Abstract
To investigate whether obstructing the cysteinyl leukotriene receptor-1 (CYSLTR1) with zafirlukast diminishes experimentally induced gastric ulcer (GU) in rats by modulating inflammation and apoptosis. Gastric ulcers affect approximately 10% of the global population and can lead to serious complications such as gastrointestinal perforation and bleeding. Leukotrienes are proinflammatory compounds, and cysteinyl leukotrienes, such as LTC4, LTD4, and LTE4, that are potent proinflammatory mediators. Rats were orally administered a single oral dose of 80 mg/kg of indomethacin to induce GU. The rats were administered an oral dose of 20 mg/kg Zafirlukast. Gastric tissues were collected for macrostructural and microstructural analyses. A portion of gastric tissue was used to assess the genetic expression and protein levels of CYSLTR1, NFκB, TNF-α, IL-1β/4/10, JNK, PKB, and caspase-3. The gastric sections were subjected to hematoxylin/eosin and Masson trichrome staining and immunohistochemical staining with anti-TNF-α and anti-caspase-3 antibodies. Zafirlukast blocked the expression of CYSLTR1. Analysis of micro-images of GU rats revealed damage to surface cells and glandular epithelial cells caused by inflammatory cell infiltration, which was mitigated by Zafirlukast. Additionally, Zafirlukast treatment significantly reduced NFκB, TNF-α, IL-1β, JNK, PKB, and caspase-3 while increasing IL-4 and IL-10. Zafirlukast successfully reduced experimentally induced gastric ulcers in rats. Its mechanism of action includes inhibition of CYSLTR1, diminishing the inflammatory pathway. This is demonstrated by a decrease in the levels of NFκB, TNF-α, and IL-1β, along with an increase in the levels of IL-4 and IL-10. Additionally, Zafirlukast exerted anti-apoptotic effects by downregulating the expression of JNK, PKB, and caspase-3.
Keywords
Introduction
Gastric ulcer (GU) is an inflammatory condition that specifically targets the stomach lining and often arises from a combination of immune-mediated factors and infectious agents, particularly Helicobacter pylori. This condition is typically characterized by upper abdominal discomfort that may present as a dull ache or burning sensation. Patients often report nausea, with the pain usually felt along the lesser curvature of the stomach and potentially radiating beyond the mucosa, indicating deeper tissue involvement. 1 The body initiates a sophisticated and multi-faceted healing process in response to ulceration. This process includes the mobilization and proliferation of various cell types, angiogenesis (formation of new blood vessels) necessary for supplying nutrients and oxygen to the healing tissue, and deposition of extracellular matrix components, critical for structural support. Key regulatory factors such as cytokines, growth factors, and hormonal signals orchestrate this healing cascade, ensuring that repair is efficient and effective. 2
Several interrelated factors have been identified as the primary causes of GU. Helicobacter pylori is A significant contributor, a spiral-shaped bacterium colonizing the stomach lining. This pathogen creates inflammation and damages the protective mucus layer, making the stomach more vulnerable to acidic gastric secretions. 3 Additionally, excessive secretion of gastric acid plays a crucial role in the development of ulcers. Factors such as stress, diet, and lifestyle choices can lead to acid hypersecretion, which erodes the stomach lining and contributes to ulcer formation. 4 Moreover, the prolonged use of non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen and aspirin, is a well-documented risk factor. These medications inhibit the production of prostaglandins, which help maintain the stomach’s protective lining. Without these protective mechanisms, the mucosal barrier is compromised, increasing the risk of injury from gastric acids. 5 Together, these factors create a perfect storm for developing gastric ulcers, emphasizing the importance of understanding their underlying mechanisms for effective prevention and treatment. This will enhance the management and treatment of gastric ulcers and ultimately improve patient outcomes and quality of life.
The standard medical treatment for peptic ulcers, which include both gastric and duodenal ulcers, generally follows a multifaceted approach aimed at promoting healing, reducing symptoms, and preventing complications. Proton pump inhibitors (PPIs) such as omeprazole or lansoprazole, which effectively inhibit gastric acid secretion, thereby creating a less acidic environment in the stomach conducive to ulcer healing. 6 In addition to PPIs, bismuth compounds such as bismuth subsalicylate, are often included in treatment plans. These compounds help protect the gastric lining by forming a protective barrier over the ulcer and exhibit antibacterial properties that can aid in the treatment process. 7 A critical component of ulcer management is the treatment of Helicobacter pylori infection. Standard therapy typically involves a combination of two antibiotics, clarithromycin and amoxicillin, to effectively eradicate this bacterium. Treatment regimens may also include metronidazole or tetracycline as alternatives, depending on antibiotic resistance patterns and patient-specific factors. 8 This combination of medications eliminates the Helicobacter pylori infection, promotes a faster ulcer healing rate, reduces the risk of recurrence, and alleviates associated symptoms, thus providing a holistic approach to managing peptic ulcers.
