Abstract
Introduction
SARS-CoV-2, the virus that is responsible for causing COVID-19, was declared a Public Health Emergency in January 2020, and a pandemic in March of 2020. 1 Mannose binding lectin (MBL) is a component of innate immunity as well as the coagulation cascade. 2 Mannose binding lectin levels and genotype have been reported to be both protective and potentially increase risk for a severe course with COVID-19.3–6 Blood clots during COVID-19 infection are known to be a common co-morbid condition.3,7–9 Depending on the type of infection, those with MBL deficiency have been reported to be either more or less susceptible to a severe course of illness.10–16 We hypothesize that those with MBL deficiency will be at more risk of a moderate to severe COVID-19 infection, but not mortality.
Methods
IRB approval was granted for this retrospective cross-sectional analysis. An electronic medical records search was conducted for MBL deficiency and COVID-19 positive tests from March 2020-August 2022. Inclusion criteria were adults and children aged 3 and older with known mannose binding lectin deficiency and positive COVID-19 test between March 2020 and August 2022. Exclusion criteria were individuals without MBL deficiency and those without a positive COVID-19 test. MBL deficiency was defined as MBL level less <100. 17 MBL genotypes were not investigated. Individualized chart analysis was conducted, and additional known immunodeficiencies were tracked along with co-morbid health conditions. COVID-19 positive results were determined by test result and patient reported messages during the infection and at routine health care visits. Statistical analysis was performed using Graphpad Prism 9 with 95% confidence level. Given the rarity of MBL deficiency, power analysis was not performed. Public data on rate of hospitalization during January 2021 for the general public was used to compare the rate of hospitalization with the MBL deficient cohort. Mild COVID-19 was defined as anyone that did not seek care at an emergency department or require hospitalization. Moderate COVID-19 infection was defined as anyone that went to the Emergency Department and severe infection was defined as anyone that was hospitalized. Critical was reserved for those that required mechanical ventilation (MV), extracorporeal membrane oxygenation (ECMO) or care in intensive care unit (ICU). Treatment plans for each COVID-19 infection were determined by various providers assuming care of these patients based on national guidelines at the time of infection, therefore, there was no standardized treatment for our study population.
