Abstract
Background
HMGA2 can promote the development of multiple malignancies. This study aimed to determine whether a putative functional genetic polymorphism (rs968697 T>C) in the HMGA2 gene promoter region was associated with malignancy susceptibility in the Chinese population.
Methods
The rs968697 genetic polymorphism was genotyped using the SNaPshot method in the case-control study. The odds ratios and 95% confidence intervals were calculated using a logistic regression model. STATA software was used to conduct the meta-analysis.
Results
The rs968697 genetic polymorphism was associated not only with susceptibility to malignant tumors [CC versus TT: OR = 0.45, 95%CI = 0.28–0.73, p = .001; CC versus (CT + TT): OR = 0.47, 95%CI = 0.29–0.75, p = .003] including gastric cancer (GC), but also with TNM stage and survival prognosis of GC patients. Genotype-tissue expression analysis, luciferase assay and bioinformatics analysis revealed that the rs968697 genetic polymorphism might affect the binding of transcription factors, especially POLR2A, which in turn regulate the expression of HMGA2.
Conclusions
The current research suggests that the rs968697 genetic polymorphism may be used as a biomarker for malignancy susceptibility and GC prognosis in the Chinese population.
Introduction
As one of the main factors contributing to death worldwide, malignant tumors pose a serious threat to public health. 1 A malignant tumor is a complex and heterogeneous disease, and its occurrence is linked to genetic, environmental, and lifestyle factors.2,3 Previous research has shown that malignant tumor development and progression are significantly influenced by single nucleotide polymorphisms (SNPs).4–6 An in-depth examination of SNPs in cancer-related genes is beneficial in determining an individual’s susceptibility to malignancies and their prognosis.
A chromosomal protein with three AT-hooks known as high mobility group protein A2 (HMGA2) can bind to the minor grooves of AT-rich DNA regions. 7 It can influence chromatin architecture and alter transcription by extensively binding to chromatin and generating a multiprotein complex. 8 HMGA2 overexpression has been related in numerous studies to increased invasion, stemness, and a poor prognosis in malignant tumors, including gastric cancer (GC).9–13 Due to the important role of HMGA2 in malignant tumors, genetic polymorphisms in the HMGA2 gene may contribute to the formation and growth of malignant tumors. It is worth noting that previous case-control studies have indicated that the rs968697 T>C genetic polymorphism located in the HMGA2 gene promoter region is associated with susceptibility to colorectal cancer and hepatoblastoma.14,15 However, no study has reported the association of the rs968697 genetic polymorphism with GC susceptibility. Additionally, it has also been suggested that the rs968697 genetic polymorphism is not linked to malignancy susceptibility.16–18 Therefore, there is a need to further investigate the role of the rs968697 genetic polymorphism in malignancy.
In the current investigation, we looked first at the relationship between the rs968697 genetic polymorphism and GC susceptibility and the TNM stage and survival prognosis of GC patients, and then explored its potential function, as well as its relationship with malignancy susceptibility.
Material and methods
Study population
This observational case-control study enrolled 330 GC patients and 430 healthy controls from Shanghai Xuhui District Central Hospital. The individuals, who were all unrelated, all identified as being of the Han Chinese ethnicity. Two pathologists confirmed the presence of GC in patients by analyzing biopsies of gastric tumors. Patients with a history of other tumors, or prior radiotherapy/chemotherapy were excluded. The sociodemographic and medical data of patients was gathered through on-site interviews, direct calling, or medical chart review. Healthy controls were individuals without cancer who resided in the same neighborhood and sought out routine physical exams. The characteristics of healthy controls, including age, gender, smoking, and drinking habits, were collected via a questionnaire. Individuals who smoked daily for more than a year were classified as smokers, whereas those who drank one or more alcoholic beverages per week for at least a year were classified as drinkers. The Xuhui District Central Hospital in Shanghai's Ethics Committee granted approval to the study protocol (No. 2017035-011). Each participant in this study provided their written, informed permission.
DNA extraction and genotyping
DNA was extracted from peripheral blood samples using a TIANamp genomic DNA kit that is available for purchase. Using a NanoDropTM 2000 Spectrophotometer, DNA concentration and purity were evaluated. For later use, all DNA samples were stored at −20°C. The SNaPshot technique was used to analyze the genotypes of the rs968697 genetic polymorphism. 10% of the samples were chosen at random for DNA sequencing in order to assure quality control. The outcomes were entirely in line with those of the SNaPshot.
