Abstract
The aim of this article is to study T-helper (Th) cell differentiation in the progression of acute, subacute, and chronic atopic dermatitis. Skin biopsies from 48 patients with acute, subacute, and chronic atopic dermatitis were studied using immunohistochemistry with antibodies to TARC/CCL17, CTACK/CCL27, and RANTES/CCL5. Peripheral blood mononuclear cells were studied in 17 patients using flow cytometry to measure the content of Th1/Th2 cells and Th17/Treg cells. Levels of interferon (IFN)-γ, interleukin (IL)-4, IL-17A, and transforming growth factor (TGF)-β1 were evaluated with enzyme-linked immunosorbent assay (ELISA). Distinctive expressions of T-cell-specific chemokines TARC/CCL17, CTACK/CCL27, and RANTES/CCL5 were observed at different stages of atopic dermatitis, which were consistent with the differentiation of the Th cell subsets, Th2/Th1, and Th17/Treg. Th2 and Th17 were acute-phase subsets, while Th1 and Treg were chronic-phase subsets. At an early stage of atopic dermatitis, Th17 and Th2 cells were found in peripheral blood mononuclear cells, followed by Th1 cells, Treg cells, and eosinophils; in late-stage or subacute and chronic atopic dermatitis, Th17 and Th2 cell numbers decreased. The levels of the IFN-γ and TGF-β1 increased during the progression of atopic dermatitis from acute to chronic forms. The levels of IL-17A and IL-4 decreased during the progression of atopic dermatitis from acute to chronic forms. The differentiation of Th subsets at distinct phases in atopic dermatitis may form the basis for further studies on the classification or control of this increasingly common clinical condition.
Introduction
Atopic dermatitis is a relapsing, non-contagious, and itchy skin disorder that is now believed to be due to altered regulation in the control of the innate immune system. 1 It has increased in incidence rate in recent decades and is more prevalent in developed countries. 2 Histology of the skin lesions shows a mixed acute and chronic inflammatory cell infiltrates in the skin in which CD4+ and T-helper (Th) cells predominate. 3 The Th cells play an important role in the adaptive immune system and can regulate the activity of other immune cells by releasing immune modulating cytokines. The CD4+ T cells can be activated and mainly developed to four subsets of Th1, Th2, Th17, and regulatory T cells (Treg). 4 Th1 and Th 2 cells have important roles in phagocyte-dependent and cell-mediated immunity,5,6 and a novel and unique subset of IL-17-producing CD4+ Th17 cells can mediate the host immunity against extracellular bacteria and fungi. 7 Treg cells have a role in the control of the immune response, and their dysregulation may result in excessive and autoimmune responses. 8 Mature T cells can be attracted by chemokines by recognition through their distinct receptors. Selective recruitment of the chemokine receptor, CCR4, expressed by Th2 cells is mediated by the macrophage-derived chemokine (CCL22) and thymus and activation-regulated cytokine (TARC/CCL17). 9 CTACK/CCL27 is a skin-specific chemokine exclusively produced by epidermal keratinocytes, which binds to CCR10 expressed on skin-homing lymphocytes. 10 Th1 cells migrate to the affected area by the response to RANTES/CCL5 signal. 11
In this study, the differentiation of Th cells, specifically Th1/Th2 and Th17/Treg, in different stages of atopic dermatitis in patients and a range of associated chemokines were investigated.
Materials and methods
Patients studied
A total of 48 patients (Table 1), who had clinically or histologically confirmed atopic dermatitis at different stages, including three patients with asthma, were studied at the First Affiliated Hospital, Guangxi University of Chinese Medicine. All the patients signed a written informed consent and the study was approved by the local ethical committee.
The information of patients in difference stages.

The characteristics indicate the location of the patient’s symptoms.
