Abstract
Objective: Considering the published manuscripts on this subject, it is thought that IL-1B and azurocidin may be diagnostic and prognostic factors in severe COVID-19 disease. This study aimed to determine whether azurocidin and IL-1β are useful biomarkers and are associated with disease exacerbation in patients needing advanced treatment options. Material and Method: Our study was performed retrospectively. The data of a total of 291 patients followed in the hospital due to COVID-19 were collected. Among these patients, 66 randomly selected patients were included in this study. The control group consisted of 24 healthy individuals referred to the infectious disease outpatient clinic who weren’t no diagnosis of COVID-19 infection or any other infection. After the samples were taken into tubes without anticoagulant, they were kept at room temperature for 30 min. Afterwards, it was centrifuged at 1000xg for 15 min at +4° according to the instructions of the ELISA kit we used. The remaining serum was transferred to Eppendorf tubes and stored at −80° throughout this study. Results: A statistically significant strong positive correlation was determined between IL1β and azurocidin. A statistically significant weak positive correlation was found between IL1β and CRP, ferritin and neutrophil count, weak negative correlation with albumin, and moderately strong positive correlation with leukocyte count. Moreover, a statistically significant weak positive correlation was found between azurucidin and CRP, ferritin, and neutrophil counts. Conclusion: Azurocidin and IL-1β may serve as a potential therapeutic target for patients at risk of developing systemic multi-organ failure, with improved patient prognosis and prevention of death in severely ill patients. Although these results may contribute to the improvement of the follow-up and treatment of the patients and reduce mortality rates, there is a need for randomized controlled studies with a larger number of patients to be performed in the future.
Key Messages
1. Our results show that azurocidin and IL-1β values were higher in each group defined according to disease severity compared to the control group. 2. These results may contribute to the improvement of the follow-up and treatment of the patients and to the reduction of deaths, there is a need for randomized controlled studies with a larger number of patients to be conducted in the future.
Introduction
In December 2019, authorities in Wuhan, China, reported a series of pneumonia cases caused by an agent of unknown etiology. The pathogen was soon identified, classified as a novel coronavirus related to the causative agent of the severe acute respiratory syndrome (SARS), and later named SARS Coronavirus-2 (SARS-CoV-2). The infection, which spread to all continents within the next 3 months, was declared a pandemic by the World Health Organization (WHO). 1 In our country, cases started to be reported as of the second week of March. 2 Globally, as of March 7, 2022, there have been 445.096.612 confirmed cases of Coronavirus-19 (COVID-19), including 5.998.301 deaths reported to WHO. 3
This new type of coronavirus specifically affects the lungs and can cause serious pneumonia. Patients with severe pneumonia may suddenly progress to severe respiratory failure and may need respiratory support treatments. 1 Most researchers use sepsis as the critical illness prototype to understand the pathogenesis of severe COVID-19. This is mainly because severe COVID-19 is associated with hypercytokinaemia. 4 As a result, it was reported in some studies that some SARS-CoV-2 positive patients with stable clinical status and whose nucleic acid tests converted negative might suddenly worsen. Therefore, biomarkers associated with both the post-viral infection stage and the exacerbation of COVID-19 positive patients, as well as potential therapeutic targets, would be extremely valuable in this regard.5–7
Azurocidin (also known as Heparin-binding protein=HBP, or CAP-37=cationic antimicrobial protein of 37 kDa) is stored in azurophilic granules and secretory vesicles in the cell membrane of polymorphonuclear neutrophils (PMN).8,9 Endothelial cells are the primary targets of azurocidin, which plays an important role in capillary leakage, which provides cytoskeletal reorganization, increases vascular endothelial permeability and inflammatory effect.8,10 Antigens, cytokines, inflammatory factors, and various enzymes may cause azurocidin to be released from PMN.11,12 Azurocidin has been associated with organ dysfunction in sepsis, acute respiratory dysfunction and acute kidney injury, and has been recommended for triage of patients in the emergency department. 8
Interleukin 1 (IL-1) proteins are small secretory proteins with critical roles in the immune response. 13 This family belongs to a broader family of ligands with 11 members identified to date (IL-1α, IL-1β, IL-18, IL-33, IL-36a, IL-36b, IL-36g, IL-1RN, IL-36Ra, IL-38, and IL-37). 14 IL-1β is expressed as a 31-kDa inactive precursor, primarily released in response to inflammatory stimuli, particularly other cytokines including IL-1α, and even IL-1β secretion itself, microbial infection, and other factors. 15 IL-1β is not expressed in cells of healthy individuals and requires a cascade of intracellular events for cytokines to trigger inflammation. 15 SARS-CoV-2 activates an inflammasome called NLRP3, whose activation increases both production and secretion of IL-1β and IL-18. The increase in these interleukins causes a cytokine storm.16,17
Considering the main role of azurocidin in systemic vasculitis that may develop in severe infections and the roles of IL-1β in the cytokine storm, we predicted that elevated azurocidin and IL-1β may mediate the pathogenesis of disease exacerbation in severe COVID-19 patients. This study aimed to determine whether azurocidin and IL-1β are useful biomarkers and ultimately associated with disease exacerbation for patients needing advanced treatment options.
