Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative disease characterized by dopaminergic neuronal loss and α-synuclein (α-syn) aggregation. With the acceleration of population aging process, the incidence of PD is expected to increase, putting a heavy burden on the whole society. Recent studies have found the alterations of gut microbiota (GM) in PD patients and the clinical relevance of these changes, indicating the underlying relationship between GM and PD. Additionally, elevated inflammatory responses originating from the gut play a crucial role in the initiation and progression of PD, which is closely associated with GM. In this review, we will summarize recent studies on the correlation between GM and PD, and discuss the possible pathogenesis of PD mediated by GM and subsequent inflammatory cascades. We will also focus on the promising GM-based therapeutic strategies of PD, including antibiotics, probiotics and/or prebiotics, fecal microbiota transplantation, and dietary interventions, aiming to provide some new therapeutic insights for PD.
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disease, ranking only after Alzheimer’s disease (AD). With accelerated aging process of the population, the incidence of PD is expected to increase. In the most populous nations, it is estimated that nearly 9 million people may suffer from this progressive disorder of central nervous system (CNS) by 2030 (more than twice as many as the number in 2005). 1
Characterized by the loss of dopaminergic neurons and aberrant accumulation of α-synuclein (α-syn) in the substantia nigra and striatum, 2 PD presents with both motor and non-motor symptoms. Other than the commonly known motor symptoms (e.g., tremor, bradykinesia, and rigidity 3 ), non-motor symptoms also severely affect the quality of life of PD patients. Moreover, some of those non-motor symptoms may precede the classical motor ones of PD by several years. 4 Gastrointestinal (GI) symptoms of PD include constipation, abdominal pain, bloating, and defecation dysfunction, 5 and they have been well recognized in PD patients, especially in the presymptomatic ones. Many (up to 80%) patients experienced constipation and delayed gastric empty several years before diagnosed with PD, 6 and the occurrence of GI symptoms or gut microbial dysbiosis in healthy people is linked to an increased risk of PD. 7
As a complex and multifactorial disease, the exact pathogenesis of PD remains unclear. However, some mechanisms have been identified to be related to PD, including α-syn aggregation, neuroinflammation, and mitochondrial dysfunction. 8 Meanwhile, due to the limited knowledge of PD pathogenesis, there has been no comprehensive therapeutic strategy to cure or even prevent the progression of this disease. As the most commonly used drug for PD, levodopa can cross the blood–brain barrier (BBB) and be converted to dopamine in the CNS to alleviate some of the motor symptoms. 9 However, it has no effect on both the non-motor symptoms and the dopaminergic neurodegeneration in PD. 10 In addition, it has some possible side effects, such as orthostatic hypotension and dyskinesia.11,12 In recent years, special attention has been paid to the role of GI tract and the inside gut microbiota (GM) as well as the inflammatory hypothesis which considers PD as a systemic disease. GM and its metabolites have been reported to be involved in the modulation of GI functions and the activity of enteric nervous system (ENS), thus affecting the CNS through the microbiota-gut-brain axis (MGBA) . 13 Moreover, the peripheral inflammation including GM-induced intestinal inflammation may initiate the accumulation of α-syn and lead to systemic and CNS inflammation, finally contributing to the initiation and progression of PD. 14 Therefore, GM-targeted treatment to attenuate intestinal inflammation may become a potential therapeutic strategy for PD.
In this review, we will summarize recent studies on the correlation between GM and PD, discuss the possible pathogenesis of PD mediated by GM and inflammation, and focus on the GM-targeted treatment of PD.
Gut microbiota alterations in PD
Gut microbiota
Gut microbiota (GM), predominated by bacteria, fungi, viruses, and protozoa, 15 is the generic term for more than 100 trillion microbes existing in the human GI tract. Firmicutes, Bacteroidetes, Actinobacteria, Proteobacteria, Fusobacteria, and Verrucomicrobia are the main phyla of human GM, in which Firmicutes and Bacteroidetes account for 90%. 16 As a highly complex microbial community, GM has co-evolved with humans for millions of years, forming a mutually beneficial relationship. 15 Physiologically, GM and its metabolites play a key role in the development of both the immune and nervous system. 15 After the fetus is born, GM obtained from the mother as well as the environment start to colonize his or her GI tract. Through the bi-directional communication between GM and brain, termed as MGBA, GM will have an impact on the both the GI functions and CNS development. 17 A balanced and stable GM is vital for the integrity of intestinal barrier, the mucosal immune function, and the motility of gut. 18 Besides, GM and its metabolites can modulate the activity of ENS and the processes in CNS, such as the permeability of BBB and biochemical reactions in the brain. 19 They can also influence humans’ behaviors, including emotional ones and the stress responsiveness.20,21 However, healthy individuals only share one third of GM, leaving the remaining two thirds varying from person to person. 22 The composition of GM can be affected by the delivery type as well as feeding mode of infants, lifestyle (especially the dietary habits), antibiotic treatment, and other factors which can alter either the internal or external environment. 16
Alterations of gut microbiota in PD
Gut dysbiosis refers to the alteration of structure and/or function of GM, disrupting the gut homeostasis. 23 Like the shaping factors of normal composition of GM, gut dysbiosis can be caused by lifestyle changes (especially the dietary alterations), antibiotic exposure, inflammation, and other factors. 16 Recent study by Wallen et al. 24 suggested the potential role of genetics on GM. They found that alterations of the host genotype at α-syn locus may influence the abundance of some opportunistic pathogens in PD gut, further indicating that PD-related genetic variants may increase susceptibility of PD via GM alterations. As an essential part of the MGBA, gut dysbiosis is correlated to several diseases, including GI diseases, autoimmune diseases, metabolic disorders, and CNS-related diseases. 25 Hager et al. 26 studied GM in patients with Crohn’s disease, finding that their levels of Candida tropicalis, E. coli, and Serratia marcescens were higher as compared to healthy controls. These microbes work in symbiosis to form biofilms, leading to the gut inflammatory responses. Studies have also shown the potential role of gut dysbiosis in CNS-related conditions, such as AD, 27 depression, 20 schizophrenia, 28 multiple sclerosis, 29 and autism. 30 Similarly, recent studies have highlighted the correlation of gut dysbiosis and PD,31-33 while some results seem inconsistent, possibly due to the differences in methodology (i.e., sampling parts and methods), geographical regions, and patients’ dietary habits.2,34,35
Alterations of gut microbiota at the family level in Parkinson’s disease(PD) patients and animal models.
