Abstract
Objectives
We present two clinical cases of association between symptomatic free-floating thrombus (FFT) in thoracic aorta and rheumatoid arthritis (RA).
Methods
In the first patient, we observed a recent onset of peripheral and visceral signs of embolization: after a first treatment with anticoagulation, our Aortic team scheduled the coverage of FFT (sited in zone 1 of the aortic arch) with an anatomical debranching of anonymous trunk and left carotid artery, a left carotid-subclavian bypass, and a TEVAR of the aortic arch with proximal landing in zone 0 of the arch. The second case was characterized by chest pain, left upper limb ischemia, and CTA evidence of an FFT in zone 3 of the aortic arch; we planned a chimney-TEVAR on the left subclavian artery and descending thoracic aorta (with proximal landing in zone 2 of the aortic arch) to exclude the FFT.
Results
No complications resulted and no new embolic episodes were registered.
Conclusions
Evaluating the aorta is warranted in all patients with peripheral emboli of uncertain pathogenesis. In our opinion, the endovascular treatment of a symptomatic FFT could represent an effective and safe solution in a patient fit for endovascular surgery, but larger studies are required to define a personalized treatment strategy.
Objectives
Rheumatoid arthritis (RA) patients have an increased risk of cardiovascular disease, estimated to be as high as in diabetes mellitus. 9 A recent study about arterial wall inflammation in RA observed the thoracic aorta would have different response to anti-inflammatory drugs than the other arterial segments. 10 In this paper, we will report two cases of symptomatic free-floating thrombus (FFT) in thoracic aorta in RA patients.
Methods and results
First patient
A 45-year-old woman was transferred from a Rheumatology Clinic to our Unit due to distal left foot ulcers of recent onset in a thoracic aortic thrombosis. Use of corticosteroids, RA, Takayasu’s arteritis, hypertension, dyslipidemia, sarcopenia, steroid-induced osteoporosis, smoking habit, and recent SARS-CoV-2 infection were present in her medical history. Blood analysis showed a hyperleukocytosis (25’730/μL) and an