Leukotrienes (LTs) are generated by the inhibition of COX enzymes using NSAIDs. Cysteinyl leukotrienes, particularly LTC4, LTD4, and LTE4, possess significant proinflammatory properties. LTB4 governs neutrophil functions such as chemotaxis and adhesion. The involvement of LTs in inducing gastric injury is driven by the oxidative stress of their active metabolites and the effects of lipid peroxidation, culminating in tissue inflammation and ischemia. 9 LTs are considered pivotal contributors to peptic ulcers, and the reversible blockage of cysteinyl LT D4 receptors aids in mitigating damage to the stomach mucosa. Furthermore, externally administered LTs results in minimal damage to the gastric mucosa; however, when combined with other harmful agents such as ethanol and aspirin, this effect is associated with severe gastric lesions. 10
Zafirlukast, an FDA-approved drug, is used to treat asthma in both adults and children. It functions as a selective and competitive antagonist of cysteinyl LT-1 receptor (CYSLTR1). Its mechanism involves competing with LTC4, LTD4, and LTE4 at the CYSLTR1 to impede inflammation induced by LTs. 11 Our research focused on investigating the potential effects of inhibiting CYSLTR1 with Zafirlukast on the reduction of gastric ulcers induced experimentally in rats. We sought to determine whether this approach influences inflammation and apoptosis. We evaluated the gene expression and protein levels in the stomach for caspase-3, cysteinyl leukotriene receptor 1 (CYSLTR1), interleukin (IL)1β/4/10, Jun N-terminal kinase (JNK), nuclear factor (NF)κB, protein kinase B (PKB), and tumor necrosis factor-α (TNF-α).
Methods
Animals and treatment outlines
The study involved 40 Sprague-Dawley rats, each weighing 180 200 g. The rats were meticulously housed in a controlled environment designed to maintain optimal living conditions, with temperature regulated between 20 and 22 °C. Furthermore, a carefully structured light cycle consisting of 12 h of bright light, followed by 12 h of complete darkness, was used to mimic the natural circadian rhythms of animals, thereby enhancing the validity of the experimental outcomes. Before the research was initiated, the study protocol was thoroughly scrutinized and approved by the Research Ethics Committee of the Faculty of Pharmacy at Delta University for Science and Technology (approval number FPDU19/2022). The inclusion criteria were rats between 8 and 12 weeks of age, exhibiting lively and vigorous movement, possessing a pristine coat of fur devoid of any lesions, and demonstrating normal, consistent stool production. The exclusion criteria included rats that exhibited weight loss exceeding 10% of their baseline body weight during the acclimatization period or displayed any symptoms of illness following the adaptation phase. Rats were divided into four groups (Refer to Figure 1 for timeline of the animal treatment in the study):

Timeline of the animal treatment in the study.
Control group
Ten rats underwent a 24-h food deprivation period while being provided with water. Subsequently, 0.5% carboxymethyl cellulose (CMC) was orally administered via gavage, and the rats remained untreated for the experiment.
Control treated with Zafirlukast
In the initial phase, rats in the control group received CMC treatment. Subsequently, they were orally administered a daily dose of 20 mg/kg Zafirlukast (Selleck Chemicals GmbH, Cologne, Germany) for seven consecutive days, commencing one day after CMC treatment.
GU group
The rats were subjected to a 24-h food deprivation period with access to water, followed by oral administration of 80 mg/kg indomethacin to induce gastric ulcers.12,13
GU treated with Zafirlukast
Following the induction of gastric ulcers, a group of ten rats were orally administered 20 mg/kg zafirlukast once daily for seven days. This treatment commenced one day after indomethacin administration of indomethacin. 14
Sample collection
The entire stomach was excised, measured, and weighed after the animals were sacrificed. Subsequently, a segment of the glandular portion of the stomach was immersed in 10% buffered formalin solution for fixation and later utilized for morphological and immunohistochemical analyses. Another stomach segment was homogenized in a sodium potassium phosphate buffer solution at pH 7.4. The resulting supernatant was stored at -80°C for future use.
Morphologic analysis and immunohistochemistry
The stomach tissue was meticulously sectioned into five-micrometer-thick slices to ensure precise analysis. A subset of these segments was subjected to hematoxylin and eosin (H&E) staining to highlight cellular morphology and tissue structure. In addition, some sections were stained with Masson trichrome to investigate tissue fibrosis. In contrast, another subset underwent immunohistochemical staining using a monoclonal antibody against transforming growth factor (TGF)-β (Sigma Aldrich Chemicals Co., St. Louis, MO, USA – Cat number SAB5702296). This staining was performed at a controlled temperature of 4 °C, in accordance with the established protocols of our research group to maintain antibody integrity and maximize binding efficacy. To capture and analyze the resulting stained sections, a digital camera-assisted computer system (Nikon Digital Camera, Japan) was used to enable high-resolution imaging and facilitate an in-depth investigation of the histological features and molecular markers present within the tissue.15–18
Enzyme-linked immunosorbent (ELISA) assay
The study employed ELISA kits that are commercially available from MyBioSource, Inc., situated in San Diego, CA, US, to evaluate caspase-3 (Cat number MBS450533), CYSLTR1 (Cat number MBS2511358), IL-1β (Cat number MBS2023030), IL-4 (Cat number MBS2023426), IL-10 (Cat number MBS2020828), JNK (Cat number MBS760947), PKB (Cat number MBS263708), and TNF-α (Cat number MBS175904). The assays were conducted according to the manufacturer’s instructions using a UV-VIS Double-Beam PC Scanning Spectrophotometer (Labomed Inc., Los Angeles, CA, US.