Results
MBL deficient individuals with COVID-19 characteristics and outcomes.
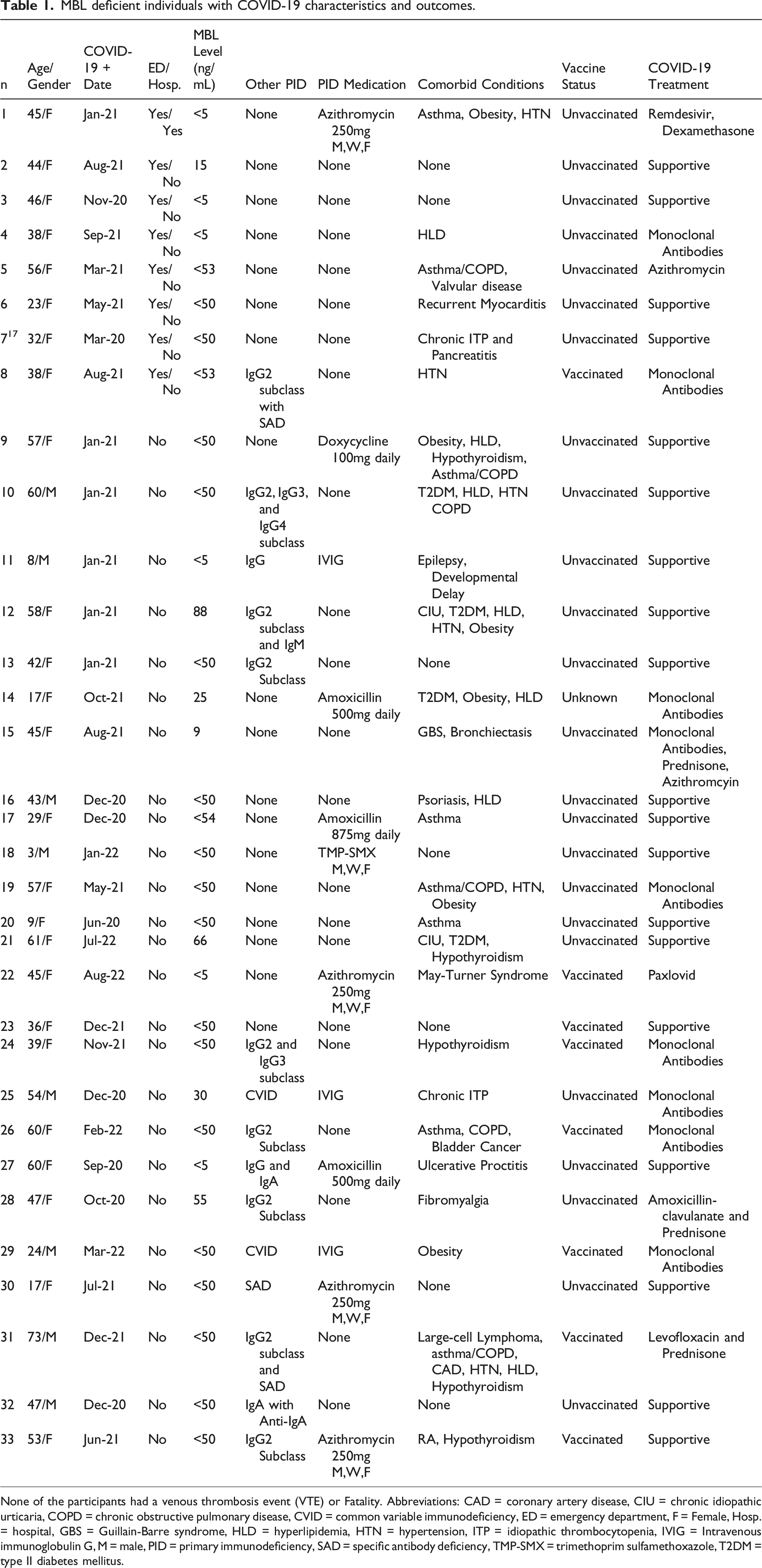
None of the participants had a venous thrombosis event (VTE) or Fatality. Abbreviations: CAD = coronary artery disease, CIU = chronic idiopathic urticaria, COPD = chronic obstructive pulmonary disease, CVID = common variable immunodeficiency, ED = emergency department, F = Female, Hosp. = hospital, GBS = Guillain-Barre syndrome, HLD = hyperlipidemia, HTN = hypertension, ITP = idiopathic thrombocytopenia, IVIG = Intravenous immunoglobulin G, M = male, PID = primary immunodeficiency, SAD = specific antibody deficiency, TMP-SMX = trimethoprim sulfamethoxazole, T2DM = type II diabetes mellitus.
Of the thirty-three individuals, twenty-five did not require an emergency department (ED) visit or hospitalization. Eight individuals did require an ED visit and one required further hospitalization. There were no mortalities and zero of the thirty-three individuals had a blood clot or thrombosis. For the general population, the risk of VTE with COVID-19 ranges from 3-8%8,9 Seven of the eight people that required ED visits had only MBL deficiency. The one hospitalized was among the seven with only MBL deficiency.
The hospitalized individual is a 45-year-old female with MBL level <5ng/mL with no other immunodeficiencies. Her comorbid conditions included asthma, obesity, and hypertension. She was diagnosed with COVID-19 in January 2021 and was unvaccinated at the time of diagnosis. It is unclear if emergency medicine physician was aware of her MBL deficiency diagnosis upon decision to admit to the hospital, but the diagnosis of MBL deficiency had previously been documented in the electronic medical record prior to ED presentation. She was treated with high dose dexamethasone for ten days as well as Remdesivir and was placed in a double-blinded trial for an unknown monoclonal antibody. She did not require intubation or ICU level care. Her maximum supplement oxygen requirement was 5L. She was admitted for a total of three days and has since made a full recovery.