Matrial-method
Study design and participants
Our study was performed retrospectively between October 1 and December 31, 2020. The data of a total of 291 patients followed in the hospital due to COVID-19 were collected. Among these patients, 66 randomly selected patients were included in this study. COVID-19 disease severity was classified by the findings of the patients at the time of hospitalization, using the WHO criteria. 18 Of the 291 patients evaluated during this study, 116 had moderate, 105 severe, and 70 critical COVID-19 infections at admission. No financial support was received for this study and due to our limited resources, azurocidin and IL-1ß could not be evaluated in all of the patients. Therefore, patients from each group were randomly selected using the “SPSS program random sample of cases.” The diagnosis of COVID-19 was made by detecting SARS-CoV-2 in respiratory tract samples by real-time-polymerase chain reaction (RT-PCR) in laboratories authorized by the Turkish Ministry of Health, together with clinical findings. All patients over the age of 18 who were diagnosed with COVID-19 with PCR positivity and followed up in the hospital were included in this study. Patients diagnosed with computed tomography findings, negative PCR, or outpatient follow-up were excluded from this study. The patients included in this study were grouped as moderate, severe, and critical according to disease severity and as dead-alive during the analysis. The control group consisted of 24 healthy individuals who admitted to the Infectious Disease Outpatient Clinic within the scope of various screening programs and were not diagnosed with COVID-19 infection or any other infection.
Data collection
A data set was created with demographic data, clinical, laboratory, and radiological findings, which are defined as mortality risk factors in COVID-19 patients in the literature. Age, gender, time from the beginning of the complaint to the hospital admission, complaints, vital signs, comorbid diseases, and laboratory findings of the patients were obtained retrospectively from the patient files and the hospital electronic information system. Fever, shortness of breath, sore throat, cough, loss of appetite, nausea, weakness, myalgia, loss of taste and smell, diarrhea, headache were recorded from the hospital application forms in which complaints were questioned. Fever, body temperature ≥38,3°C; hypoxia, ≤ 93% of O2 saturation in room air; tachycardia, heart rate ≥100 beats/min and hypotension was determined by arterial blood pressure ≤90/60 mg/Hg. Ground glass appearance, pleural effusion, pulmonary embolism, consolidation, crazy-paving, interlobular septal thickening were noted in radiological imaging. C-reactive protein (CRP) (mg/L), ferritin (ng/mL), creatinine (mg/dL), albumin (g/L), aspartate aminotransferase (AST) (U/L), alanine aminotransferase (ALT) (U/L), troponin (pg/ml), D-dimer (ng/mL), fibrinogen (mg/dL), INR, prothrombin time (PT) (sec), activated partial thromboplastin time (aPTT) (sec), and platelet, leukocyte, neutrophil and lymphocyte counts (109/L) from the laboratory values of the day of hospitalization were recorded.