Immunohistochemical staining
Punch biopsies of 4 mm were taken from skin with active eczema in 48 patients with atopic dermatitis recruited in this study. Tissue sections of skin biopsies were de-paraffinized, rehydrated, immersed in 3% hydrogen peroxide solution for 30 min, heated in 0.01 M citrate buffer, pH 6.0, at 95°C for 25 min, and cooled to room temperature. After three washes in phosphate-buffered saline (PBS), the tissue sections were incubated with 10% normal goat serum at 37°C for 1 h at room temperature and then incubated with the primary antibody against TARC/CCL17 (monoclonal mouse IgG1 clone # 124308; R&D Systems, USA), CTACK/CCL27 (monoclonal mouse IgG1 clone # 124308; R&D Systems), RANTES/CCL5 (Monoclonal Mouse IgG1 Clone # 21418; R&D Systems) overnight at 4°C. After washing with PBS, the slides were incubated with goat anti-rabbit IgG biotinylated secondary antibody (R&D Systems) for 30 min at 37°C, followed by streptavidin–peroxidase incubation at 37°C for 30 min. Immunolabeling was visualized with a mixture of diaminobenzidine (DAB) solution. Counterstaining was done using hematoxylin and eosin (H&E).
Flow cytometry assay
Analysis of the Th cell subsets by flow cytometry was performed as previously described. 11 Briefly, peripheral blood mononuclear cells (PBMCs) were isolated from patients and control subjects by Ficoll-hypaque density gradient centrifugation. Cells were then washed and stained, using a direct immunostaining method, with fluorescein isothiocyanate (FITC)-labeled FITC Mouse anti-Human CD4 (No. 566320; BD Biosciences, USA) for 20 min at room temperature. After centrifuging at 1000g for 5 min, 100 mL of Cytofix/Cytoperm buffer (BD Biosciences) was added to the cell suspension that was incubated for 20 min at 4°C and washed with Perm/Wash solution as per the protocol (BD Biosciences). Cells were stained with a allophycocyanin (APC) Mouse anti-Human CD25 (No. 560987; BD Biosciences), phycoerythrin (PE) Mouse anti-Human IL-17A (No. 560436; BD Biosciences), PE Mouse Anti-Human IL-4 (No. 554516; BD Biosciences), and PerCP-Cy™5.5 Mouse Anti-Human IFN-γ (NO. 560704; BD Biosciences), for 20 min at 4°C. Fluorescence profiles were analyzed by flow cytometry using the FACSCanto (BD Biosciences). Data were analyzed with FlowJo software.
Enzyme-linked immunosorbent assays
Enzyme-linked immunosorbent assay (ELISA) kits for detection of human interferon (H-IFN)-γ, human interleukin (H-IL)-4, H-IL-17A, and human transforming growth factor (H-TGF)-β1 were purchased from Multisciences Ltd. ELISA was performed as per the manufacturer’s protocol. Briefly, diluted antibody solution was incubated with plasma collected from peripheral blood of atopic dermatitis patients at different stages for 1 h, after several washes, horseradish peroxidase (HRP) solution was added and incubated for a further 1 h. The chromogenic substrate 3,3′,5,5′-tetramethylbenzidine (TMB) was added and incubated for 15 min in the dark, and the reaction was then quickly stopped. All incubations were done at room temperature. The absorbance in each microplate well was determined at 450 nm within 5 min of the end of the reaction.
Statistical analysis
All statistical analyses were performed using SPSS13.0 software. Kolmogorov–Smirnov goodness-of-fit test was used to determine the normality (Gaussian-shaped distribution) of the data. Analysis of variance (ANOVA) was used for comparisons among multiple groups, and data were expressed as mean ± standard deviation (SD) of three independent experiments. A statistically significant difference was regarded as P < 0.05.
Results
TARC/CCL17, CTACK/CCL27, and RANTES/CCL5 were expressed at different stages of atopic dermatitis
Skin biopsies were examined at the acute, subacute, and chronic stages of atopic dermatitis, which were confirmed by histology or clinical appearance. Immunohistochemical staining (Figures 1 and 2) demonstrated that CTACK/CCL27 and TARC/CCL17 were predominantly expressed during the acute and chronic stages, which were significantly higher than during sub-acute stage. Also, expression sites of CTACK/CCL27 moved from epidermis in acute stage to dermis in chronic stage; however, TARC/CCL17 expressions were mainly observed in stratum spinosum and dermis during all stages. For the RANTES/CCL5, it was predominantly expressed in the sub-acute stage and distributed in stratum spinosum and dermis. Interestingly, there was nearly no expression of RANTES/CCL5 was observed in chronic stage. Meanwhile, in skin biopsies at the subacute stage of atopic dermatitis, lymphocyte infiltration was observed in the superficial layer of the dermis. When atopic dermatitis progressed to the chronic stage, hyperkeratosis of the stratum corneum was seen, with dense collections of blood capillaries in the upper dermis (data not shown). The distinctive expression of chemokines at different stages of atopic dermatitis suggested different subsets of T cells were responsible for the progression of atopic dermatitis.