Of the other seven individuals that required only ED visits, all were female with an average age of 40 years. Six of them were unvaccinated at the time of diagnosis. One received Johnson & Johnson vaccine, and two vaccination statuses are unknown. The vaccinated patient was a 38 year old female who tested positive in August 2021. She also had IgG2 subclass and specific antibody deficiency. Following her ED visit, she was treated with monoclonal antibodies and made a full recovery.
Hospitalization rate for COVID-19 infection in the state of West Virginia in January of 2021 was 2.4%. 19 Of the MBL deficient participants who were COVID-19 positive in January 2021, 16.7% (1/6) were hospitalized. When compared to the state-wide hospitalization rate in this time period, MBL deficient patients had 7 times the odds of hospitalization (p = .1506, OR 7.06 (CI 1.117-44.52)). However, the Fisher exact test did not achieve statistical significance. Eight of thirty-three (24%) of our MBL deficient cohort required advanced care, whether that was an ED visit or hospitalization. Overall hospitalization rate for our population was 3%.
Among the total MBL deficient cohort, 33% of those without another immunodeficiency went to either the ED or were hospitalized. Only 7% of those with low MBL levels and a humoral co-morbid immunodeficiency required advanced care. The Mann-Whitney two tailed t-test was statistically significant between the two groups with a p = .0463. Only 11.1% of those with MBL deficiency alone were vaccinated prior to infection compared to 42.9% of those with MBL def. and humoral immunodeficiency. Use of monoclonal antibodies between the two groups was nearly the same with 22% (4/18) and 33% (5/15). The difference in vaccination status and monoclonal antibody use among the two groups was not statistically significant using the Mann-Whitney two tailed t-test (p = .1015 and p = .6968 respectively). The average age of the two groups (MBL only, 38 years old verse MBL and humoral immunodeficiency, 45 years old) was also not statistically different using an unpaired student t-test (p = .2387). Among those with co-morbid humoral immunodeficiencies, three were on IVIG at the time of their COVID-19 infection. The difference between the two groups was not statistically significant however, with respect to the use of IVIG in a Mann-Whitney two tailed t-test p = .0834.
Discussion
Mannose-binding lectin’s role in protection against infection has long been known. When one is deficient in MBL, associated increased number of infections and severity of infections have been reported. It has also been reported that too much Mannose-binding lectin can lead to increased undesirable inflammation. MBL has been shown to cause microvascular injury in COVID-19 viral infections. In a study of critically ill COVID-19 patients, extensive deposits of the terminal complement complex C5b-9 as well as C4d and MASP2 were found in the lungs on histology. 7 These findings are consistent with sustained systemic activation of the complement pathway. 7 A cohort study revealed activation of the MBL complement pathway in severe COVID-19. A similar study showed patients with higher MBL levels were at higher risk for thromboembolic events. 3 In our study, none of the 33 individuals had VTE, critical course, or fatality with their COVID-19 infections.
Our population did however have a higher percentage of hospitalization than the general public. However, statistical significance was not achieved. This is likely secondary to the low number of those known to have MBL deficiency. We could also not control for anyone in the general population who may have undiagnosed MBL deficiency. The odds ratio was calculated using general public data from January 2021, since the vast majority of the general public had yet to be vaccinated in West Virginia. However, our findings to some extent do support the idea MBL deficiency confers risk for an initially more moderate to severe COVID-19 infection.