Genotype-tissue expression analysis
Using the RNAsimple total RNA kit (Tiangen), total RNA was isolated from 25 GC tissues and 15 normal paracancerous tissues in accordance with the manufacturer’s instructions. The NanoDropTM 2000 Spectrophotometer was used to determine the RNA content and purity. The extracted RNA was reverse transcribed into cDNA using the ReverTra Ace qPCR RT kit (TOYOBO). Quantitative real-time PCR was performed with a Roche FastStart Universal SYBR Green Master (Rox) using a StepOnePlus PCR instrument. The primer sequences are displayed in Table S1. 14 The mRNA expression levels were calculated using the 2−ΔΔCt approach. GAPDH expression levels were used as internal controls. All assays were repeated three times in parallel.
Luciferase assay
RPMI 1640 media (GIBCO) with 10% fetal bovine serum, 100 U/mL penicillin, and 100 g/mL streptomycin was used to cultivate two GC cell lines (BGC-823 and SGC-7901) at 37°C in an incubator with humidified 5% CO2. The rs968697 genetic polymorphism-related human HMGA2 promoter sequences were created and built into the pGL3-basic vector (Promega). To guarantee 90% confluence at the time of transfection, the cells were planted in 24-well plates. By using Lipofectamine 2000 (Invitrogen), the cells were transfected with 0.8 g of each generated vector, either with the T or C allele. Additionally, 10 ng of pRL-SV40 was simultaneously transfected into each well as an internal control for adjusting transfection effectiveness. The Dual-Luciferase Reporter Assay System (Promega) was used to assess luciferase activity 24 h after transfection and to calculate the ratio of firefly to renilla luciferase activity. The identical conditions were used in triplicate for all cells.
Bioinformatic analysis
The impact of the rs968697 genetic polymorphism in the HMGA2 gene promoter region on transcription factor binding was examined using the databases EnhancerDB and GVAT.19,20 Also, it was determined using the EnhancerDB database if transcription factors might regulate HMGA2 expression.
Meta-analysis
A literature search of Embase, Pubmed, Web of Science, and China National Knowledge Infrastructure was conducted using the following keywords: “High mobility group protein A2 or HMGA2”, “polymorphism” and “malignancy or cancer”. The final day of the search was February 25, 2023. The studies that were eligible for inclusion in the current meta-analysis had to satisfy two criteria: (a) they had to use a case-control study design to investigate the association between the rs968697 polymorphism and cancer susceptibility, and (b) they had to have comprehensive genotype data for calculating odds ratios (ORs) and 95% confidence intervals (CIs). Using a standardized data form, two researchers independently extracted information on the first author, publication year, cancer kind, nation, genotyping technique, and the quantity of the genotype distribution between cases and controls. Discussions were used to settle disagreements. The combined ORs and 95% CIs were calculated by selecting an appropriate effect model based on the heterogeneity test results. The I2 test and the χ2-based Q test were used to evaluate heterogeneity between studies. The fixed-effect model was applied if I2 <50% or the p-value for heterogeneity >0.1, indicating no heterogeneity; otherwise, the random-effect model was used. Sensitivity analyses were conducted with a leave‐one‐out method to investigate the quality and consistency of the results. Egger’s and Begg's tests were utilized to evaluate possible publication bias.
Trial sequential analysis
To determine whether the cumulative data were strong enough to draw conclusions, TSA was carried out using the TSA software. We set the type I error to 5%, the statistical test power to 80%, and the relative risk reduction to 20% in order to establish the necessary sample size and create the trial sequential monitoring border.
Statistical analysis
The χ2 test or the Student’s t-test was used to compare the distinctions in characteristics between GC patients and controls. Hardy-Weinberg equilibrium (HWE) was determined by analyzing the genotype frequencies of the rs968697 genetic polymorphism in controls using a goodness of fit χ2 test. A logistic regression model with adjustment for age, gender, smoking, drinking, and H.pylori infection was utilized to calculate ORs and 95% CIs to examine the relationship between the rs968697 genetic polymorphism and the susceptibility and TNM stage of GC. The Cox proportional hazards regression model was employed to calculate the hazard ratios (HRs) and 95% CIs. A t-test was used to assess the expression of HMGA2 mRNA between different genotype groups and the relative luciferase activity between different alleles. The above analysis was completed using SPSS 20.0 software. p < .05 was regarded as statistically significant. In addition, a meta-analysis was performed by STATA 12.0 software.
