Abstract
Background:
Portal hypertension is a complication of liver cirrhosis. The portal vein drains the spleen and the intestines, which are both rich in inflammatory mediators. Portal hypertension- induced stress within these organs that may result in pro-inflammatory changes. The association of these changes with regulatory T cells was not addressed before.
Aim:
Our aim is to investigate the involvement of some subsets of regulatory T cells in portal hypertension.
Methods:
In the current study we used the partial portal vein ligation model to demonstrate differences in the distribution of regulatory T cells within the portal vein and the inferior vena cava associated with portal hypertension.
Results:
We show that CD4+CD25+FoxP3+ regulatory T cells are significantly (
Conclusions:
These novel findings support the involvement of regulatory T cells in the inflammatory signals accompanied with acute portal hypertension.
Introduction
Portal hypertension (p-HTN) is a major complication of chronic liver disease that can lead to severe outcomes.1–3 The most common cause of p-HTN in Western countries is liver cirrhosis,1,2 in which hepatic compliance is reduced, constituting increased resistance to the portal blood flow and resulting in increased portal pressure. This pressure increase promotes splenomegaly, the engorgement of the vascular collaterals and the development of varices. Traditionally, the pathogenesis and treatment options for p-HTN are considered to be hemodynamic terms and include medical, angiographic, or surgical interventions, either to reduce splanchnic blood flow or to create porto-systemic shunts.4,5 The portal vein drains the spleen and the intestines into the liver. Both of these organs are rich in lymphatic tissues containing various inflammatory cells. Mast cells, for example, have been implicated in the development of portal hypertensive enteropathy, which in turn is induced by pre-hepatic portal hypertension.6,7 The ability of mast cells to support the synthesis and release of angiogenic mediator molecules 8 and cytokines, such transforming growth factor (TGF)-β1 and tumor necrosis factor (TNF)-α,9,10 may explain their role in the splanchnic remodeling associated with experimental p-HTN. In response to the various stress conditions that could occur in p-HTN, these cells have the potential to generate an inflammatory response that could affect other organs. This inflammatory response may be most pronounced within the portal vein, and if the hepatic uptake of this signal is sufficient, it may go undetected if only the systemic blood is sampled. Partial portal vein ligation (PPVL) in various animals, but particularly in the rat, has been widely used for portal hypertension studies. 11 Early and chronic PPVL rats show hemodynamic and metabolic impairments with an inflammatory nature,11,12 including the infiltration of the mast cells and bacterial translocation.13,14 Bacterial translocation secondary to portal hypertension could lead to the development of a “leaky gut”, as reflected by the bacterial overgrowth and disruption of gut homeostasis. 15 Various T cell lineages are involved in these inflammatory processes. CD4+CD25+FoxP3+ regulatory T cells (Tregs) are key elements in immunological self-tolerance, which is crucial for preventing autoimmune diseases and maintaining immune homeostasis.16,17 For example, Tregs play a key role in limiting liver injury in Con A-induced hepatitis via a TGF-β-dependent mechanism. 18 Similar results were found also in patients with autoimmune hepatitis.19,20 Also in other liver diseases such hepatits C virus (HCV) and hepatitis B virus (HBV) and the role of Tregs is well documented.21,22 Th17 cells were established as an independent subset of T helper cells. 23 These cells, which can produce IL-17, IL-17F, and IL-22, thereby inducing massive tissue inflammation, drive different effector functions in different tissues. 23 Studies have shown increased numbers of Th17 cells in inflammatory bowel disease (IBD) patients. 24 The balance between Tregs and Th17, both of which are enriched in mucosal surfaces, has been shown to be a fundamental element in mucosal immunity.25,26 Although inflammation is increasingly recognized in the understanding of p-HTN evolution, the involvement of various T cell subsets in the splanchnic and systemic impairments produced throughout p-HTN was not investigated. In the current study, we investigated the involvement of Tregs and Th17 in p-HTN.
Aim
To demonstrate the local and systemic inflammatory changes within the portal vein (PV) and inferior vena cava (IVC) associated with p-HTN. Additionally, this study aimed to characterize the differences in the distributions of Tregs and Th17 cells between the systemic and portal blood.