In one study, a more severe COVID-19 disease course and ICU care was associated with the BB genotype MBL. 4 Mortality at 28 days was not found to differ across MBL variants (AA, AB, BB, highest to lowest levels of MBL respectively) in this study. Another study suggests that any genetic variations that included a B gene in MBL2 could also play a role in determining susceptibility to severe COVID-19 with respiratory failure and pneumonia development in univariate analysis. 6 In multivariate analysis, the B gene’s presence lost statistical significance with respect to hospitalization and pneumonia. Additionally, the multivariate analysis showed that other co-morbid variables such as: age and obesity retained their statistical significance for both hospitalization and pneumonia. In neither study was mortality associated with MBL deficiency.4,6 There were no reports of the presence or absence of blood clots in either study. The latter study also did not discuss mortality in their cohort. 6
Our study differs from the two above in that this cohort consists of those known to have MBL deficiency in the general population prior to infection; and venous thrombus events, as well as mortality incidences were tracked. Additionally, co-morbid associated humoral immunodeficiencies were also taken into account in our study, whereas the other two studies4,6 focused on only the MBL genotypes in those hospitalized without accounting for co-morbid immunodeficiencies. In our study, those with MBL def. alone had a higher percentage of ED visits and hospitalization and were slightly statistically significant compared to those with the addition of a humoral immunodeficiency. The difference is likely secondary to a combination of higher vaccination rate in those with additional humoral immunodeficiencies, slightly higher percentage of monoclonal antibody use, as well perhaps the use of IVIG by some. Intravenous immunoglobin IgG has been reported to help with COVID-19 infections, particularly as more antibodies to COVID-19 are found in IgG infusion stock.20,21
In a study on the MBL recognitions of SARS-CoV-2, the authors concluded that MBL’s role may represent a “double edge sword” owing to increased late-stage uncontrolled inflammation. 5 Other findings suggest that high activity of the classical complement pathway and high levels of MBL may be associated with an increased odds of unprovoked VTE. 2 For the general population, the risk of VTE with COVID-19 ranges from 3-8%8,9 The rate of VTE in our population was zero, and we postulate that the lack of VTE incidences from late-stage uncontrolled inflammation is related to the counterintuitive protective effect observed in our study as well as others. Of course, given the small numbers, it may be simply explained by chance.
Correspondingly, treatment against COVID-19 using drugs that target the MASP-2 pathway are beginning to be studied. Narsoplimab, monoclonal antibody against MASP-2, inhibits lectin pathway activation and has anticoagulant effects. This study showed higher mortality in the control groups than the narsoplimab-treated group. All narsoplimab-treated patients recovered and survived. Narsoplimab may be an effective treatment for COVID-19 by reducing COVID-19-related endothelial cell damage, inflammation, and thrombotic risk. 22 MBL deficient individuals naturally are inhibited in their lectin pathway activation and our study’s findings are in line with the pharmaceutical study’s creation of transient pseudo-MBL deficiency or tempering of MBL levels.
In regards to MBL’s role in other infections, the data varies and MBL deficiency has had protective role against fatality with other viruses as well. Studies with influenza H1N1 revealed that MBL knockout mice developed mild disease as evidenced by lower cytokine and chemokine production. [12] In an observational study, MBL levels were assessed in patients with an H1N1 diagnosis within 24–48 h following ICU admission. Serum MBL was significantly higher (3741 ng/ml) in non-survivors of severe infection compared to survivors (215 ng/ml), as well as in the control group (1814 ng/ml). The results showed a relation between serum MBL levels and mortality in ICU patients with serious infection. 13 In a case-control study, there was no association between MBL deficiency and predisposition to H1N1 2009 influenza in 63 pairs of seropositive and seronegative participants.14
There have been several studies on MBL deficiency and the coronavirus that emerged in 2003 that was responsible for SARS. A Chinese study from 2005 revealed that MBL gene polymorphisms, associated with decreased serum MBL, were significantly associated with susceptibility to SARS-CoV-1 infection; however, the authors did not observe an association between MBL polymorphisms and SARS disease severity.15,16
Limitations of this study included the small sample size, given the small number of patients know to have MBL deficiency in the local population; and the inability to control for MBL deficiency (known and unknown) in the general population.
Conclusion
This retrospective cross-sectional analysis of a small cohort of persons with known MBL deficiency and COVID-19 infection suggest a lower risk of fatalities and VTE (blood clots). A potential higher rate of hospitalization was observed compared to the general public, however this finding was not statistically significant. Those with MBL deficiency without a humoral immunodeficiency had a statistically significant higher rate of emergency department visits that those with low MBL levels and a humoral immunodeficiency. Further prospective studies are needed to determine if persons with MBL deficiency alone or in combination with other humoral immunodeficiency have either a decreased risk for VTE or poorer survival for COVID-19 infection, despite ostensibly more severe disease at presentation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from West Virginia University Institutional Review Board # 2107378483.
Informed consent
Informed consent was waived by WVU IRB for the present study because de-identified data was used for study.