Quantitative real-time polymerase chain reaction (RT-PCR)
The gene expression levels of caspase-3, CYSLTR1, IL-1β/4/10, JNK, PKB, NFκB, and TNF-α mRNA in rat gastric lysate were quantified following established protocols.19–23 Total RNA was obtained using the RNeasy Mini kit (Qiagen, USA), with its concentration measured by Maxima® SYBR Green/Fluorescein Master Mix (Fermentas, USA). Following this, 1 µg of RNA was converted into complementary DNA (cDNA) using the QuantiTect® Reverse Transcription Kit (Qiagen, USA). The mRNA expression levels of caspase-3, CYSLTR1, IL-1β/4/10, JNK, PKB, NFκB, and TNF-α in rat gastric lysates were quantified with Maxima® SYBR Green/Fluorescein qPCR Master Mix alongside the Rotor-Gene Q (Qiagen, USA). The primer sets for proteins used in the study were summarized in Table 1. The thermal cycling conditions for RT-qPCR consisted of one cycle of reverse transcription at 55°C for 10 min, followed by enzyme inactivation at 59°C for 2 min, and then forty amplification cycles of 95°C for 10 s, 55°C for 10 s, and finally 72°C for 30 s. A final extension was conducted at 72°C for 5 min. Rat glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used for housekeeping and internal reference. The results from the RT-PCR analysis were expressed as Cycle threshold (Ct) values. The PCR data sheet includes the Ct values for the target genes about the reference housekeeping gene, GAPDH. A control sample was incorporated to assess the gene expression of each specific gene. The Relative Quantification (RQ) for each target gene was calculated and normalized to the housekeeping gene using the delta-delta Ct (ΔΔCt) methodology. The RQ for each gene was determined using the formula 2-∆∆Ct.
The primer set used to detect gene expression in rats.

Statistical analysis
In this study, quantitative variables were presented using mean values and standard errors. To evaluate the normality of the sample distribution, we employed the Kolmogorov-Smirnov (K-S) test to ensure that the critical assumptions for parametric analyses were satisfied. One-way analysis of variance (ANOVA) was conducted to assess potential differences between groups. In instances where the ANOVA indicated statistically significant differences, the post hoc Bonferroni correction test was used to adjust the p-values, helping to control for Type I errors in multiple comparisons. All statistical analyses were performed using SPSS software version 20 (IBM, Chicago, IL, USA). The significance threshold was established at p < .05.
Results
Effect of Zafirlukast on the expression of CYSLTR1
GU remarkably enhanced gene expression and gastric protein levels of CYSLTR1 in experimental rats. Specifically, gene expression increased by a factor of 3.87, whereas the associated protein levels rose by 4.21 times compared with baseline measurements. Notably, treatment with the leukotriene receptor antagonist Zafirlukast effectively mitigated GU-induced upregulation of CYSLTR1 in the experimental group of rats, demonstrating a significant reduction in expression levels. Importantly, Zafirlukast did not exert any observable effects in the control group that did not receive GU treatment (see Figure 2a and b for detailed observations).

Effect of GU and 20 mg/kg Zafirlukast on gene expression of CYSLTR1 (a) and its gastric protein levels (b). Representative images of the whole stomach from different treated groups (c). The images of control rats exhibited a normal appearance without any observable abnormalities. The image of GU’s stomach exhibited mucosal hemorrhagic lesions and areas of ulceration. In contrast, the stomach from the GU treated with Zafirlukast showed a substantial reduction in hemorrhage and ulcer areas, indicating a positive impact of the treatment. Finally, statistical analysis of mucus production (d), and stomach/body weight ratio (e). The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p<0.05. # Significant difference as compared with GU group at p<0.05. CYSLTR1, cysteinyl leukotriene receptor-1; GU, gastric ulcer.
Effect of Zafirlukast on GU
Morphological analyses of the entire stomach in the control group demonstrated a well-preserved structure characterized by smooth and continuous mucosal plicae with no signs of edema or inflammation. In contrast, examination of the entire stomach in the GU group revealed notable swelling and vascular congestion, accompanied by the presence of mucosal hemorrhagic lesions, highlighting significant pathological changes compared with the control group. Additionally, treatment with indomethacin resulted in a remarkable 9.25-fold increase in mucus production, which correlated with an increased stomach-to-body weight ratio, indicating a physiological response to the irritative effects of the drug. However, when rats in the GU group were treated with Zafirlukast, mucosal hemorrhagic lesions were significantly alleviated, with a 61% reduction in mucus production, indicating its protective effects on the gastric mucosa. Furthermore, Zafirlukast treatment improved the stomach-to-body weight ratio in these rats compared to the untreated GU group rats, suggesting a recovery in gastric function and morphology (Figure 2c–e).
Effect of Zafirlukast on gastric tissue structure
Histopathological examination revealed that the gastric glands in the control group exhibited a closely arranged and orderly configuration, with epithelial cells remaining intact and devoid of signs of congestion or edema. In contrast, the gastric mucosa of the GU group demonstrated significant disorganization of the epithelial cell structure, characterized by pronounced edema and infiltration by inflammatory cells, as indicated by the black arrows in the accompanying images. Additionally, extensive mucosal necrosis was evident in this group, highlighting the severity of the alterations induced by the gastrointestinal insult. Remarkably, the administration of Zafirlukast to indomethacin-treated animals resulted in the partial restoration of the mucosal layer architecture, suggesting its potential protective efficacy against the damage observed. This finding is illustrated in Figure 3.

Gastric sections stained with hematoxylin/eosin in the control group (a), the control group treated with Zafirlukast (b), the GU group (c), and the GU treated with Zafirlukast (d). Yellow arrows represented focal mucosal inflammation, and black arrows represented extensive mucosal necrosis in the GU group. Treatment of GU rats with Zafirlukast reduced mucosal inflammation and necrosis. Scale bar 50 μm. GU, gastric ulcer.
Microscopic images of glandular gastric sections stained with Masson’s trichrome demonstrate a clear distinction between the control group and those treated with zafirlukast, with neither group exhibiting any signs of excessive collagen deposition. In contrast, the glandular gastric sections obtained from the peptic ulcer group reveal a significant presence of bluish collagen deposition within the mucosal layer, indicative of fibrosis. Additionally, these sections display pronounced mucosal necrosis accompanied by extensive infiltration of leukocytic cells, highlighting the inflammatory response associated with peptic ulcers. Sections from GU treated with zafirlukast revealed reduction in the mucosal necrosis (Figure 4).

