants for the control group. Bilateral mandibular samples from both groups were removed after 2 and 4 weeks of implantation to investigate the attachment and spreading of human gingival fibroblasts (HGFs) on the surface of the samples, their proliferative and migratory behaviors as well as the expression levels of related gene proteins (CoL-1 and TGF-β), and HE staining of the soft tissues of the samples and inflammatory cell counts. After 2 and 4 weeks of implantation, the experimental group’s HGFs adhesion and spreading were significantly better than that of the control group, the migration and proliferation of HGFs were significantly greater than that of the control group (p < .05), and the expression of CoL-1 and TGF-β genes was significantly higher than that of the control group (p < .05), and the inflammatory cell count was significantly smaller than that of the control group (p < .05). Surface modification by magnesium and zinc ions enhances the sealing performance and antimicrobial performance of 3D printed titanium implants in oral soft tissues, with significant effects.
Introduction
Peri-implantitis is a disease in which the tissues surrounding oral implants and other tissues undergo an inflammatory reaction and destruction. It is a common complication of oral implant surgery and can lead to functional failure of the implant and tissue damage. Peri-implantitis is usually caused by bacterial infection, which enters the peri-implant tissues through the fiber bundle gap at the gingival margin or other injuries. It is a biological complication of implants characterized by connective tissue inflammation and progressive marginal bone resorption, and is related to a variety of factors such as oral hygiene habits, implant performance, and surgical operation, etc., and its specific pathogenesis is still controversial .1,2 Pure titanium and titanium alloy have the advantages of good biocompatibility, mechanical properties and corrosion resistance, and are one of the widely used oral implant materials in clinical practice. However, titanium is a biologically inert material, which does not have biological activity and antibacterial properties, and with long-term contact between the oral cavity and the external environment, bacteria are prone to gather and adhere to the titanium surface, causing implant-related infections, leading to delayed closure of the implant denture, or even restoration failure. Implant surface modification can improve the success rate of implant denture restoration by changing the surface morphology or introducing antimicrobial substances, so that the surface of the implant has antimicrobial ability while not affecting or even promoting osseointegration.3,4 It has been found that magnesium and zinc, as important constituent elements of the human body, are involved in regulating the body’s metabolism and other life activities. Magnesium has good biocompatibility and osseointegration; zinc exhibits excellent antimicrobial capacity and osteogenic activity. The combination of magnesium and zinc is expected to promote accelerated healing and repair of soft tissue injury wounds. 5 Based on this background, the present study introduced magnesium and zinc into the surface of 3D printed titanium materials to systematically investigate the role of surface properties of 3D printed titanium materials on the regulation of the behavior of HGFs, with the aim of making a theoretical and experimental foundation for improving the soft tissue closure performance of 3D printed personalized titanium implant surfaces, and further expanding the range of clinical indications for 3D printed personalized titanium implants.
Materials and methods
Laboratory animal
Eighteen 6-month-old healthy adult New Zealand Large White rabbits, male and female, with body mass of 2.5∼3.5 kg, provided by the Animal Experiment Center of Xi’an Jiaotong University (XJTU) were used as experimental animals and kept in the Animal Room of the Animal Experiment Center of the Faculty of Medicine, Xi’an Jiaotong University (XJTU). Comprehensive consideration of manpower, resources, financial resources into and research on the degree of tolerance for error, this study to determine the sample size is calculated as follows: N = Z2x (Px (1-P))/E2, where N is the sample size; Z is the amount of statistics, the confidence level of 95%, Z = 1, 96; when the confidence level of 90%, Z = 1, 64; E is the value of the error; p is the probability of the value. The ethic approval was reviewed and approved from The College of Stomatology, Xi’an Medical University, Research Center for Tooth and Maxillofacial Tissue Regeneration and Restoration (Approval no. XYLS2022271). This study followed mainland China’s guidelines for the humane treatment of animals and complied with relevant laws.
Main reagents
S-3000N scanning electron microscope (SEM; Hitachi, Japan), E300CP/400CS Hard Tissue Cutting and Grinding System (EX-AKT Vertriebs GmH, Germany), TCS.SP8 Laser Scanning Confocal Microscope (Leica, Germany). Fluorescence quantitative PCR instrument (Roche, LightCycler480). Sodium pentobarbital, methylene blue, acidic magenta (Sigma, USA), 4,6-amidino-2-phenylindole (DAPI, Sigma, USA), ghost pen cyclic peptide (FITC-Phalloidin, Sigma, USA), alamarBlueTM (AbD serotec Ltd, UK) kit.