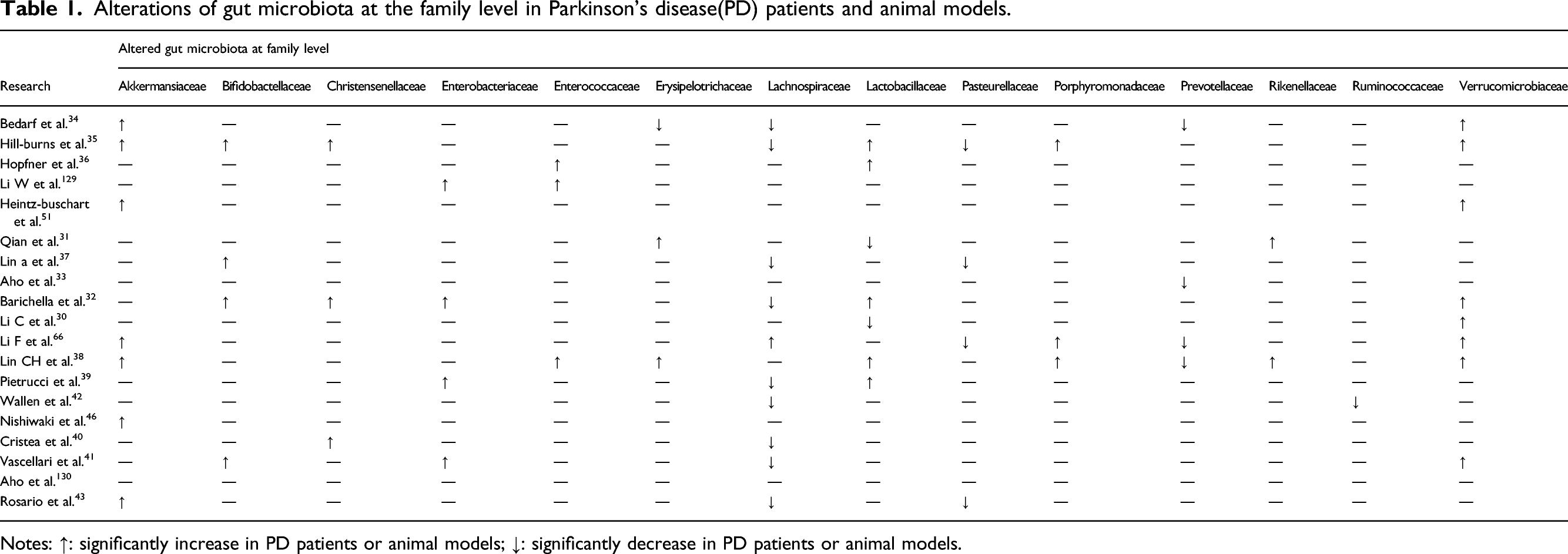
Notes: ↑: significantly increase in PD patients or animal models; ↓: significantly decrease in PD patients or animal models.
Alterations of GM composition are closely related to the metabolism and function of these microbes. Roseburia and Faecalibaterium were reported to decrease in PD patients. 48 Known as the health-promoting genera, Roseburia and Faecalibaterium can produce butyrate, a kind of short chain fatty acids (SCFAs). SCFAs can induce the expression of anti-inflammatory cytokines genes, exerting anti-inflammatory and neuroprotective effect through the MGBA. 49 Therefore, a decrease of Roseburia and Faecalibaterium in PD patients will create an inflammatory environment, contributing to the aggregation of α-syn and the cascading neuroinflammation. 50 Similarly, both Wallen et al. 44 and Cristea et al. 42 have reported lower abundance of SCFA-producing bacteria in PD patients, while they also have their own points. Wallen et al. 44 revealed that there was a significant increase in Lactobacillus and Bifidobacterium, two carbohydrate-metabolizing bacteria. Cristea et al. 42 also found an increase in Bifidobacterium, and they pointed that GM in PD patients demonstrated a lower capacity of carbohydrate fermentation and butyrate synthesis, with the process of proteolytic fermentation enhanced and production of deleterious amino acid metabolites increased.