Statistical analysis
Data of 66 randomly selected patients from the study cohort were used. Normality distribution for quantitative variables was tested with the Kolmogorov-Smirnov test (p > .50). The Chi square test or Fisher’s exact test was used for categorical variables. Comparison of continuous variables between groups with and without mortality was made with Student’s t-test or Mann-Whitney U test, depending on suitability. The correlation between biomarkers was determined using the Pearson correlation test. If the correlation coefficient was between 0.2–0.39, it was considered that there was a weak correlation, a moderate correlation between 0.4–0.59, a strong correlation between 0.6–0.79, and a very strong correlation between 0.8 and 1. A receiver operating characteristic (ROC) curve demonstrated the predictive ability of the variables used for survival estimation. Youden index was used to determine the optimal cut-off value. The area under the curve (AUC) was calculated by ROC analysis to determine the diagnostic power of risk factors. Before performing this study, necessary permission was obtained from the Scientific Research Commission of the TR Ministry of Health and permission for data use from center involved. Ethics committee approval was obtained from the Erzurum Regional Education and Research Hospital Non-Invasive Clinical Studies Ethics Committee with No:E37732058–514.10.
IL-1β and azurocid quantification
After the samples were taken into tubes without anticoagulant, they were kept at room temperature for 30 min. Afterwards, it was centrifuged at 1000xg for 15 min at +4° according to the instructions of the ELISA kit we used. The remaining serum was transferred to Eppendorf tubes and stored at −80° throughout this study.
Azurocidin analysis was performed with the Human Azurocidin ELISA Kit (Bioassay Technology Laboratory, China, Cat.No: E0715Hu) per the manufacturer’s procedures (Azurocidin unit: ng/mL. Sensitivity: 0.05 ng/ml).
Interleukin 1 Beta analysis was performed with the Human Interleukin 1 Beta ELISA Kit (Bioassay Technology Laboratory, China, Cat.No:E0143Hu) per the manufacturer’s procedures (Interleukin 1 Beta unit: pg/L. Sensitivity: 10.07 pg/L).
Results
Baseline characteristics
Characteristics during hospital admission.

*Chi-Square and Mann Whitney -U tests were performed.CKF: Chronic Kidney Failure, COPD: Chronic Obstructive Pulmonary Disease, CAD: Coronary Artery Disease, CRP: C reactive protein, ALT: Alanine aminotransferase, AST: Aspartate aminotransferase, INR: International normalized ratio, PT: Prothrombin time, aPTT: Activated partial thromboplastin time, IL-1β: Interleukin 1 beta.
IL1β and azurocidin values by disease severity
The distribution of azurocidin and IL-1β values evaluated at the time of hospital admission by the severity of the disease according to the moderate, severe, and critical patients and the healthy control group is demonstrated in Figure 1. Azurocidin and IL-1β were statistically significantly higher in each group defined by disease severity than the control group (p < .001). Also, IL-1β values were significantly higher in critically ill patients than in severe patients and in severe patients compared to those with moderate disease (p < .001). Among the patient groups, azurocidin values was statistically significantly higher in critically ill patients than in severe and moderate patients (p < .001). However, there was no statistical difference between those with severe and moderate disease (p = .308). Mean distribution of azurocidin and IL-1β by disease severity.
