Abstract
Inflammatory bowel diseases are idiopathic relapsing disorders characterized by chronic inflammation of the intestinal tract. The aim of the present study was to examine the effect of ultramicronized palmitoylethanolamide (PEAultra), underlining its correlation with PPARα and TLR4; in particular, we aimed at evaluating its anti-inflammatory effect in mice subjected to experimental colitis. Colitis was induced in mice by intracolonic instillation of dinitrobenzene sulfonic acid (DNBS), PEAultra was administered daily intraperitoneally (10 mg/kg) for 4 days. On day 4, animals were sacrificed and tissues were taken for histological and biochemical analysis. Four days after DNBS administration, TNF-α and IL-1β productions were increased in association with colon damage. Neutrophil infiltration, evaluated by MPO activity, in the mucosa was associated with upregulation of ICAM-1 and P-selectin. Immunohistochemistry for nitrotyrosine and PARP showed an intense staining in the inflamed colon. Treatment with PEAultra significantly reduced the appearance of colon damage and the loss of body weight. These effects were associated with a remarkable amelioration in the disruption of the colonic architecture and reduction in colonic MPO activity. PEAultra also reduced the pro-inflammatory cytokine release, the appearance of nitrotyrosine and PARP immunoreactivity as well as the upregulation of ICAM-1 and P-selectin; moreover, pro-MMP-9 and MMP-2 expressions were significantly inhibited in the colon of DNBS-treated mice. Furthermore, we studied PEAultra correlation with PPARα and TLR4, demonstrating that PEAultra inhibited TLR4 pathway through a PPARα independent pathway. Taken together, our results clearly show that this new formulation of PEA may be considered as a possible therapeutic approach against Th1-induced colitis.
Inflammatory bowel disease (IBD) is a chronic inflammatory disorder of the intestine.1,2 In humans, as well as in several different animal models of intestinal inflammation, the contractile force generated by the smooth muscle is significantly reduced, resulting in altered motility leading to diarrhea or constipation. 3 In IBD, intestinal dysfunctions as well as critical morphological changes have been reported. 4 IBD includes two subtypes, ulcerative colitis (UC) and Crohn’s disease (CD).5,6 Studies conducted on experimental models of colitis also suggest that IBD-associated tissue damage is due to an exces-sive immune response directed against normal constituents of the bacterial microflora, 7 in which dysregulation of mucosal CD4+ T-helper 1 (Th1) effector cell responses to the normal enteric bacterial flora plays a crucial role. Several animal models of IBDs have been developed, 8 among which the hapten model of colonic inflammation induced by dinitrobenzene sulfonic acid (DNBS) intrarectally delivered to normal mice displays human Crohn’s disease-like features, notably predominant nuclear factor κB (NF-κB)-dependent Th1 activation.7,9,10
Conventional therapy uses anti-inflammatory and immunosuppressive corticosteroids to treat acute-phase symptoms; however, there is a high medical need for new therapeutic strategies. Recent articles have shown the many beneficial actions of palmitoylethanolamide (PEA), an endogenous amide belonging to the family of fatty acid ethanolamides (FAEs).11 –13 Over time, it has been shown that PEA inhibits peripheral inflammation and mast cell degranulation, 14 these actions are accompanied by a decrease in nitric oxide (NO) production, 15 neutrophil influx, 16 and expression of inducible nitric-oxide synthase (iNOS). Anti-inflammatory effects of PEA have been associated with peroxisome proliferator-activated receptor-(PPAR-)α activation, 17 a nuclear receptor fundamental in the control of inflammatory responses, and expressed in various cells of the immune system. 18 Also, it has been recently demonstrated that PPAR-α is additionally expressed in the digestive tract, mainly localized in the intestinal mucosa in the small intestine and colon. 19 Recent data report that PEA selectively, and mainly via a PPARα-dependent mechanism, reduces the expression of S100B protein, a pivotal signaling molecule participating in the onset and progression of the inflammatory status of gut degenerative processes. 20 Based on these facts, the aim of our study was to investigate the effects of PEAultra in IL-10KO mice, spontaneously developing a Th1-dependent chronic enterocolitis, and compare in a mouse model of DNBS-induced colitis the effects of PEAultra with other formulations (PEA and PEA micronized). In order to identify the PEAultra mechanism of action and to prove the involvement of different pathways like PPAR-α and Toll-like Receptors 4 (TLR4), the same investigations were performed in PPARαKO as well as TLR4KO mice.
Materials and methods
Animals
Male CD1 mice (25–30 g; Harlan Nossan, Milan, Italy), IL-10KO mice (4–5 weeks old, 20–22 g), and littermates wild-type (WT) controls were purchased from Jackson Laboratories (Charles River, Italy); male C57BL/10ScNJ mice (20–22 g; Charles River, Milan, Italy) with a targeted disruption of the Toll like receptor 4 gene (KO) and littermate WT controls and mice (4–5 weeks old, 20–22 g) with a targeted disruption of the PPAR-α gene (PPAR-α WT) were purchased from Jackson Laboratories (Harlan Nossan, Italy) and were used for all studies. Mice were housed in individual cages (five per cage) and maintained under 12-h light/dark cycle at 21 ± 1°C and 50 ± 5% humidity. Standard laboratory diet and tap water were available ad libitum. Animal care was in compliance with Italian regulations on protection of animals used for experimental and other scientific purposes (DM 116192) as well as with the EEC regulations (OJ: of ECL 358/1 12/18/1986).
Induction of experimental colitis
Colitis was induced with a very low dose of DNBS (4 mg per mouse) by using a modification of the method first described in rats. 21 In preliminary experiments, this dose of DNBS was found to induce reproducible colitis without mortality. Mice were anesthetized by Enflurane. DNBS (4 mg in 100 μL of 50% ethanol) was injected into the rectum through a catheter inserted 4.5 cm proximally to the anus. Vehicle alone (100 μL of 50% ethanol) was administered in control experiments (sham). Thereafter, the animals were kept for 15 min in a Trendelenburg position to avoid reflux. After colitis and sham-colitis induction, the animals were observed for 4 days. On the 4th day, the animals were weighed and anaesthetized with chloral hydrate, and the abdomen was opened by a midline incision. The colon was removed, freed from surrounding tissues, opened along the anti-mesenteric border, rinsed, weighed, and processed for histology and biochemical studies.
