Abstract
Objectives
To investigate the differences in aneurysmal sac regression after endovascular aortic repair (EVAR) of abdominal aortic aneurysms (AAAs) in patients with and without dyslipidemia.
Methods
This was a retrospective analysis of 3453 patients from an international prospective registry (Europe, United States of America, Brazil, Australia, and New Zealand) of patients treated with the GORE® EXCLUDER® endograft. All scheduled EVARs for infrarenal AAAs between 2014 and 2016 with complete 6-year follow-up imaging data were included. Logistic regression analysis was performed to assess changes in aneurysm diameter based on dyslipidemia after adjusting for sex, age, body mass index, tobacco use, hypertension, coronary artery disease, aneurysm diameter, aneurysm neck length, and aneurysm neck angle. The secondary outcomes included all-cause mortality, stroke/transient ischemic attack (TIA), paraplegia/paraparesis/spinal cord ischemia, reintervention, endoleaks, and aortic rupture. A control group of patients without dyslipidemia with similar age and comorbidities was selected using propensity scores and matched using a 1:1 scheme.
Results
Of the 3453 patients, 85.3% in the non-dyslipidemia group and 85.5% in the dyslipidemia group were men. The mean age was 73.5 ± 8.8 years for the non-dyslipidemia group and 73.4 ± 8.2 years for the dyslipidemia group (p < .001). The mean body mass index was 26.7 ± 4.9 kg/m2 for the non-dyslipidemia group, whereas it was 28.0 ± 5.2 kg/m2 for the dyslipidemia group (p < .001). Overall, 2269 patients (64.0%) were identified as having dyslipidemia. After propensity score matching (PSM), the dyslipidemia group had higher rates of hypertension (p < .001), TIA (p = .006), carotid disease (p = .002), coronary artery bypass grafting (p = .005), congestive cardiac failure (p = .018), and peripheral artery disease (p < .001). The indications for device placement were similar between the groups. After PSM, the non-dyslipidemia group was more likely to be treated off-label (p = .030) and to have an aneurysm neck length of <15 mm (p = .035). There was no significant difference in the sac size changes within 1 year (p = .7) or 6 years (p = .14) between the groups. After PSM, all-cause mortality was 6.7% in individuals with dyslipidemia and 10.6% in those without (p = .018). However, there were no significant differences in aortic-related mortality between the overall (p = .571) and the matched (p = .662) populations. The rates of reintervention, stroke/transient ischemic attack, and spinal cord ischemia were not significantly different. Among patients with dyslipidemia, larger aneurysm size at 1 year (p = .037) and increased age at 6 years (p < .001) were associated with sac expansion.
Conclusion
In this comprehensive real-world study of patients who underwent EVAR with the GORE® EXCLUDER endoprosthesis found that dyslipidemia did not increase endoleak rates or worsen imaging outcomes over 6 years. Overall, these findings suggest that dyslipidemia does not negatively impact EVAR outcomes and may indicate improved long-term care.
Keywords
Introduction
Endovascular aortic repair (EVAR) is the gold standard treatment for abdominal aortic aneurysms (AAAs). 1 Aneurysm sac regression indicates successful treatment and better outcomes,2–4 whereas stability or expansion is associated with higher long-term mortality. 5 The Global Registry for Endovascular Aortic Treatment (GREAT) cohort showed that conical aneurysm necks and larger EVAR device diameters promoted sac regression, whereas older age and a larger initial AAA diameter correlated with sac expansion. 6 Sac expansion post-EVAR is influenced by endoleaks, anatomical characteristics, age, and comorbidities.5,7 In contrast, smoking, cerebrovascular disease, and statin therapy may reduce this risk.5,8 Device choice also affects sac regression, though mechanisms remain unclear.9,10 Diabetes mellitus appears to have little impact on the sac size after EVAR. 11
Dyslipidemia, characterized by high low-density lipoprotein and low high-density lipoprotein levels, is associated with AAA incidence.12–14 Paradoxically, a previous review showed an inconsistent link between dyslipidemia and its potential influence on sac regression after EVAR in patients with atherosclerosis.8,15 Statin therapy may explain this link owing to its benefits in sac regression, though the overall role of dyslipidemia remains unexplored.5,8
This study aimed to evaluate the potential differences in 6-year aneurysm sac regression rates after EVAR using the GORE® EXCLUDER® stent graft for AAA in patients with and without dyslipidemia examining differences in endoleak and mortality rates using a prospective cohort registry.
Materials and methods
GREAT (https://www.clinicaltrials.gov, NCT01658787) started in 2010 and encompassed 114 centers worldwide, including the United States of America, Europe, Australia, New Zealand, and Brazil. Institutional review board and/or ethics committee approval was granted at all participating facilities prior to enrollment and guidelines from the Declaration of Helsinki and Good Clinical Practice were followed. It includes approximately 5000 patients with thoracic, abdominal, or thoracoabdominal aortic diseases. The technical procedures and methodologies for the GREAT registry have been published previously. 16 The study protocol was approved by the institutional review board, and all patients provided informed consent. The GREAT collects data on aortic devices from W.L. Gore and Associates, including demographics and outcomes. Participation in the registry did not mandate strict compliance with a follow-up protocol; rather, it was at the discretion of the enrolling or participating site. It was advisable to conduct follow-up assessments at intervals of 30 days, 6 months, 1 year, and then annually for up to 10 years. For these follow-up evaluations, the recommended imaging modalities include computed tomography (CT), magnetic resonance imaging (MRI), and duplex ultrasound. The Rave web-based platform (Medidata Solutions, New York, NY, USA) was used for data collection and was monitored by Gore Clinical Research. The focus was on infrarenal EVAR procedures performed between January 2014 and December 2016, excluding cases of ancillary procedures or inflammatory conditions. All cases used the GORE® EXCLUDER® device for EVAR. Anesthesia techniques varied by center, and documentation included access methods and supplementary procedures. Complete angiography was routinely performed postoperatively.
Definitions
Technical success was defined as effective endograft deployment without type 1 or 3 endoleaks or unintentional branch coverage with successful delivery system removal. Major adverse cardiovascular events included myocardial infarction, stroke, transient ischemic attack (TIA), and aortic-related death, as documented using the MedDRA standardized medical query code based on site descriptions. This study identified patients with dyslipidemia by analyzing their medical records and hospital coding systems rather than relying solely on laboratory values.
Outcomes
The primary outcome was the proportion of patients with aneurysm sac regression one and 6 years post-EVAR. Secondary outcomes included the incidence of all-cause mortality, stroke/TIA, paraplegia/paraparesis/spinal cord ischemia, and reintervention rates, in addition to endoleak and aortic rupture. The annual incidence of these events was recorded. Device-related re-interventions were determined by the GREAT advisory committee.