The results of studies on PD animal models are similar to most of those on PD patients. Sun et al. 51 reported an increase in phylum Proteobacteria in fecal samples of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD mice, which is consistent with the findings in humans. Likewise, Lai et al. 52 found the decreased family Lachnospiraceae in a MPTP-induced mouse model, just like the results in PD patients. However, there are some inconsistencies at both families and genera levels, which may be ascribed to the differences between humans and mice, especially the dietary habits of PD patients and healthy controls.
In a word, GM in PD patients and animal models are significantly altered, which can possibly be regarded as biomarkers for early diagnosis or targets for PD treatment. However, due to the differences in the design of studies as well as the selection of subjects, the exact rules of alterations including the altered types and timepoints remain unclear.
Clinical correlations between alterations of gut microbiota and PD
Clinical correlations between gut microbiota or its metabolites and Parkinson’s disease(PD).
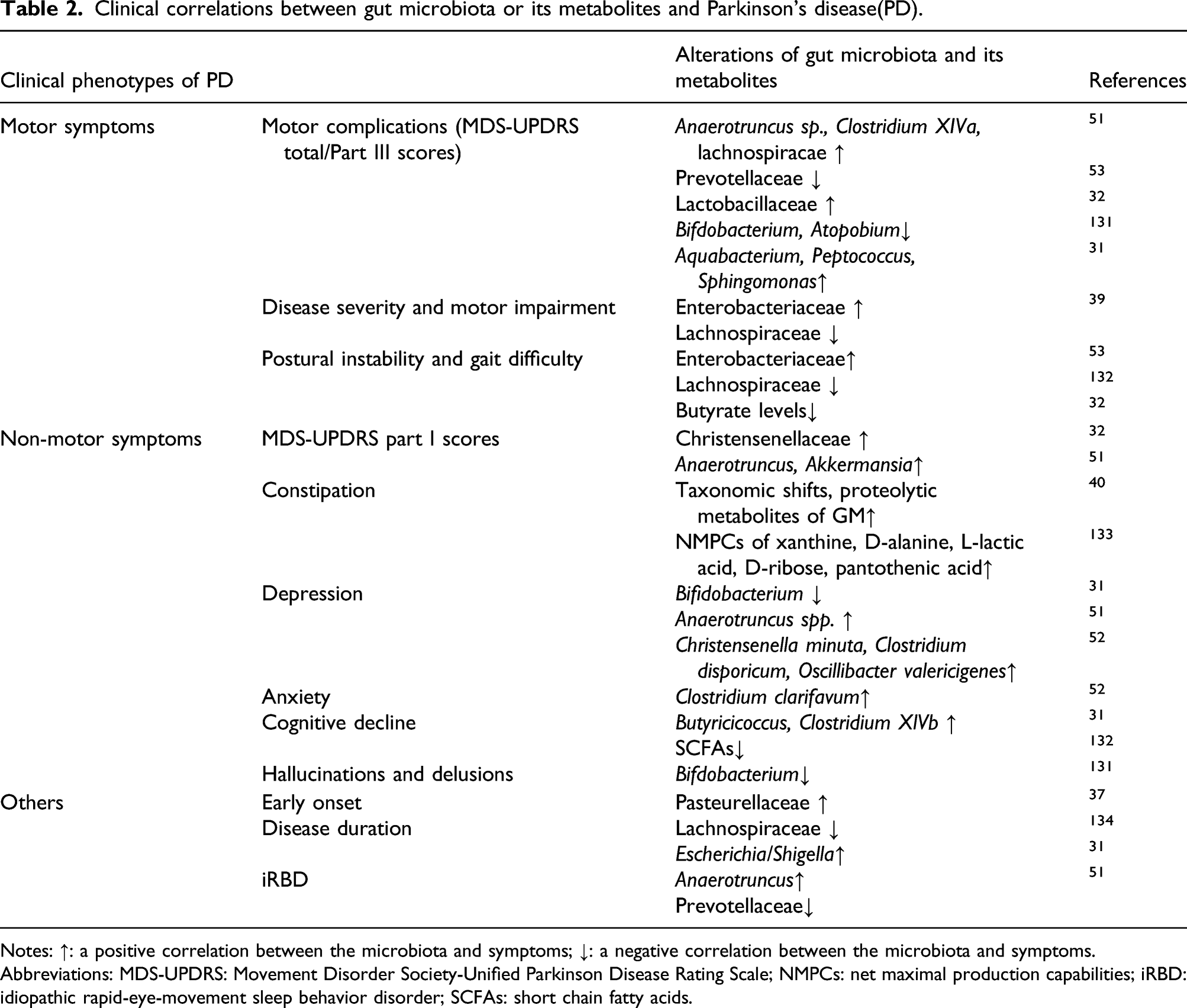
Notes: ↑: a positive correlation between the microbiota and symptoms; ↓: a negative correlation between the microbiota and symptoms.