Materials and methods
Animals
Male Sprague-Dawley rats (Harlan Jerusalem, Israel), aged 10–11 weeks, weighing 275–325 gr, were used for all experiments. The rats were housed in regular cages and maintained on standard rat chow (Teklad, USA) and tap water
Surgical procedures
Two separate experiments were performed. In experiment 1, rats were randomly allocated to receive either PPVL or a sham operation. After overnight fasting, all of the rats were anesthetized with ketamine and xylazine. Surgery within the PPVL group (n = 9) was performed as follows. The portal vein was freed from surrounding tissue after a midline abdominal incision. A ligature (3/0 silk) was placed around the portal vein. A 20-gauge blunt needle (0.889 mm diameter) was placed alongside the vein, and the ligature was tied securely to the needle and vein. The needle was then removed from the ligature, creating a calibrated constriction of the portal vein. At this point, the abdominal wall incision was closed with 3-0 silk. A control group (n = 6) for the PPVL rats underwent a sham operation, which included a midline excision, exposure of the PV and subsequent closure of the abdominal wall without performing PPVL. After surgery, all of the animals were housed in individual cages and were allowed free access to standard diet and water. All of the rats were sacrificed after 1 week. Sacrifice was performed via terminal bleeding from the portal vein and the supra-hepatic IVC. Blood sampling from the IVC vessels was achieved immediately following the PV. Next, the spleens were removed and weighed.
In experiment 2, naïve rats were sacrificed in a similar way as in experiment 1, without any prior surgical intervention. The PV group was punctured first, and then the IVC group went, or vice versa, in order to compare the blood taken from these two vessels.
Blood tests
After the blood was collected, plasma bilirubin, ALT, AST, total and HDL-cholesterol, hemoglobin, and triglycerides were measured using a Reflotron Plus clinical chemistry analyzer (Roche Diagnostics, GmbH, Mannheim, Germany). Blood glucose was measured by a glucometer (Accu-Chek Performa, Roche, Mannheim, Germany).
Flow cytometry analysis
To determine the regulatory cell phenotype in PV and IVC, flow cytometry was performed. Briefly, 50 μL of whole blood were incubated with appropriate amounts of fluorochrome-labeled Abs in the dark at 4°C for 30 min, washed once, and analyzed. We used for surface staining the following anti-rat antibodies: PE-CD8b, FITC-CD4, APC-CD25 and PE-CD3. A fluorescence threshold based on the CD45-FITC fluorescence was used to select the CD45+ population for analysis. All antibodies were purchased from eBioscience (San Diego, CA, USA). FOXp3 and IL-17A were determined by intracellular staining as follows. After surface staining, the cells were permeabilized using the FoxP3 buffer solution (eBioscince) and then stained with APC-IL-17A and PerCP-Cy5.5- Foxp3 at 4°C for 30 min, washed once, and analyzed, all according to the manufacturer’s instructions. The total cell numbers were calculated by gating on reference beads, and at least 100 positive cells were counted per staining. Flow cytometry was performed on a BDcanto2 apparatus, and BD FACSDiva v6.3 software was used for analysis.
Histology and pathological examination
Both the liver and spleen biopsies were dissected upon sacrifice. Sections of 5 µm were cut from the formalin-fixed paraffin-embedded tissue blocks of liver and spleen. Slides stained with hematoxylin-eosin (H&E) were evaluated by a pathologist in a blinded fashion.
Statistical analysis
The data are expressed as the means
Results
Model establishment
Partial ligation of the portal vein resulted in splenomegaly and congestion of the spleen, as demonstrated by the significant spleen-to-body weight ratio of PPVL rats compared to sham-operated rats (Figure 1a). Additionally, vascular collaterals at the gastroesophageal junction were clearly visible in the PPVL rats (Figure 1b). We then further evaluated the histopathology of the spleens and livers taken from the treated and sham-operated animals through a blinded pathologist. The features of congestion and dilatation splenic sinusoids, together with hemosiderin deposits, were observed in spleens taken from PPVL rats. In contrast, the normal appearance of the red and white pulp and the Mantle zone could be seen in the spleens taken from sham-operated rats (Figure 1c). The partial portal vein ligation model in the rat is known for the lowest degree of hepatic impairment, as portal hypertension is pre-hepatic. 11 Therefore, the liver biopsy in our experiment showed a normal histology in PPVL rats, as expected. Figure 1c shows representative images of the spleens and livers taken from sham and PPVL rats after being sacrificed.

Partial portal vein ligation model-establishment. (a) One week after the portal vein was partially ligated portal hypertensive (n = 9) and sham (n = 6) operated rats were sacrificed. After their sacrifice, the spleens were dissected and weighed, and then the body-to-spleen weight ratios were calculated for all rats. The results are expressed as means
Two-vessels’ blood puncturing
Next, we demonstrated in naïve rats the immunological differences between the portal and systemic circulation using blood samples that were drawn from the PV and the IVC. Synchronous sampling could not be technically performed, as PV exposure could not be achieved while keeping the supra hepatic IVC accessible to puncture. Therefore, the PV was punctured first, immediately followed by the IVC puncture. Hemolysis during blood sampling was ruled out because the AST, LDH, and bilirubin levels did not differ between the IVC and PV in either sham or PPVL rats. The triglyceride levels were also similar between the groups, ruling out thoracic duct aspiration during IVC puncture (Supplementary Table S1). To rule out a possible bias of blood vessel sampling associated with the stress of sequentially puncturing two blood vessels, another group of rats (n = 6) was sacrificed, in which the IVC was sampled prior to the PV. In this group, neither biochemical nor immunological differences between the IVC and PV were detected.