Statistical analysis
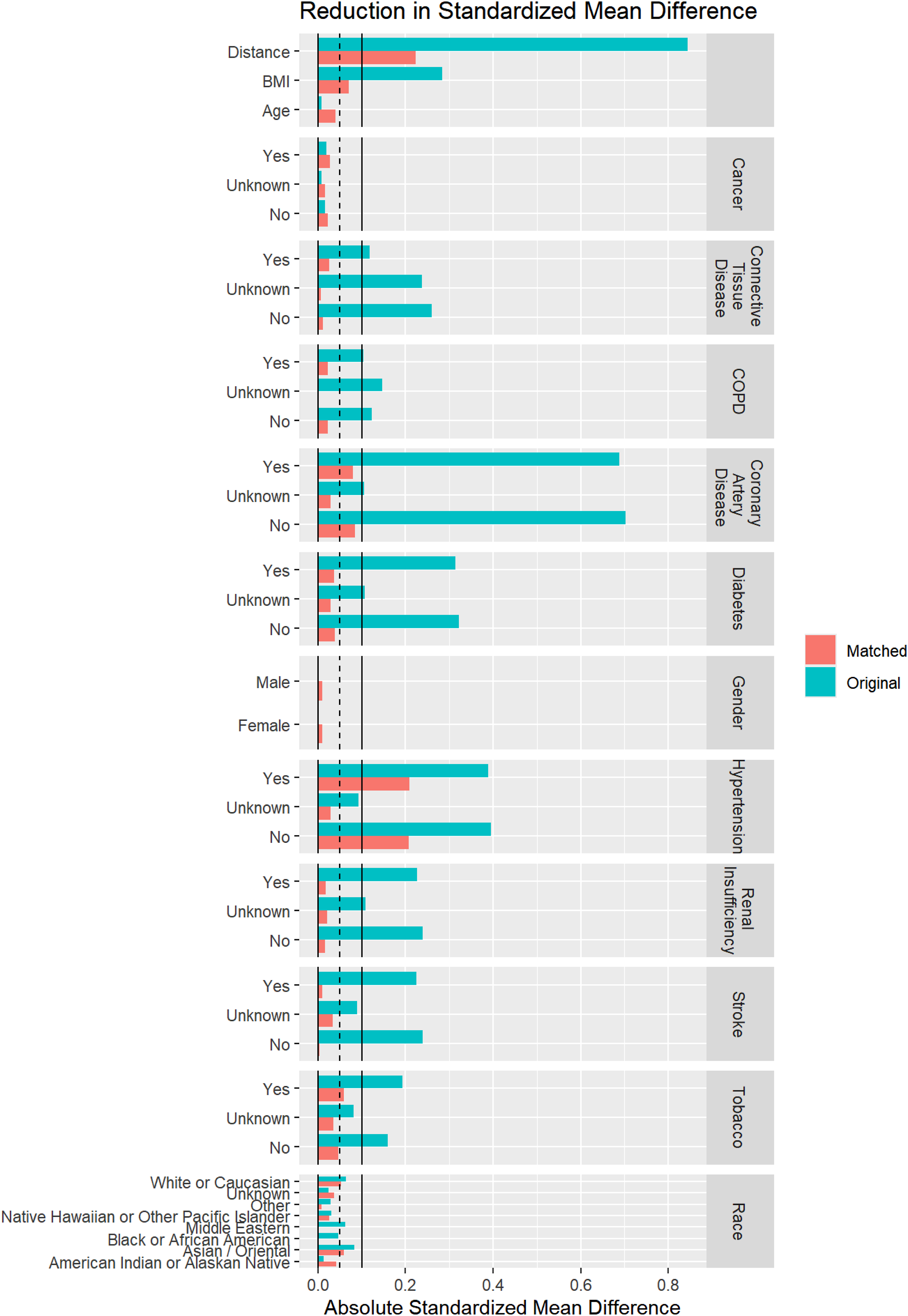
Categorical variables were expressed as numbers and percentages, while continuous variables were reported as mean ± standard deviation or median (range) for non-normal distributions. Normality was assessed using Q–Q plots and the Shapiro–Wilk test. Kaplan–Meier survival estimates were generated and compared using the Mantel–Cox log-rank test. Single-predictor models evaluated the relationship between risk factors and outcomes, retaining significant interaction terms with p-values of 0.1 or less. A multivariable Cox proportional hazards model was developed through forward selection, focusing on risk factors significant at p < .1, and was refined to include significant interactions to address the proportional hazards assumption. We established a propensity score-matched (PSM) cohort of individuals without dyslipidemia (controls) to account for baseline differences. The distribution of propensity scores effectively distinguished between the groups, with individuals without dyslipidemia peaking at approximately 0.5, whereas those with dyslipidemia peaked at 0.4. Most participants without dyslipidemia were retained and those with lower scores in the dyslipidemia group were excluded (Figure 1). The post-matching C-statistics were 0.5, indicating a balance with negligible standardized mean differences (Figure 2). All calculations were conducted using R version 4.0.5 (R Project, Vienna, Austria), with a significance threshold of p < .05. Distribution of propensity scores for the total cohort of patients with dyslipidemia and non-dyslipidemia (before propensity score matching) and matched populations (after propensity score matching). Standardized mean difference between the diabetic and non-diabetic groups, pre-(blue) and post-matching (red), using the generated propensity scores in 3453 patients with AAA treated with EVAR.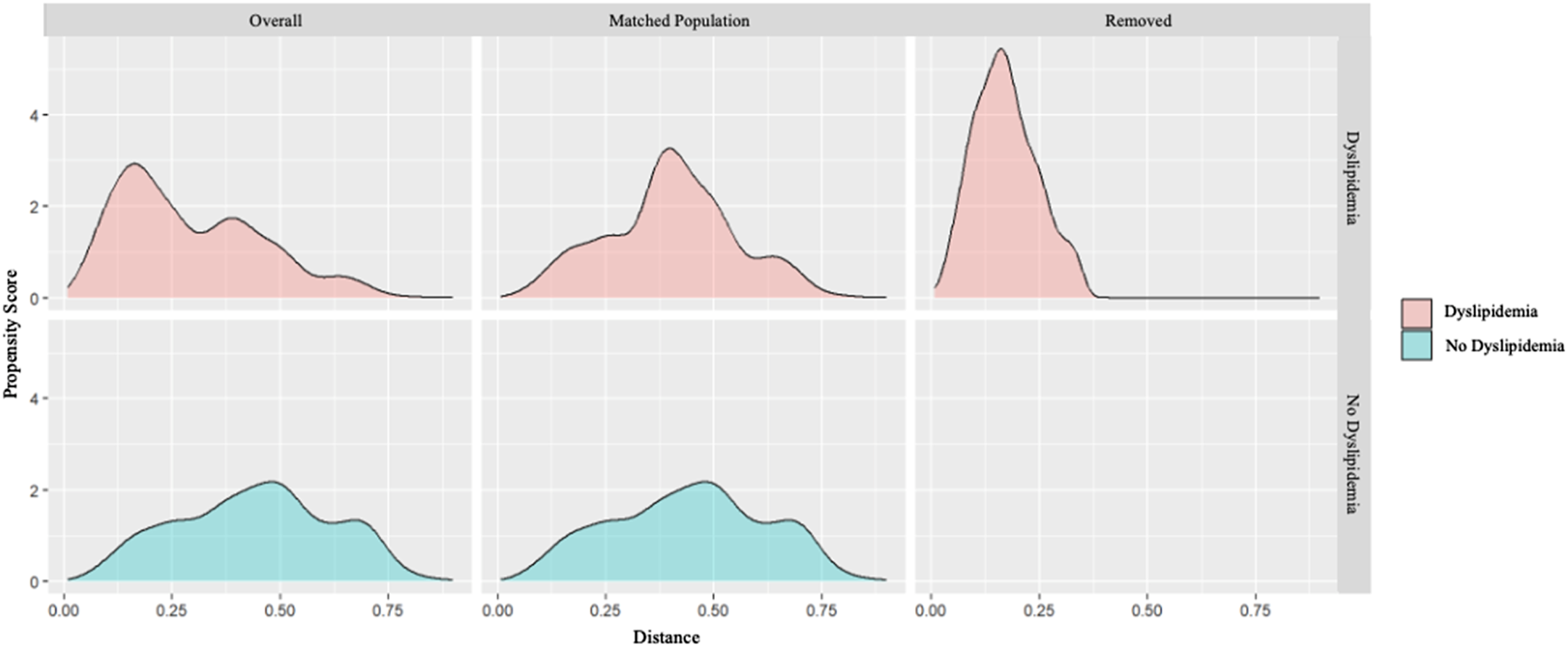
Results
Demographics and comorbidities of patients with and without dyslipidemia and abdominal aortic aneurysm in the general sample (n = 3453) and propensity score-matched (PSM) cohort (n = 2354).