Abbreviations: MDS-UPDRS: Movement Disorder Society-Unified Parkinson Disease Rating Scale; NMPCs: net maximal production capabilities; iRBD: idiopathic rapid-eye-movement sleep behavior disorder; SCFAs: short chain fatty acids.
Recently, clinical correlations between GM and PD have been extended to other aspects, including the motor symptoms, staging and duration as well as the onset time of PD.34,39,61 For example, Tan et al. found that increased Enterobacteriaceae and decreased Lachnospiracae was associated with motor impairment.41,62 However, abundant Lachnospiracae was related to motor symptoms by Heintz-Buschart et al., 53 which might be attributed to different selection of cohorts. Studies by Zhang et al. 63 revealed that 15 genera are related to the Hoehn and Yahr stage while 11 genera are linked to the duration of PD. Furthermore, 8 inflammation-associated genera are positively associated with the Hoehn and Yahr stage or the disease duration, indicating the potential role of inflammation in PD pathogenesis.
Other than correlated with the clinical phenotype and severity of PD, alterations of GM can also serve as a marker for the initiation and progression of PD. For example, idiopathic rapid-eye-movement sleep behavior disorder (iRBD) is known to predispose to neurodegenerative diseases including PD. 64 Nishiwaki et al. 50 found that there was an increase in Akkermancia but not a decrease in SCFA-producing bacteria in iRBD. Thus, a decrease in a SCFA-producing genus Faecalibacterium might be a hallmark for the transition from iRBD to PD, indicating GM as a potential therapeutic target to retard the progression of PD. However, due to the difficulties in identifying non-motor symptoms of PD in clinical practice, the number of clinical studies among newly diagnosed or pre-motor patients are limited. Yan et al. 65 found elevated Sybergistetes, Akkermansia, and Eggerthella lenta and decreased Prevotella in A53T transgenic monkey models with early stages of PD. Based on existing data of 423 newly diagnosed PD patients, Jones et al. 66 concluded that gastrointestinal symptoms, which is associated with GM dysbiosis, may predict cognitive function in de novo PD. Further clinical studies focusing on pre-motor stages of PD are warranted.
In general, alterations in GM are clinically correlated with the symptoms and development of PD. However, mechanisms behind the correlations have not been clarified, and evidence of the relationship between the specific changes of GM and the development of PD is limited. Therefore, we expect more cohort studies with data supporting the association between GM alterations and the progression of PD as well as the specific alterations of GM composition.
Possible GM-inflammation-PD pathogenesis
Emerging studies have revealed that the initiation and progression of PD can be attributed to the inflammation originating from the gut. For instance, inflammatory bowel diseases (IBD) share some risk genes such as LRRK2 67 and CARD15 68 with PD, indicating the potential role of genetic risk factors in gut inflammation through MGBA. Besides, PD patients were reported to have increased bowel inflammation. 69 Devos et al. 70 found an increase in some pro-inflammatory cytokines (IL-1β, IL-6, IFN-γ, TNF-α) and glial markers (GFAP, Sox-10) in colonic biopsies of PD patients. Stool immune profiles of PD patients also revealed an elevated expression of several pro-inflammatory cytokines and chemokines such as IL-1α, IL-1β, and IL-8. 69 In this part, we will discuss the possible pathogenesis of PD represented by GM dysbiosis, intestinal barrier, gut inflammation, and neuroinflammation, highlighting the two mediators: α-syn and pro-inflammatory substances produced by GM.