Results
A case-control study for gastric cancer
330 patients with GC and 430 healthy controls participated in the study. Table S2 displayed the sociodemographic and clinical characteristics of the participants. No significant difference was observed in age (cases: 63.3 ± 10.2 years, healthy controls: 64.1 ± 10.8; p = .302), gender (cases: male 72.1%, healthy controls: male 73.3%; p = .733), smoking (cases: 24.8%, healthy controls: 26.3%; p = .651), and drinking (cases: 38.5%, healthy controls: 33.5%; p = .153) between cases and controls. However, the ratio of H. pylori infection in cases (56.4%) was higher than that in healthy controls (47.0%) (p = .012). Furthermore, there were 154 (46.7%) and 176 (53.3%) patients classified as TNM stage I + II and III + IV, respectively.
Association of the rs968697 genetic polymorphism with GC susceptibility.
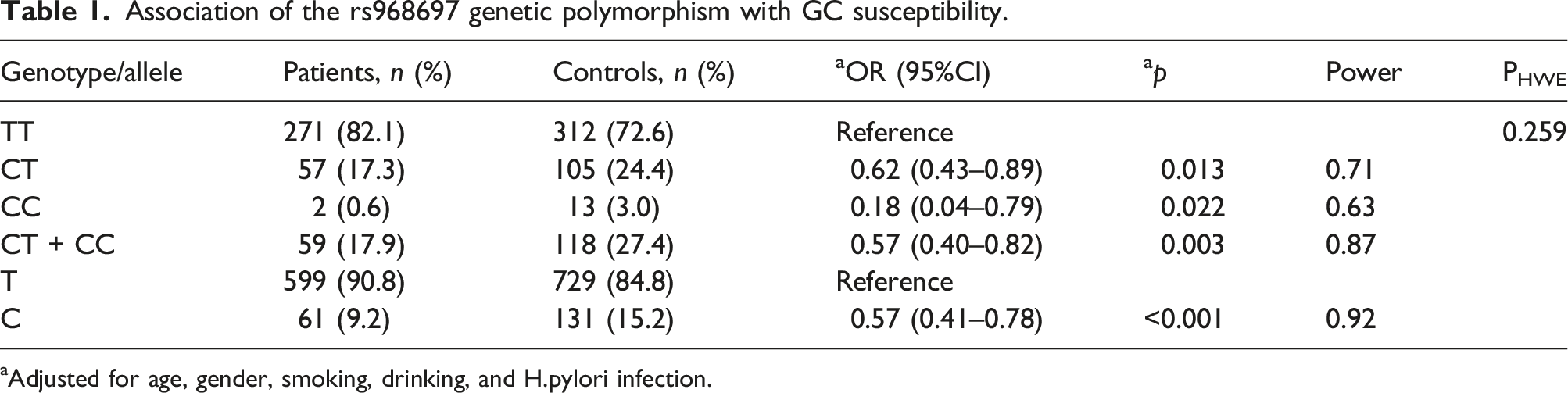
aAdjusted for age, gender, smoking, drinking, and H.pylori infection.
Association of the rs968697 genetic polymorphism with TNM stage of GC.

aAdjusted for age, gender, smoking, drinking, and H.pylori infection.
Associations between the rs968697 genetic polymorphism and overall survival in GC patients.

aAdjusted for age, gender, smoking, drinking, TNM stage, and H.pylori infection.
Potential functions of the rs968697 genetic polymorphism
Genotype-tissue expression analysis revealed that the expression of HMGA2 mRNA was significantly reduced in GC tissues carrying the rs968697 CT and CC genotypes and in normal paracancerous tissues carrying the rs968697 CT and CC genotypes (Figure 1). Luciferase assay showed that C allele was able to significantly reduce the promoter activity of the reporter gene more than T allele (Figure 2). According to bioinformatics investigation, the rs968697 genetic polymorphism could interfere with the binding of the transcription factors including POLR2A, ESRRA, ESRRG, MSC, PKNOX2, and TGIF2LX. Among them, POLR2A might regulate the expression of HMGA2 (Table S3). The relative expression of HMGA2 mRNA between different genotype groups (a): normal paracancerous tissues; (b) GC tissues. *: p < .05;**: p < .01). Promoter activity of different alleles of HMGA2 rs968697 genetic polymorphism (**: p < .01).