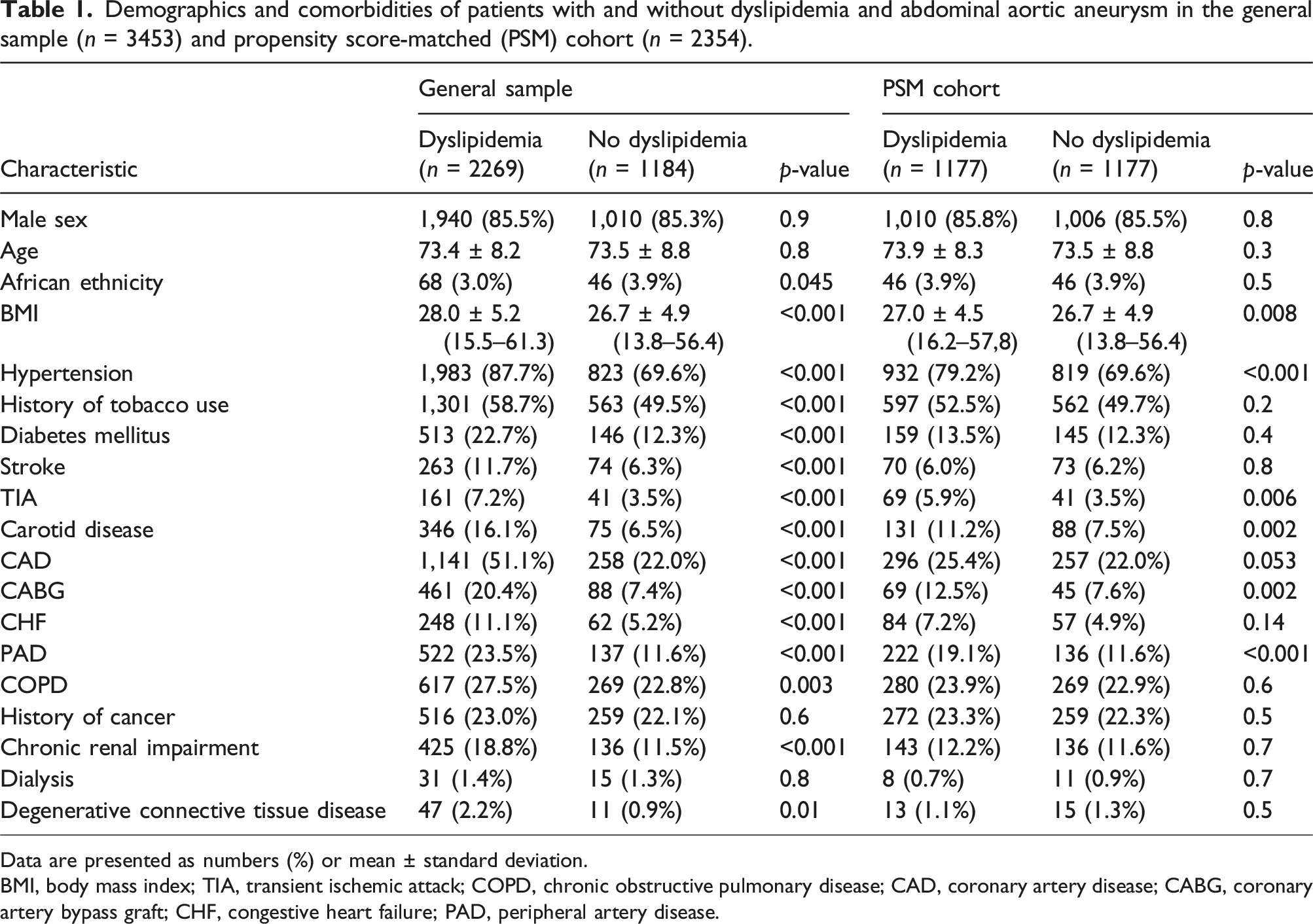
Data are presented as numbers (%) or mean ± standard deviation.
BMI, body mass index; TIA, transient ischemic attack; COPD, chronic obstructive pulmonary disease; CAD, coronary artery disease; CABG, coronary artery bypass graft; CHF, congestive heart failure; PAD, peripheral artery disease.
Operative details
Abdominal aortic aneurysm details of patients in the general sample (n = 3453) and the propensity score-matched (PSM) cohort (n = 2354).
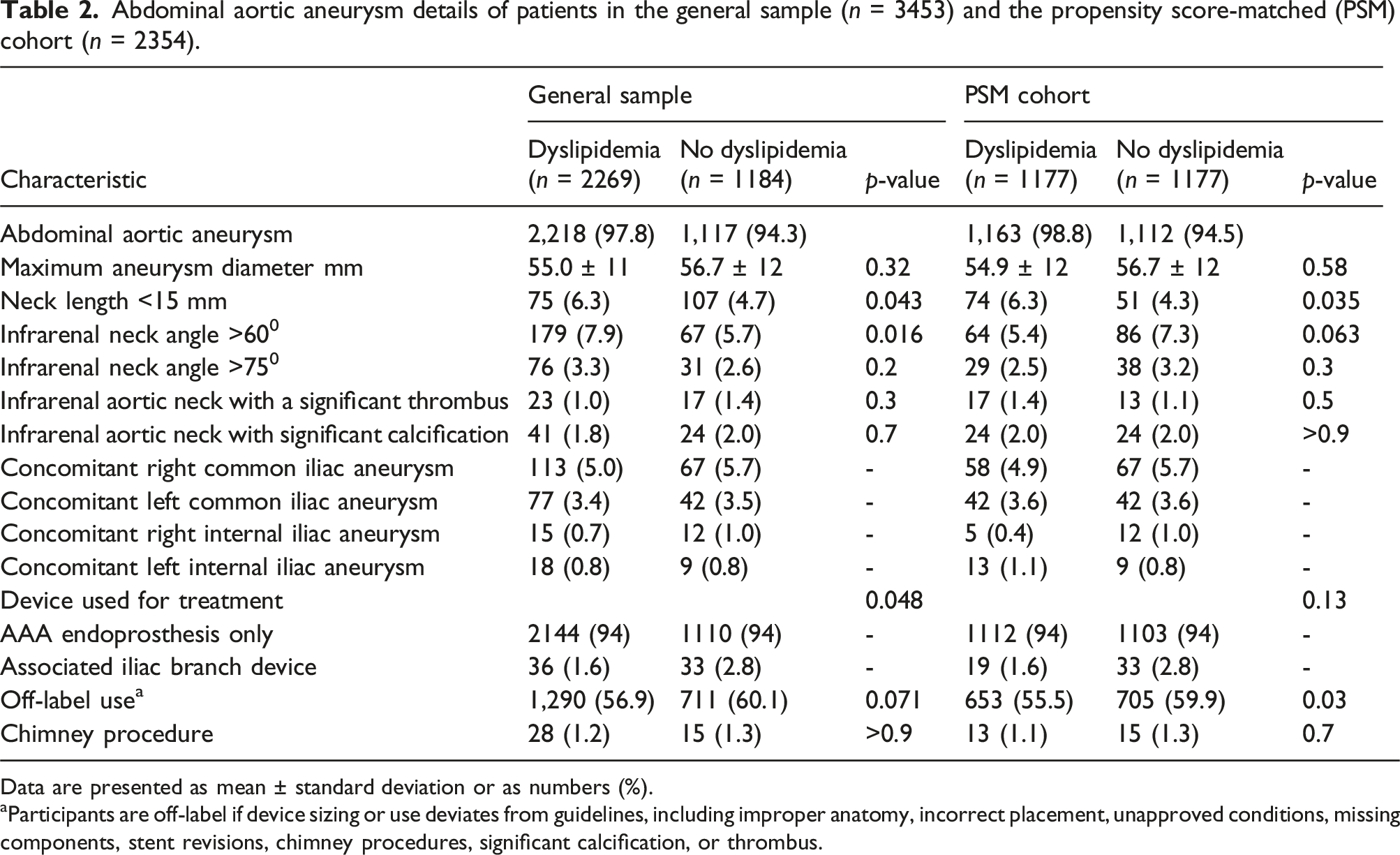
Data are presented as mean ± standard deviation or as numbers (%).