elevated value of C-reactive protein (29.7 mg/dL). In addition, she showed a high level of D-dimer (1387 ng/mL), high level of activated partial thromboplastin time (ratio 1.62,) and a low level of anti-thrombin activity (41%), low level of C-protein activity (53%) and low level of Factor-VII activity (24.7%). A hematological consultation defined this alteration of some blood coagulation parameters as possibly due to only heparin inference (the patient was treated with intra-venous heparin for two months). At the clinical examination, the left leg presented erythema on the forefoot and gangrene of the second and third toes. Ultrasound-Color-Doppler examination showed a total occlusion of the distal popliteal artery and the tibial-peroneal trunk. Contrast-enhanced computed tomography angiography (CTA) showed a partial thrombosis in a “bovine” aortic arch (starting from less than 1 cm from common ostium of anonymous trunk and left carotid artery) with an FFT and signs of multi-embolism in both kidneys and in the spleen (Figure 1). A left leg popliteal–tibial thrombectomy was performed. After our Aortic team discussed the treatment of the FFT, a left carotid–subclavian bypass and a TEVAR of the aortic arch with proximal landing in zone 0 of the aortic arch (Bolton Medical Relay NBS Plus 32-32-160 and Relay Pro 30-26-160) were scheduled (Figure 1). After intervention, the patient was transferred in the Cardiovascular Intensive Care Unit for 48 hours and then moved back to our Unit; we administrated double anti-aggregation therapy, and she was discharged on post-operative day (POD) 13 without any complications and she was referred to the outpatient Rheumatology Unit of our Hospital. At one-year-follow-up, she underwent a CTA scan that showed good patency of the grafts and total exclusion of the FFT. 1. CTA shows an FFT of aortic arch and descending thoracic aorta. 2. Surgical preparation of supra-aortic trunks. 3. Hybrid reparation type I of the aortic arch: debranching of supra-aortic trunks. 4. Hybrid reparation type I of the aortic arch: final angiogram after TEVAR.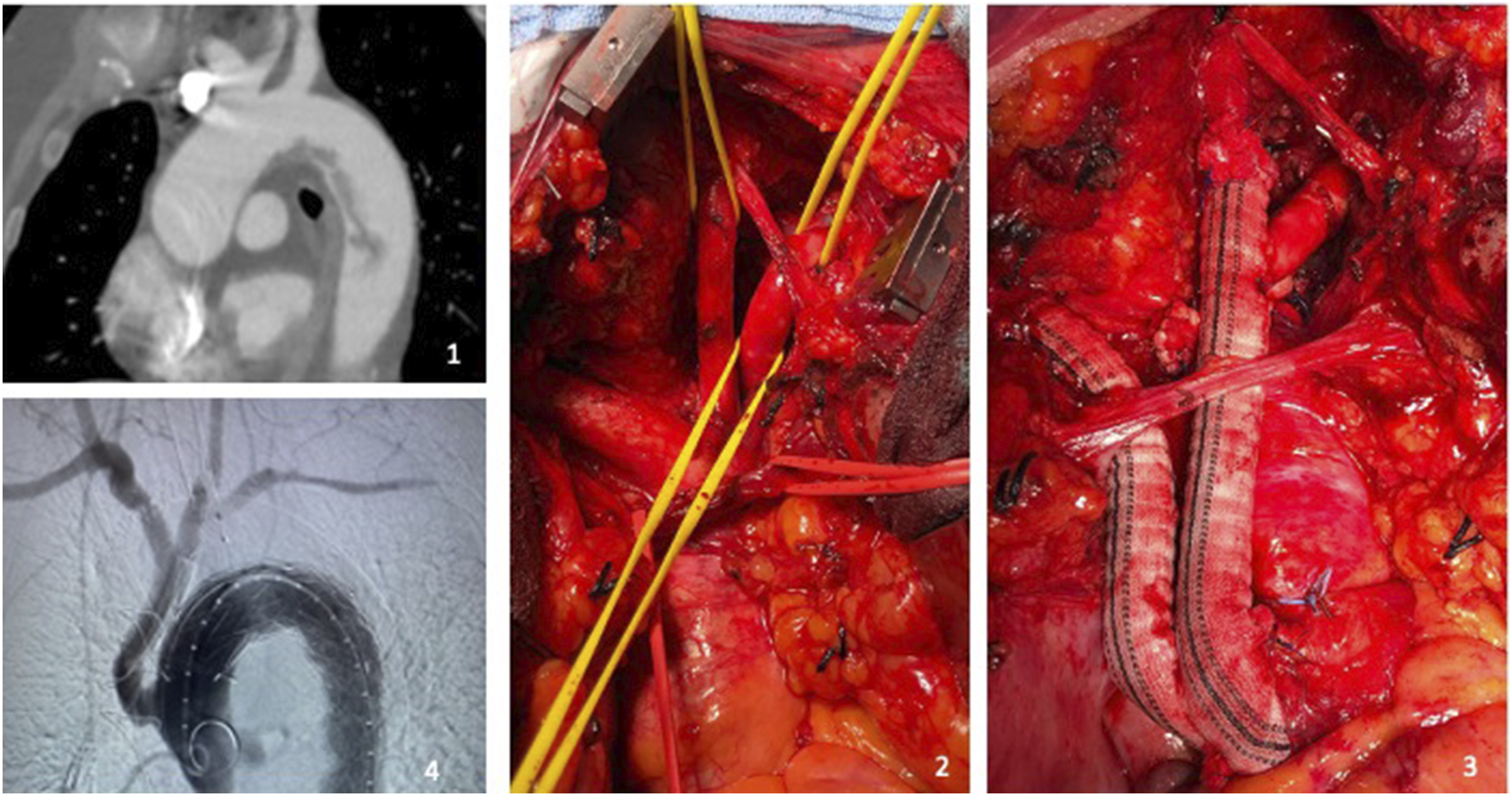
Second patient