Personalized implant design and fabrication
A CT of rabbit head was taken and the obtained DICOM data were imported into the medical image processing software MIMICS 15.0 to obtain a 3D geometric surface model of the jawbone and teeth. The 3D geometric surface model of the mandible with teeth was exported as a point cloud file in STL format and imported into Geomagic 12.0 inverse software, and a personalized root-shaped titanium implant was designed according to the morphology of the bilateral mandibular first premolar, which was printed out by using 3D printing technology (SLM, EOS-M290, Germany, average particle size of 20 μm). The printed titanium implant material was placed on the vacuum chamber target stage of the plasma immersion ion implantation (PIII) equipment, and when the vacuum reached 5 × 10-2 Pa, magnesium and zinc ions were injected individually and magnesium/zinc ions were co-injected, respectively, and finally rinsed with distilled water, gamma sterilized, dried and stored for use. 6
Experimental grouping and treatments
The left and right mandibular teeth of each experimental animal were randomly divided into two groups, and the bilateral first premolar teeth were extracted after general anesthesia, and 1 implant was implanted in each of the magnesium/zinc ion surface-treated group (experimental group) and the surface-untreated group (control group) respectively, with a total of 36 implantations, i.e., 18 implantations in the experimental group and 18 implantations in the control group. The specific methods of soft tissue implant surgery were as follows: routine preoperative toweling, skin preparation, disinfection, and general anesthesia by using 3% sodium pentobarbital at a dose of 1 mL/kg in auricular marginal vein. After satisfactory anesthesia, the bilateral mandibular first premolar teeth were extracted by minimally invasive extraction, the sterilized implants were implanted, and the gingiva and implants were closed by mattress suture. Penicillin was used for 1 week postoperatively to prevent infection at a dose of 20,000 U/kg. The operation methods were standardized to ensure that there was no significant difference between the experimental subjects in each group before the test observation. The experimental animals in each group were executed by air embolization of the marginal ear vein at 2 and 4 weeks postoperatively, and HGFs from bilateral mandibular specimens were taken for testing and detection.
Cell culture
HGFs were cultured using fibroblast medium (FM, ScienCell Research Laboratories, USA) in a constant temperature incubator at 37°C in a humidified atmosphere with a
Cell adhesion and spreading assay
The experimental samples were sterilized with 75% ethanol for 2h (changing ethanol every 0.5 h) and dried on an ultra-clean bench. The aseptically dried samples were placed in 24-well cell culture plates, the samples were washed twice with PBS (10 mM), 1 mL of HGFs suspension with a cell density of 2 × 104 cells/mL was added to each well, the samples were submerged, the cell culture plates were shaken slightly to homogenize the cell suspension, and then placed in a thermostatic incubator with a
Cell migration assay
75% ethanol sterilized and dried 10 mm × 10 mm×1 mm samples (three in each group, one at each time point) were put into 24-well cell culture plates, the samples were washed twice with PBS, and 1 mL of HGFs cell suspension with a cell density of 5 × 104 cells/mL was added to each well. The cell suspension was homogenized by slight shaking, and the plates were placed into a constant temperature incubator in a humidified atmosphere of 5%
Cell proliferation assay
The cell proliferation activity on the surface of the samples was evaluated using alamarBlueTM (AbD serotec Ltd, UK) kit. The 10 mm × 10 mm×1 mm samples (four parallel samples in each group) sterilized by 75% ethanol and air-dried were put into 24-well cell culture plates, the samples were washed twice with PBS, and the samples were submerged by adding 1 mL of HGFs suspension with a cell density of 3 × 104 cells/mL to each well, and then the cell suspensions were shaken gently to homogenize the cell suspensions and put into a constant temperature incubator in a humidified atmosphere of 5%
Gene expression tests
Real-time quantitative PCR was used to detect the expression level of soft tissue closure-related genes of cultured HGFs on the sample surface. 75% ethanol sterilization and drying of 20 mm × 20 mm × 1 mm samples (three parallel samples in each group) into a 6-well cell culture plate, wash the sample twice with PBS, add 4 mL of HGFs suspension with a cell density of 1 × 105 cells/mL to each well to submerge the sample, and then gently shake the cell suspension uniformly and place it in a constant temperature incubator in a humidified atmosphere of 5%
HE staining test
In both groups, a 5 mm × 3 mm square incision was made at the mucosal healing point of the flap area to reach the surface of the hard tissue, the mucosa was peeled off and cut, fixed in paraformaldehyde solution, and the tissue specimen was made into a tissue specimen, dehydrated step by step by alcohol, put into xylene transparent, dipped in wax, and embedded buccolingually. Wax blocks were serially sectioned at a thickness of 3 μm, routinely deparaffinized with xylene and gradient alcohol to water, stained with Eosin staining solution for HE staining, and sealed with neutral gum. The histology was observed under light microscope, and the area of interest was the interface between hard and soft tissues. 3 fields of view were randomly selected at 400×, and the inflammatory cell count was performed by ImageJ software.