aParticipants are off-label if device sizing or use deviates from guidelines, including improper anatomy, incorrect placement, unapproved conditions, missing components, stent revisions, chimney procedures, significant calcification, or thrombus.
Right common iliac aneurysms were found in 67 and 113 patients with and without dyslipidemia, respectively. Left common iliac aneurysms were noted in 42 patients without dyslipidemia and in 36 with dyslipidemia. An iliac branch endoprosthesis was needed in 33 patients without dyslipidemia and in 36 with dyslipidemia (Table 2).
Aneurysm sac evolution
AAA changes and incidence of adverse outcomes in patients with AAA treated with EVAR at 1-year follow-up.
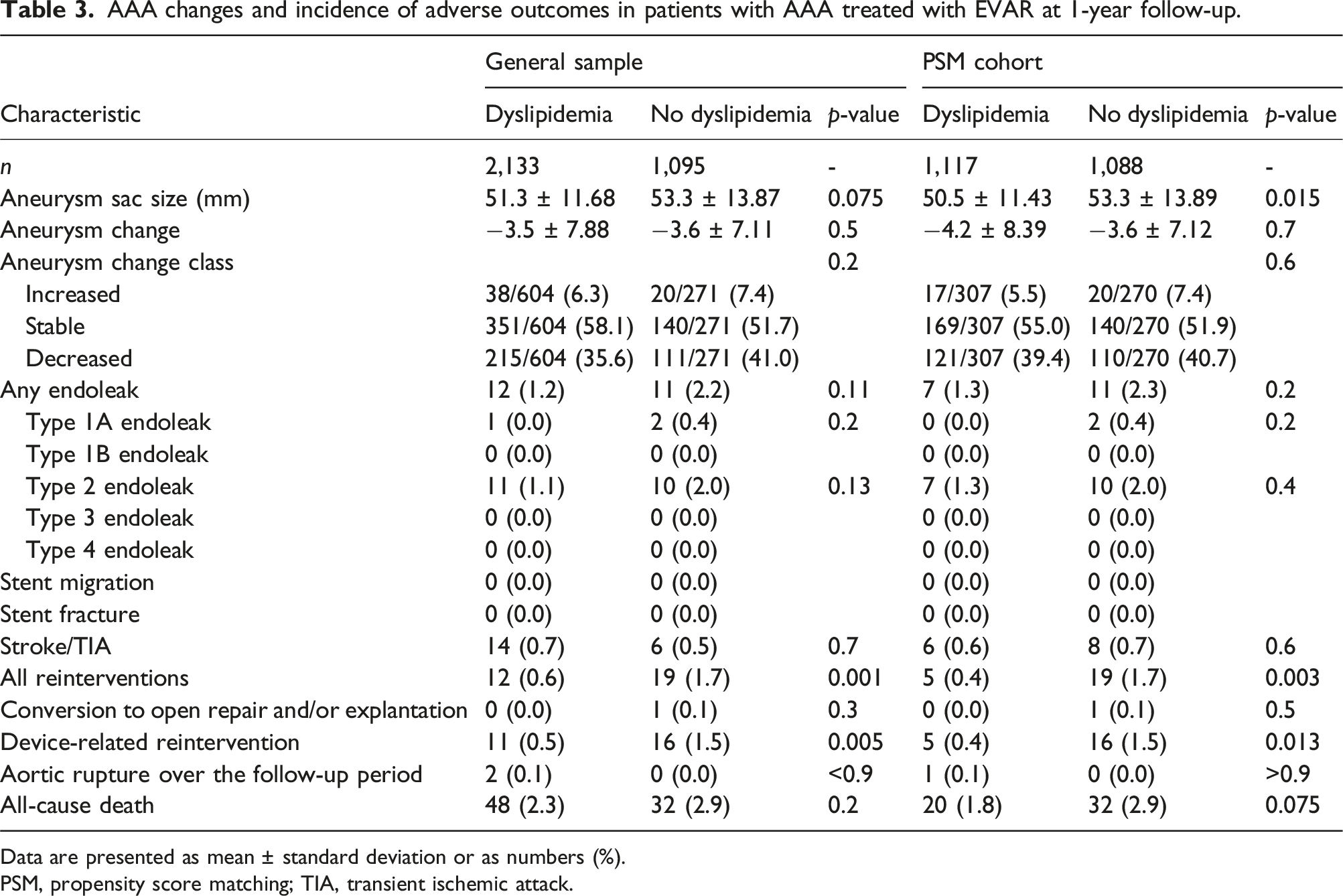
Data are presented as mean ± standard deviation or as numbers (%).
PSM, propensity score matching; TIA, transient ischemic attack.
AAA changes and incidence of adverse outcomes in patients with AAA treated with EVAR at 6-year follow-up.