Gut microbiota, intestinal barrier, and gut inflammation
There is a mutual relationship between GM, intestinal barrier, and gut inflammation (Figure 1 The link between gut microbiota dysbiosis, intestinal barrier and gut inflammation. There is a mutual relationship between gut microbiota dysbiosis, intestinal barrier and gut inflammation. Alterations of gut microbiota may increase the permeability of intestinal epithelial barrier, exposing the enteric nervous system and the immune system to the bacteria and their metabolites and thus contributing to the gut inflammation. The inflammatory environment will aggravate the dysfunction of intestinal barrier and the dysbiosis of gut microbiota.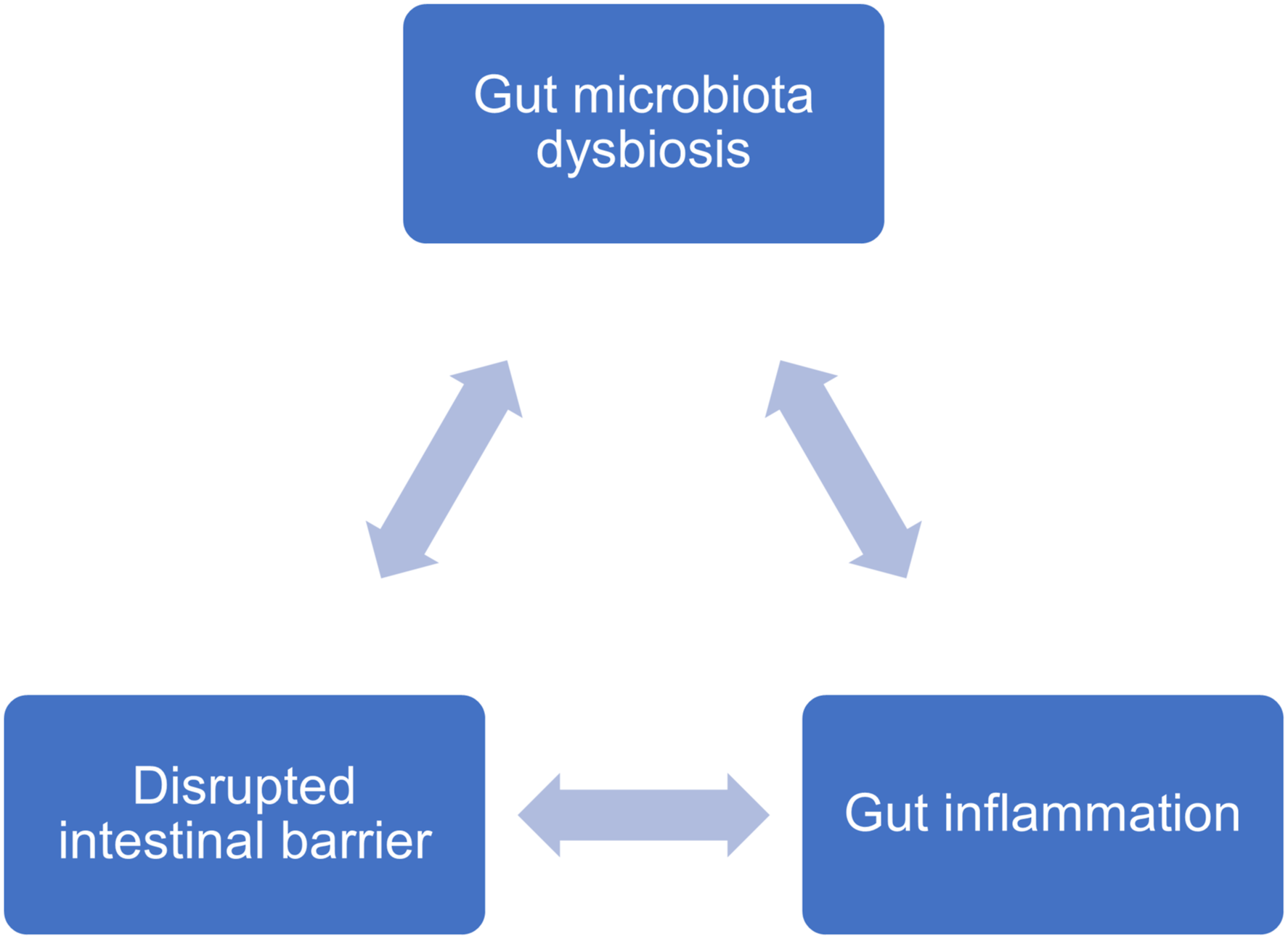
GM dysbiosis in PD are shown to be associated with the destructed intestinal barrier. Involved in the mucin synthesis and the production of SCFA, Prevotellaceae were reported to decrease in PD patients, 77 suggesting the possible role of increasing intestinal permeability in PD pathogenesis. Due to the gastroparesis and impaired GI motility in PD, small intestinal bacterial overgrowth (SIBO) is common in PD patients, 78 which will disrupt the integrity of the small intestinal barrier, resulting in the immune stimulation and/or alterations in L-dopa absorption. This was supported by the observation that motor fluctuations were improved after treating PD patients with rifaximin. 79
Increased permeability of intestinal barrier is closely associated with gut inflammation, which has been proved in both PD patients 80 and animal models. 81 The intestinal epithelial barrier prevents the invading pathogenic microbes and other environmental factors from altering gut homeostasis, thus alleviating gut inflammation. The possible mediators between the impaired intestinal barrier and gut inflammation include the ENS and the immune system. As the first level of the neural network of the GI system, 82 ENS is represented by neurons of the myenteric (Auerbach’s) plexi, submucosal (Meissner’s) plexi, and enteric glial cells (EGCs). 83 The neurons are responsible for the local reflexes such as the relaxation and contraction of the smooth muscle, while EGCs are essential for the development and differentiation of the neurons. 84 Additionally, EGCs are involved in the modulation of the intestinal barrier and gut inflammation. 70 Glial-derived neurotrophic factors (GDNFs) were shown to be fundamental in maintaining the integrity of the intestinal epithelial barrier, 84 and some pro-inflammatory cytokines and glial markers were increased in colonic biopsies of PD patients, which is link with the disease duration. 70 Pro-inflammatory cytokines secreted by GM activate the EGCs, contributing to the subsequent inflammation in the ENS and even the CNS. Besides, the neurotoxic metabolites of GM can stimulate the afferent neurons of ENS, thus resulting in the neurological dysfunction through direct neural communication 73 and affecting the function of the intestinal barrier as well as gut inflammation.