Statistical methods
Data were analyzed using the statistical analysis software SPSS 17.0, and measurements were expressed as arithmetic mean ± standard deviation and statistically tested using t-test. Each group of variables contained at least three valid values, and the significant difference level was set at p < .05 to indicate that the data were statistically significantly different.
Results
Comparison of the properties of the two groups of nanocoatings
From the scanning electron microscope observation, it can be seen that there is no obvious difference between the coating morphology of the experimental group and the control group, and the inner layer of the micro-arc oxidized film of the two groups is dense, while the outer layer shows a rough multi-stage pore-like structure. See Figure 1. ①:Observation of the indicated morphology of the control implant nano-coating (SEM, ×1000);②: Observation of the indicated morphology of implant nanocoatings in the experimental group (SEM, ×1000).
Cell adhesion and spreading
Through the naked eye, after 2 and 4 weeks of implantation, it was observed that the experimental group showed significantly more red dots than the control group, indicating that the experimental group showed stronger cell adhesion and fuller spreading than the control group. In addition, within-group comparisons showed that both groups had more red dots after 4 weeks of implantation than after 2 weeks of implantation in this group. See Figure 2. (a): Cell adhesion and spreading pictures of control group after 2 weeks; (b): Cell adhesion and spreading pictures of control group after 4 weeks; (c): Cell adhesion and spreading pictures of experimental group after 2 weeks; (d): Cell adhesion and spreading pictures of experimental group after 4 weeks; Note: Larger red dots indicate better cell adhesion and spreading.
Cell migration
By t-test, after 2 and 4 weeks of implantation, the number of migrated HGFs cells in the experimental group was significantly greater than that in the control group [(40.52 ± 2.56) versus (15.45 ± 2.12) and (88.76 ± 2.97) versus (35.53 ± 2.64)], and the difference was significant (p < .05), suggests that the experimental group showed greater cell migration than the control group. In addition, within-group comparisons showed that both groups migrated more HGFs cells after 4 weeks of implantation than after 2 weeks of implantation in this group. See Figures 3 and 4. (a): Pictures of cell migration after 2 weeks in the control group; (b): Pictures of cell migration after 4 weeks in the control group; (c): Pictures of cell migration after 2 weeks in the experimental group; (d): Pictures of cell migration after 4 weeks in the experimental group; Note: Larger blue dots indicate greater cell migration. Comparison of cell migration between the two groups after 2 and 4 weeks; *p < .05 compared to the control group at the same time point.

Cell proliferation
By t-test, after 2 and 4 weeks of implantation, the cell counts of HGFs in the experimental group were significantly greater than those in the control group [(25,520.38 ± 210.89) versus (10,505.67 ± 202.36), (170,342.32 ± 438.27) versus (70,650.44 ± 378.36)], with a significant difference (p < .05), suggests that the experimental group showed greater cell proliferation than the control group. In addition, within-group comparisons showed that both groups had more HGFs cells after 4 weeks of implantation than after 2 weeks of implantation in this group. See Figure 5. Comparison of cell proliferation between the two groups after 2 and 4 weeks; *p < .05 compared to the control group at the same time point.
