Abstract
Previous works have demonstrated the involvement of cytotoxic T-lymphocyte antigen 4 (CTLA-4) and nuclear factor kappa B (NF-κB) in maintaining the normal physiology of the heart. Polymorphisms in genes encoding these proteins may affect their normal functions, which could subsequently lead to chronic heart failure (CHF). In this work, we examined the association of CTLA4 and NFKB1 polymorphisms with CHF in a Chinese population. The −318C>T and +49A>G polymorphisms of CTLA4 and −94 insertion/deletion ATTG polymorphism of NFKB1 were genotyped on 538 patients with CHF and 1076 healthy controls. Our data indicate that the CTLA4 +49A>G and NFKB1 polymorphisms could confer susceptibility to CHF among Chinese. A significantly increased CHF risk was observed for the mutant CTLA4 +49GG genotype (P = 0.0093), and both heterozygous ATTG1/ATTG2 (P = 0.0142) and mutant ATTG2/ATTG2 (P = 0.0018) genotypes of NFKB1. Our data suggest that there is a potential use of these polymorphisms for early genetic screening for CHF.
Keywords
Introduction
Chronic heart failure (CHF) is a complex multifactorial syndrome with substantial morbidity and mortality all over the world. 1 Genetic hereditary is known to play an important role in the etiology of heart failure. 2 Thus, early genetic screening can potentially be used for early identification of at-risk individuals. 2 Genes involved in maintaining the normal cardiac physiology, including CTLA4 and NFKB1, represent promising candidate biomarkers for early screening of presymptomatic individuals.
CTLA4 encodes cytotoxic T lymphocyte antigen 4 (CTLA-4), which inhibits activation of either pro- or anti-inflammatory T cells. It has been known that inflammation is an important mediator of CHF. 3 Thus, any changes in the activation of T cells can affect the state of inflammation in the heart, which promotes the development of CHF. 4 There are two commonly studied polymorphisms in the CTLA4 gene. The first one is located in the promoter region (the −318C>T polymorphism), and can affect its protein expression. In addition, the exon 1 (+49A>G) polymorphism of CTLA4 may affect the interaction between the protein and its ligand, B7.1, which is required for its T-cell inhibitory activity. 5 As such, these polymorphisms could potentially influence the risk of CHF. Several studies have investigated the association between these polymorphisms and CHF risk, but the reproducibility of the results in a large-scale population is not known.6,7
Besides CTLA-4, there is an emerging role of nuclear factor kappa B (NF-κB) in maintaining the normal heart physiology. Apart from mediating the inflammatory process in cardiomyocytes, NF-κB serves as an important regulator of immune cell maturation and cell survival in the heart. 8 Although NF-κB is essential for the normal heart function, sustained activity of NF-κB has been shown to induce molecular pathways that lead to CHF. 9 The −94 insertion/deletion ATTG polymorphism of NFKB1, which encodes NF-κB, is a functional polymorphism that can influence the activity of the gene and the protein product. Two previous studies have investigated the role of this polymorphism in heart failure, but the results were conflicting.10,11
The present work aimed to examine the association of the −318C>T and +49A>G polymorphisms of CTLA4 and –94 insertion/deletion ATTG polymorphism of NFKB1 with risk of CHF in a large-scale Chinese population.
Materials and methods
Ethics
The study protocol was approved by the Human and Research Ethics Committee of First Peoples Hospital Baiyin City (approval number: 023-RQU-2014) and the Board of Ethics for Research of Fu Ping Township Public Health Center (approval number: 2014-1000813). Study participants were briefed on the study procedures on recruitment. Written informed consent was obtained from all participants before sample collection.
Study population
To increase the statistical power of this case-control study, we used a case-to-control ratio of 1:2. The study population comprised 538 patients with CHF and 1076 healthy controls. The participants were recruited from First Peoples Hospital Baiyin City and Fu Ping Township Public Health Center between July 2014 and June 2018. Cases were defined according to the New York Heart Association (NYHA) Functional Classification system. Patients with Classes III and IV were included in the study. Diagnosis of CHF was done based on established guidelines. Patients with congenital heart disease or other chronic diseases were excluded from the study. Healthy controls were recruited among those who attended a health screening program in the same hospital. They were free from cardiovascular diseases, hypertension, coronary heart disease, and diabetes mellitus. All study participants were Han Chinese.
