Abstract
Objective:
The aim was to evaluate early and medium-term outcomes of double fenestrated physician-modified endovascular grafts for total endovascular aortic arch repair.
Methods:
This single-center retrospective analysis of prospectively-collected data included 100 patients, from January 2017 to December 2021, undergoing thoracic endovascular aortic repair (TEVAR) for zone 0. The fenestrations were a proximal larger fenestration that incorporated the brach2iocephalic trunk and left common carotid artery and a distal smaller fenestration for the left subclavian artery (LSA). Only the LSA fenestration was stented.
Results:
The median duration for stent-graft modification was 23±6 minutes. Of the 100 patients, 70 were men. The mean patient age was 70±10.5 years. Indications for treatment included degenerative aortic arch aneurysm (n=32), dissecting aortic arch aneurysm after type A dissections (n=23) and (n=19) after type B dissections, acute complicated type B dissection (n=16), and other pathologies (n=10). Technical success rate was 97%. The 30 day mortality was 2% (n=2). Four patients (4%) had minor stroke with full recovery. One patient (1%) had a type IA endoleak, 1 patient (1%) had a type IB endoleak, and 2 patients (2%) have a type II endoleak from the LSA. Eight patients (8%) required reintervention: 1 type IA endoleak, 1 type IB endoleak, 1 retrograde type A dissection, and 5 because of access-related complications. During a mean follow-up of 24±7.2 months, there were no aortic rupture, paraplegia, and all supra-aortic trunks were patent.
Conclusions:
Double homemade fenestrated TEVAR is both feasible and effective for total endovascular aortic arch repair avoiding the need for anatomical and extra-anatomical surgical revascularization. The long-term durability will need to be assessed in studies with long-term follow-up.
Clinical Impact
Double homemade fenestrated TEVAR is effective for total endovascular aortic arch repair avoiding the need for anatomical and extra-anatomical surgical revascularization. The standout feature of this double fenestrated device is its simple handling during operation with the proximal fenestrations being directed to the orifices of the BT and LCCA automatically when the LSA fenestration is catheterized and secured by covered stent placement. The deployment algorithm actively steers the operator away from superfluous manipulations of the device within the arch and avoids guidewire manipulation in carotid arteries. The long-term durability will need to be assessed in studies with long-term follow-up.
Introduction
Open surgery of the aortic arch represents the gold standard for pathologies of the aortic arch. Open aortic arch surgery continues to become increasingly common with data from the Society of Thoracic Surgeons Adult Cardiac Surgery Database (STS-ACSD) demonstrating a nearly 25% increase in case volumes from 2011 to 2014. 1 Despite increasing operator experience, however, mortality and stroke, rates of 12% and 8% respectively, in that study were not significantly improved from an earlier STS-ACSD report spanning the time interval 2004 to 2009. 2 Therefore, there remains significant apprehension over subjecting a patient to these procedures. This is particularly so for patients who are elderly, requiring emergency repair or with major comorbidities. 3
Total arch thoracic endovascular aortic repair (TEVAR) can be accomplished using branched endografts or fenestrated endografts.4,5 The evolution of stent-graft technology recently has meant the development of custom-made branched stent-grafts to achieve total endovascular aortic arch repair. The disadvantages of custom-made include manufacture and deliver times for urgent cases and the associated high costs. Unfortunately, there is also a high rate of embolization associated with this approach probably related to the complexity of deploying a multibranched unibody stent-graft.
A further alternative is physician-modified endografts (PMEGs) for zone 0 TEVAR involving the deployment of a conventional stent-graft device ex vivo, fashioning of customized fenestrations and re-constrainment into the delivery system. The authors have previously reported their early experience in 50 patients unfit for open repair, needing rapid treatment, with good short-term results. 6 The objectives of this study were to evaluate the medium-term results for the current series of 100 patients treated with PMEGs for total endovascular aortic repair.
Methods
Patients
Protocol and informed consent were approved by the Institutional Review Board. All patients gave written consent, and the local authorities approved the study. From January 2017 to December 2021, all patients presenting with aortic arch lesions deemed either unfit for open thoracic surgery or in need of emergent repair, and with suitable anatomy for a double fenestrated PMEG were enrolled and treated in 1 tertiary referral center (Arnaud de Villeneuve Hospital, Montpellier). During the same study period, there were 142 open surgical procedures of the aortic arch, of which 9 were frozen elephant trunks.
Multidisciplinary teams, including cardiovascular surgeon, were involved in the decision-making. Patients with zone 2 and zone 1 aortic arch lesions or zone 0 saccular aneurysms at the lesser curvature of the arch were suitable with full inclusion criteria detailed in Table 1. Zone 0 aortic arch lesions, except saccular aneurysms at the lesser curvature of the arch, were an anatomical exclusion criterion. Demographic, anatomic, intraoperative, and postoperative data were prospectively recorded.
Inclusion and Exclusion Criteria Used for Double Homemade Fenestrated Physician-Modified Endovascular Grafts for Total Endovascular Aortic Arch Repair.