Gene expression
By t-test, after 2 and 4 weeks of implantation, CoL-1 gene expression was significantly greater in the experimental group than in the control group [(2.23 ± 0.19) versus (0.82 ± 0.12), (3.52 ± 0.17) versus (1.21 ± 0.11), p < .05]; and TGF-β gene expression was significantly greater in the experimental group than in the control group [(3.55 ± 0.10) versus (0.42 ± 0.11), p < .05]. 0.08), (6.02 ± 0.11) versus (2.48 ± 0.10), p < .05], it was shown that the experimental group showed stronger implant closure characteristics than the control group. In addition, the results of within-group comparison showed that both groups had more CoL-1 and TGF-β gene expression after 4 weeks of implantation than after 2 weeks of implantation in this group. See Figures 6 and 7. Comparison of CoL-1 gene expression between the two groups after 2 and 4 weeks; *p < .05 compared with the control group at the same time node. Comparison of TGF-β gene expression between the two groups after 2 and 4 weeks; *p < .05 compared with the control group at the same time node.

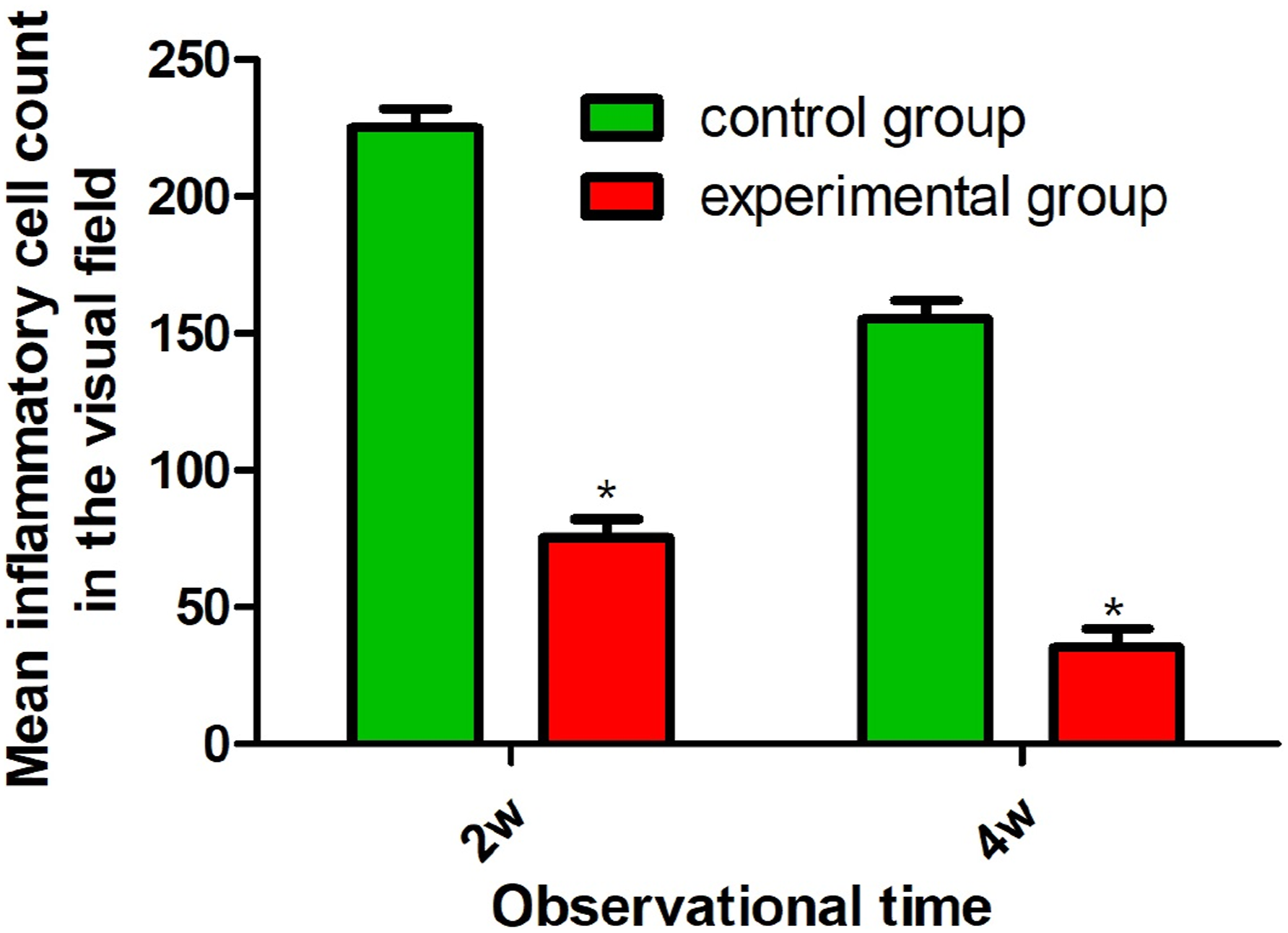
HE staining and inflammatory cell count
HE staining observed areas of inflammatory cell infiltration of varying degrees within the gingival tissue surrounding the implants. By t-test, the results of inflammatory cell counts showed that after 2 and 4 weeks of implantation, the inflammatory cell counts in the experimental group were significantly smaller than those in the control group [(76.87 ± 6.15) versus (224.35 ± 8.36), (34.28 ± 5.23) versus (153.38 ± 8.87), p < .05], it was shown that the experimental group showed stronger antimicrobial properties of the implants than the control group. In addition, within-group comparisons showed that inflammatory cell counts in both groups were less after 4 weeks of implantation than after 2 weeks of implantation in this group. See Figures 8 and 9. (a): HE staining pictures after 2 weeks in the control group; (b): HE staining pictures after 4 weeks in the control group; (c): HE staining pictures after 2 weeks in the experimental group; (d): HE staining pictures after 4 weeks in the experimental group; magnification: ×400. Comparison of inflammatory cell counts in peri-implant tissue between the two groups after 2 and 4 weeks; *p < .05 compared to the control group at the same time point.
Discussion
Since the first pure titanium Branemark implant was implanted into the human body in 1965, over the past 40 years, pure titanium implants have been more and more widely used in the field of dentistry and have become one of the main treatments for restoration of defective and missing teeth, although titanium implants have better biocompatibility, mechanical properties, corrosion resistance and good processability, which makes them widely used in the field of dental implants. Although titanium implants have good biocompatibility, mechanical properties, corrosion resistance and good processability, which make them widely used in the field of dental implants, titanium itself does not have antimicrobial activity, and after implantation and restoration, it is prone to form an acquired film of microbial attachment, which gradually forms plaque accumulation around titanium implants, easily leading to the occurrence of peri-implantitis and affecting the efficacy of oral implantation.7–9 Titanium is chemically very active and easily combines with other elements to form stable compounds. Studies have shown that titanium surfaces can be modified by showing that the method of modification can improve the ability to bind with soft tissues and enhance the antimicrobial properties of titanium surfaces.10,11 In this study, magnesium/zinc ions were injected into the surface of 3D printed titanium materials using PIII technology to investigate the effects of magnesium and zinc ion injection on the surface physicochemical properties of titanium implant materials, and the results are as follows.