Regulatory T cell characterization in p-HTN
We further examined the systemic inflammatory aspects associated with p-HTN that were reflected by the distribution of regulatory T cells in the IVC and in the PV. Figure 2a shows that CD4+CD25+FOXP3+ cells were significantly increased within the IVC of PPVL rats compared to sham operated rats (7.9% vs. 5.4%, respectively;
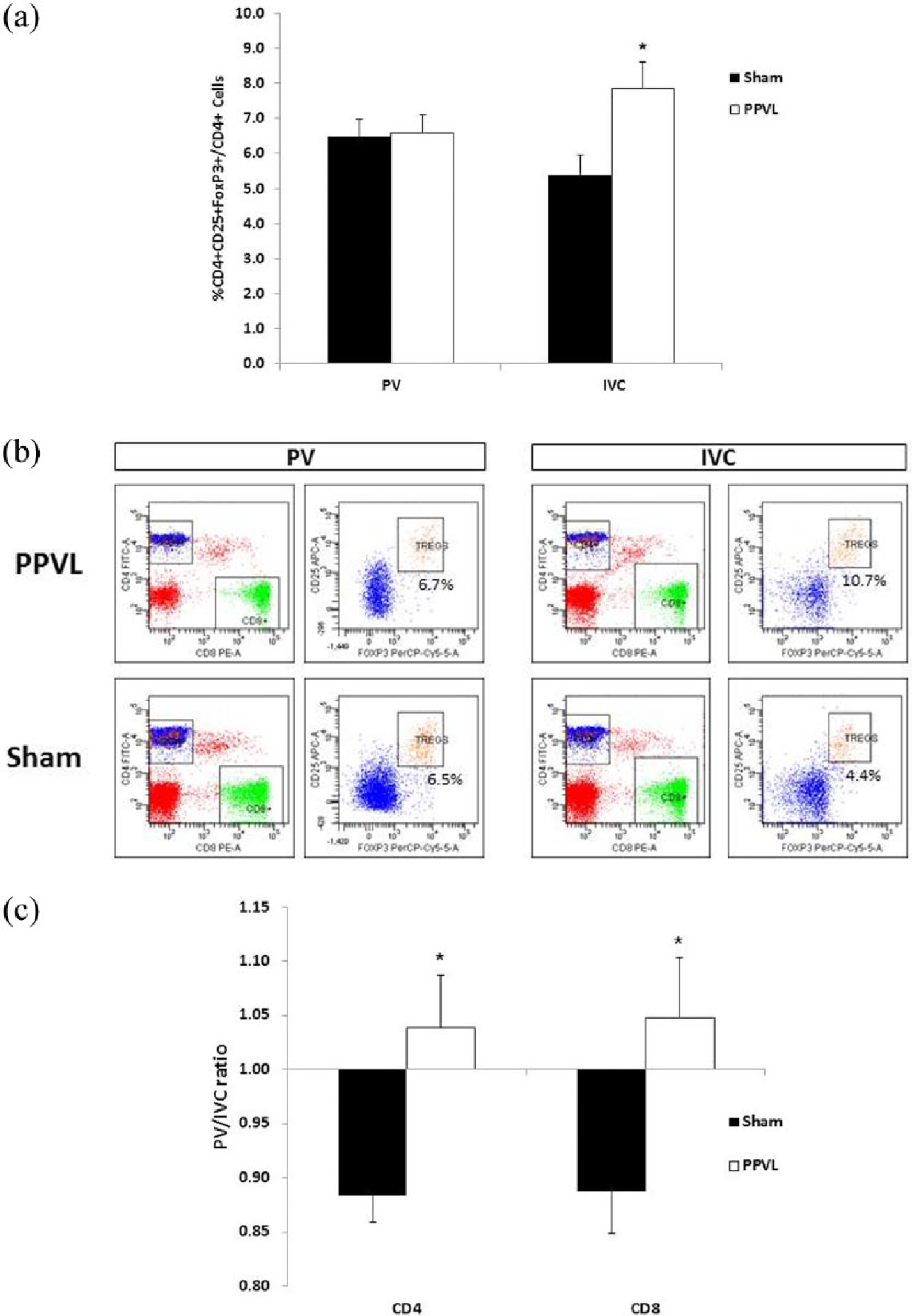
Flow cytometry characteristics of the portal vein and inferior vena cava in sham and partial portal vein ligated rats. (a) Circulating blood CD4+CD25+Foxp3+ cells were analyzed by flow cytometry in the portal vein and inferior vena cava of partial portal vein ligated (n = 9) and sham rats (n = 6). The results are expressed as means
Regulatory T cell characterization in naïve rats
Next, we explored whether certain immunological properties of the IVC could differ from those of the PV without any relationship with p-HTN. Figure 3a shows that in the blood samples taken from the IVC and the PV of naïve rats, CD4+IL17A+ cells (Th17) were significantly increased within the IVC compared with the PV (1.98% vs.0.67%, respectively;

Flow cytometry characteristics of the portal vein and inferior vena cava in naïve rats (n = 6). (a) Circulating blood CD4+IL17A+ cells were analyzed by flow cytometry in the portal vein and inferior vena cava. The results are expressed as means
Discussion
Our work serves as proof of concept for the involvement of Tregs in p-HTN. We showed the immunological changes associated with p-HTN that occur at the local (portal system) and systemic circulation (IVC). Using the PPVL model, these changes can be attributed to p-HTN directly, even in the absence of hepatic injury.