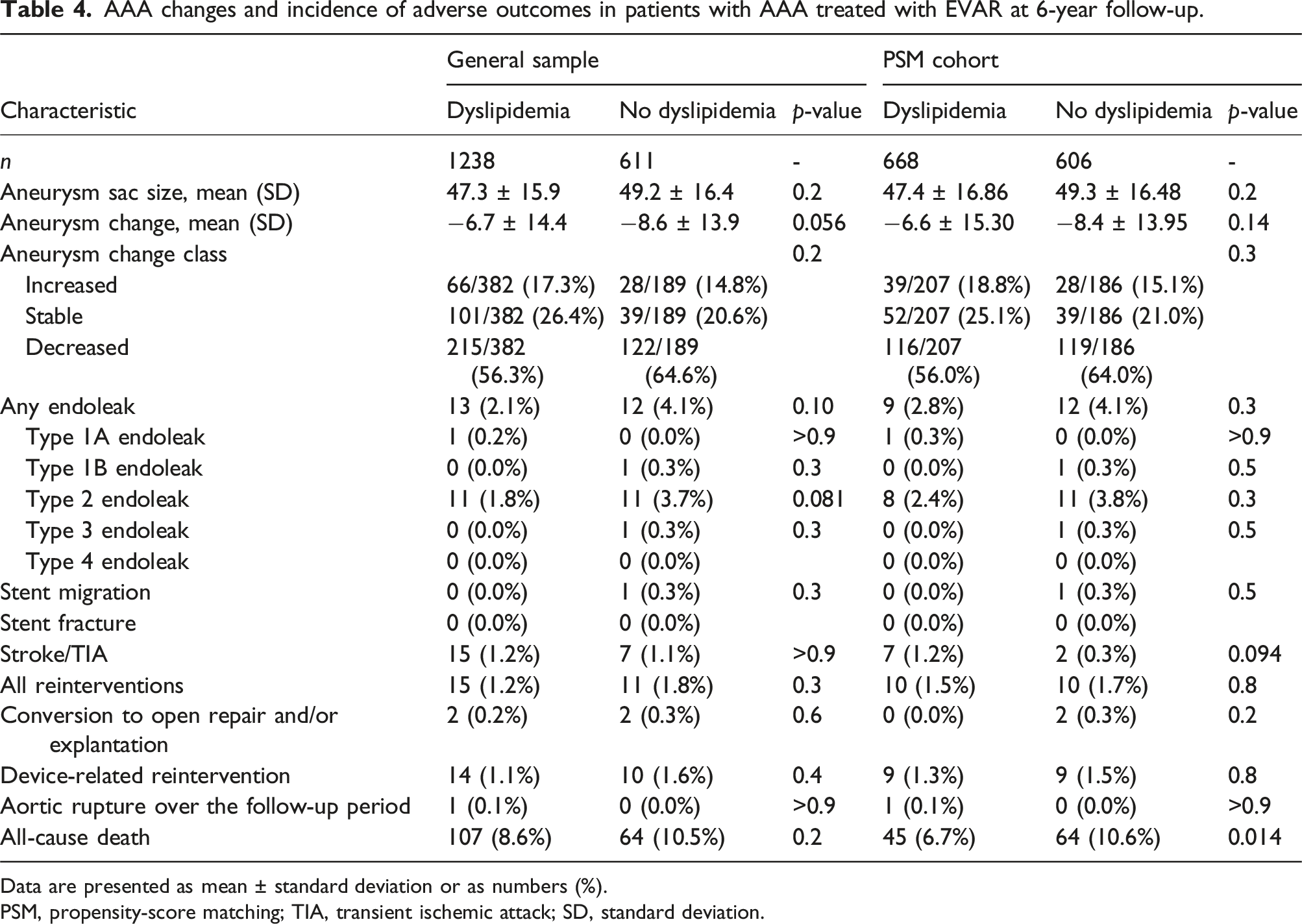
Data are presented as mean ± standard deviation or as numbers (%).
PSM, propensity-score matching; TIA, transient ischemic attack; SD, standard deviation.
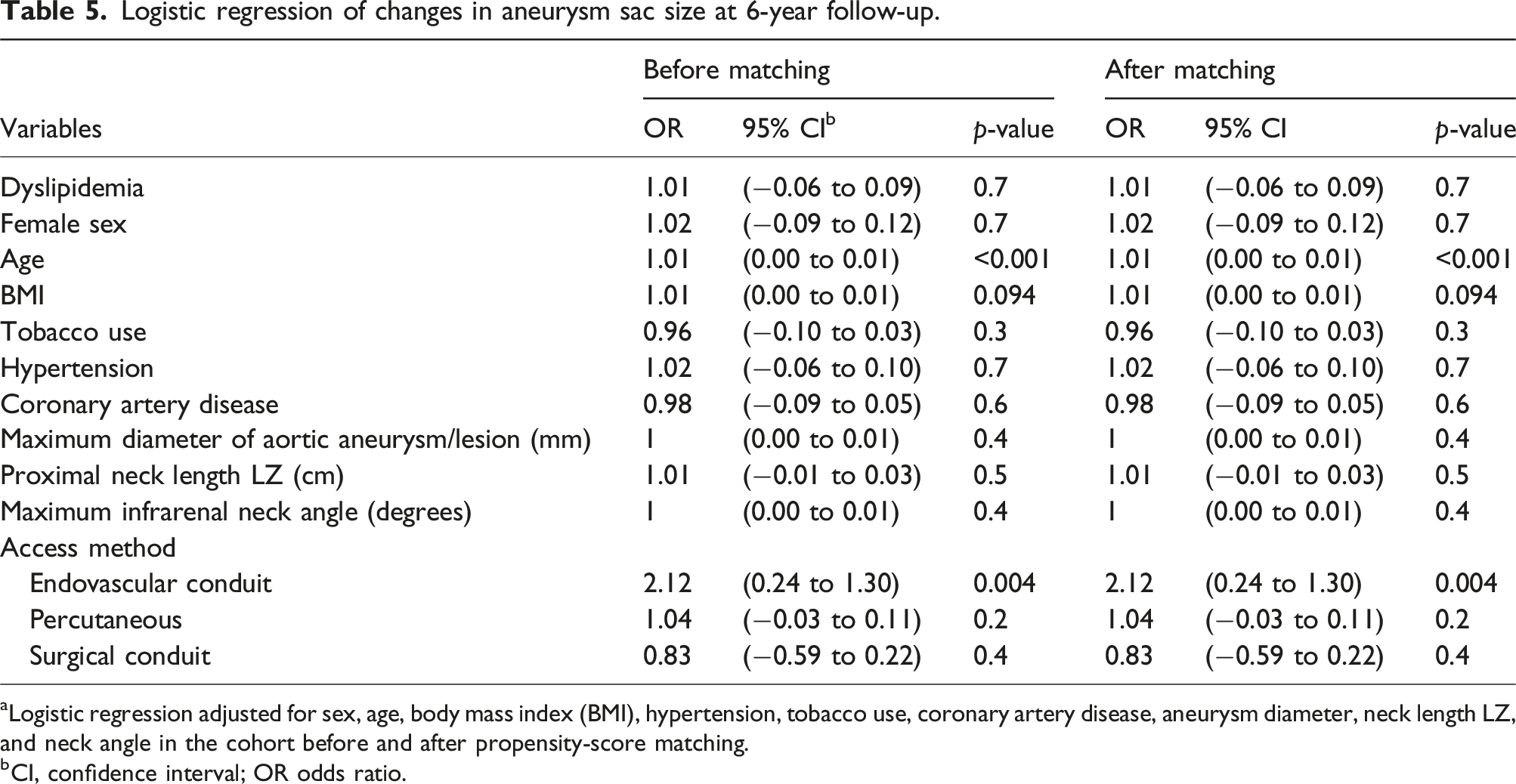
The dyslipidemia and non-dyslipidemia groups showed that a larger baseline aneurysm diameter correlated with a greater reduction in sac size over 6 years. Neither group showed significant differences in sac enlargement (Figures 3(a) and 3(b)). Logistic regression analysis revealed that older age and endovascular conduit access were associated with sac expansion (p < .001 and p = .004; see Table 5). (a) Kaplan–Meier curve for freedom from abdominal aortic aneurysm increases in the full population. (b) Kaplan–Meier curve for freedom from abdominal aortic aneurysm increases in the matched population. Logistic regression of