Gastric sections stained with Masson trichrome in the control group (a), the control group treated with Zafirlukast (b), the GU group (c), and the GU treated with Zafirlukast (d). The black arrow represents mucosal necrosis, and the yellow arrows represent mucosal leukocytic cell infiltration. Treatment of GU rats with Zafirlukast reduced mucosal inflammation and necrosis. Scale bar 50 μm. GU, gastric ulcer.
Effect of Zafirlukast treatment on GU-induced alteration in NFκB and IL-1β expression
In the comprehensive analysis depicted in Figure 5, GU group demonstrated a significant upregulation in the expression levels of the genes NFκB and IL-1β, which increased by 3.69-fold and 2.89-fold, respectively, in comparison to the control group. This pronounced escalation in gene expression was paralleled by an increase in gastric protein content, with NFκB and IL-1β proteins exhibiting impressive rises of 4.13-fold and 3.39-fold, respectively. Furthermore, the administration of Zafirlukast, a leukotriene receptor antagonist, resulted in a marked attenuation of the heightened expression levels of NFκB and IL-1β in the GU group, effectively reverting them towards baseline levels. Importantly, this intervention did not appear to influence the control group, indicating a specific modulatory effect of zafirlukast on inflammatory pathways activated in the context of gastric ulceration.

Effect of GU and 20 mg/kg Zafirlukast on gene expression of NFκB (a), and IL-1β (c), as well as the gastric level of NFκB (b), and IL-1β (d) representing an increase in the expression of both NFκB and IL-1β in GU rats that was ameliorated by Zafirlukast treatment. The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p < .05. # Significant difference as compared with GU group at p<0.05. GU, gastric ulcer; IL, interleukin; NFκB, nuclear factor κB.
Effect of Zafirlukast on GU-induced expression of TNFα
In the assessment of gastric tissues, a striking increase of 3.29-fold and 3.83-fold in the expression levels of TNF-α genes, along with a corresponding elevation in gastric protein concentrations, was detected in the GU group when compared to the control group. Notably, these inflammatory effects were significantly attenuated following zafirlukast treatment, suggesting its potential therapeutic benefit for managing gastric inflammation. Immunohistochemical analysis further corroborated these findings, revealing a marked increase in TNF-α expression in the gastric tissues of rats suffering from GU. The immunostaining results illustrated a clear distinction, highlighting elevated levels of TNF-α in the GU-affected rats. Conversely, Zafirlukast treatment resulted in a notable reduction of TNF-α immunostaining in the GU group, effectively lowering the inflammatory marker back towards baseline levels, while importantly, this treatment did not alter TNF-α expression in the control group (Figure 6).

Effect of GU and 20 mg/kg Zafirlukast on gene expression of TNF-α (a) and its protein level in gastric tissues (b). Gastric sections were stained with anti-TNF-α in the control group (c), the control group treated with Zafirlukast (d), the GU group (e), and the GU treated with Zafirlukast (f). Scale bar 100 μm. The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p < .05. # Significant difference as compared with GU group at p < .05. GU, gastric ulcer; TNF-α, tumor necrosis factor-α.
Effect of Zafirlukast on GU-induced downregulation of anti-inflammatory cytokines
GU resulted in a 63% reduction in the gene expression levels of IL-4 and an impressive 59% decrease in IL-10 gene expression. These changes in gene expression correlated with notable reductions in the protein levels of these cytokines in the gastric tissue, with IL-4 levels decreasing by 67% and IL-10 levels by 56% compared to the control group of rats. Furthermore, administration of Zafirlukast to the GU-afflicted rats effectively restored the previously diminished levels of IL-4 and IL-10 gene expression to near-normal levels. Importantly, administration of Zafirlukast did not exert any significant effects on the control rats (Figure 7).

Effect of GU and 20 mg/kg Zafirlukast on gene expression of IL-4 (a) and IL-10 (c) as well as the gastric level of IL-4 (b) and IL-10 (c) representing a reduction in the expression of IL-4 and IL-10 in GU rats which was reversed by treating with Zafirlukast. The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p < .05. # Significant difference as compared with GU group at p < .05. GU, gastric ulcer; IL, interleukin.
Effect of Zafirlukast on GU-induced expression of PKB and JNK
GU treatment resulted in a remarkable upregulation in the gene expression levels of JNK and PKB, showing increases of approximately 3.97-fold and 4.37-fold, respectively, compared to the control group. Additionally, there was a significant rise in the gastric tissue protein concentrations of JNK and PKB, with elevations noted at 4.23-fold and 4.12-fold, respectively. Notably, the administration of Zafirlukast to the GU rat model effectively reversed these biochemical alterations, bringing the expression levels back to baseline, without exerting any observable effects on the control animals (Figure 8).

Effect of GU and 20 mg/kg Zafirlukast on gene expression of JNK (a) and PKB (c) and the gastric protein levels of JNK (b) and PKB (d) representing an increase in the expression of JNK and PKB that was reduced by treating with Zafirlukast. The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p < .05. # Significant difference as compared with GU group at p < .05. GU, gastric ulcer; JNK, Jun N-terminal kinase; PKB, protein kinase B.