Studies have also revealed both the innate and the adaptive immune system are dysregulated in neurodegenerative diseases including PD. 85 As an essential part of the innate immunity, toll-like receptors (TLRs) are closely related to the early recognition of infection defensing against the invading microbes. 86 Evidence has shown that the disruption of TLR signaling may be involved in PD. For instance, expressed in the ENS and the gut smooth muscle, TLR2 can regulate not only the integrity of the intestinal barrier but also the activation of the ENS, even the microglial cells in the CNS. 87 Moderate activation of TLR2 was reported to restore the neurogenesis in the gut and increase the number of nitrergic neurons in GF mice, 88 while excessive activation of TLR2 can overstimulate the microglial cells, leading to the subsequent neuroinflammation and PD development. 87 TLR4 also plays a crucial role in the process of inflammation. Increased permeability of intestinal barrier contributes to the translocation of some GM metabolites including LPS to the internal environment, thus activating the TLR4-mediated inflammatory cascades. The cascades then induce the systematic inflammation and/or the neuroinflammation, aggravating the neurodegeneration in PD. 89 Another supporting study found that the knockdown of TLR4 substantially alleviated the dysfunction of intestinal barrier, decreased the activation of microglia and the loss of dopaminergic cells in the CNS, explaining the relief of some motor symptoms in rotenone-induced PD mice. 90 Alternatively, elevated gut inflammation will destruct the intestinal barrier, contributing to a more severe leakage of bacteria and their metabolites to the blood circulation. 91 The dysfunction of intestinal barrier and the inflammatory environment can also disrupt the homeostasis of GM, affecting the diversity and function of these microbes. 92
CNS inflammation
Neuroinflammation presented by the activation of microglia and the dysfunction of BBB can be essential in PD pathology.93, 94 As the most substantial immunocytes in the CNS, microglia are at the first line of defensing against the pathogenic infection in the brain, initiating the downstream immune responses. 95 Moderate microglial activation was proved to be beneficial, 96 while overactivation of microglia will lead to the excessive production of pro-inflammatory cytokines (including IL-1β, IL-6, IFN-γ, and TNF), exerting negative effect on the dopaminergic neurons. 97 Consisting of endothelial cells, the basal membrane, tight junction and the end feet of astroglia, BBB is essential for the homeostasis of brain. Disruption of the integrity of BBB was proved to be related to several neurodegenerative diseases including PD. 98 Permeability of BBB in germ-free (GF) mice was increased due to the decreased expression of tight junction proteins such as claudin-5 and occludin, while administration of butyrate can strengthen the tight junctions and preserve BBB permeability in GF mice. 99 Likewise, sodium butyrate alleviated BBB lesion in MPTP-induced PD mice by upregulating the expression of occluding and ZO-1, 100 indicating the impact of GM and their metabolites on BBB integrity, neuroinflammation, and consequent neurodegeneration. Other metabolites like quinolinic acids can also serve as modulators of BBB, influencing the pathology of neurodegenerative diseases like PD. 101 BBB dysfunction has also been assessed in the midbrain and striatum of PD patients.102-104 Kortekaas et al. 102 found PD patients have reduced P-glycoprotein function in the BBB of midbrain, facilitating the translocation of pro-inflammatory substances. Striatal BBB permeability also increased in PD patients, allowing for the potential therapeutic usage. 103
Potential mediators: α-syn and pro-inflammatory substances
There has been substantial evidence on the association between gut inflammation and PD.68,70,74,75 However, the specific mechanism remains unclear. The most promising mediators contain α-syn and GM-originating pro-inflammatory substances, and a potential mechanism is summarized in Figure 2. A proposed schematic representation of gut microbiota(GM) dysbiosis-inflammation-Parkinson’s disease(PD) mechanisms. GM dysbiosis contributes to the disruption of intestinal barrier and gut inflammation, promoting the production of pro-inflammatory cytokines and the accumulation as well as misfolding of α-syn. Misfolded α-syn may propagate to the brain through the vagus nerve or the blood circulation, while pro-inflammatory cytokines reach the central nervous system(CNS) via the humoral pathway. Both α-syn and pro-inflammatory cytokines may destruct the blood–brain barrier and activate the microglia in the CNS, which will result in the neuroinflammation and neurodegeneration in PD.
As one of the hallmarks of PD, α-syn is much more widespread in PD patients as compared to the physical condition. 105 Physically, α-syn is crucial for the regulation for neurotransmission, while in neurodegenerative diseases, α-syn may have an impact on triggering and/or enhancing the activation of microglia and astroglia. 106 The abundance of α-syn in the gut is tightly correlated with the GM alterations which contribute to the disruption of intestinal barrier and gut inflammation. Gut inflammation may lead to the misfolding of α-syn or downregulate the clearance of it, resulting in the accumulation of α-syn and its inflammatory cascades. Numerous studies have supported that the propagation of α-syn through the vagus nerve, known as the Braak hypothesis. 107 It was inspired by the finding that lesions in the ENS precede those in the CNS. 108 In PD animal models, the transmission of aberrant α-syn was prevented by vagotomy, 109 thus prohibiting the misfolding of α-syn in intermuscular neurons and relieving the symptoms of PD. Recently. there has been some controversy on the results of vagotomy, indicating another pathway of α-syn propagation. Arotcarena et al. 110 observed an increase in the concentration of α-syn in the whole blood of the monkeys that were enteric-injected with α-syn-containing Lewy body extracts from PD patients, thus proposing the general circulation pathway for the long-distance transmission of α-syn between the gut and CNS. However, more studies are needed to confirm this hypothesis.