changes in aneurysm sac size at 6-year follow-up. aLogistic regression adjusted for sex, age, body mass index (BMI), hypertension, tobacco use, coronary artery disease, aneurysm diameter, neck length LZ, and neck angle in the cohort before and after propensity-score matching. bCI, confidence interval; OR odds ratio.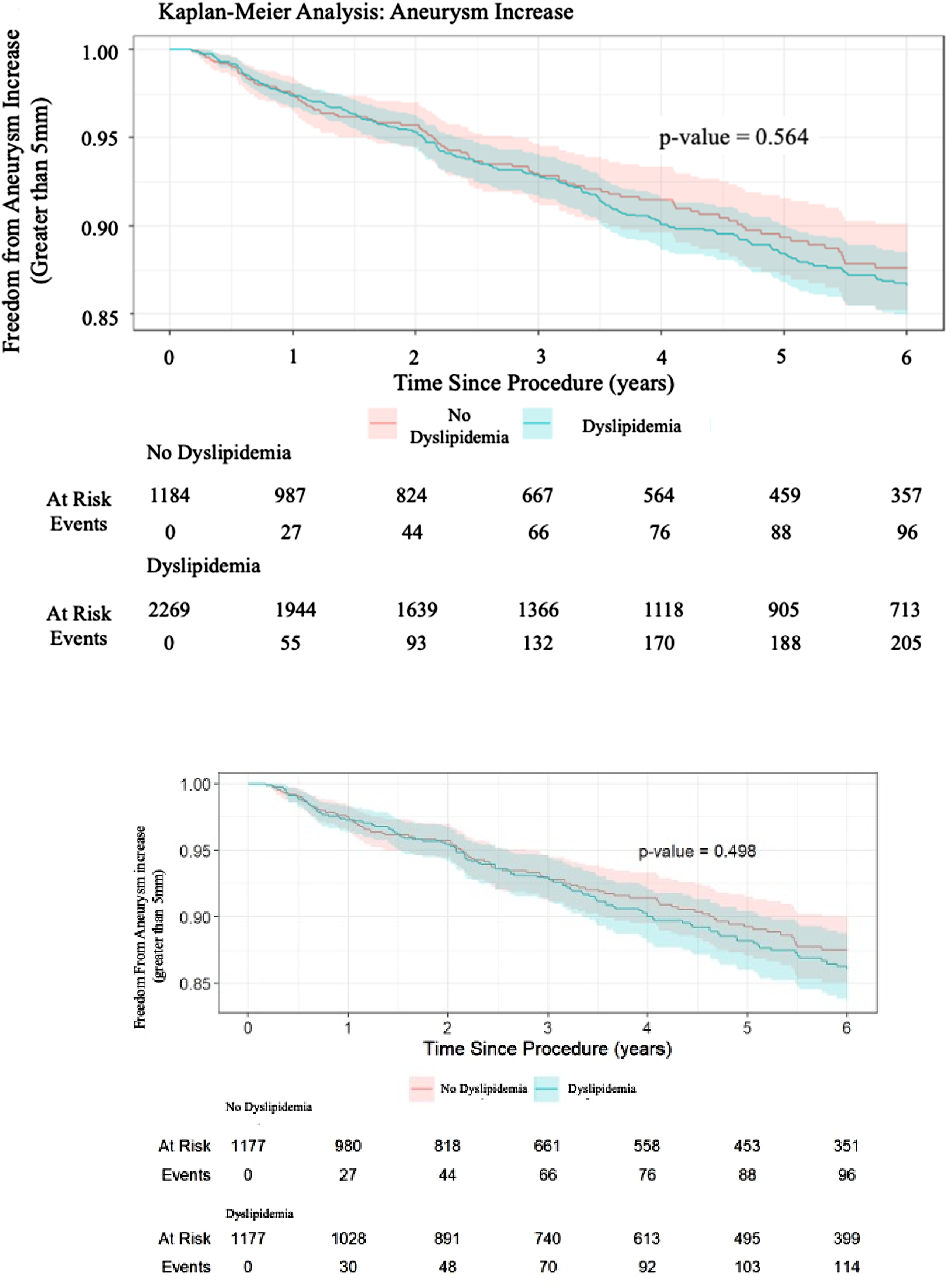
Secondary outcomes
Cohort follow-up.
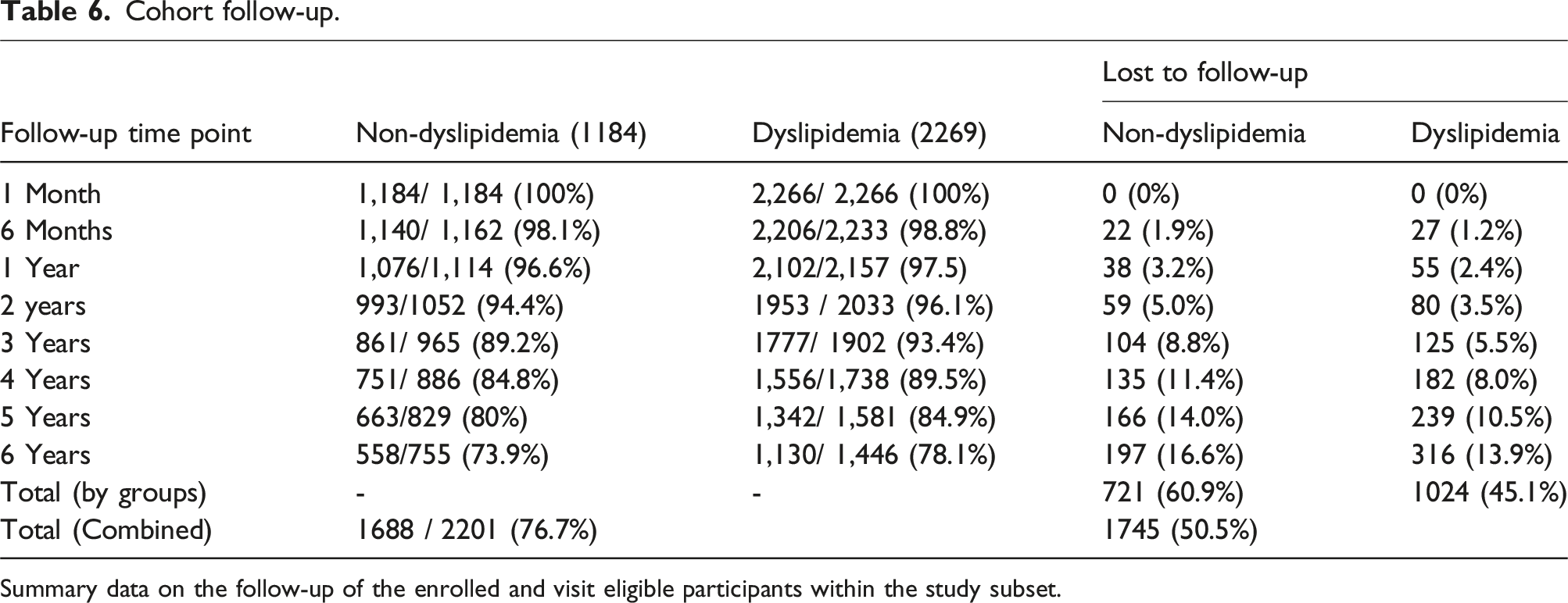
Summary data on the follow-up of the enrolled and visit eligible participants within the study subset.
Propensity score matched cohort analysis
PSM resulted in 1177 patients in each group. The dyslipidemia group had a higher BMI and a greater prevalence of comorbidities, including hypertension, TIA, carotid disease, history of coronary artery bypass surgery, congestive heart failure, and peripheral arterial disease (Table 1).