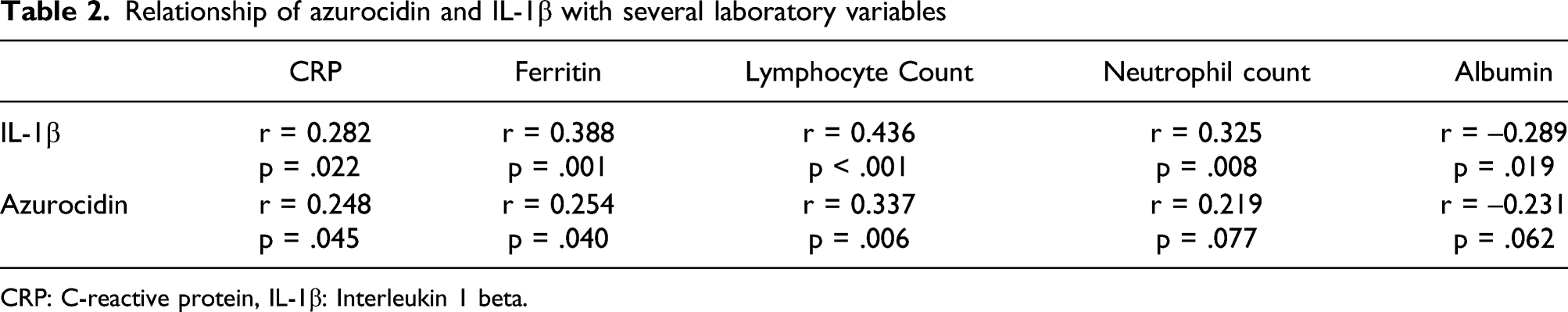
Relationship of IL-1β and azurocidin with some biomarkers
A statistically significant very strong positive correlation was determined between IL-1β and azurocidin (r = 0.800; p < .001) (Figure 2). The relationship of azurocidin and IL-1β with some biomarkers with a statistically significant relationship is presented in Table 2. A statistically significant weak positive correlation was found between IL-1β and CRP, ferritin and neutrophil count, weakly strong negative correlation with albumin and moderately strong positive correlation with leukocyte count. In addition to, a statistically significant weak positive correlation was found between azurocidin and CRP, ferritin, and neutrophil counts. Relationship between IL1β and azurocidin (r = 0.800; p < .001). Relationship of azurocidin and IL-1β with several laboratory variables CRP: C-reactive protein, IL-1β: Interleukin 1 beta.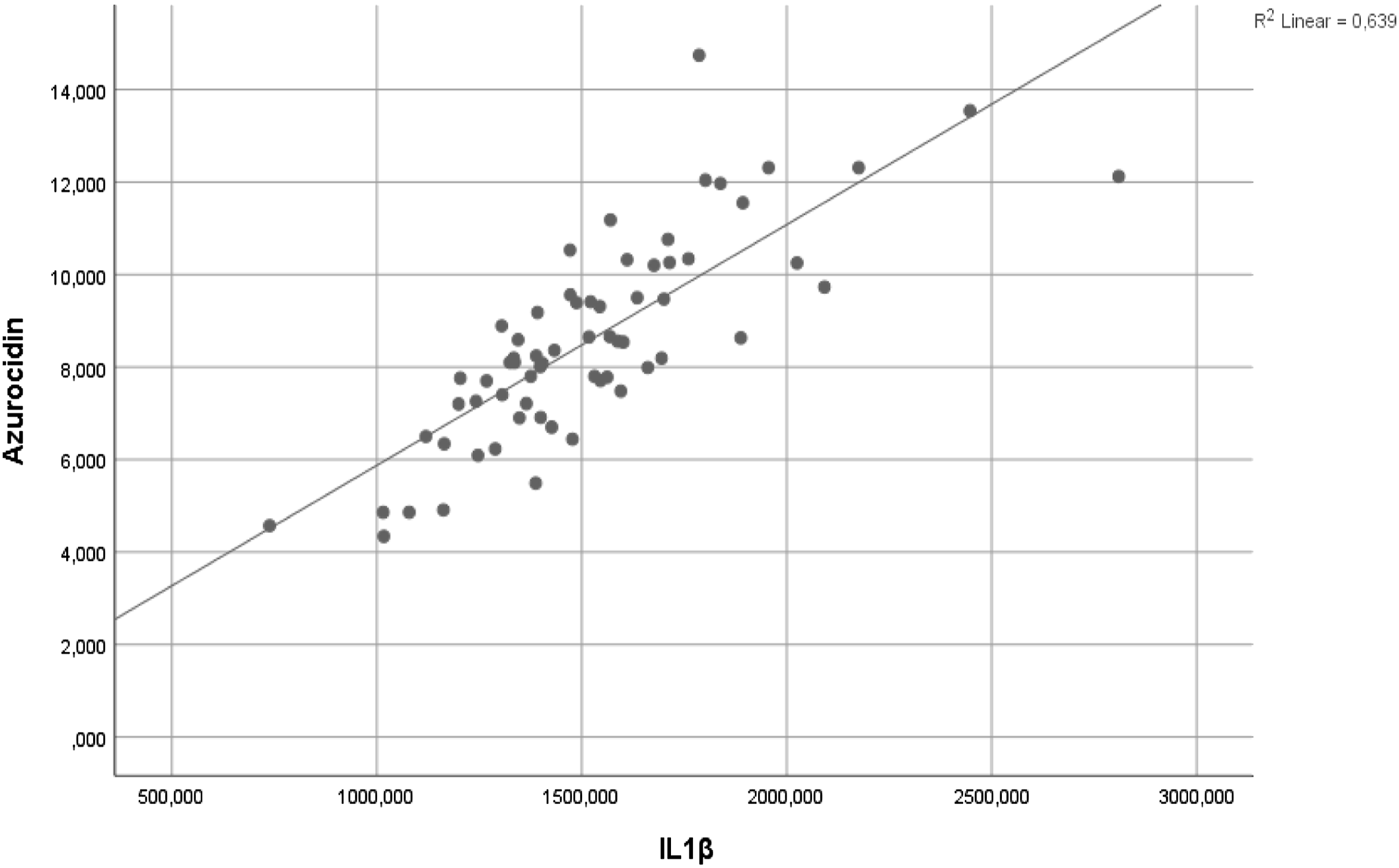