TARC/CCL17, CTACK/CCL27, and RANTES/CCL5 expressed in atopic dermatitis. Representative photomicrographs of 4-mm punch biopsies taken from skin with active eczema in 48 patients with atopic dermatitis stained with antibodies to chemokines TARC, CTACK, and RANTES. Arrows indicate specific staining for the different antibodies. Sections are from acute, subacute, and chronic atopic dermatitis. Counterstained with hematoxylin and eosin (H&E, bars = 20 µm).
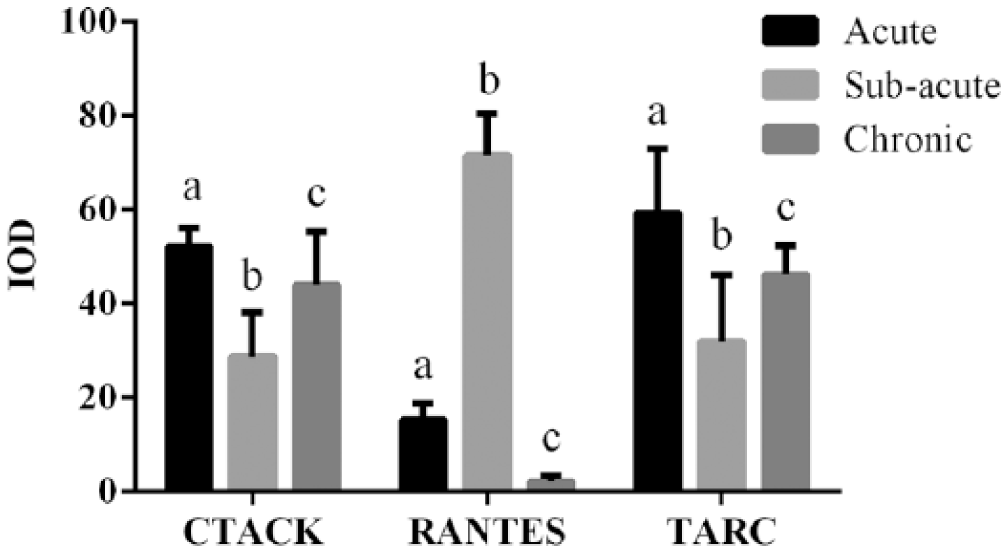
Quantitative immunohistochemical staining results of chemokines TARC, CTACK, and RANTES in different stages (different letters indicate significant differences between groups). A total of 17 patients (6 acute stage patients, 5 subacute stage patients, and 6 chronic stage patients) were quantified.
Th1/Th2 and Th17/Treg varied from acute to chronic atopic dermatitis
Four subsets of CD4+ T-cells were further analyzed. The cell populations were identified by IL-17, IL-4, CD25, and IFN-γ staining using the flow cytometry assay (Figure 3) in six acute stage patients, five subacute stage patients, and six chronic stage patients randomly selected from 48 patients recruited in this study.