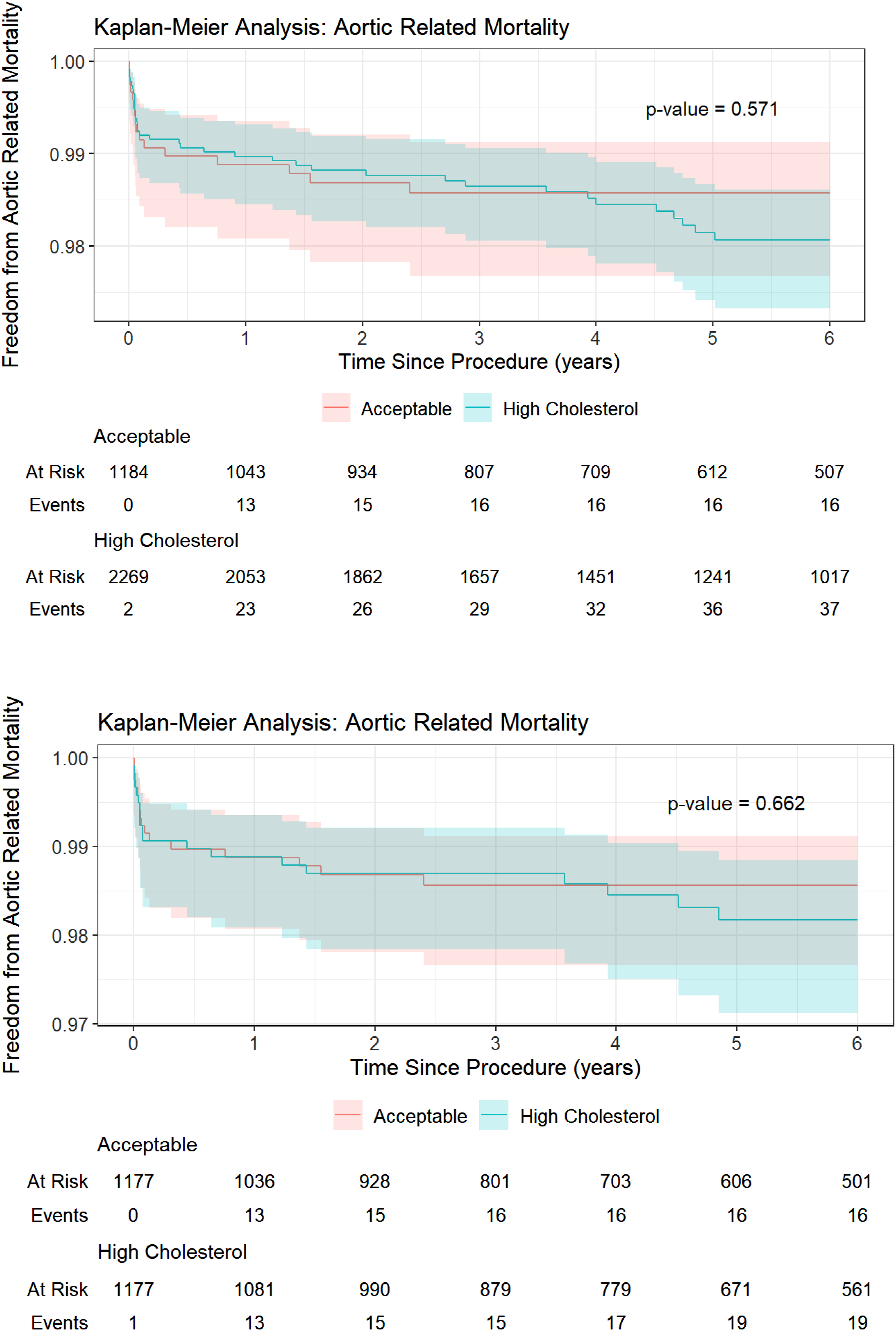
Patients without dyslipidemia were more likely to undergo off-label stent grafting (59.9% vs 55.5%, p = .03) and have a higher incidence of aneurysm necks measuring <15 mm (6.3% vs 4.3%, p = .035) (Table 2). The differences in the endoleak or aortic rupture rates were not significant. The only significant difference was in all-cause mortality rate between the matched (p = .018) and non-matched populations (p = .413) (Figures 4(a) and (b)). Our analysis revealed no significant differences in aortic-related mortality between the two populations (Figure 5(a) and (b)). Specifically, the matched population had a p-value of 0.662, whereas the non-matched population had a p-value of 0.571 (Figure 5(a) and (b)). Additionally, there were no notable differences in the changes in aneurysm size over the 6-year period between the non-dyslipidemia and dyslipidemia groups. In the non-matched population, the non-dyslipidemia group experienced an average change of −8.6 ± 13.94 mm, whereas the dyslipidemia group showed a change of −6.7 ± 14.42 mm (p = .056). In the matched population, the non-dyslipidemia group had an average change of −8.4 ± 13.95 mm compared with −6.6 ± 15.30 mm in the dyslipidemia group (p = .14). (a) Kaplan–Meier analysis of all-cause mortality in the entire population. (b) Kaplan–Meier analysis of all-cause mortality in the matched population. (a) Kaplan–Meier analysis of aortic-related mortality in the entire population. (b) Kaplan–Meier analysis of aortic-related mortality in the matched population.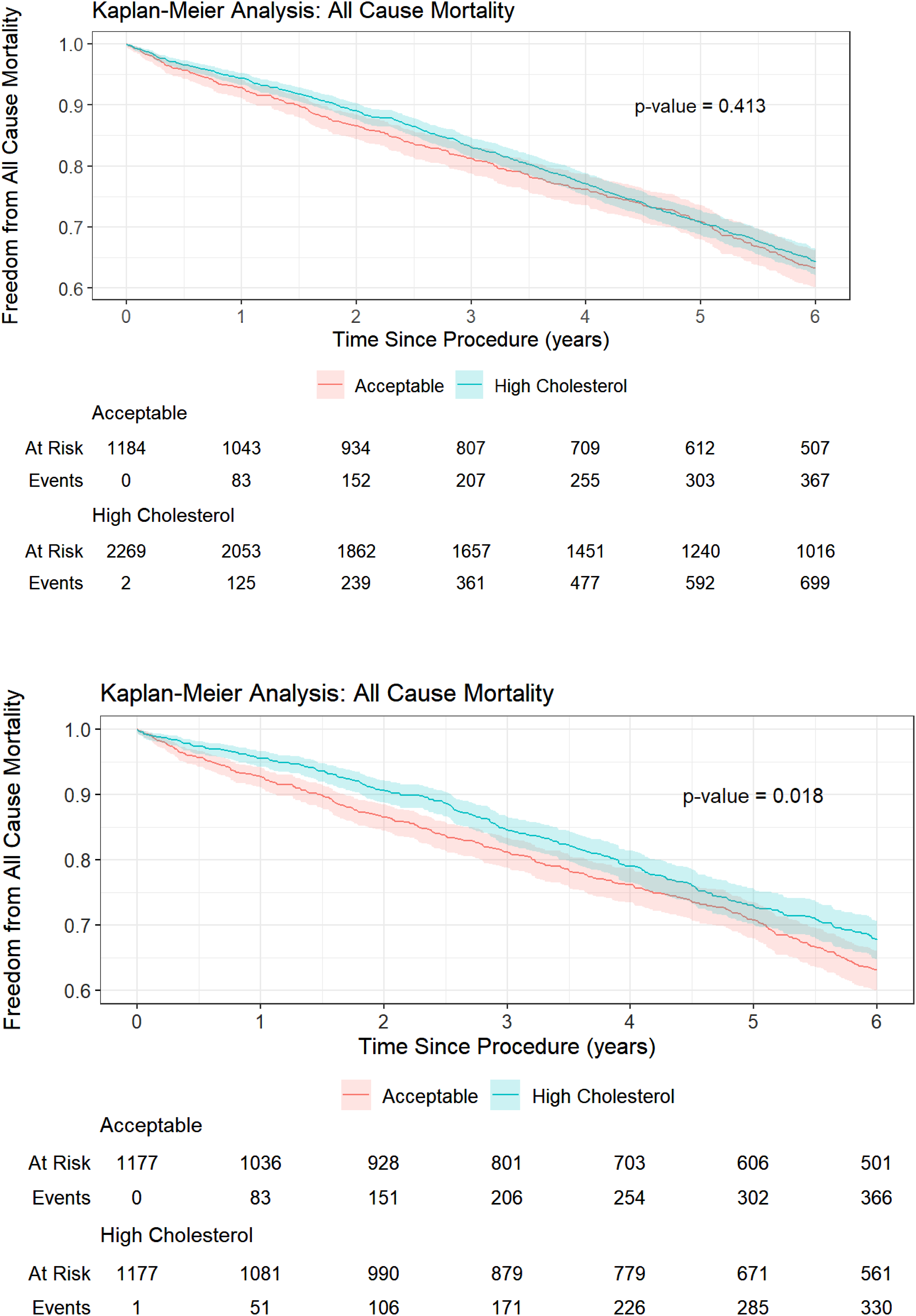
Discussion
This study found no significant effect of dyslipidemia on sac regression 6 years after EVAR with the GORE® EXCLUDER® device. This finding contrasts with that of Lalys et al., who suggested that dyslipidemia might contribute positively to sac regression after EVAR. 8 Their systematic review utilized multiple studies with different devices, of which 11.9% used the GORE® EXCLUDER® devices, and found that dyslipidemia was associated with higher rates of sac regression. However, studies examining this effect have not clearly defined the type or degree of dyslipidemia,17–19 and device-specific outcomes have not been reported. 8 A previous study showed that the GORE® EXCLUDER® device is less likely to result in sac regression post-EVAR without an increased risk of mortality, reintervention, or aneurysm-related complication.9,10 However, these studies were dated, and updates in the device and delivery system were made by the manufacturer. The device-specific effects of dyslipidemia on sac regression were unclear in this study.