Ultramicronization process of PEA
The ultramicronization process was performed using jet mill equipment (Sturtevant Inc., Hanover, MA, USA) with a chamber of 300 mm in diameter, operated with ‘spiral technology’ and driven by compressed air at 10–12 bars. Crashing was determined by the high number of collisions that occurred among particles as a result of the high level of kinetic (not mechanical) energy. This process is effective not only in reducing product particle size, but also in modifying crystalline structure. Scanning electron microscopy showed an intimate intermixing of PEA, while analysis by differential scanning calorimetry and X-ray diffraction indicated transformation into a crystalline form different from the original two, definable as a higher energy content form. The composite showed the following particle size distribution: 100% <10 μm; 99.9% <6 μm; and 59.6% <2 μm.
Experimental groups
The animals were randomly divided into groups (n = 10 for each group):
Sham + vehicle group: vehicle solution (saline) was administered daily intraperitoneally and animals were sacrificed on day 4 after intrarectal administration of vehicle.
DNBS + vehicle: vehicle was administered daily intraperitoneally and animals were sacrificed on day 4 after administration of DNBS.
DNBS + PEA: mice were subjected to DNBS administration described as above, and PEA (10 mg/kg, i.p., 10% ethanol) was administered every 24 h, starting from 1 h after the administration of DNBS and animals were sacrificed on day 4 after intrarectal administration of DNBS.
DNBS + PEAmicro: mice were subjected to DNBS administration described as above, and micronized PEA (10 mg/kg, i.p., 10% ethanol) was administered every 24 h, starting from 1 h after the administration of DNBS and animals were sacrificed on day 4 after intrarectal administration of DNBS.
DNBS + PEAultra: mice were subjected to DNBS administration described as above, and PEAultra (10 mg/ kg, i.p., 10% ethanol) was administered every 24 h, starting from 1 h after the administration of DNBS and animals were sacrificed on day 4 after intrarectal administration of DNBS.
PPAR-α KO Sham + vehicle group: vehicle solution (saline) was administered daily intraperitoneally and animals were sacrificed on day 4 after intrarectal administration of vehicle.
PPAR-α KO DNBS + vehicle: vehicle was administered daily intraperitoneally and animals were sacrificed on day 4 after administration of DNBS.
PPAR-α KO DNBS + PEAultra: mice were subjected to DNBS administration described as above, and PEAultra (10 mg/kg, i.p., 10% ethanol) was administered every 24 h, starting from 1 h after the administration of DNBS and animals were sacrificed on day 4 after intrarectal administration of DNBS.
TLR4 KO Sham + vehicle group: vehicle solution (saline) was administered daily intraperitoneally and animals were sacrificed on day 4 after intrarectal administration of vehicle.
TLR4 KO DNBS + vehicle: vehicle was administered daily intraperitoneally and animals were sacrificed on day 4 after administration of DNBS.
TLR4 KO DNBS + PEAultra: mice were subjected to DNBS administration described as above, and PEAultra (10 mg/kg, i.p., 10% ethanol) was administered every 24 h, starting from 1 h after the administration of DNBS and animals were sacrificed on day 4 after intrarectal administration of DNBS.
To address whether PEAultra treatment was beneficial in treating established colitis (therapeutic protocol), 9-week-old IL-10KO mice, with fully developed chronic colitis, were treated intraperitoneally with PEAultra three times per week for 7 consecutive weeks, until week 16. The doses of PEA, PEAmicro, and PEAultra (10 mg/kg) used here were based on previous dose-response and time-course studies by our laboratory.11,22
Evaluation of colon damage
After its removal, the entire colon was gently rinsed with saline solution, opened by a longitudinal incision, and immediately examined under a microscope. Colon damage (macroscopic damage score) was evaluated and scored by two independent observers as described previously23 –25 according to the following criteria: 0, no damage; 1, localized hyperemia without ulcers; 2, linear ulcers with no significant inflammation; 3, linear ulcers with inflammation at one site; 4, two or more major sites of inflammation and ulceration extending >1 cm along the length of the colon; and 5–8, one point is added for each centimeter of ulceration beyond an initial 2 cm.
Myeloperoxidase assay
Myeloperoxidase (MPO) activity in the colon was used as an indicator of polymorphonuclear (PMN) cell infiltration using a method previously described. 26
Colon tissue were homogenized in a solution containing 0.5% hexa-decyl-trimethyl-ammonium bromide and 10 mM 3-(N-morpholino)-propane-sulfonic acid dissolved in 80 mM sodium phosphate buffer, pH 7, and centrifuged for 30 min at 20,000g at 4°C. An aliquot of the supernatant was then allowed to react with a solution of tetra-methyl-benzidine (16 mM) and 1 mM hydrogen peroxide. The rate of change in absorbance was measured by a spectrophotometer at 650 nm. MPO activity was defined as the quantity of enzyme degrading 1 μmol peroxide/min at 37°C and was expressed in units per gram weight of wet tissue.
Histological examination
For histological examination, tissues were removed, fixed in 10% buffered formalin phosphate, embedded in paraffin sectioned and stained with hematoxylin and eosin. The degree of inflammation on microscopic cross-sections of the colon was graded semi-quantitatively from 0 to 4 (0, no signs of inflammation; 1, very low level; 2, low level of leucocyte infiltration; 3, high level of leucocyte infiltration, high vascular density, thickening of the colon wall; 4, transmural infiltration, loss of goblet cells, high vascular density, and thickening of the colon wall). Grading was made in a blinded fashion, by two experienced pathologists using an Olympus BX60 microscope (Olympus, Tokyo, Japan).
Measurement of cytokines
The levels of tumor necrosis factor-α (TNF-a), interleukin-1β (IL-1b), and monocyte chemotactic protein-1α (MCP-1α) were evaluated in colon samples at 4 days after DNBS treatment. The assay was carried out by using a commercial colorimetric kit (Diaclone Research Cell Science Cedex, France), according to the manufacturer’s instructions. All TNF-a, IL-b, and MCP-1α determinations were performed in duplicate serial dilutions. The ELISA detection limit is <25 pg/mL.
