Abstract
Background and study aim:
The aim of the present study was to investigate the effects of
Materials and methods:
Group 1 was the control. In group 2 (TAA), rats received TAA daily for 3 days. In group 3 (TAA+LB),
Results:
In our study,
Conclusions:
In conclusion, the effects of
Introduction
Bacterial translocation (BT) is defined as the migration of bacteria or bacterial products from the gut to the extra-intestinal space. 1 The liver plays a central role for prevention of translocation of gut-derived microbial products to the systemic circulation by cleansing and detoxifying microbial products.2,3 It is well known that bacterial translocation is closely associated with the development of complications in many liver diseases. Intestinal bacterial overgrowth (IBO), increased permeability of intestinal mucosa, and impaired immunity may favor BT. 4
The fruit of
Materials and methods
Animals
Healthy male Wistar rats weighing 250–300 g were used in this study. Animals were housed on a 12-h light/dark cycle (lights on from 08:00 h) at a constant ambient temperature (24±1°C) with normal rat chow and water available
Induction of liver injury
Liver injury was induced by intraperitoneal (ip) injection of 350 mg/kg/day TAA (Merck, Germany) for 3 days as previously described. 14 Supportive therapy of subcutaneous administration of 5% dextrose (25 mL/kg) and 0.9% NaCl with potassium (20 mEq/L) every 6 h was given to avoid weight loss, hypoglycemia, and renal failure, as previously described. 14
The preparation of Lycium barbarum extract
The extraction of
Experimental design
A total of 28 male rats were divided into four groups. Group 1 (n = 7) was the control group, which received 0.1 mL/day 0.9% NaCl ip for 3 days. Group 2 (n = 7) was the TAA group, in which rats received 350 mg/kg/day TAA ip for 3 days at intervals of 24 h. Group 3 (n = 7) was the TAA+LB group.
Tissue samples
On day 24 of the study, laparotomy was performed under anesthesia with 50 mg/kg ketamine (Ketalar, Parke-Davis, Eczacibasi, Turkey) and 10 mg/kg xylazine HCl (Alfazyne 2%; Alfasan, Woerden, the Netherlands) under strictly sterile conditions. After the skin was sterilized with iodine and shaved, the abdomen was opened wide. The mesenteric lymph nodes (MLNs) from the ileocecal area and the spleen were aseptically dissected, removed, weighed, and liquefied in sterile saline for bacterial culture. The liver was aseptically dissected and the left lobe was used for bacterial cultures. The other lobes of liver was excised for histopathological and biochemical analyses including measurement of thiobarbituric-acid-reactive substances (TBARS) and reduced glutathione (GSH) levels. The ileum was also excised for histopathological and biochemical analyses including measurement of TBARS and GSH levels. We aspirated 0.1 mL ileal fluid with a sterile needle for assessment of IBO. Blood was taken from the right ventricle for biochemical determinations (ammonia, aspartate transaminase [AST], and alanine transaminase [ALT]).
Liver histology
Midsections of the liver were processed for light microscopy. This processing consisted of fixing the specimen in 5% neutral formol solution, embedding the specimens in paraffin, slicing sections of 5 µm thickness, and staining the sections with hematoxylin and eosin. The tissue slices were scanned and scored by an experienced histologist who were not aware of sample assignment to experimental groups. The degree of inflammation and necrosis was expressed as the mean of 10 different fields within each slide, classified on a scale of 0 to 3 (0, normal; 1, mild; 2, moderate; and 3, severe). 14
Intestine histology
The ileum samples taken from the rats were fixed for 24 h in 5% neutral formol solution. Samples were divided into pieces of 0.5 × 0.5 × 0.5 cm3 and were processed for routine examination. These samples from each animal were obtained in separate blocks. Sections of samples were cut at 6–7 μm, mounted on slides, stained with hematoxylin and eosin (H&E) and examined by a Leica DFC280 light microscope and Leica Q Win Image Analyses System (Leica Microsystems Imaging Solutions, Cambridge, UK). The tissue slices were scanned and scored by two expert pathologists, who were unaware of the sample assignment to an experimental group. A scoring system in the range of 0–3 (0, normal; 1, mild; 2, moderate; 3, severe) was used for intestinal histopathological evaluations. Mucosal integrity, edema, inflammatory cellular infiltration of the lamina propria, and vessel vasodilation were scored accordingly. 18