In this study, it was found that after 2 and 4 weeks of implantation, the amount of HGFs adhesion spread, migration, proliferation, CoL-1 and TGF-β gene expression of the experimental group was significantly larger than that of the control group, and the inflammatory cell count was significantly smaller than that of the control group, suggesting that the surface modification of transmagnesium and zinc ions can enhance the closure performance and antimicrobial performance of the 3D-printed titanium implant in the soft tissues of the oral cavity, and the effect is significant. To explore the mechanism, on the one hand, HGFs are the main cells involved in soft tissue regeneration and closure, for the formation of the keratinocyte layer, and play a key role in oral wound healing and soft tissue regeneration. 12 Studies have shown that magnesium can effectively improve the adhesion and spreading of HGFs on the surface of the material and increase the potential of implant materials to promote soft tissue closure. 13 The study of the migration rate of HGFs on the surface of pure magnesium revealed that a specific concentration of magnesium ions could significantly enhance the adhesion of HGFs on the surface of the material although it did not have a significant effect on the proliferation and activity of HGFs. 14 Zheng J et al 6 discussed the effect of magnesium ion concentration on the directional migration ability of human fibroblasts and found that this property was related to integrins in extracellular matrix proteins. On the other hand, zinc, as one of the essential trace elements for the vital activities of the organism, plays an important role in the growth and development of bones throughout the body and in the maintenance of the physiological and structural functions of bones. Zinc is an important component of the cell membrane and is involved in the synthesis of a variety of enzymes. Zinc plays an important role in the maintenance of cell structure and physiological function, and is a component or activator of more than 200 enzymes such as DNA, RNA polymerase, thymidine kinase, alkaline phosphatase, lactate dehydrogenase, carbonic anhydrase, carbonyl peptidase and other enzymes, and is directly involved in synthesis of nucleic acids, proteins and other synthesis and tissue metabolism. 15 If the activity of zinc-containing enzymes is reduced, the metabolism of cysteine and lysine is disturbed, the synthesis of glutamate is reduced, and the process of synthesis of mucin and soft tissue proteins is blocked. 16 All of the above mechanisms may have profound effects on the activities of HGFs cell proliferation, migration, and gene expression. In addition, zinc has a strong oxidizing ability, can be directly or indirectly with almost all the gaseous elements to generate the corresponding compounds, and the formation of ZnO in contact with air has good antibacterial properties . 