Association of the rs968697 genetic polymorphism with malignancy susceptibility
Meta-analysis of the association between the rs968697 genetic polymorphism and malignancy susceptibility.

Publication bias analysis on the association between the rs968697 genetic polymorphism and malignancy susceptibility.

In the comprehensive analysis using the homozygote model, the TSA was carried out (Figure S3). The cumulative Z-curve crossed the threshold for significance, which was in line with the findings of the meta-analysis. The cumulative data were not sufficiently powerful to conclude as it failed to cross any trial sequential monitoring boundaries or reach the necessary sample size.
Discussion
Several studies have pointed to the important role of HMGA2 in the development of multiple malignancies such as colorectal cancer, gallbladder cancer and GC.9,10,13,21,22 For instance, through the recruitment of tumor-associated macrophages mediated by STAT3, HMGA2 could promote the growth of colorectal cancer. 9 HMGA2 could promote the migration, invasion, epithelial-mesenchymal transition and angiogenesis of gallbladder cancer cells. 10 By upregulating Twist1, HMGA2 could increase vasculogenic mimicry and tumor aggressiveness in GC. 13 The increased HMGA2 levels were linked with the TNM stage, lymph node status, vascular invasion, and poor overall survival in GC patients. 21 Silencing of HMGA2 gene expression could induce apoptosis and suppress the proliferation of GC MKN-45 cells. 22 These findings indicated that HMGA2 might serve as a promising therapeutic target and prognostic marker in GC.
In the current study, we found that the HMGA2 rs968697 genetic polymorphism was not only associated with susceptibility to GC, but also with TNM stage and survival prognosis of GC patients after adjusting for confounders such as age, gender, smoking, drinking, TNM stage, and H.pylori infection. Individuals carrying the rs968697 C allele had a noticeably reduced risk of developing GC. GC patients with the C allele had a significantly lower risk of developing stage III + IV tumors. GC patients carrying the rs968697 CT and CC genotypes had a better overall survival prognosis. Furthermore, the rs968697 CC and CT genotypes were associated with low HMGA2 mRNA expression. The rs968697 genetic polymorphism might associate GC occurrence and development by affecting the binding of transcription factors, especially POLR2A, which in turn regulate the expression of HMGA2.
Recent studies have shown that the HMGA2 rs968697 genetic polymorphism could reduce susceptibility to digestive tumors, including hepatoblastoma and colorectal cancer.14,15 However, the findings in other malignancies were inconsistent and needed to be clarified by meta-analysis. The current meta-analysis showed that the rs968697 genetic polymorphism was linked to malignancy susceptibility in the Chinese population. Individuals carrying the rs968697 CC genotype had a noticeably reduced risk of developing malignancy under the homozygote comparison and recessive model.
Although some meaningful results had been achieved, there were still some shortcomings. First, because the subjects came from a small single-center, selection bias was unavoidable in the current case-control study. Second, no precise mechanisms of the rs968697 genetic polymorphism in the occurrence and progression of GC were studied, which can be further explored in in vitro and in vivo experiments based on single-base editing techniques. Third, TSA results suggested that more observational case-control studies were required to assess the role of the rs968697 genetic polymorphism in malignancy susceptibility.
Conclusions
The current investigation discovers that the HMGA2 rs968697 genetic polymorphism may have a protective impact against malignancy incidence and progression of GC in the Chinese population, which can lead to a better understanding of molecular genetics in malignancy.
Supplemental Material
Supplemental Material - The relationship between rs968697 genetic polymorphism and gastric cancer susceptibility, TNM stage and survival prognosis
Supplemental Material for Change in The relationship between rs968697 genetic polymorphism and gastric cancer susceptibility, TNM stage and survival prognosis by Jun Hu and Xiaoting Wang in European Journal of Inflammation
Footnotes
Author contributions
WXT designed the study and collected data. HJ performed experiments and wrote the paper. HJ and WXT interpreted and discussed the results. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by project of natural science research in universities of Jiangsu province (20KJB310029).
Ethical statement
Data availability statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