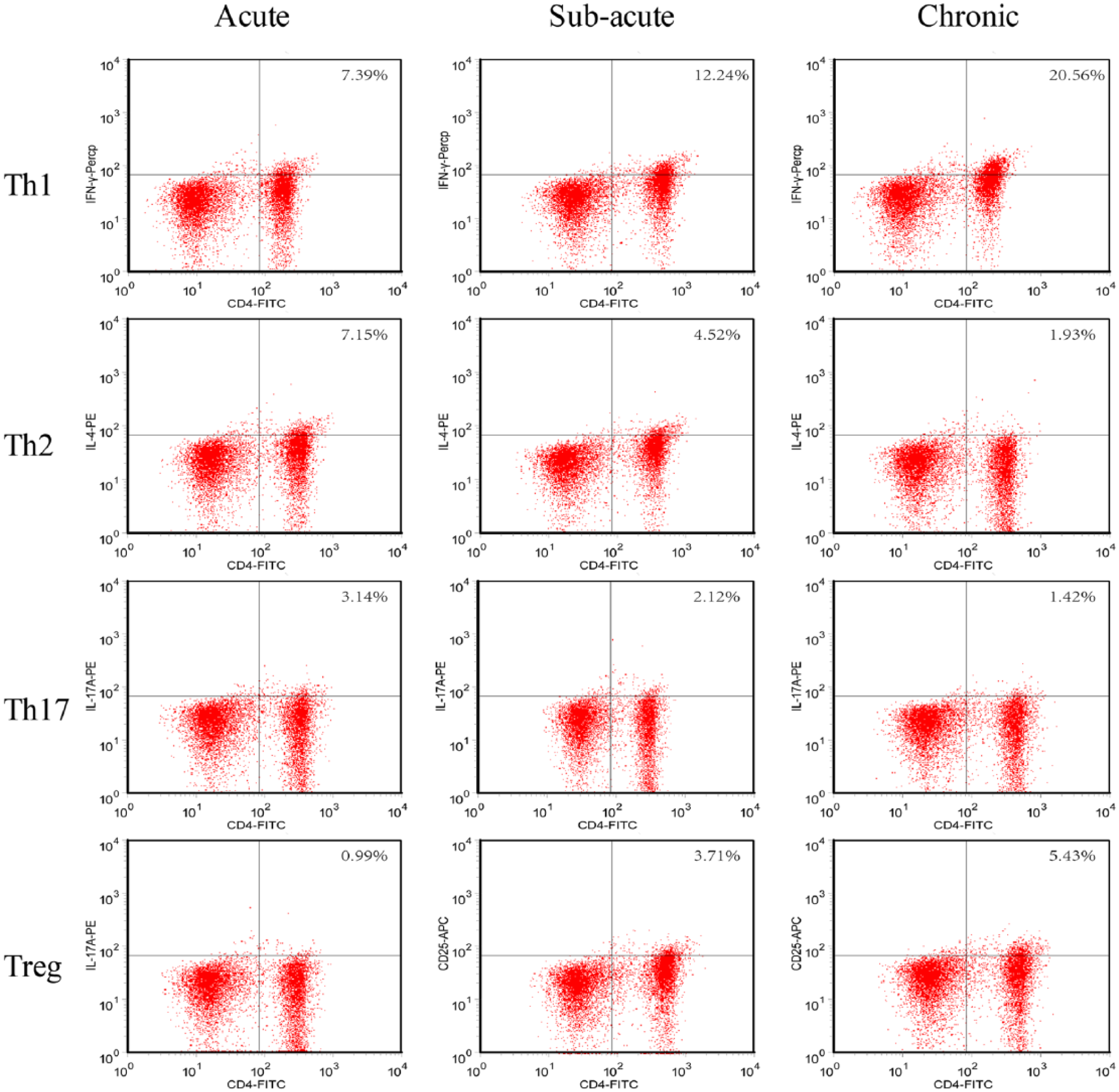
Identification of Th1/Th2 and Th17/Treg in acute, sub-acute, and chronic atopic dermatitis with flow cytometry assay. Flow cytometry assay were performed on CD4+ cells population isolated from peripheral blood. Cells were labeled with PE- or APC- or PerCP-Cy™5.5-conjugated IL-17A, IL-4, IFN-γ, and CD25.
Th17 cells were present proportionally as 3.063% (±0.653%) in the acute stage of atopic dermatitis, 1.775% (±0.211%) in the subacute stage, and 1.195% (±0.151%) in the chronic stage (P < 0.05; P < 0.05) (Figure 4(a)).

Quantification of Th1/Th2 and Th17/Treg in acute, subacute, and chronic atopic dermatitis. Flow cytometry was performed in six acute stage patients, five subacute stage patients, and six chronic stage patients randomly selected from 48 patients recruited in this study. For each individual, we tested the sample for three times. Statistical data of cell populations are shown by columns, with data from the average of six acute stage, five subacute stage, or six chronic stage patients. (a and b) Th17/Treg cells, (c and d) Th1/Th2 cells (different letters indicate significant differences between groups).