Biochemical analysis
Blood samples of approximately 4 mL were taken from each rat. A 1-mL aliquot from each blood sample was transferred to centrifuge tubes that contained 1 mL 10% TCA for deproteinization, and centrifuged at 3000
Blood ammonia analysis
Blood levels of ammonia were analyzed using the colorimetric Berthelot (indophenol) reaction. 19 In this reaction, free ammonia in previously deproteinized samples generates a blue-colored indophenol molecule by reacting with sodium nitroprusside in an alkaline environment that contains phenol and hypochlorite ions. The color intensity of the chromogen, spectrophotometrically measured against blank samples at 625 nm, increases in direct proportion to the concentration of ammonia. (NH4)2SO4 was used as a standard. Results are expressed in µg ammonia nitrogen/dl.
Serum AST and ALT levels
Serum levels of AST and ALT were measured using a kinetic UV method defined by the International Federation of Clinical Chemistry (IFCC) using pyridoxal phosphate and NADH as co-factors. An Olympus auto-analyzer and commercial kits of the same brand were used for two analyses in the routine biochemistry laboratories of Turgut Ozal Medical Center. AST and ALT levels were expressed as U/L.
Determination of lipid peroxidation
TBARS and other products of lipid peroxidation considered to be indicators of oxidative stress were analyzed using the method described by Buege and Aust.
20
Briefly, 250 μL of tissue homogenate, 500 μL of thiobarbituric acid (TBA) reactant (3.7 g/L TBA dissolved in 0.25 mol/L HCl), and 1.5 mL of 15% trichloroacetic acid were added to screw-topped Pyrex centrifuge tubes (~10 mL) and mixed. The tubes were placed in a hot water bath at 95°C for 30 min and then immediately cooled under tap water.
GSH assay
GSH was measured in the tissue homogenates using the method described by Ellman.
21
A total of 250 mL of 10% trichloroacetic acid was added to 250mL of tissue homogenate containing 6mM disodium EDTA. The mixture was vortexed vigorously for 2 min, followed by centrifugation at 3000
BT and IBO
BT was defined as positive bacterial cultures from MLN, liver, or spleen samples. From each animal group, 1 g tissue samples (liver, spleen, and MLN) were homogenized in 1 mL saline. This homogenized solution was then inoculated onto blood agar, eosin methylene blue (EMB) agar, and Sabouraud agar, and incubated at 37°C for 24 h. On the following day, the bacterial growth was checked and the colonies were counted. Gram staining was carried out for each colony. According to the Gram stain results, specific biochemical tests, including those for catalase, oxidase, coagulase, indole, citrate reduction, urease, and sugar fermentation on triple sugar iron agar, were carried out for primary identification of the isolated bacteria. An Api 20 E test (bioMereux, Marcy L’Etoile, France) was used to confirm the identification. To grow yeasts, the Remel RapID™ Yeast Plus System 8311007 (Remel Inc., Lenex, KS, USA) was used.
IBO of a specific organism was defined as a bacterial count in the ileal aspirate higher than the mean + 2 SD for the same organism in normal rats. The ileal aspirate (0.1 mL) was inoculated onto blood agar, EMB agar, and Sabouraud agar and incubated at 37°C for 24 h. The bacterial growth was checked on the following day and the colonies counted. Colony counts for ileal aspirates are presented as × 103 CFU/mL ileal aspirate (median and range). 22
Statistical analyses
Results are expressed as mean ± SD. Comparisons of quantitative variables among groups were made with the one-way ANOVA or the corresponding non-parametric test (Kruskal–Wallis), as required. Post-hoc comparisons were performed with the least significant difference test. Proportion comparisons were carried out using the χ2 or Fisher test. For all comparisons, a statistically significant difference was defined as
Results
Survival rates
The survival rates in the TAA (85.7%) and the TAA+LB groups (71.4%) were lower than in the control and LB group on the day of sacrifice. There was no statistically significant difference between groups.
Serum ammonia, AST, and ALT levels
Table 1 shows the mean + SD values for serum ammonia, AST, and ALT levels. Serum ammonia, AST, and ALT levels in the TAA group were significantly higher than in the control and LB groups (all
Mean + SD values of serum ammonia, AST, and ALT levels of the groups.