Immunohistochemical localization of ICAM-1, P-selectin, nitrotyrosine, and PAR
At 4 days after DNBS treatment, colon tissues were fixed in 10% (w/v) PBS buffered formaldehyde, and 7-μm sections were prepared from paraffin embedded tissues. After deparaffinization, endogenous peroxidase was quenched with 0.3% H2O2 in 60% methanol for 30 min. The sections were permeabilized with 0.1% Triton X-100 in PBS for 20 min. Non-specific adsorption was minimized by incubating the section in 2% normal goat serum in PBS for 20 min. Endogenous biotin or avidin-binding sites were blocked by sequential incubation for 15 min with avidin and biotin. The sections were then incubated overnight with primary anti-P-selectin antibody (BD PharMingen, San Diego, CA, USA; CD62P 1:500), anti-ICAM-1 antibody (BD PharMingen; CD54, 1:500), anti-nitrotyrosine rabbit polyclonal antibody (Upstate Biotechnology, Lake Placid, NY, USA; 1:500 in PBS, v/v), anti-PAR antibody (1:500 in PBS, v/v), or control solutions. Controls included buffer alone or non-specific purified rabbit IgG. Immunohistochemistry photographs were assessed by densitometric analysis by using Optilab Graftek software on a Macintosh personal computer.
Western blot analysis for IκBα, NF-κB p65, MMP-2, MMP-9, COX-2, and iNOS
In brief, tissue samples from the terminal colon were suspended in extraction buffer A containing 0.2 mM PMSF, 0.15 μM pepstatin A, 20 μM leupeptin, and 1 μM sodium orthovanadate; homogenized at the highest setting for 2 min; and centrifuged at 1000 g for 10 min at 4°C. Supernatants represented the cytosolic fraction. The pellets, containing enriched nuclei, were resuspended in buffer B containing 1% Triton X-100, 150 mM NaCl, 10 mM Tris-HCl, pH 7.4, 1 mM EGTA, 1 mM EDTA, 0.2 mM PMSF, 20 M leupeptin, and 0.2 mM sodium orthovanadate. After centrifugation at 15,000 g for 30 min at 4°C, the supernatants containing the nuclear protein were stored at −80°C for further analysis. The levels of IκBα, Matrix Metalloproteinases 9 (MMP9), Matrix Metalloproteinases 2 (MMP-2), Cyclooxygenase-2 (COX-2), and iNOS were quantified in a cytosolic fraction from colon tissue collected after DNBS injection, whereas NF-κB p65 levels were quantified in a nuclear fraction. The filters were blocked with 1X PBS, 5% (w/v) non-fat dried milk for 40 min at room temperature, and they were subsequently probed with specific antibodies, anti-IκBα (1:1000, Santa Cruz Biotechnology), anti-NF-κB p65 (1:1000, Santa Cruz Biotechnology), anti-iNOS (1:500, Santa Cruz Biotechnology), anti-MMP-2 antibody (1:500; Chemicon International), and with anti-MMP-9 antibody (1:500, Calbiochem), anti-COX-2 (1:500, Santa Cruz Biotechnology) in 1X PBS, 5% (w/v) non-fat dried milk, and 0.1% Tween 20 at 4°C overnight. Membranes were incubated with peroxidase-conjugated bovine anti-mouse IgG secondary antibody or peroxidase-conjugated goat anti-rabbit IgG (1:2000, Jackson ImmunoResearch Laboratories) for 1 h at room temperature. To ascertain that blots were loaded with equal amounts of protein lysates, they were also incubated in the presence of the antibody against β-actin protein (1:2000, Sigma-Aldrich). Signals were detected with enhanced chemiluminescence detection system reagent according to the manufacturer’s instructions (SuperSignal West Pico Chemiluminescent Substrate, Pierce Thermo Scientific). The relative expression of the protein bands was quantified by densitometric analysis with Gel Logic 200 PRO software and standardized to β-actin or lamin A/C levels. Images of blot signals (8 bit/600 dpi resolution) were imported to analysis software (Image Quant TL, v2003). A preparation of commercially available molecular weight in the range of 10–250 kDa was used to define molecular weight positions and as reference concentrations for each molecular weight.
Statistical evaluation
All values in the figures and text are expressed as mean ± standard error (SEM) of the mean of n observations. For the in vivo studies, n represents the number of animals studied. In the experiments involving histology or immunohistochemistry, the figures shown are representative of at least three experiments (histological or immunohistochemistry coloration) performed on different experimental days on the tissue sections collected from all the animals in each group. The results were analyzed by one-way ANOVA followed by a Bonferroni post-hoc test for multiple comparisons. A P value less than 0.05 was considered significant, and individual group means were then compared with Student’s unpaired t-test. A P value of less than 0.05 was considered significant.
Results
Effect of different PEA formulation in experimental colitis
To study the possible effect of different formulations of PEA in Th-1 inflammatory bowel disease, we induced colitis to CD1 mice, by DNBS intrarectum administration.
The macroscopic inspection of cecum, colon, and rectum showed presence of mucosal congestion, erosion, and hemorrhagic ulcerations (Figure 1a, see macroscopic score 1b), as compared to the sham group (Figure 1a). Macroscopic analysis showed a visible reduction in the extent of macroscopic sign with PEAultra treatment compared to other formulations (PEA and PEAmicro) (Figure 1a, see macroscopic score 1b). Four days after colitis induction by DNBS treatment, all mice had diarrhea and a significant reduction in body weight (compared with the sham group) (Figure 1c). PEAultra significantly restored the loss of body weight induced by DNBS-administration in mice (Figure 1c). Therefore, we analyzed the inflammatory cell profile in colon sections. Noteworthy, the colon infiltration with PMN appeared to correlate with the leukocyte influx, thus we investigated the effect of PEA and its formulations on MPO activity, a well-known marker of neutrophil infiltration. MPO activity was significantly increased after DNBS injection in vehicle-treated mice (Figure 1d). Treatment with PEAultra significantly attenuated neutrophil infiltration into the colon tissue in respect to the other formulation (Figure 1d). Statistics showed that PEAultra treatment correspondingly decreased by 74.36 % (P <0.05) neutrophil infiltration compared with PEAmicro and PEA (67.9% and 69.24%, respectively; P <0.05).