Treg cells were present proportionally as 0.91% (±0.0242%) in the acute stage of atopic dermatitis, 3.575% (±0.3125%) in the subacute stage, and 5.25% (±0.005%) in the chronic stage (P < 0.05; P < 0.01) (Figure 4(b)).
Th1 cells were present proportionally as 7.55% (±0.405%) in the acute stage of atopic dermatitis, 12.45% (±0.005%) in the subacute stage, and 19.2% (±0.08%) in the chronic stage (P < 0.05; P < 0.05) (Figure 4(c)).
Th2 cells were present proportionally as 7.0% (±0.02%) in the acute stage of atopic dermatitis, 4.4% (±0.08%) in the subacute stage, and 1.8% (±0.02%) in the chronic stage (P < 0.001; P < 0.001) (Figure 4(d)).
In summary, these data showed that Th1 and Treg cells increased while Th2 and Th17 cells decreased during the progression from acute to subacute to chronic atopic dermatitis.
IFN-γ, IL-4, IL-17A, and TGF-β1 expression in peripheral blood of patients with atopic dermatitis
The finding that the T-cell subsets varied during the progression of atopic dermatitis indicated that cytokine expression may also vary.
Cytokine levels were measured in the peripheral blood of 48 patients (8 acute, 12 sub-acute, 10 Acute to sub-acute, 11 sub-acute to chronic, and 7 chronic stage patients). The levels of the Th1-specific cytokine, IFN-γ, and the Treg-specific cytokine, TGF-β1, increased during the progression of atopic dermatitis from acute to chronic forms.
The levels of the Th17-specific cytokine, IL-17A, and Th2-specific cytokine, IL-4, decreased during the progression of atopic dermatitis from acute to chronic forms (Figure 5).

IFN-γ, IL-17A, IL-4, and TGF-β expression in peripheral blood of patients with atopic dermatitis. Samples were isolated from peripheral blood from 17 patients with atopic dermatitis. Enzyme-linked immunosorbent assay (ELISA) measurements of (a) IFN-γ, (b) IL-17A, (c) IL-4, and (d) TGF-β1 are shown by representative columns, with data from the average of the 8 acute, 12 sub-acute, 10 acute to sub-acute, 11 sub-acute to chronic, or 7 chronic stage patients (different letters indicate significant differences between groups).
Discussion
The findings of this study have shown that distinctive expressions of T-cell-specific chemokines TARC/CCL17, CTACK/CCL27, and RANTES/CCL5 were observed at different stages of atopic dermatitis, which were consistent with the differentiation of the Th cell subsets. These findings are supported by previously reported studies on cytokines and T cells in inflammatory dermatoses, including atopic dermatitis.3,7,10,11 Atopic dermatitis, asthma, and other Th2-setting diseases are demonstrated to be correlated with high level expression of chemokines. Nakazato et al. 12 proved Th2-specific chemokines TARC/CCL and MDC/CCL22 was responsible to severity of Alzheimer’s disease (AD) in affected children. Werfel 13 characterized immune responses at different stages of AD and demonstrated, when immune response is dominated by Th1 cells, the expressions of CXCL9, CXCL10, CXCL11, CCL5, and other chemokines are increased and play a role in IFN-γ-mediated Th1 type immune response. Also, Rebane et al. 14 demonstrated that IFN-γ-mediated apoptosis in keratogenesis, as well as high level of CCL5 and CCL8, contributes to edema and exudation by acanthocyte in AD patients. Moreover, skin-specific chemokine CTACK/CCL27 is vital in inflammatory permeation, by regulating IL-1β, tumor necrosis factor (TNF-α), and lymphocyte homing. 15 Increasing knowledge of the specific immune mechanisms involved at each stage of the development and progression of this disease may allow for the development of more specific and targeted treatments. The Th1/Th2 and Th17/Treg differentiation patterns in the progression of atopic dermatitis in this study indicate the dominant role of CD4+ cells in atopic dermatitis. Treatments that target these T cells according to the stage of atopic dermatitis may be a promising direction for future clinical studies.
Footnotes
Acknowledgements
C.S. and T.Y. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the National Natural Science Fund Program from the National Natural Science Foundation of China (No. 81360599); the Natural Science Foundation of Guangxi (NO. 2013GXNSFAA019148).