The pro-inflammatory substances produced by GM include pro-inflammatory cytokines and other metabolites like LPS. Other than the traditional blood circulation pathway, these substances can possibly propagate through lymphatic circulation, which was supported by the discovery of a network of lymphatic vessels existing in dural sinuses and connecting the GI tact with CNS. 111 Pro-inflammatory cytokines and LPS may contribute to neuroinflammation by disrupting the BBB and activating microglia, which resembles the process in the gut. 112 In addition, LPS can interact with α-syn by binding to it and initiating the fibrilization in the gut that facilitates the propagation of α-syn to the CNS. 91 LPS-induced neuroinflammation and memory loss in mice was reduced after administration of Lactobacilli-fermented cow’s milk, indicating the possible effects of probiotics on LPS-mediated pathology of PD. 113
GM-targeted therapeutic strategies for PD
Due to the limited knowledge of PD pathogenesis, there has been no comprehensive therapeutic approach to prevent the progression of PD. Since the crucial role of GM in PD pathophysiology, altering the composition as well as metabolites of GM may be a promising intervention to slow the progression and relieve the symptoms of PD. These GM-targeted interventions mainly include antibiotics, probiotics and/or prebiotics, fecal microbiota transplant (FMT) and dietary changes.
Antibiotics
Known as one of the most powerful tools to alter GM composition, antibiotics has been reported in various studies on PD. More recently, Koutzoumis et al. 114 studied the influence of antibiotics on PD. After injecting with broad-spectrum antibiotics together with oxidopamine, 90% of the GM richness was reduced, of which the abundance of Firmicutes decreased, while the abundance of Proteobacteria, Verrucomicrobia, Bacteroidetes, and Cyanobacteria increased. These alterations of GM decreased the levels of IL-1b and TNF-a in the striatum, reduce the damage to dopaminergic neurons and alleviated motor deficits in PD rodent models. 115 Emerging studies focused on the role of minocycline. It was shown to reduce the Firmicutes/Bacteroidetes ratio, re-establishing the GM homeostasis to the normal condition 116 and exerting a beneficial influence on the CNS via MGBA. For example, minocycline was observed to prohibit the activation of microglia, decelerate the depletion of dopamine in striatum and nucleus accumbens and prevent the neurodegeneration of nigrostriatal dopaminergic neurons in MPTP-induced PD mice . 117 In addition, minocycline may serve as a scavenger for peroxynitrite detoxification and thus inhibit the aggregation and misfolding of α-syn. 117 However, there are some contradictions on the specific effect of minocycline on PD. A study found an amplified damage to dopaminergic neurons in spite of the prohibited microglial activation, 118 and similar results were reported in several other animal models. 119
Considering the unknown specific correlation between GM and PD, as well as the side effects of antibiotics, further fundamental studies and in-vivo experiments are expected before the application on PD patients.
Probiotics, prebiotics, and synbiotics
Probiotics are microbes conferring healthy benefits on human hosts. 120 Living in the GI tract, probiotics are involved in the homeostasis of GM, modulation of immune responses and the integrity of intestinal barrier. 121 Recent studies have found that probiotics might reduce the neuroinflammation in the CNS via MGBA and inhibit the subsequent loss of dopaminergic neurons, 122 indicating their possible application in PD. Various studies focused on Lactobacilli and Bifidobacteria. More recently, Magistrelli et al. 123 further demonstrated the mechanisms. L. salivarius LS01 and L. acidophilus LA02 significantly reduced the production of pro-inflammatory cytokines while increased the production of anti-inflammatory ones in PD patients. Furthermore, most strains of genus Lactobacillus and Bifidobacterium also regulate the membrane integrity and inhibit the overgrowth of several pathogenic bacteria, such as E. coli and Klebsiella pneumoniae, which may lead to GM dysbiosis and the consequent inflammatory cascades. Additionally, Castelli et al. 124 found that a mixture of nine probiotic strains was involved in the modulation of neuroinflammation and brain-derived neurotrophic factor (BDNF) signaling pathway. It can also increase the expression of several neuroprotective proteins and might decrease the counteracting ones, exerting a neuroprotective effect. In addition to their direct impact of neuroprotection, probiotics may also influence the synthesis and transformation of levodopa. Bacillus sp. JPJ can directly synthesize levodopa from L-tyrosine in-vitro. 125 Besides, more than 50 Enterococcus strains, several Lactobacillus and Staphylococcus may convert levodopa to dopamine through the tyrosine decarboxylase encoded in their genomes. 126 Combination of these probiotics and levodopa may improve the efficacy of traditional treatment. However, the exact influence of probiotics on the pharmacodynamics and pharmacokinetics of levodopa remains to be studied.