Genotype determination
Genomic DNA was obtained from the blood specimens of the study participants. The −318C>T and +49A>G polymorphisms of CTLA4 were genotyped on the genomic DNA via PCR-RFLP (polymerase chain reaction–restriction fragment length polymorphism) analysis, while the −94 insertion/deletion ATTG polymorphism of NFKB1 was genotyped by using PCR method. The PCR primers used were described elsewhere.7,11 After PCR amplification, the 226 bp CTLA4 –318C>T products were incubated with MseI restriction enzyme overnight, while the 192 bp CTLA4 +49A>G products were incubated with BvbI. The genotype was determined through gel electrophoresis. The T allele of the CTLA4 –318C>T polymorphism was cleaved into two fragments of 110 bp and 116 bp, and the G allele CTLA4 +49A>G polymorphism was cleaved into two fragments of 112 bp and 80 bp. The wild type alleles were uncleaved.
For NFKB1 –94 insertion/deletion ATTG polymorphism, the PCR products were visualized directly under electrophoresis, without incubating with any restriction enzymes. The ATTG1 allele yielded a 154 bp band, while the ATTG2 allele yielded a 158 bp band.
For all the three polymorphisms, DNA sequencing was performed on 10% of randomly chosen samples to confirm the genotype. All sequencing results concurred with the PCR-based genotyping results.
Statistical analysis
SPSS version 18 was used for data analysis. Continuous data were presented as mean ± standard deviation and categorical data were presented as frequencies. Chi-square test (for categorical data) or t test (for continuous data) was performed to evaluate the differences between patients and controls and also to assess deviation from Hardy-Weinberg equilibrium. Linkage disequilibrium (LD) was analyzed for the −318C>T and +49A>G polymorphisms of CTLA4 (as they are located in a same chromosome) by using Lewontin’s standardized disequilibrium coefficient D and the square of correlation coefficient (r2) method. The association between the polymorphisms and CHF was examined by calculating the odds ratios (ORs) and 95% confidence intervals (CIs) using a logistic regression method. The wild type genotype was used as the reference in the logistic regression model. Classic risk factors of CHF, and other factors which could potentially confound the results (age, sex, smoking habits), were also considered in the logistic regression. A P value < 0.05 was considered significant.
Results
Baseline characteristics
The baseline characteristics of the study participants are shown in Table 1. There were 538 cases and 1076 controls, which corresponded to a ratio of 1 case to 2 controls. In cases, the number of male participants was slightly higher, but the difference was not statistically significant (Table 1). The ages of cases and controls were comparable (Table 1). Cases had a higher systolic and diastolic blood pressure compared to controls (P < 0.0001), but this is expected, because hypertension is linked to CHF and we excluded controls who had a high blood pressure. Cases and controls were very similar to each other in other characteristics (total cholesterol, triglycerides, high-density lipoprotein) (P > 0.05, Table 1). Other echocardiographic and biochemical parameters of the patients are shown in Table 1. Among the patients, 259 (48.1%) had advanced atherosclerosis, 126 (23.4%) had dilated cardiomyopathy, and 153 (28.4%) had ischemic heart disease.
Baseline characteristics of the study participants.