A 75-year-old man was admitted to our Emergency Department for acute chest pain and left arm weakness. Urgent blood analysis showed an elevated C-reactive protein level (27.3 mg/dL) and D-dimer level (2241 ng/mL) and a mild low hemoglobin rate (13.9 g/dL). In his medical history, he reported rheumatoid arthritis (in treatment with oral corticosteroids), previous skin cancer, and a former smoking habit. Ultrasound color Doppler examination showed an occlusion of the left axillary artery greater than 5 cm and no-flow in the brachial artery. A CTA revealed a focal dissection of the isthmic thoracic aorta, very close to the subclavian ostium (less than 1 cm), with an FFT (Figure 2). We scheduled an urgent thrombectomy of the upper left limb and a subclavian chimney-TEVAR with a landing in zone 3 of the aortic arch (VBX, W.L. Gore & Associates Inc, BXA083902E and Medtronic International Trading Sarl, Valiant VAMF3434C100). We positioned the aortic stent graft and the proximal stent markers at the same line, 5 mm in advance from the subclavian ostium. At the final angiogram, we observed the patency of left carotid artery, a total exclusion of floating thrombus, and a good patency of the aortic graft and the subclavian graft, without signs of endoleak (Figure 2). After the operation, the patient was moved to our Unit: no complications were observed. He was discharged to home on POD 5, with a double anti-aggregation therapy, and he was referred to the outpatient Rheumatology Unit. 1-2. CTA shows a semilunar shape floating thrombus attached on the thoracic isthmic aortic wall and a mobile dissecting flap. 3. Intraoperative angiogram: the chimney-TEVAR is realized to exclude the FFT.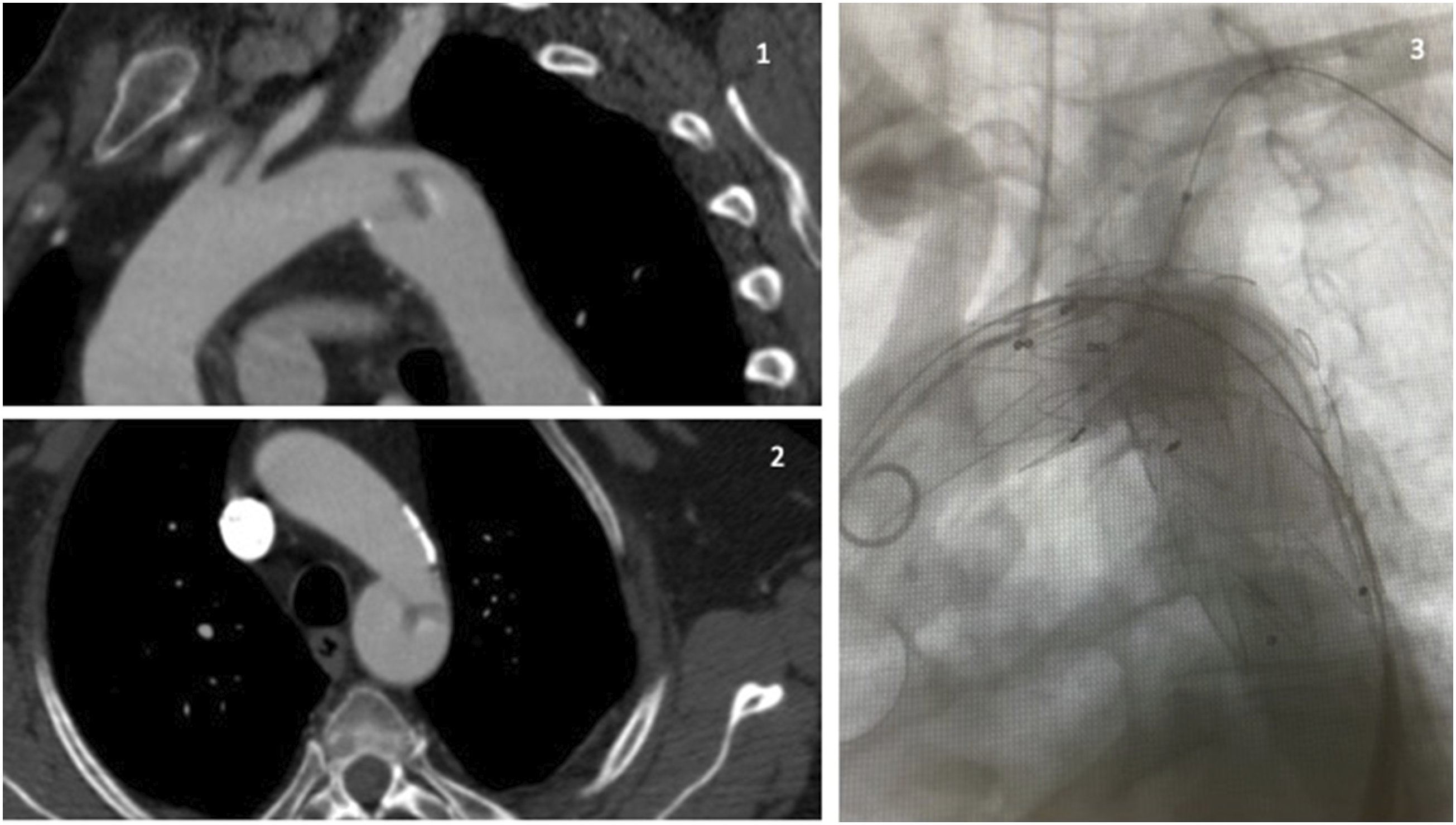
Discussion
Patients with rheumatoid arthritis have an increased risk of cardiovascular disease compared to the general population. 2 Interestingly, a recent study on RA showed a significant reduction of inflammation in the aortic wall when treated with anti-inflammatory drugs, but not in the thoracic district. The reason appears uncertain. 3 Many studies have revealed that steroids can damage the endothelium directly and cause thickening of the intima. 