P-HTN is considered to be the most severe complication among patients with chronic liver disease. 3 This clinical complication, as defined by a pathological elevation of blood pressure in the portal system, is usually described in hemodynamic terms. The role of inflammation in the initiation and progression of vascular diseases is increasingly recognized; 27 therefore, inflammation could be the basic mechanism that drives the essential nature of p-HTN.28,29 Growing evidence shows that inflammation contributes to the initiation and maintenance of p-HTN. 12 Pre-hepatic portal hypertension induces the infiltration of inflammatory cells, particularly that of mast cells.6,7 In PPVL rats, the enhanced aortic mRNA expression of oxidative and inflammatory mediators was found to be associated with features of fibrosis, as reflected by the increased aortic expression of collagen I and connective tissue growth factor.30,31 Although inflammation was proposed to play a key role in the pathophysiological mechanisms involved in the complications of p-HTN,28,32,33 the role of Tregs, key mediators in controlling inflammation, was not investigated. The involvement of Tregs in p-HTN was recently implied by the study of Guo et al., who explored the p-HTN, splenomegaly, and level of various Tregs in 20 patients with liver cirrhosis. 34 In this study, the CD4+CD25+ regulatory cells were increased in the peripheral blood. However, the FoxP3+ regulatory cells were not defined within the circulation, but only in the splenic marginal of patients with hypersplenism. 34 In contrast, we demonstrated by flow cytometry the presence of CD4+CD25+FOXP3 cells within the IVC and PV of sham and PPVL rats. Furthermore, in PPVL rats, the levels of these cells within the IVC were significantly increased compared to those of the PV. This increase may be attributed to the presence of related cytokines in the IVC or to changes in cytokine balance between the IVC and PV, but more studies are warranted in order to establish this hypothesis. The PPVL is a pre-hepatic model for portal HTN that allowed us to characterize the inflammatory changes associated with portal HTN as a stand-alone instigator without the possible effect of any primary liver disease. Immunological alterations regarding p-HTN have been previously described. Tokushige and colleagues reported an increase of serum TNF-α and altered Th1/Th2 balance. 35 Although lymphocyte density, especially in the spleen, is decreased; the total amount of lymphocytes is increased due to p-HTN. 36 Long-term contact between exogenous molecules and the splenic environment promotes lymphocytes response. 37 However, to the best of our knowledge, our study is the first to show divergence in the T cell distributions between the portal and systemic circulation. In our study, we used the PPVL model to demonstrate that CD4+CD25+FoxP3+ cells were significantly increased within the IVC of PPVL rats, in contrast to similar level of these cells among the PV in sham and in portal hypertensive rats. Altogether, enhancement of CD4+CD25+FOXP3+ T regulatory cells may lead to immune dysfunction in p-HTN. However, further investigations are warranted to elucidate the exact implications of these observations. CD4+ T cells develop into different T helper cell subsets with distinct cytokine profiles and effector functions. Th17 cells were established as an independent subset of T helper cells. Th17 cells are in close relationship to CD4+CD25+FoxP3+ cells, as TGF-β also induces the differentiation of naïve T cells into Foxp3+ Tregs in the peripheral immune compartment. 38 It should be noted that, in our study, Th17 cells within the PV of PPVL rats were not decreased compared with the IVC, as was observed in naïve rats. However as sham-operated rats also did not display these changes in the distribution of Th17 cells, this effect may be associated with p-HTN. We also showed in naïve mice that the increase of Th17 cells within the IVC compared to the PV is unique to this subset, as no changes in any other T cell subsets were detected (data not shown).
In conclusion, our study suggests an active role for Tregs in the inflammation associated with p-HTN. Further understanding of this role in the unique milieu of the portal vein may present opportunities for therapeutic interventions. Nevertheless, more studies especially in chronic models of p-HTN are warranted in order to establish the role of Tregs in p-HTN.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by internal grants.