Effect of Zafirlukast on GU-induced expression of caspase-3
In the evaluation of gastric tissues, a significant increase in the expression levels of caspase-3 genes and associated gastric protein concentrations was noted in the GU group, exhibiting enhancements of approximately 3.82-fold and 3.73-fold, respectively, compared with the control group. Importantly, the administration of Zafirlukast effectively mitigated the elevated expression levels in the GU group, suggesting a protective effect against gastric tissue damage. These findings were further substantiated by caspase-3 immunostaining, which revealed a pronounced increase in gastric caspase-3 expression in GU-affected rats. Conversely, post-treatment with Zafirlukast resulted in a notable decrease in the levels of gastric caspase-3 in the GU group, indicating the reversal of pathological changes. Notably, no significant alterations in caspase-3 expression were observed in the control group, reinforcing the specificity of the effects of zafirlukast on gastric ulcers (Figure 9).
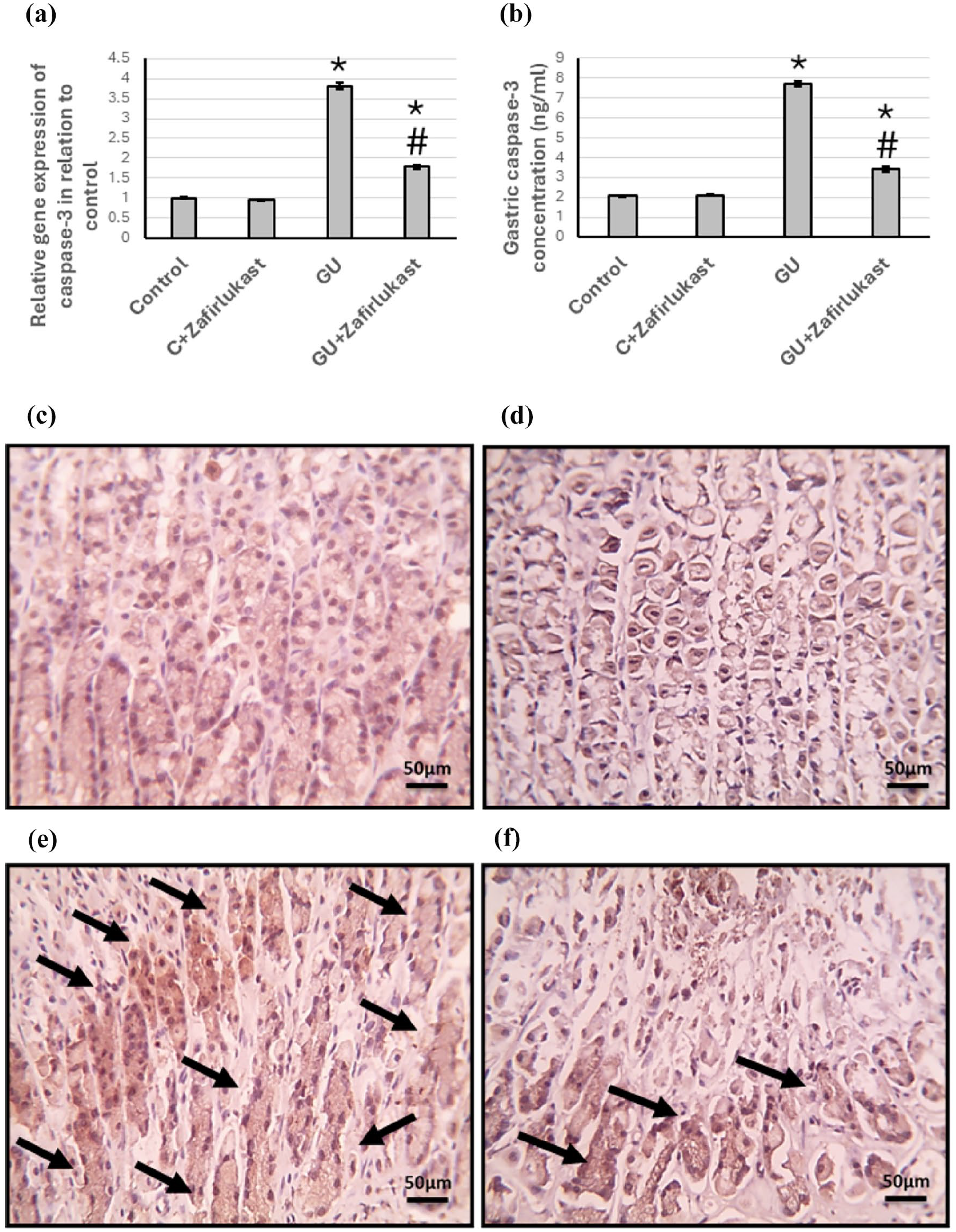
Effect of GU and 20 mg/kg Zafirlukast on gene expression of caspase-3 (a) and its protein level in gastric tissues (b). Gastric sections were stained with anti-caspase-3 in the control group (c), the control group treated with Zafirlukast (d), the GU group (e), and the GU treated with Zafirlukast (f). Scale bar 100 μm. The data are presented as Mean ± SEM, utilizing ten rats per group. Statistical analysis was conducted using ANOVA, followed by post hoc Bonferroni correction testing. * Significant difference as compared with the control group at p < .05. # Significant difference as compared with GU group at p < .05. GU, gastric ulcer.