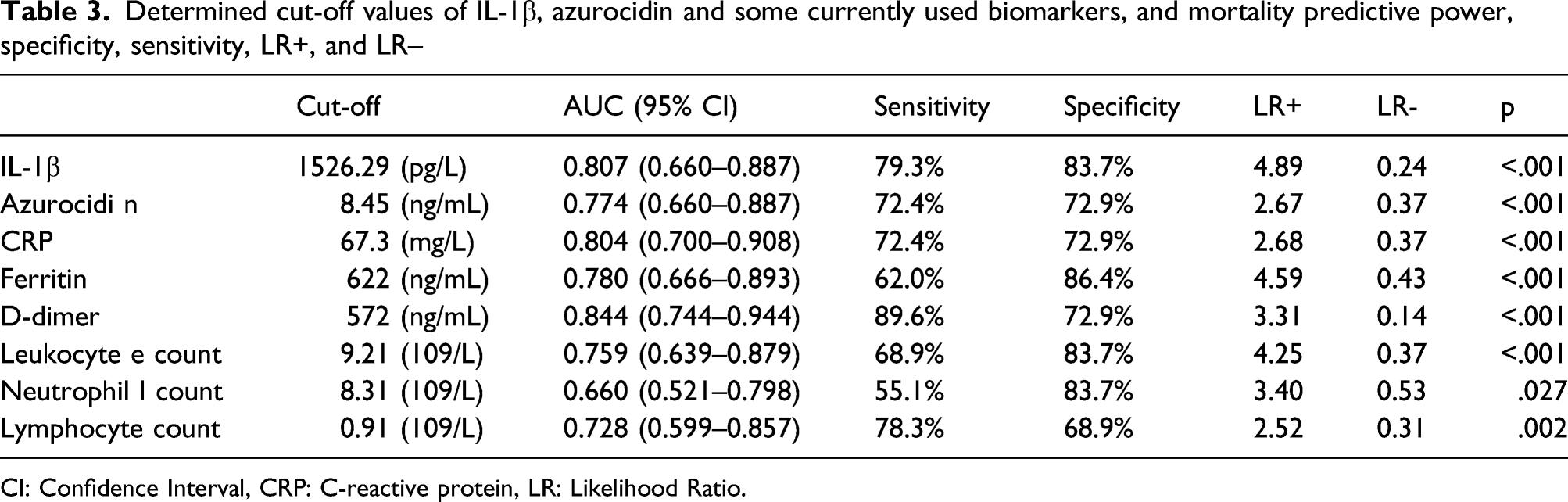
Mortality predictive power of IL-1β, azurocidin, and some biomarkers
Determined cut-off values of IL-1β, azurocidin and some currently used biomarkers, and mortality predictive power, specificity, sensitivity, LR+, and LR–
CI: Confidence Interval, CRP: C-reactive protein, LR: Likelihood Ratio.

Mortality predictive power of IL-1β and azurocidin.
Among the biomarkers evaluated initially, D-dimer had the highest predictive power of mortality, followed by IL-1β. Likewise, the sensitivity of IL-1β was determined as the highest biomarker together with D-dimer. The sensitivity of IL-1β in the prediction of mortality was 79, 3%, the specificity 83, 7%, the sensitivity of azurocidin was 72, 4%, and the specificity was 72, 9%. IL-1β has the highest positive probability among biomarkers (x4, 89).