Four days after intra-colonic administration of DNBS, the colon appeared flaccid and filled with liquid stool. The macroscopic inspection of cecum, colon, and rectum showed the presence of mucosal congestion, erosion, and hemorrhagic ulcerations compared to sham colon (a). PEA and PEA micro treatment reduced the macroscopic damage (a); however, PEAultra significantly reduced macroscopic damage (a). The macroscopic damage score was made by two independent observers (b). Myeloperoxidase (MPO) activity was significantly increased in DNBS-treated mice in comparison to sham-treated mice (d). PEAultra treatment significantly reduced the degree of PMN infiltration in inflamed colon compared to PEA and PEAmicro treatments (d). Six days after colitis induced by DNBS treatment, all mice had a significant reduction in body weight compared with sham groups (c). PEAultra significantly increased body weight during the 6 days after DNBS injection in mice (c). This figure is representative of at least three experiments performed on different experimental days. Data are expressed as mean ± SEM from n = 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Microscopic studies of hematoxylin and eosin-stained colon tissue sections of DNBS control group of animals showed extensive formation of ulcer with infiltration of inflammatory cells mostly neutrophils with moderate lymphoid tissue hyperplasia (Figure 2). In the PEA and PEAmicro groups, acute inflammation partially subsided with partial healing of ulcer were observed (Figure 2); on the contrary, the infiltration of inflammatory cells and severity of colitis was reduced significantly in the PEAultra-treated group (Figure 2). However, no ulcer formation was observed in the colonic tissue of the sham group (Figure 2)
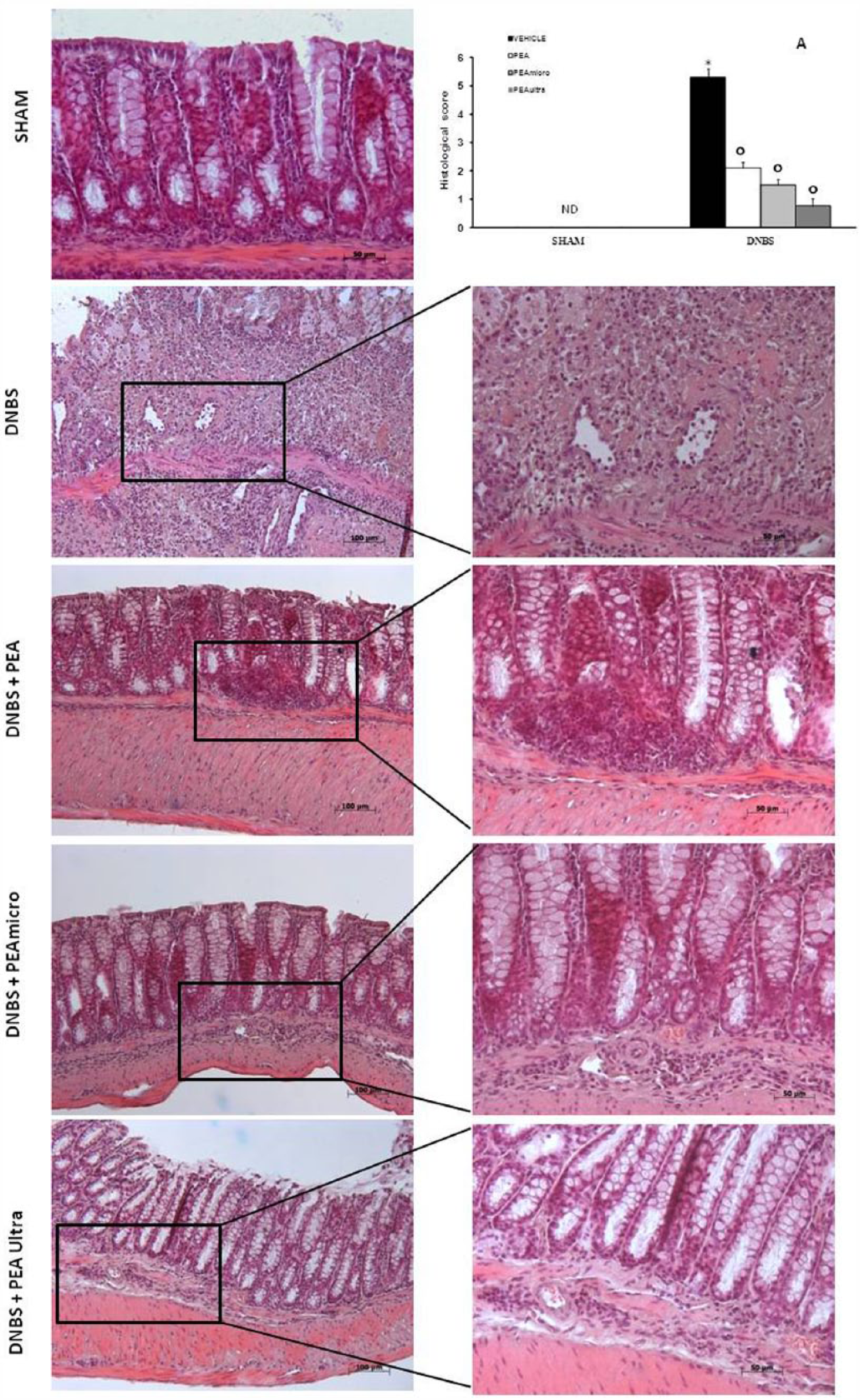
Representative histological appearance of mouse colon. Normal colon of healthy mice at 10× magnification (SHAM), colon of DNBS-induced colitis group at 10× magnification and 20× magnification (DNBS), PEA-treated colon of mice at 10× magnification and at 20× magnification (PEA), PEAmicro-treated colon of mice at 10× magnification and at 20× magnification (PEAmicro), PEAultra-treated colon of mice at 10× magnification and at 20× magnification (PEAultra). The histological score was made by two independent observers (a). This figure is representative of at least three experiments performed on different experimental days. Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Based on these findings, all further experiments were performed on mice subjected to DNBS-induced colitis treated with PEAultra.