1 It is accepted that atherosclerosis is a chronic inflammatory disease like RA and the inflammation associated with rheumatoid arthritis increases the incidence of vascular insults and the progression of atherosclerosis. 1 Glucocorticoids can effectively suppress RA-related inflammation. 1 However, there are numerous adverse cardiovascular effects associated with their use, including stroke, myocardial infarction, and heart failure; the adverse effects on lipid metabolism may account in part for their detrimental cardiovascular effects. 1 In addition, the SARS-CoV-2 4 infection was present in the recent history of the first patient: this condition represents an additional risk factor 1 in the first patient.
Many reports5,6 indicate anticoagulation as a successful strategy to solve the thrombus, but in our first case the conservative treatment was not effective; in the literature,1,6 this clinical situation appears as an indication for intervention. TEVAR appears as the most used endovascular strategy to exclude an FFT, 6 while the open surgery is often used as the last resort when other less invasive approaches fail or in cases of diagnostic uncertainty when a concomitant surgical pathology cannot be ruled out. 6 The management of the second patient was influenced by the chest pain episode and acute left arm ischemia. In an endovascular strategy, a complete entrapment of the thrombus between the stent graft and the aortic wall is fundamental. 5
Conclusions
Larger studies are required to investigate a possible relationship between rheumatoid arthritis, aortic wall inflammation, and thoracic floating thrombus. The procoagulant nature of RA is correlated with thrombosis and should be considered in patients with cardiovascular events. Evaluating the aorta is warranted in all patients with peripheral emboli of uncertain pathogenesis. We presented two endovascular strategies for a symptomatic FFT. In both cases, these strategies were safe and effective.7–9
Footnotes
Acknowledgments
We would like to thank Chiara Di Giorgio (Fondazione IRCCS Casa Sollievo della Sofferenza) for editing and reviewing this manuscript for English language.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