Discussion
Our results show that azurocidin and IL-1β values were higher in each group defined according to disease severity compared to the control group. In addition, IL-1β values increased significantly between the groups as the severity of the disease increased, while azurocidin levels were found to be higher only in critically ill patients than in severe and moderate patients.
Considering the potent inducer of azurocidin in endothelial dysfunction and systemic vasculitis in severe infections, we were not surprised by the high levels in critical COVID-19 patients, consistent with the literature.19–21 In addition, the correlation of azurocidin with biomarkers used in routine practice to predict the poor prognosis of severe infections and similar mortality predictive power support previous results in the literature.20,22 In our study, azurocidin predicted mortality with a cut-off value of 8.45 ng/ml, sensitivity of 72.4% and specificity of 72.9%. Sarıdaki et al. 20 showed azurocidin as an independent risk factor for mortality in SARS-CoV-2 infection with higher specificity and lower sensitivity values compared to our study. This result was thought to be associated with a higher cut-off value (>35 ng/ml). In the same study, elevation of azurocidin was shown as an independent risk factor for severe respiratory failure. This was explained by the correlation of azurocidin with positive fluid balance in the lung as a result of increased vascular endothelial leakage. 20 Xuel M et al., 19 on the other hand, found that azuroside was correlated with lung involvement and that azuroside increased before lung involvement progressed. 19 The higher mortality rates in patients with pulmonary involvement in our study support this finding.
Known as a proinflammatory cytokine, IL-1β has been shown to be associated with inflammation in the respiratory tract in acute respiratory distress syndrome characterized by severe hypoxemia and alveolar-capillary barrier disruption. 23 In the study of McElvaney OJ et al., IL-1β was found to be higher in the plasma of COVID-19 patients compared to the healthy control group, and as the severity of the disease increased, the rate of IL-1β in the plasma was found to increase. These results are similar to the results in our study. 24 In the study of Evangelos et al., 1 hyper-inflammatory responses with immune dysregulation or macrophage activation syndrome, both characterized by pro-inflammatory cytokines, were demonstrated in all patients with pneumonia caused by SARS-CoV-2 and who developed respiratory failure. They suggested that the immune dysregulation here was not caused by IL-6 but by IL-1β. 1 In the study of Ye Q et al., high levels of IL-1β and other cytokines were found in patients with COVID-19. As a result, an excessive specific immune response occurs and causes a cytokine storm. 25 Consistent with the literature, in our study, found that the elevation of IL-1β increased with the severity of the COVID-19 disease and was more significant than azurocidin as a predictor of mortality. In addition, the very strong correlation of azurocidin with IL-1β, which is an indicator of vascular endothelial damage seen in the course of severe SARS-Cov-2 infection, is another proof that it can be used as an indicator of poor prognosis.
Consistent with the literature, we showed more deaths in men in the presence of diabetes mellitus, hypertension, coronary artery disease, and chronic obstructive pulmonary disease.20,26 In addition, high CRP, ferritin, D-dimer and neutrophil count and low lymphocyte count as indicators of poor prognosis supported the previous literature in our study.27,28
The main limitations of this study are that it was conducted in a single center and the number of patients was limited. Another limitation of this manuscript is the lack of power analysis. The strengths of this study are that it is the first study in the literature to evaluate the two indicators together in COVID-19 patients and is among the limited number of studies in which they are evaluated separately.
Conclusion
Azurocidin and IL-1β appear to be potential biomarkers as prognostic indicators. It is thought that these results may contribute to the improvement of the follow-up and treatment of the patients and to the reduction of deaths. However, future randomized controlled studies with larger numbers of patients are needed.
Footnotes
Acknowledgements
We thank to all dedicated health care workers caring for COVID-19 patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The case series was approved by Ethics Committee of Ministry of Health, Erzurum Regional Education and Research Hospital (Date: 15.02.2021 No:37,732,058–514.10). Written informed consent was obtained from all patients.
Informed consent
Written informed consent for publication was obtained from all authors.