Effects of PEAultra on NF-kB-related pro-inflammatory mediators
In order to understand the molecular mechanisms implicated in PEAultra effects on DNBS colonic inflammation, we investigated the activation of the NF-kB pathway. In particular, we analzyed by western blot the expression levels of IκBα and NF-κB p65 subunit in protein extracts from colon homogenates by means of specific antibodies. As shown in Figure 3a, a basal level of IkBa was detected in the colon from sham-mice while in mice subjected to DNBS injection, the levels of IkBa were significantly reduced. PEAultra was able to restore IkBa expression levels by preventing its degradation. Accordingly, the translocation of p65 subunit protein into the nuclei of the colon tissue homogenates was increased after DNBS injection, compared with the sham group. PEAultra administration significantly reduced the translocation of p65 in nuclear extracts from colon homogenates (Figure 3b), suggesting that PEAultra prevented the activation of the NF-kB pathway. Recent evidence links the colonic inflammation to the TLR4 pathway and the downstream signaling cascade linked to p38-pERK-pJNK. 27 Thus, we evaluated the effects of PEAultra on TLR4 expression by immunohistochemical analysis. A significant increase in TLR4 expression in colon tissue section was observed in the DNBS-treated mice (Figure 3c, see densitometric analysis) compared to sham (Figure 3c, see densitometric analysis). PEAultra administration was able to significantly reduce TLR4 expression in the DNBS group (Figure 3c, see densitometric analysis). To confirm the implication of TLR4 and the capability of PEAultra to interfere with the TLR4 signaling cascade, we evaluated by western blot analysis the expression of phospho-p38 in colon homogenates. A significant increase in phospho-p38 levels was observed in the colon from DNBS-injected mice compared to sham (Figure 3d). PEAultra notably reduced phospho-p38 expression, thus corroborating the importance of TLR4/p-p38 in this inflammatory pathway. Moreover, several pro-inflammatory mediators involved in colonic inflammation are transcriptionally induced by the NF-kB pathway; therefore, we tested whether PEAultra treatment of mice modulated the inflammatory secretion of TNF-α and IL-1β cytokines by measuring their protein levels by ELISA. The level of TNF-α was 12 ± 2.5 pg/100 g wet tissue in intact control mice. However, in the DNBS-treated mice there was an increase in TNF-α production (95 ± 1.0 pg/100 g wet tissue); treatment with PEAultra significantly inhibited this increase (43 ± 3.3 pg/100 g wet tissue, P <0.05; Figure 4a). Accordingly, IL-1β level was 13 ± 1.0 pg/100 g wet tissue in DNBS-treated mice, which was significantly higher than the levels in the mice treated with PEAultra (42 ± 1.1 pg/100 g wet tissue, P <0.001), indicating that these treatments significantly inhibited the increase in IL-1β level following colitis induction. The serum IL-6 level of the normal intact group was 13 ± 1.5 pg/100 g wet tissue (Figure 4b).

Representative western blots showing the effects of PEAultra on IkB-a degradation (a), p65 translocation (b), and phospho-p38 expression (d) after DNBS injection. A representative blot of lysates (a, b, and d) obtained from 10 animals/group is shown, and densitometric analysis of all animals is reported. Immunohistochemical analysis for TLR4 showed a positive staining in DNBS-treated mice compared to sham mice (c); PEAultra reduced the staining for the receptor. The results are expressed as mean ± SEM from 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
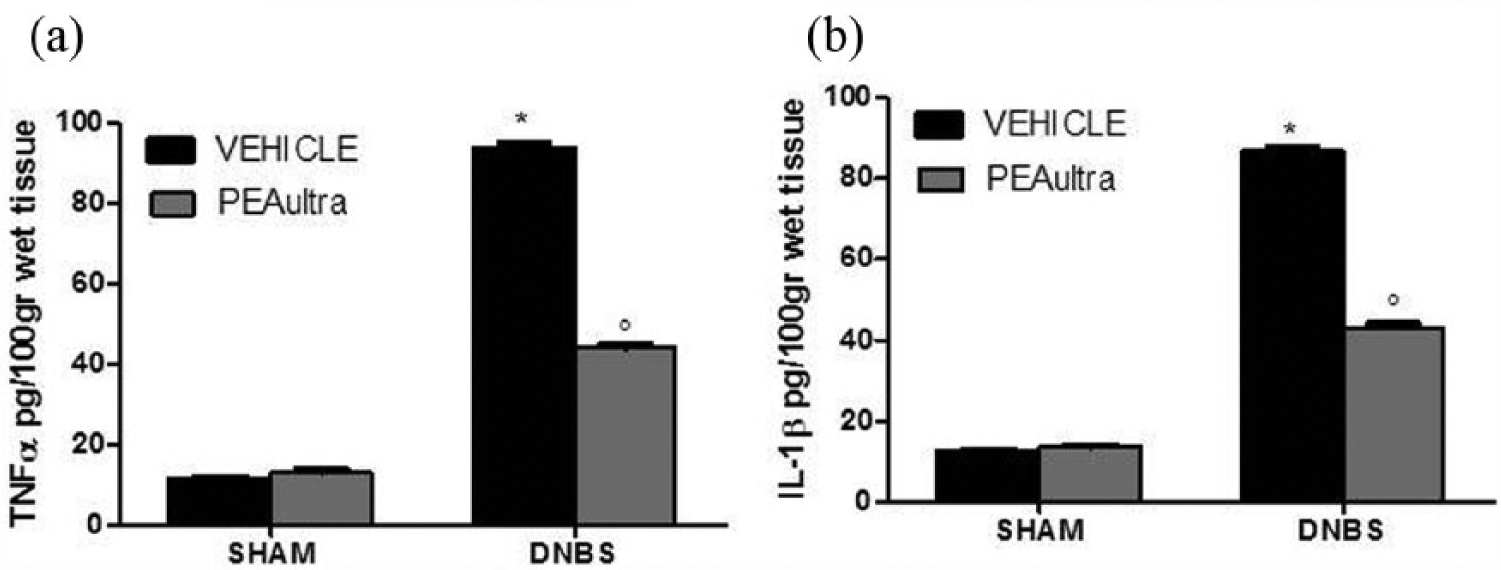
TNF-α and Il-1β colon levels were significantly elevated after 4 days from DNBS injection if compared with sham mice (a and b, respectively). PEAultra significantly reduced TNF-a and IL-1β levels (a and b, respectively). Data are expressed as mean ± SEM from n = 10 mice for each group. *P <0.01 vs. sham group. °P <0.01 vs. DNBS.
Effect of PEAultra on metalloproteinases, iNOS and COX-2 expression
Colonic inflammation is accompanied by the degradation of extracellular matrix. 28 Among the pro-inflammatory mediators related to NF-kB, we studied the expression of MMP-9 and MMP-2 by western blot analysis. An undetectable basal level of MMP-2 expression was identified in tissue homogenates from control mice, as shown by the immunoreactive band migrating at approximately 72 kDa (Figure 5a, see densitometric analysis A1). In contrast, a significant upregulation of MMP-2 expression was observed in the colon tissue homogenates from mice subjected to DNBS treatment (Figure 5a, see densitometric analysis A1). Conversely, a remarkable reduction of MMP-2 protein expression was observed in colon tissue homogenates from PEAultra-treated mice (Figure 5a, see densitometric analysis A1). A similar profile of results was observed for MMP-9 expression (Figure 5b, see densitometric analysis B1).