Discussion
Gastric ulcers (GU) are well-documented medical conditions with a range of serious complications, including gastrointestinal bleeding, perforation of the stomach lining, and intestinal obstruction. The incidence of gastric ulcers has seen a notable rise globally, a trend largely attributed to the increased consumption of non-steroidal anti-inflammatory drugs (NSAIDs) and excessive alcohol intake. 24 In our investigation, we utilized established animal models to effectively induce gastric ulcers in a cohort of fasting rats. Specifically, we administered a single oral dose of indomethacin, a potent NSAID known for its ability to induce gastrointestinal inflammation, ulcer formation, and hemorrhagic conditions in the gastric mucosa. This method has been validated in previous studies as an effective means of simulating GU. 24 Post-administration, we assessed morphological alterations within the stomach. Our findings revealed significant pathological changes characterized by extensive hemorrhage and large ulcerative lesions distributed throughout the gastric lining. Furthermore, there was a marked increase in mucus production in response to injury and a significant elevation in the stomach-to-body weight ratio. This increase indicated gastric distension secondary to inflammation and hemorrhage, while the reduction in overall body weight suggested systemic effects, including decreased appetite and metabolic wasting, commonly resulting in discomfort and a burning sensation in affected subjects. Microscopic analysis of gastric tissue sections from indomethacin-treated rats stained with hematoxylin and eosin revealed pronounced focal mucosal inflammation and widespread necrotic areas within the mucosa, while sections stained with Masson trichrome revealed increased collagen deposition within the mucosal layer, indicative of fibrosis, mucosal necrosis accompanied by extensive infiltration of leukocytic cells.
The pharmaceutical treatment of GU disorders can incur substantial costs and may be associated with significant adverse effects. Recognizing these challenges, our study aimed to investigate the potential therapeutic advantages of CYSLTR1 receptor inhibition as a novel intervention for GU. Specifically, we assessed the efficacy of zafirlukast, an FDA-approved medication for the treatment of asthma, as a selective and competitive inhibitor of CYSLTR1 for the management of GU disorders. Zafirlukast functions by competitively inhibiting the actions of leukotrienes, including LTC4, LTD4, and LTE4, at the CYSLTR1 receptor, thereby attenuating the inflammatory response driven by these mediators. 11 Following the administration of Zafirlukast, we observed significant improvements in gastric morphology, evidenced by a reduction in mucus hypersecretion and a favorable decrease in the stomach-to-body ratio. Histological examination using hematoxylin and eosin staining revealed a marked enhancement in the structural integrity of the gastric tissue, highlighting the potential of the drug to ameliorate tissue damage associated with inflammatory processes. Sections stained with Masson trichrome revealed a reduction in the mucosal necrosis. Given these promising findings, we proceeded to the subsequent phase of our investigation, which focused on elucidating the precise mechanism of action of Zafirlukast in GU.
Inflammation is a critical factor in GU pathogenesis. Increased gut permeability, which results from a breakdown of the gastrointestinal barrier, serves as a catalyst for the initiation of inflammation. 25 This process activates macrophages, which are key immune cells, subsequently leading to an upsurge in the production of pro-inflammatory cytokines, such as NFκB and TNF-α.26,27 The activation of NFκB, an essential regulator in the development and progression of GU, plays a pivotal role in modulating inflammation within the gastric lining. 28 It exerts its influence by regulating the secretion of various cytokines and chemokines, which are signaling molecules that mediate and regulate immunity and inflammation. 29 As a consequence of this inflammatory cascade, tissue damage in the gastric mucosa becomes exacerbated. Compounds that possess anti-inflammatory properties and demonstrate efficacy in treating acetic acid-induced gastric ulcers function primarily by inhibiting NFκB activity. 30 This inhibition helps to mitigate the inflammatory response, thereby promoting healing. Furthermore, TNF-α, a potent pro-inflammatory cytokine, initiates an acute inflammatory response by recruiting neutrophils to the site of injury within the gastric mucosa. This influx of neutrophils can disrupt gastric microcirculation and hinder angiogenesis, the formation of new blood vessels, thereby impeding the vital process of cell proliferation at ulcer sites and delaying ulcer healing. 31 Hence, targeting TNF-α in the ulcerated area has the potential to significantly enhance the healing process. Additionally, IL-1β, another key inflammatory marker predominantly produced by activated inflammatory cells such as monocytes and macrophages, plays a significant role in various inflammatory responses. The elevated levels of IL-1β in the gastric mucosa during episodes of GU can further exacerbate the condition by promoting neutrophilic infiltration into areas of scarred mucosa. 32 This phenomenon may contribute to an increased recurrence of experimental GU.
In our study, we observed that administration of Zafirlukast to rats with GU significantly attenuated the overexpression of critical inflammatory mediators, specifically NFκB, TNF-α, and IL-1β, which are known to play key roles in the pathophysiology of gastric ulceration. Previous research has demonstrated that Zafirlukast effectively diminishes the levels of IL-1β in models of indomethacin-induced gastric ulcers in rats, suggesting its potential therapeutic application in managing inflammation associated with gastric mucosal injury. 14 Furthermore, prior investigations have indicated that Zafirlukast also reduces the expression of NFκB and TNF-α in various animal models, including those involving hepatic ischemia-reperfusion injury in rats. 33 These findings highlight Zafirlukast’s broad anti-inflammatory properties across different pathological conditions. However, it is noteworthy that no earlier studies have specifically explored the efficacy of Zafirlukast in downregulating NFκB and TNF-α expression in the context of GU. This research gap underscores the significance of our findings and suggests that zafirlukast could be a novel therapeutic agent for mitigating GU-related inflammation.