Ammonia was expressed in µg ammonia nitrogen/dL.
Oxidative stress results
Table 2 shows the mean + SD values of liver and intestine TBARS and GSH levels in the groups. Liver TBARS levels in the TAA group were significantly higher than in the control group (
Mean + SD values of liver and intestine TBARS (nmol/g wet tissue) and GSH (mmol/g wet tissue) levels in the groups.

Intestine TBARS levels in the TAA group were higher than in the control group but difference was not significant. Intestine TBARS levels in the TAA+LB group were significantly lower than in the TAA group (
Results of liver histology
Table 3 shows mean ±/SD values for hepatic necrosis and inflammation scores, which were significantly higher in the TAA group than in the control group (both
Mean ±/SD values for hepatic necrosis and inflammation scores.

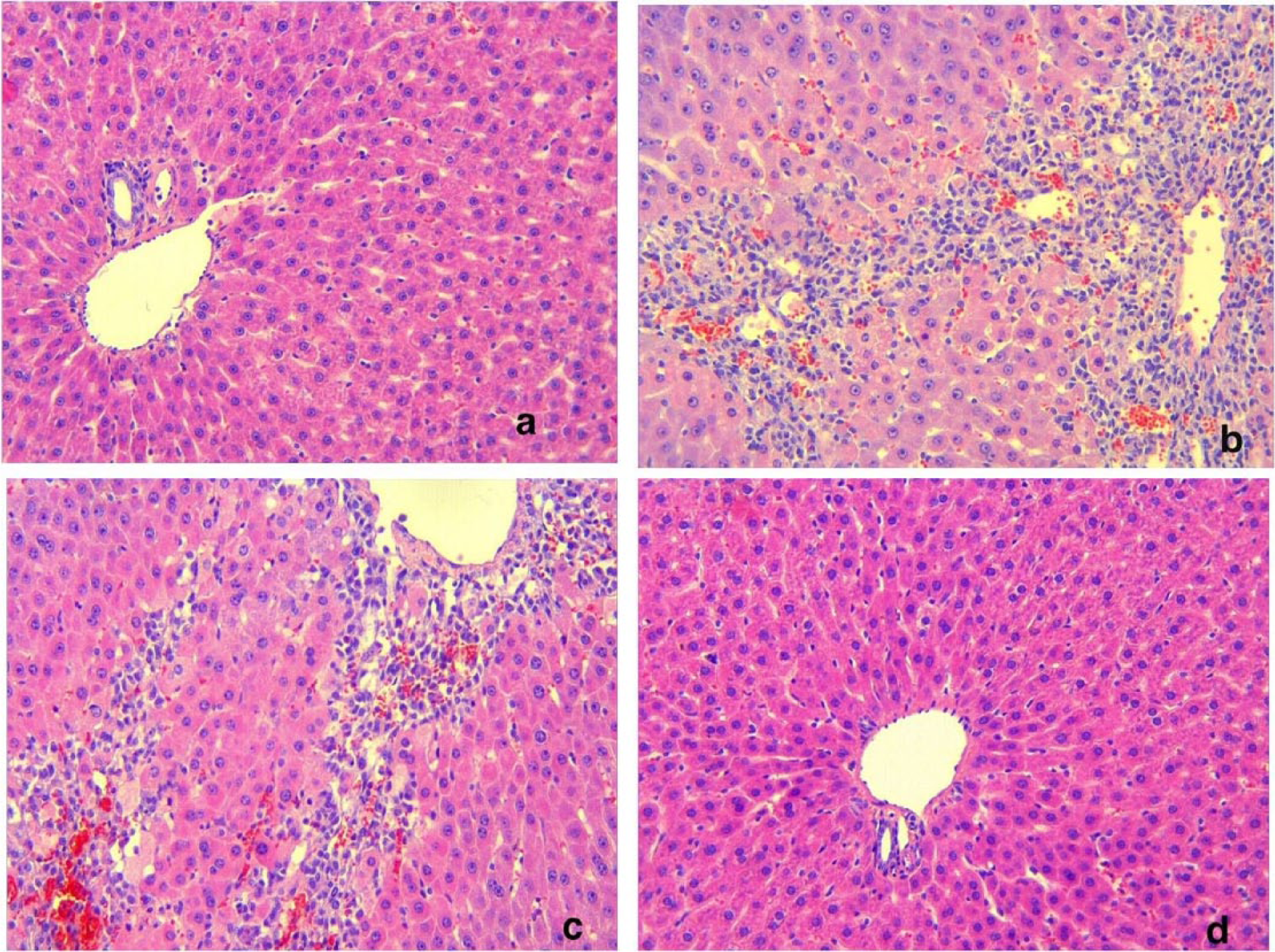
Liver histology in the control animals (a), TAA group (b), TAA+ LB group (c), and the LB group (d) (Hematoxylin and eosin, ×20). Bridging necrosis and inflammatory cell infiltration in the liver parenchyma area was noted in (b) and (c).
Results of intestine histology
Intestinal damage scores in the TAA group were significantly higher than in the control group (

Intestinal histology of the control animals (a), the TAA group (b), the TAA+LB group (c), the LB group (d) (Hematoxylin and eosin, ×20). Villous blunting, degeneration of surface epithelium and inflammatory cell infiltration was noted in (b). (c) Minimally inflammatory cell infiltration in the lamina propria. Also, height of the villi was normal, surface epithelium was shapely and uninterrupted. Marked lacteal dilation in the apical region of villi was noted.
Comparison of intestinal mucosal integrity, edema, inflammatory cellular infiltration of the lamina propria, and vasodilation scores between the groups.

Values are given as mean ± SD.
INF, inflammatory cellular infiltration of the lamina propria; MI, mucosal integrity; V, vasodilation.
IBO and BT
Counts of

Discussion
In this study, TAA led to severe liver damage (Figure 1b). We found that liver enzymes (AST and ALT) and ammonia levels significantly increased in the TAA group. Additionally, ileal aspirate
The
In this study,
It has been shown that intestinal bacterial overgrowth plays an important role in BT in the TAA-induced hepatic failure model.
35
In our study,
Bacterial translocation (BT) includes the migration of viable microorganisms but also all microbial products across an even, anatomically intact intestinal barrier from the intestinal lumen to mesenteric lymph nodes (MLN) and other extraintestinal organs and sites. IBO, increased intestinal permeability, and impaired immunity are main factors in development of BT.
37
We previously have reported that intestinal oxidative damage plays a major role in the development of BT by disrupting the barrier function of intestinal mucosa in TAA-induced liver injury.
23
Intestinal TBARS levels in the TAA group were higher than in the control group, but difference was not significant. Intestinal GSH levels in the same group were significantly lower than in the control group (
In conclusion,
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