Western blot analysis of colonic homogenates showed basal levels of total MMP-2 from control mice, as shown by the immunoreactive band migrating at approximately 72 kDa (a, see densitometric analysis A1). A significant upregulation of MMP-2 expression was observed in the colon tissues from mice treated with DNBS when compared with controls. PEAultra treatment significantly prevented DNBS-induced MMP-2 expression (a, see densitometric analysis A1). Similarly, a basal level of total MMP-9 was detected in tissues from control mice, as shown by the immunoreactive band migrating at approximately 95 kDa (b, see densitometric analysis B1). A significant upregulation of MMP-9 expression was observed in the colon tissues from mice treated with DNBS when compared with controls. PEAultra treatment significantly prevented DNBS-induced MMP-9 expression (b, see densitometric analysis B1). Also, a significant increase of iNOS (c, see densitometric analysis C1) and COX-2 (d, see densitometric analysis D1) levels was observed in the colon from DNBS- injected mice. The treatment with PEAultra prevented the DNBS-induced iNOS (c, see densitometric analysis C1) and COX-2 (d, see densitometric analysis D1) expression. β-actin was used as internal control. Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Several reports demonstrated high levels of nitric oxide (NO) in colitis. 29 To determine the effect of PEAultra, we evaluated iNOS expression by western blot analysis. Interestingly, PEAultra treatment significantly prevented the iNOS protein expression (Figure 5c, see densitometric analysis C1) observed in colon tissue homogenates of mice subjected to DNBS (Figure 5c, see densitometric analysis C1). A similar profile was observed with COX-2 expression, where DNBS injection markedly upregulated its expression as compared sham-treated mice (Figure 5d, see densitometric analysis D1). Treatment with PEAultra reduced COX-2 protein expression (Figure 5d, see densitometric analysis D1).
Effect of PEAultra on ICAM-1 and P-selectin expression
Since expression of selectins are necessary for leukocyte and T-cell migration, 30 we evaluated the intestinal expression of ICAM-1 and P-selectin. Positive staining for P-selectin and for ICAM-1 was substantially increased in the vessels of the lamina propria and submucosa as well as in epithelial cells of injured colon and in infiltrated inflammatory cells in damaged tissues from DNBS-treated mice as compared to sham animals (Figure 6a and b, respectively, see densitometric analysis 6c). PEAultra treatment reduced the levels of positive staining for P-selectin (Figure 6a, see densitometric analysis 6c) and ICAM-I (Figure 6b, see densitometric analysis 6c) in the colon tissues collected from DNBS-treated mice.

Immunohistochemical analysis for P-selectin and ICAM-1 showed positive staining mainly localized on endothelial cells and in the inflammatory cells in the injured area from DNBS-treated mice compared to sham mice (a and b, respectively). PEAultra treatment reduced the staining for P-selectin (a) and for ICAM-I (b) in the colon tissues collected from DNBS-treated mice. Densitometric analysis of immunocytochemistry photographs (n = 5 photos from each sample collected from all mice in each experimental group) for P-selectin and ICAM-1 from colon tissues was assessed (d). The assay was carried out by using Optilab Graftek software on a Macintosh personal computer (CPU G3-266). Data are expressed as % of total tissue area. This figure is representative of at least three experiments performed on different experimental days. Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Effect of PEAultra on nitrotyrosine formation and PAR formation
Nitrotyrosine, a specific marker of nitrosative stress abundantly found in colonic inflammation, 31 was measured by immunohistochemical analysis in the terminal colon sections to determine the localization of ‘peroxynitrite formation’ and/or other nitrogen derivatives produced during experimental colitis. Colon sections from sham-treated mice did not stain for nitrotyrosine (Figure 7a, see densitometric analysis 7c), whereas colon sections obtained from DNBS-treated mice exhibited positive staining for nitrotyrosine (Figure 7a, see densitometric analysis 7c). The positive staining was mainly localized in inflammatory cells as well as around the vessels. PEAultra treatment markedly reduced the degree of positive staining for nitrotyrosine (Figure 7a, see densitometric analysis 7c) in the colon. A similar profile of expression was observed for PAR, an indicator of in vivo PARP activation that revealed the occurrence of positive staining for PAR localized in nuclei of inflammatory cells in the colon tissues from DNBS-treated mice (Figure 7b, see densitometric analysis 7c). PEAultra treatment reduced the degree of positive staining for PAR (Figure 7b, see densitometric analysis 7c) in the sections of colon tissue. No positive staining for PAR was found in the colon tissues from sham-treated mice (Figure 7b, see densitometric analysis 7c).