GU development occurs when there is an imbalance between the protective and destructive factors in the body. Key protective factors include anti-inflammatory cytokines such as IL-4 and IL-10, which play crucial roles in counteracting the deleterious effects of pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6. 34 This counteraction is essential for reducing damage to gastric mucosal tissues. IL-4 is a crucial cytokine synthesized in response to the activation of T helper type 2 (Th2) cells and plays a pivotal role in the regulation and orchestration of the immune response. This pleiotropic cytokine influences the differentiation and function of various immune cells, particularly in promoting the proliferation and activity of B cells, which are responsible for antibody production. 35 IL-4 exerts its biological effects through interaction with a specific receptor complex that comprises the interleukin-4 receptor alpha chain (IL-4Rα) and the common gamma chain (γc). This receptor binding initiates a cascade of intracellular signaling events, primarily via the Janus kinase-signal transducer and activator of the transcription (JAK-STAT) signaling pathway. Upon receptor engagement, Janus kinases (JAK1 and JAK3) are activated, leading to the phosphorylation and activation of transcription factors in the STAT family, particularly STAT6. 36 The activated STAT6 dimers translocates to the nucleus where it promotes the transcription of genes associated with Th2 cell responses, including those involved in B cell activation, IgE class switching, and the production of other Th2 cytokines. This intricate signaling mechanism underscores IL-4’s essential role in defining the adaptive immune response and its implications in allergic reactions and various immune disorders. This pathway is crucial for promoting B cell differentiation and enhancing immunoglobulin production, thus contributing to an adaptive immune response. 37
Complementarily, IL-10 functions as a potent anti-inflammatory cytokine that plays a crucial role in modulating the immune response. IL-10 effectively suppresses the pro-inflammatory response by inhibiting the activation of macrophages, which are key players in inflammatory process. This inhibition leads to a significant reduction in the production of pro-inflammatory cytokines, such as TNF-α and IL-6, helping to prevent excessive tissue damage often associated with chronic inflammation. 38 Moreover, IL-10 contributes to its anti-inflammatory effects by downregulating the expression of major histocompatibility complex (MHC) class II molecules on antigen-presenting cells. This downregulation decreases the ability of these cells to present antigens to T cells, thereby attenuating the activation of helper T cells, which typically drive inflammatory responses. Additionally, IL-10 reduces the expression of co-stimulatory signals, such as CD80/CD86, which are necessary for T cell activation, further curbing the immune response. 39 Through these mechanisms, IL-10 plays a protective role in the gastric epithelium, mitigating the risk of injury from inflammatory processes and promoting tissue healing and regeneration. The multifaceted action of IL-10 is vital for maintaining homeostasis and preventing the progression of inflammatory diseases.
In our study, conducted on rat models of GU, we observed a significant reduction in IL-4 and IL-10 levels. Notably, administration of Zafirlukast led to a remarkable upregulation of IL-4 and IL-10 expression. This increase was associated with a marked improvement in the histopathological features of the GU, indicating enhanced healing and recovery of the gastric mucosa. This pivotal study is the first to establish the potential therapeutic role of Zafirlukast in modulating the cytokine profiles in GU, suggesting that its use may extend beyond respiratory disorders to gastrointestinal applications.
MAPK pathways are integral to mediating a variety of cellular responses, particularly those associated with inflammation. These pathways effectively integrate signals derived from intracellular sources, such as growth factors and cytokines, as well as extracellular stimuli, including stressors and pathogens. 40 The MAPK are comprised of several key members, notably extracellular signal-regulated kinases 1 and 2 (Erk1/2), JNK, and p38 MAPK. Upon phosphorylation and subsequent activation, these kinases can trigger a wide array of biological processes, including inflammatory responses, cellular proliferation, and programmed cell death (apoptosis). 41 Selective activation of specific subgroups within the MAPK family has been shown to regulate the synthesis and release of pro-inflammatory mediators, an effect that has been especially well-documented in ethanol-induced GU in murine models. 42 This selective activation can lead to the upregulation of various cytokines and chemokines, as well as the enhancement of cell survival and proliferation pathways that contribute to the pathology of inflammation. Furthermore, therapeutic interventions aimed at inhibiting the activation of the MAPK pathways are promising for alleviating gastric mucosal injury. Targeting these pathways might reduce the production of inflammatory mediators and protect the gastric epithelium from damage. Such interventions could prove beneficial not only in the context of ethanol-induced injuries but may also have broader implications in managing various inflammatory conditions affecting the gastrointestinal tract. 43 The findings of this study demonstrated that Zafirlukast significantly diminishes the overexpression of JNK triggered by GU. This study represents a pioneering investigation into the capacity of zafirlukast to attenuate JNK expression, specifically within the context of GU.
Our investigation focused on the intricate role of PKB in GU, emphasizing its significance in various pathological processes. AKT is composed of three distinct isoforms: PKBα (also known as AKT1), PKBβ (AKT2), and PKBγ (AKT3), each with unique functions and regulatory mechanisms. 44 The AKT/PKB signaling pathway is crucial for mediating various cellular responses. They play key roles in regulating the cell cycle, promoting cellular proliferation, and orchestrating autophagy and apoptosis. Within the context of GU conditions, AKT serves as a critical modulator of the interaction between autophagy, the process by which cells degrade and recycle components, and apoptosis, the programmed cell death mechanism. 45 The activation of the AKT signaling pathway is vital for facilitating cell proliferation and enhancing cellular motility, which is essential for tissue homeostasis and repair. Previous studies have indicated that activation of the PKB signaling pathway can lead to significant disruptions in autophagic flux, thereby impairing the body’s ability to eliminate damaged or unnecessary cellular components. Our study demonstrated that Zafirlukast effectively reduced GU-induced JNK overexpression, potentially contributing to the survival of damaged cells undergoing programmed cell death. 46 Our study demonstrated that Zafirlukast effectively reduced the overexpression of GU-induced JNK. This investigation represents the initial evidence of the potential of Zafirlukast to lower JNK expression in the context of GU.