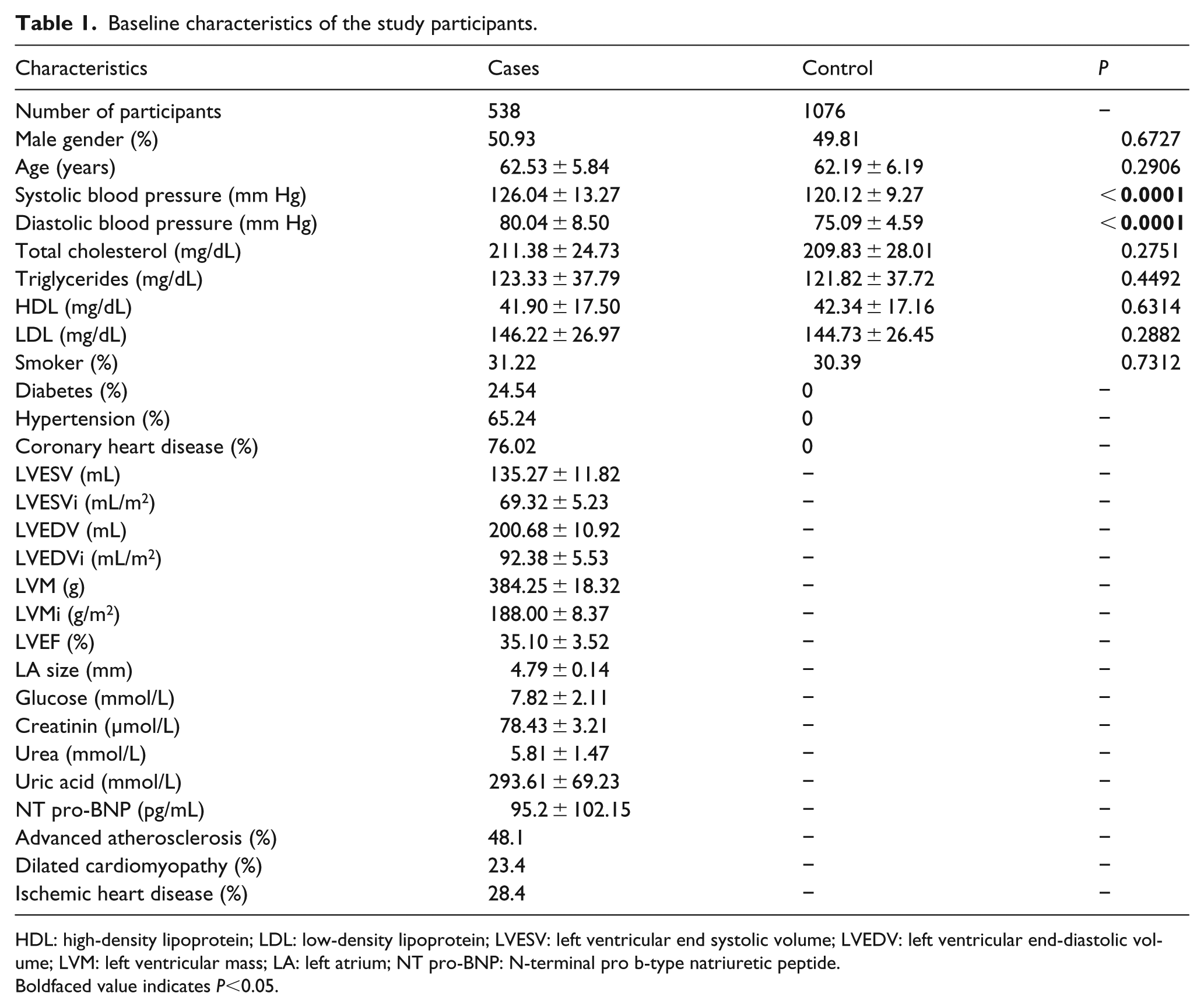
HDL: high-density lipoprotein; LDL: low-density lipoprotein; LVESV: left ventricular end systolic volume; LVEDV: left ventricular end-diastolic volume; LVM: left ventricular mass; LA: left atrium; NT pro-BNP: N-terminal pro b-type natriuretic peptide.
Boldfaced value indicates P<0.05.