The role of dyslipidemia in sac regression post-EVAR may be linked to dyslipidemia or statin therapy. 8 Statin use has been strongly associated with sac regression,18,20 likely because of statin-induced reduction in matrix metalloproteinases (MMPs), particularly MMP-2 and MMP-9, which degrade collagen and elastin in vessel walls. 18 Elevated MMP-9 levels are also associated with a higher incidence of endoleaks, suggesting that statin therapy may reduce endoleak rates and contribute to sac regression.21,22 Paradoxically, dyslipidemia may promote sac regression 8 owing to its association with a prothrombotic state,23–25 which may be protective against endoleaks. However, the present study did not find any differences in endoleak or sac regression rates between the groups, thus failing to support either theory. The rate of radiologically undetected endoleaks in the current literature is unclear. 26
In this study, among patients with dyslipidemia, a larger aneurysm size was associated with sac expansion at 1 year, although this effect did not persist. This finding is consistent with that of a previous study that showed that an aneurysm size of ≥60 mm is an independent predictor of sac expansion 6 months post-EVAR, although the proportion of patients with dyslipidemia is unknown. 27 Similarly, increasing age was associated with sac expansion 6 years post-EVAR among patients with dyslipidemia, consistent with a previous study that recognized age as an independent risk factor for sac expansion. 5 These findings suggest an additive effect of dyslipidemia on sac expansion, which may be an important consideration for clinicians when assessing patients with dyslipidemia to determine the suitability of EVAR.
One study found that violations of neck angulation instructions were associated with increased all-cause mortality, whereas neck length violations were not.28,29 The impact of off-label treatment on mortality remains uncertain. Additionally, statins and other medications may have reduced the incidence of adverse events in the dyslipidemia group.30–33 The higher off-label treatment rates in the non-dyslipidemia group may reflect a more conservative surgical approach for patients with multiple comorbidities. Furthermore, we have observed in the early stages, follow-up rates exceeded 95% in both groups; MEDICAL HISTORY Notably, patients without dyslipidemia experienced a significantly higher cumulative loss to follow-up at 60.9%, in contrast to 45.1% for those with dyslipidemia at 6 years follow-up. This finding suggests that individuals with established cardiovascular risk profiles may exhibit greater long-term engagement. The disparity in loss to follow-up could be attributed to more frequent clinical interactions or an increased perception of risk among those with dyslipidemia. By the end of the 6 years, loss to follow-up rose to 16.6% in the no-dyslipidemia group and 13.9% in the dyslipidemia group, raising concerns about potential surveillance bias.
Limitations
This study had several limitations. First, retrospective analysis of observational data is inherently associated with a higher risk of bias. Nevertheless, the GREAT registry upholds a high standard of data quality supported by a robust monitoring schedule and methodologies that effectively integrate real-world conditions into clinical studies. Second, the classification of dyslipidemia relied on hospital coding systems rather than analytical values, which may limit the precision of the findings. Additionally, the impact of anatomical factors such as neck length, angulation, and thrombus burden on sac behavior over time has not been fully explored. A significant limitation of this study was the lack of data on lipid-lowering therapies and other cardiovascular medications. Given the recognized effects of statin therapy on sac regression, this lack of information raises concerns about the potential for type 1 and 2 errors. Furthermore, only 61% of the patients underwent follow-up imaging within 1 year, risking underreporting complications. Long-term data on the effect of dyslipidemia on sac regression are valuable for guiding clinical decision making in the management of AAA. Finally, although propensity score matching aims to minimize baseline differences between groups, some residual confounding may persist, particularly regarding anatomical and procedural characteristics that are not fully captured by clinical covariates. In the matched cohort, the prevalence of proximal neck lengths less than 15 mm was higher in patients with dyslipidemia (6.3% vs 4.3%; p = .035), while off-label Instructions for Use (off-IFU) devices were used more frequently by those without dyslipidemia (59.9% vs 55.5%; p = .030). Despite these statistically significant differences, their magnitude was modest and unlikely to bias primary outcome comparisons. These residual imbalances underscore the limitations of propensity-based approaches in contexts where anatomical complexity and operator discretion influence device selection and procedural strategies, which should be considered when evaluating the generalizability and internal validity of the findings.
Conclusion
In this comprehensive real-world study of patients who underwent EVAR with the GORE® EXCLUDER® device, the presence of dyslipidemia was not associated with higher rates of endoleaks over 6 years. Interestingly, patients with dyslipidemia showed better adherence to long-term follow-up, likely reflecting a higher level of clinical engagement due to the additional challenges posed by their comorbidities. The overall incidence of endoleaks remained low, possibly due to the predominant use of duplex ultrasound and strict coding standards. These findings suggest that while dyslipidemia is recognized as a cardiovascular risk factor, it does not negatively affect EVAR surveillance outcomes and may even indicate improved long-term care outcomes. Future studies that incorporate biochemical data and standardized imaging protocols could provide further insights into these associations.
Footnotes
Acknowledgments
The authors acknowledge the important contributions of all GREAT registry centers and study personnel, the support of GORE in organizing the registry, and all statistical expertise. The authors would like to thank all the statisticians, particularly Dr Derek Sonderegger, who was involved in the data analysis.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from W. L. Gore & Associates, Inc., Medical Products Division, located in Flagstaff, Arizona, USA. Each research centre involved in the registry was responsible for collecting data. The steering committee, along with the contract research organisation (CRO) team, reviewed the collected data, which the sponsor subsequently analysed.
Ethical approval
This paper utilizes data from the Global Registry for Endovascular Aortic Treatment (GREAT). The study protocol was approved by the institutional review board of each center, and all patients provided informed consent.
Consent to participate
All patients participating at each center provided informed consent.
Data Availability Statement
Datasets and analyses generated during the study are available upon request.