Caspase-3 plays a crucial role as a key executioner enzyme in the intricate process of apoptosis, the programmed cell death necessary for maintaining cellular homeostasis. When mucosal tissue is damaged, elevated levels of pro-inflammatory cytokines such as TNF-α and reactive oxygen species (ROS) can accumulate. This surge often results from inflammatory cell infiltration, which exacerbates tissue damage and disrupts normal cellular functions. 47 The presence of these pro-inflammatory mediators initiates a complex cascade of molecular signaling events. This series of interactions activates various caspases, a family of cysteine proteases, ultimately activating caspase-3. Once activated, caspase-3 catalyzes the breakdown of critical cellular substrates, resulting in lipid peroxidation within the cellular membranes. This peroxidative damage is detrimental, as it compromises membrane integrity, triggers mitochondrial dysfunction, and ultimately leads to cell death by apoptosis. 48 These mechanisms are of particular significance in the context of nonsteroidal anti-inflammatory drugs (NSAIDs), which have been documented to induce apoptosis specifically in the gastric mucosa. Understanding the precise biochemical pathways through which caspase-3 is activated in response to NSAID exposure can provide valuable insights into the adverse effects of these medications. It could help in developing strategies to mitigate their impact on gastric health. 49 Our research findings indicate that Zafirlukast effectively mitigated the increased expression of caspase-3 triggered by GU. This study provides preliminary evidence for the potential of zafirlukast to reduce caspase-3 expression in GU.
In our discussion, we aim to highlight the potential of using selective and competitive antagonists of the CYSLTR1 receptor in treating gastric ulcers. Notably, Zafirlukast, which has been previously documented for its therapeutic effects on gastric ulcers, operates primarily through the inhibition of the prostaglandin E2 (PGE2) pathway. 14 Our manuscript also examined its effect on reducing the inflammatory pathway and promoting anti-apoptotic effects. Among the various CYSLTR1 antagonists investigated, Montelukast stands out as the most prominent agent in managing gastric ulcers. Research has demonstrated that Montelukast exhibits significant anti-ulcerogenic properties, showing marked improvements both macroscopically and microscopically compared to traditional treatments such as famotidine. 50 Montelukast’s efficacy in enhancing gastric health is attributed to several key mechanisms. It has been observed to mitigate the progression of gastric ulcers by actively inhibiting oxidative stress, which is known to exacerbate mucosal damage. 51 Additionally, Montelukast possesses antisecretory properties that help regulate gastric acid production, thereby reducing irritation and promoting healing. Furthermore, it exerts anti-apoptotic effects by preserving cellular integrity in the gastric mucosa. 52 The therapeutic advantages of CYSLTR1 antagonism represent a promising avenue for treating gastric ulcers, offering renewed hope for patients suffering from this condition. The potential to improve healing outcomes through targeted receptor modulation highlights the importance of continued research in this area.
The research presented here has limitations stemming from choosing rats as the model organism. It is essential to acknowledge that rats’ metabolic pathways and drug metabolites differ significantly from those in humans. This variation can result in discrepancies in drug absorption, distribution, metabolism, and excretion, potentially leading to different therapeutic responses and side effects in humans. Consequently, the results of this study should be interpreted with caution when extrapolating them to human applications. Furthermore, it is essential to highlight multiple established methods for inducing GU in rat models, each with its own implications for data interpretation. Additionally, although this study focused on assessing the effects of the administered dose at the gastric level, we did not quantify the broader systemic and pulmonary effects. Despite these methodological limitations, this study provided valuable insights into the potential efficacy of zafirlukast as a therapeutic agent against GU. These findings could serve as a foundational framework for future research investigating the application of Zafirlukast in clinical settings involving human patients.
Conclusion
Zafirlukast, a selective leukotriene receptor antagonist, has demonstrated significant efficacy in mitigating experimentally induced GU in rat models. Its primary mechanism of action involves specific inhibition of CYSLTR1, which plays a critical role in mediating inflammatory processes within the gastrointestinal tract. This inhibitory action leads to a marked attenuation of the inflammatory cascade, as evidenced by a substantial reduction in the expression levels of key pro-inflammatory cytokines such as NFκB, TNF-α, and IL-1β. Concurrently, there is a notable increase in the expression of anti-inflammatory cytokines, including IL-4 and IL-10, which further contributes to its therapeutic effects. Moreover, Zafirlukast exhibits significant anti-apoptotic properties, which are facilitated by the downregulation of various signaling molecules such as JNK, PKB/Akt, and caspase-3. This downregulation helps preserve cellular integrity and promotes cell survival in the context of inflammatory stress. The multifaceted and complex nature of the mechanism of action of zafirlukast underscores its promising potential as a therapeutic agent for alleviating gastroenteropathy, providing a multifactorial approach for managing inflammatory gastrointestinal conditions.
Footnotes
Acknowledgements
Authors declare none.
Author Contributions
HMH, AB, RD, AN, DA, DFB and MZN were responsible for performing the biochemical analysis. HMH and MMHA were responsible for performing the animal experiments. HMH and MMHA performed pathological and immunohistochemistry analysis. HMH, AB, RD, AN, DA, DFB and MZN performed the statistical analysis. MMHA developed the study concept and supervised the work. All coauthors helped develop and design the present study. All authors contributed to the writing of the manuscript and approved the final version.
Data availability
Data were uploaded as excel file with the submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. The research protocol received approval from the Research Ethics Committee of the Faculty of Pharmacy, Delta University for Science and Technology (FPDU-REC) under approval No. (FPDU19/2022).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