17 Magnesium and zinc synergistically can achieve complementary effects, the surface observation of titanium samples chelated with both magnesium and zinc ions revealed that magnesium ions can promote cell proliferation and differentiation, and zinc ions can be highly efficient antibacterial. In addition, studies have shown that immune cells such as macrophages and their secreted cytokines can directly or indirectly affect bone remodeling, and the immune environment within gingival tissues is closely related to the state of alveolar bone at its site. 18 Relevant studies have shown that metal ions such as magnesium and zinc may affect the microenvironment of the monocyte-macrophage system and inhibit the polarization of the relevant inflammatory cells to phenotypes such as M1 and M2, thus suppressing the immune response of the body’s intrinsic immune system to local tissues and attenuating the inflammatory response, as well as promoting wound healing and tissue repair. 19 In addition, a study in mice showed that oral implantation of foreign bodies leads to subcutaneous foreign body reaction and promotes the expression of pro-inflammatory cytokines in the tissues, generating a local inflammatory stress response. However, after subcutaneous injection of magnesium and zinc ions, the expression of pro-inflammatory factors, such as IL-1β, TNF-α, and other pro-inflammatory factors in local tissues, and the expression of anti-inflammatory, repair-promoting related cytokines, such as IL-10 and IL-35, were significantly downregulated, and the expression of the above mechanism was proved to be effective in inhibiting cell proliferation and differentiation. CoL-1, TGF-β and other genes and proteins related to cell proliferation and differentiation were significantly up-regulated in expression, and the above mechanism was proved by in vivo and in vitro experiments to have the effect of inhibiting macrophage fusion and preventing macrophage polarization in the direction of M2, which is considered to be one of the important pathways of the antimicrobial effect exerted by allogeneic seed plants. 20 Finally, the sample size of this study is small, the research sample is single, and subject to the constraints of the experimental conditions, the relevant validation experiments are not perfect, although this study provides some theoretical support for the future clinical use of surface-modified titanium implants, but the molecular mechanisms associated with implant closure and antimicrobial properties are still unknown, and the future needs to be explored further to explore the specific molecular mechanisms, more in-depth and more precise conclusions of the study are still waiting for a broad range of researchers to be confirmed by multicenter, large-sample studies.
Conclusion
Surface modification by magnesium and zinc ions enhances the sealing performance and antimicrobial performance of 3D printed titanium implants in oral soft tissues, with significant effects.
Footnotes
Acknowledgments
We would like to acknowledge the everyone for their helpful contributions on this paper.
Author contributions
Each author has made an important scientific contribution to the study and has assisted with the drafting or revising of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Innovative team for disease mechanism and prevention of oral and maxillofacial system (No. 2022TD-54); General project of Key R&D Plan of Science and Technology Department of Shaanxi Province (No. 2023-YBSF-097); Xi ‘an Medical University 2022 Research Capacity Improvement Plan Project (No. 2022NLTS090); The Youth Innovation Team of Shaanxi Universities (no number).
Ethical statement
Informed consent
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All of the authors have consented to publish this research.
Animal welfare
This study followed mainland China’s guidelines for the humane treatment of animals and complied with relevant laws.