Effects of PEAultra treatment on nitrotyrosine and PAR formation. Sections obtained from sham mice did not demonstrate positive staining for nitrotyrosine or PAR (a and b, respectively). Colon section obtained from DNBS-treated mice exhibited positive staining for nitrotyrosine; the positive staining was mainly localized in inflammatory cells as well as around the vessels (a). PEAultra treatment reduced the degree of positive staining for nitrotyrosine (a) in the colon. The same situation was visible for PAR that revealed a positive staining for PAR localized in nuclei of inflammatory cells in the colon tissues from DNBS-treated mice (b). PEAultra treatment reduced the degree of positive staining for PAR (b) in the colon. Densitometric analysis of immunocytochemistry photographs (n = 5 photos from each sample collected from all mice in each experimental group) for nitrotyrosine and PAR from colon tissues was assessed (c). The assay was carried out by using Optilab Graftek software on a Macintosh personal computer (CPU G3-266). Data are expressed as % of total tissue area. Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Effect of PEAultra on inflammatory markers in PPARα KO mice
When given to WT mice, DNBS resulted in a severe illness, as demonstrated by microscopic examination, showing a marked mononuclear cell infiltration associated with loss of goblet cells, while some parts of the mucosa layer lost crypts and were replaced by polymorphonuclear neutrophil and mononuclear cells (Figure 8a, see histological score 8b). PEAultra treatment significantly reduced the histological signs of colon injury (Figure 8a, see histological score 8b). Furthermore, it has been widely proposed that PEA effects are mostly related to the activation of the PPARa pathway. 17

No histological alteration was observed in the colon tissue from sham-treated WT and PPARα KO mice (a). The histopathological features included a transmural necrosis and edema and a diffuse leukocyte cellular infiltrate in the submucosa of colon section from DNBS-treated WT and PPARα KO mice (a); PEAultra reduced the severity of injury in WT mice (a), a similar situation was present in PPARα KO mice treated with PEAultra (a). The histological score was made by two independent observers (b). No macroscopical alteration was observed in Sham PPARα KO mice (c) while in DNBS treated PPARα KO mice a marked hemorrhagic ulcerations were noted (c); PEAultra significantly reduced macroscopic damage (c). The macroscopical score was made by two independent observers (d). MPO activity was significantly reduced in PEAultra treatment compared to DNBS-treated mice (e). Body weight was measured for 6 days (f). Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Based on these premises, we decided to investigate the effects of PEAultra on PPARα KO mice. In agreement with data collected from WT mice, no histological or macrosopical alterations were observed in the colon tissue from sham-treated PPARα KO mice (Figure 8a, see histological score B; Figure 8c, see macroscopic score 8d). The histopathological features included a transmural necrosis and edema and a diffuse leukocyte cellular infiltrate in the submucosa of colon section from DNBS-treated mice (Figure 8a, see histological score 8b). The macroscopic inspection showed presence of erosion and hemorrhagic ulcerations, and 4 days after intra-colonic administration of DNBS, the colon appeared flaccid and filled with liquid stool (Figure 8c, see macroscopic score 8d). To our surprise, the absence of PPARα did not affect the action of PEAultra, maintaining its beneficial effect (Figure 8a, see histological score 8b; Figure 7c, see macroscopic score 7d). In addition, PEAultra treatment in PPARaKO mice, significantly reduced the degree of PMN infiltration (determined as increase in MPO activity) in inflamed colon, as compared to the DNBS-treated mice (Figure 8e), suggesting that even though the PPARα receptor is lacking, PEAultra is capable of exerting an anti-inflammatory effect (Figure 8e). Four days after colitis was induced by DNBS treatment, PPARα KO mice showed a significant reduction in body weight (compared with the sham groups of mice) (Figure 8f). PEAultra administration determined a reduction of body weight induced by DNBS-administration in KO mice (Figure 8f). These results strongly favor the hypothesis that PEAultra might additionally act through other than PPARa activation pathways.
Effect of PEAultra on inflammatory markers in TLR4KO mice
In order to deepen the molecular pathways involved into PEAultra effects, and since the TLR4 pathway is known to play an important role, we analyzed the effects of PEAultra on TLR4KO mice treated with DNBS. No histological or macroscopical alterations were observed in the colon tissue from DNBS-treated mice (Figure 9a, see histological score; Figure 9b, see macroscopic score) similarly to sham-treated mice (Figure 9a, see histological score; Figure 9b, see macroscopic score). PEAultra treatment did not alter the normal architecture of colon tissue (Figure 9a, see histological score; Figure 9b, see macroscopic score). Importantly, PEAultra treatment of TLR4KO mice did not improve the beneficial effect already produced by the absence of TRL4 on PMN infiltration, measured by evaluating the MPO activity (Figure 9C). The daily control of body weight showed no weight loss in TLR4 KO mice and in PEAultra TLR4-treated mice (Figure 9d). These results suggest that the contribution of TLR4 is pivotal in inducing colonic inflammation and that the effects of PEAultra may be related to interference on this signaling pathway.

No histological and macroscopical alterations were observed in the colon tissue from DNBS-treated mice and sham-treated mice (a and b, respectively). PEAultra treatment did not alter the normal architecture of colon tissue. The histological and macroscopic damage score was made by two independent observers (a and b, respectively). PMN infiltration did not show significant difference between groups (c). The daily control of body weight showed no weight loss in TLR4 KO mice and in PEAultra TLR4-treated mice (d). Data are means ± SEM of 10 mice for each group. *P <0.01 vs. sham; °P <0.01 vs. DNBS.
Effect of PEAultra on immune-inflammatory markers in IL-10 KO mice
The DNBS animal model is well known and its ability to overlap with the human pathology is proven.9,10 However, IBD is a group of inflammatory diseases with a strong autoimmune molecular basis. Therefore, in order to assess whether PEAultra was capable of preventing the autoimmune-mediated inflammation, we decided to use the IL-10KO mice that spontaneously develop a Th1-dependent chronic enterocolitis shortly after birth, with full establishment of the clinical features at the age of 8–10 weeks. IL10KO mice aged 9 weeks were treated with PEAultra for 7 weeks. As shown in Figure 10, treatment of IL10KO mice with PEAultra significantly reduced the activity of colitis at week 16, as assessed by histologic damage scores, the MPO activity, as well as TNF-α and MCP-1α levels expression in colon tissue homogenates. These results suggest that PEAultra is capable of exerting anti-inflammatory effects in a spontaneous autoimmune colitis model.

H&E staining of colon from 9-week-old IL-10KO mice (note the presence of a fully established colitis, (a) 16-week-old IL-10 WT mice (no colitis, a), 16-week-old IL-10KO mice (thickening of the colon wall and massive inflammatory infiltrate in the LP, a), and 16-week-old IL-10 KO mice with PEAultra (subepithelial edema with no inflammatory infiltrate, a) was performed. No histological alteration was observed in 16-week-old IL-10 WT mice (a). The histological score was made by two independent observers (a). Administration of PEAultra exerts therapeutic activity in fully established colitis in 16-week-old IL-10 KO mice. PEAultra was administered i.p. for 7 consecutive weeks, starting at the age of 9 weeks. Effect of PEAultra administration on colonic colonic MIP-1α (b), MPO activity (c), and TNF-α levels (d). Data are means ± SEM of 10 mice for each group. #P <0.01 vs. 16-week-old WT mice; #*P <0.01 vs. 9-week-old IL-10 KO mice; ##*P <0.01 vs. 16-week-old IL-10 KO mice.
Discussion
Crohn’s disease and ulcerative colitis, collectively referred to as inflammatory bowel diseases, are relatively common diseases of the gastrointestinal tract. IBD are characterized by dysfunction of mucosal immune response, abnormal cytokine production with increase in TNF-α and IL-1, augmentation in adhesion molecules expression and cell infiltrate that ultimately lead to epithelial cell apoptosis and mucosal damage.7,9,32 The pathogenesis of DNBS-induced colitis is immunologic, characterized by a Th-1 response and is ameliorated in rats by well-known drugs, nitric oxide synthase inhibitors, 33 and in mice also by immunostimulatory DNA sequences; 34 DNBS is a contact-sensitizing allergen that, when intrarectally administered and dissolved in ethanol, induces transmural necrosis.