Genotype distribution
The polymorphisms were successfully genotyped in all cases and controls. The genotype distribution is shown in Table 2. The distribution of CTLA4 –318C>T polymorphism in cases was similar to controls (P = 0.4807). For CTLA4 +49A>G polymorphism, a statistically significant difference was observed (P = 0.0081), with the AA and AG genotypes being overrepresented in controls and GG genotype being overrepresented in cases. Similarly, for the NFKB1 polymorphism, a significant difference was observed. The ATTG1/ATTG1 and ATTG1/ATTG2 genotypes were found at a higher frequency in controls and the opposite was observed for the ATTG2/ATTG2 genotype.
Association of the polymorphisms with chronic heart failure risk.

NA: did not investigate as the P value for genotype distribution is not statistically significant.
Boldfaced values indicates P<0.05.
The presence of LD between CTLA4 –318C>T and +49A>G polymorphisms were measured. No significant LD was observed (D’ = 0.337, r2 = 0.0721).
Association with CHF
Logistic regression was performed to evaluate the relationship between the significant polymorphisms (CTLA4 +49A>G and NFKB1) and CHF risk. After adjusting for confounders, the results are shown in Table 2. The heterozygous and mutant genotypes of both polymorphisms showed an OR > 1.000, indicating that they were associated with increased risk of CHF. However, for CTLA4 +49A>G polymorphism, the association of the AG genotype was not statistically significant (P = 0.3129). Only the mutant GG genotype significantly increased the risk of CHF, at an OR of 1.4134 (95% CI= 1.1297–2.1139) (P = 0.0093).
For the NFKB1 polymorphism, both the ATTG1/ATTG2 and ATTG2/ATTG2 genotypes were statistically significant in relation to their association with CHF risk. The ATTG1/ATTG2 genotype was associated with an OR of 1.7843 (95% CI = 1.2640–2.6521) risk of CHF (P = 0.0142), whereas the ATTG2/ATTG2 genotype presented an OR of 2.0137 (95% CI = 1.6734–3.5007) (P = 0.0018).
Discussion
In this study, we successfully showed that the CTLA4 +49A>G and NFKB1 –94 insertion/deletion ATTG polymorphisms, but not the CTLA4 –318C>T polymorphism, were associated with risk of CHF among Chinese. The CTLA4 +49A>G polymorphism is located in exon 1 of the gene and causes an amino acid change in the protein region that interacts with its ligand, B7.1. 5 The G allele may have a reduced T cell suppression activity, which allows pro-inflammatory signals to be transmitted more efficiently. 12 This facilitates cardiac inflammation, which can lead to an increased CHF risk. Similar to CTLA4 –318C>T polymorphism, the association of the +49A>G polymorphism was also investigated by Ruppert et al. 7 before, who also found a significant association between the variant genotype and CHF risk.
To our knowledge, this is the first study which investigated the association between CTLA4 –318C>T polymorphism and CHF risk among Chinese. However, it was not the first study in the world. Previously, Ruppert et al. 7 had investigated the effect of this polymorphism on dilated cardiomyopathy among Germans. Our finding that there was no association between the polymorphism and CHF risk was similar to that of Ruppert et al. 7 As this polymorphism is known to affect its protein expression, our observation suggests that changes in CTLA-4 levels may not translate into a change in the state of inflammation.
For NFKB1, our result conformed to a previous work carried out in the Chinese population. 11 Both our work and that carried out by Zhou et al. 11 suggested that the ATTG2 allele was associated with an increased CHF risk. Another study, carried out in the Brazilian population, did not observe any significant overrepresentation of the ATTG allele in either cases or controls. 10 These conflicting findings indicate that the polymorphism may modulate CHF differently in different populations.
In conclusion, our findings indicate that the CTLA4 +49A>G and NFKB1 –94 insertion/deletion ATTG polymorphisms were associated with CHF risk. The major advantage of this study is the large sample size and thus statistical power. Through this highly powered analysis, our findings provided an important indication that there is a potential use of these polymorphisms for early genetic screening for CHF. However, the major disadvantage of our study is the lack of functional analysis and independent validation of the findings. Thus, future studies are needed to independently validate our findings and investigate the functional impact of the polymorphisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