Prebiotics refer to the complex carbohydrates facilitating the growth of probiotics, of which Galacto-oligosaccharides (GOS) and fructo-oligosaccharides (FOS) are most well-known. 127 By metabolizing them, probiotics can synthesize SCFAs. SCFAs are involved in the modulation of intestinal barrier, BBB and immune reponses, affecting the homeostasis of CNS. 49 In addition, butyrates, one type of SCFAs, was shown to induce Atg5-and PI3K/Akt/mTOR-related autophagy pathway, causing α-syn degradation in a pesticide-induced PD rat model. 128
Synbiotics are the combination of probiotics and prebiotics, 120 introducing the probiotic bacteria to the gut and promoting their growth simultaneously. Rajkumar et al. 129 found that probiotic Lactobacillus salivarius reduced the inflammatory markers in the serum of healthy subjects, and the reduction seemed more evident when combined with prebiotic FOS. Similarly, administration of a symbiotic yogurt (containing probiotic Bifidobacterium animalis together with prebiotic FOS) significantly improved the bowel movement in a patient with functional constipation, 130 indicating the possible relief of constipation in PD patients.
Since the correlation between GM and PD is still unclear, we should be more cautious when considering probiotics, prebiotics or synbiotics as therapeutic strategies for PD. Further studies in both animal models and PD patients should focus on the exact probiotic strains, the proportion of probiotics and prebiotics, treatment durations as well as dosages.
Fecal microbiota transplantation
Fecal microbiota transplantation (FMT) is a technique transplanting fecal filtrate from healthy donors to patients, which can restore the normal GM homeostasis in the gut. 131 First proposed to treat GI disorders, FMT has been used in various other diseases including neurodegenerative diseases. 131 Aside from benefits on the GI symptoms like constipation, FMT can also markedly relieve the non-GI symptoms in PD patients. 132 Possible mechanisms may include the amelioration of GM dysbiosis, which is indicated by decreased fecal SCFAs and improved motor symptoms. 51 FMT can also downregulate the TLR4/TNF signaling pathway, alleviate inflammation in the gut and CNS. 51 Based on the limited amount of studies, there are several problems remaining to be solved. Most in-vivo studies did not assess the extent of inflammation and α-syn aggregation, which features the progression of PD. 132 Considering the ethical issues and the potential side effects, large-scale applications of FMT in PD patients should be put off until further studies on the fundamental theories and long-term effects are conducted.
Diet
Considered as one of the gateways to the outer environment, 133 gut together with the inside GM is vulnerable to the environmental factors including diet. Since there is no effective therapeutic strategy, dietary interventions are necessary in both PD patients and high-risk groups. Diets with adequate fruits, rich vegetables, certain amount of high-quality olive oil and low dietary fat are commonly recommended for early intervention of PD. 134 Fruits and vegetables are rich in fibers and vitamins, which facilitates the synthesis of SCFAs, improving the intestinal barrier and exerting neuroprotective effects. 49 Vitamins are also associated with the occurrence and progression of PD, possibly by affecting GM composition.135-137 Higher level of vitamin E contributes to a lower risk of PD,135,137 while low intake of vitamin B6 increases the risk of PD. 138 Adequate intake of vitamin D3 prevents the progression of Hoehn and Yahr stage in PD patients, 136 and long-term administration of vitamin B2 improves the motor symptoms of PD patients. 139 High-quality olive oil may decrease the inflammatory mediators and increase the number of lactic acid bacteria, suppressing the potential inflammatory cascades and regulating GM composition. 140 High fat especially saturated fat may contribute to GM dysbiosis, increasing Firmicutes and Proteobacteria while reducing Bifidobacteria. 141 Besides, dietary fat can also disrupt the intestinal barrier, facilitating the intestinal, systematic and even CNS inflammation. 142 A balanced diet plays an essential role in the regulation of GM composition and immune responses, 143 suggesting diet may be a potential tool to intervene the occurrence and development of PD. However, there are various other environmental factors influencing GM and PD, such as geographical conditions and exercise, which makes it more difficult to examine the precise role of dietary habits in PD. Therefore, studies in larger cohorts and animal models are expected.
Conclusions
Recent studies have indicated the GM alterations in PD patients and the clinical relevance of these changes, which sheds light on the underlying relationship between GM and PD. However, the exact mechanisms await elucidation. GM can communicate with the CNS via humoral or direct neural pathways, in which inflammation plays a crucial role. In the inflammatory cascade, α-syn, pro-inflammatory cytokines and other GM metabolites may serve as major mediators. Further studies are expected to investigate the characteristics of GM alterations in presymptomatic stages of PD, contributing to the earlier diagnosis of PD. Besides, more emphasis should be placed on both pharmacological and dietary interventions which aim at altering GM composition, enhancing the intestinal barrier or alleviating gut inflammation. GM-based therapies for PD patients mainly include antibiotics, probiotics, prebiotics, synbiotics and FMT, while lifestyle changes especially dietary alterations can be applied in both high-risk people and PD patients. However, larger scales of microbial interventions in human require more consideration when the exact mechanism of PD pathology remains unclear. In addition to the safety and efficacy of these therapies, the specific strain types (e.g., Lachnospiraceae,41,53,79,144 Bifidobacterium,32,39,145 and Prevotellaceae36,40,53,61), dosages, durations as well as intervals of the treatment should be considered. Moreover, since the GM composition is environment- and age-affected, GM-targeted therapeutic strategies are likely to be individualized.
Footnotes
Acknowledgment
We would like to acknowledge National Natural Science Foundation of China for supporting this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (No. 81673137).