Based on studies conducted on PEA and its anti-inflammatory effects,12,22 we wanted to test a new formulation of this compound (PEAmicro and PEAultra) on a colitis model. We observed the highest effectiveness in PEA ultramicronized formulation, wherein more than 90% by weight of PEA has particle sizes lower than 6 microns. A common paradigm for several pro-inflammatory mediators over-expressed in IBD is the activation of the NF-κB pathway, leading to the increased transcription of pro-inflammatory target genes, such as those encoding for chemokines and cytokines, pro-inflammatory enzymes, thus perpetuating inflammatory responses and promoting cell survival. 35
In our study, we found that DNBS caused a significant increase in the phosphorylation of NF- κB p65 on Ser536 in the colon tissues of WT mice at 4 days, whereas treatment with PEAultra reduced this phosphorylation; these data were also supported by the augmented levels of expression of IκB-α, NF-κB inhibitor, whose degradation was prevented by PEAultra administration to mice. Moreover, our results demonstrate that TNF-α and IL-1β production in the colon after DNBS injection were reduced by PEAultra treatment. It has been demonstrated that NF-κB regulates the expression of more genes as metalloproteinases (MMPs), considered to play a pivotal role in the progression of inflammation. The role of NF-κB in regulation of MMP-9 was further demonstrated by the inhibition of MMP-9 production by proteasome inhibitors, lactacystin and MG-132, which prevented the ubiquitination and dissociation of IκB from NF-κB. 36
There is also growing evidence that MMPs can be implicated in the pathophysiology of several intestinal inflammatory disorders, in fact, MMPs can be released from almost all connective tissue cells present in the bowel in response to inflammatory stimuli including inflammatory bowel disease, necrotizing enterocolitis, collagenous colitis, and diverticulitis. 37
Interestingly, we demonstrated a significant upregulation of MMP-2 and MMP-9 expression in the colon tissues from mice treated with DNBS. Moreover, a more and significantly pronounced inhibition of MMPs expression was observed when DNBS-treated mice received PEAultra.
To encounter and to engage antigens in inflamed tissues, T cells must leave the bloodstream, and the expression of selectins, such as P-selectin, and of addressins, such as ICAM-1, is necessary for T-cell interaction with the endothelium of blood vessels, extravasation, and tissue migration. 38 In accordance, we observed that DNBS injection induced the appearance of P-selectin on the endothelial vascular wall and the expression of ICAM-1 on endothelial cells. Treatment with PEAultra significantly abolished the expression of P-selectin and ICAM-1. The absence of an increased expression of the adhesion molecule in the colon tissue from mice treated with PEAultra is, at least in part, correlated with the reduction of the number of PMN, as shown by MPO activity, and with the attenuation in the colon tissue damage.
Activation and accumulation of leukocytes are one of the initial events of tissue injury as a result of release of ROS. The decrease in ROS levels corresponds to a decrease in NO that physiologically plays an important role in local and systemic inflammation. Here, we clearly demonstrated that PEAultra treatment fully inhibited the appearance of nitrotyrosine staining in the inflamed tissue. Moreover, these data strongly correlated with iNOS expression, thus suggesting that PEAultra was capable of preventing ROS pathway generation. Various studies have demonstrated that PARP hyperactivation after single DNA strand breakage induced by ROS plays an important role in the process of acute injury. 39 In this study, we confirmed the increase in PAR formation in the colon tissues from DNBS- treated mice and we showed that PEAultra treatment attenuated PARP hyperactivation, suggesting that PEAultra may act through different pathways, beside the anti-inflammatory one.
At the histological level, in the present study we observed that epithelial disruption was significantly less in mice treated with PEAultra, showing more resistant to DNBS-induced colitis with a significant resolution of the macroscopic and histological signs of the inflammatory process.
Although the exact mechanism by which PEAultra may modulate inflammatory pathway is not fully understood, recently, a relationship through PPARα and TLR4 has been proposed, showing an inhibition of S100B/TLR4 axis mainly via PPARα in Enteric Glial cells (EGC) during UC by PEA. 40 In agreement, here we report that PEAultra inactivated TLR4 pathway and the downstream activation of phospho-p38, whose expression was markedly decreased. Therefore, in order to assess the role of TLR4 on the modulation of inflammatory process, on the one hand, we evaluated the action of PEAultra on TLR4 KO mice showing a minor development of colitis in DNBS- injected mice. Noteworthy, PEAultra had no effect, suggesting that the mechanisms of action are linked to the TLR4 pathway. On the other hand, we showed that, surprisingly, PEAultra acts also independently from PPARα binding, as demonstrated by treatment of PPARαKO mice. In fact, our results show that PEAultra was capable of preventing the loss of body weight, and the MPO activity as well as the histological features observed in DNBS-treated mice. Noteworthy, the effects of PEAultra in PPARαKO mice paralleled with those observed in WT mice.
Furthermore, interleukin-10 (IL-10) is a pluripotent cytokine that plays a pivotal role in the regulation of immune and inflammatory responses. IL-10 has been shown to suppress the production of proinflammatory mediators and downregulate costimulatory molecules that are critical for the activation of T cells. 41 In our study, we used IL10KO mice in order to highlight the efficacy of PEAultra in a spontaneous immulogical model. Interestingly, PEAultra administration fully prevented the clinical, histologic, molecular, and immunologic signs of DNBS-induced colonic inflammation and exerted a therapeutic activity in IL-10KO mice with fully developed colitis.
Our results show that PEAultra is a new potent anti-inflammatory drug as compared to PEA and PEAmicro, and suggest that the PEAultra may represent a new therapeutic strategy for the treatment of IBD.
These effects might be also related to its lower weight, as compared to PEA or PEA micronized, which guarantees a greater absorption capacity. Taken together, our results shed new light on the mechanisms at the basis of IBD and the potential therapeutic efficacy of PEAultra, although further studies are needed to fully deepen the mechanism of action of PEA. In conclusion, PEAultra may be considered as a new possible therapeutic strategy versus autoimmune/inflammatory disorders.
Footnotes
Acknowledgements
The authors would like to thank Emanuela Mazzon and Maria Antonietta Medici for their excellent technical assistance during this study, Mr Francesco Soraci and Mr Giuseppe Mancuso for their secretarial and administrative assistance, and Miss Valentina Malvagni for her editorial assistance with the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by PON01-02512 grant.