Planning, Sizing, and Device Preparation
The Valiant Captivia stent-graft (Medtronic, Santa Rosa, California) was used for all PMEGs. The technical aspects of device modification and fenestration have already been reported in detail. 7 A brief summary follows which incorporates the most recent modification, the incorporation of a preloaded guidewire. A vascular imaging workstation was used to accurately define and measure the patient’s anatomy from computed tomographic angiography images. An appropriate stent-graft was selected to achieve seal and modifications planned. These were a proximal (nearest the leading edge) larger fenestration that incorporated the brachiocephalic trunk (BT) and left common carotid artery (LCCA) and a distal smaller fenestration for the left subclavian artery (LSA). From the early experience, it became evident that the critical intraprocedural step was successful cannulation of the distal LSA fenestration, via a guidewire introduced from the brachial artery. From January 2018, the procedure was refined to include a preloaded guidewire in the stent-graft modifications for the LSA fenestration. To achieve this, the stent-graft must be completely unsheathed on the back table. Fenestrations were fashioned. 7 Subsequently, a needle hole was made in the introducer sheath (graft cover) just distal to the section bearing the endograft through which a 260 cm 0.035 hydrophilic stiff angled guidewire was passed cephalad. The guidewire traverses the inside of the sheath and the deployed endograft before being delivered through the LSA fenestration, and continues cephalad external to the endograft (Figure 1). The endograft was re-sheathed using nylon tape and snuggers, avoiding embedding or trapping the guidewire in the folds of the graft, by keeping the guidewire external to the nylon tape and snuggers.
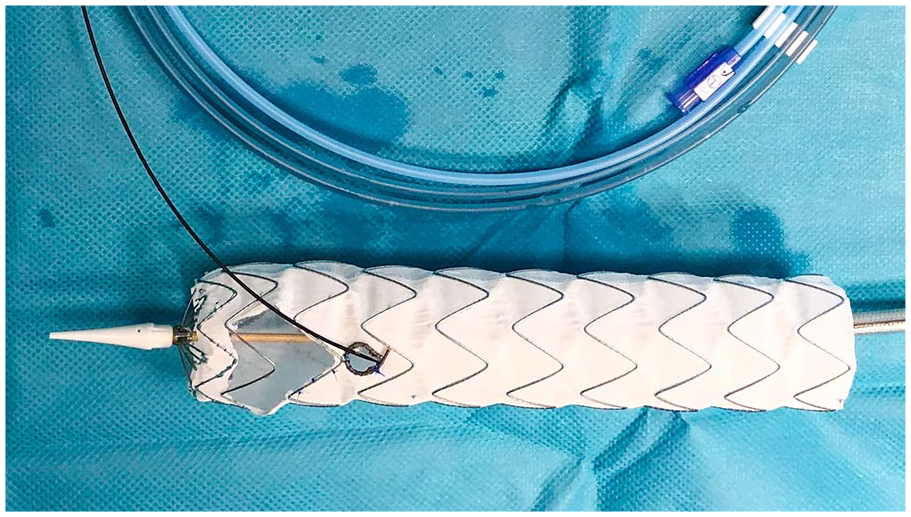
The unsheathed custom fenestrated Valiant Captivia thoracic stent-graft. The proximal large fenestration for the brachiocephalic trunk and the left common carotid artery is constructed without removing the stent-graft stent struts. The site for the distal circular fenestration for the left subclavian artery (LSA) is selected so that it is not crossed by any stent struts. A radiopaque marker is included to delineate the fenestration for the LSA under fluoroscopy. The hydrophilic guidewire is passed cephalad into the sheath trough a needle hole and advanced through the stent-graft lumen, exiting through the LSA fenestration, and continuing cephalad external to the stent-graft.
Technical Steps
All procedures were performed under general anesthesia, through either surgical exposure or percutaneous access of the common femoral arteries and left brachial artery. Systemic heparin 100 units/kg was administered after access was obtained and activated clotting time monitored. A 24F 33 cm length introducer sheath was placed retrograde through the common femoral access. A 6F 45 cm sheath introducer was placed through the retrograde left brachial access into the ostium of the LSA. A double-curved 300 cm extra-stiff 0.035 wire was positioned against the aortic valve through the femoral access. The proximal side of the preloaded guidewire was advanced through the femoral access sheath, delivered to the LSA using a 6F 25 mm snare loop and exteriorized establishing a through-and-through wire. The PMEG was advanced over the Lunderquist wire and the preloaded guidewire was progressively pulled by the second operator form the left brachial access orientating the fenestrations superiorly to face the supra-aortic trunks originating off the superior arch. The first angiographic run, perpendicular to the LSA, was performed through the left brachial sheath. Mean blood pressure is lowered to approximately 80 mmHg, deployment commenced under visualization. Rapid pacing was not used. A 9F 80 cm sheath was advanced with its dilatator through the LSA fenestration over the preloaded guidewire into the stent-graft lumen from the femoral access over the preloaded wire. An 8 to 12 mm diameter, 38 or 59 mm in length balloon-expandable covered stent (Lifestream, Bard, Tempe; Arizona) was inflated, protruding approximately 5 mm into the aortic stent-graft lumen, with the remaining length in the LSA. The covered stent was flared using a 14 mm balloon. Completion angiography was performed to verify the correct position of the PMEGs and patency of all supra-aortic vessel trunks (Figure 2). Technique of preparation and deployment for a double fenestrated PMEG for total endovascular aortic arch repair is summarized in video 1 and video 2 of the Supplemental Material.
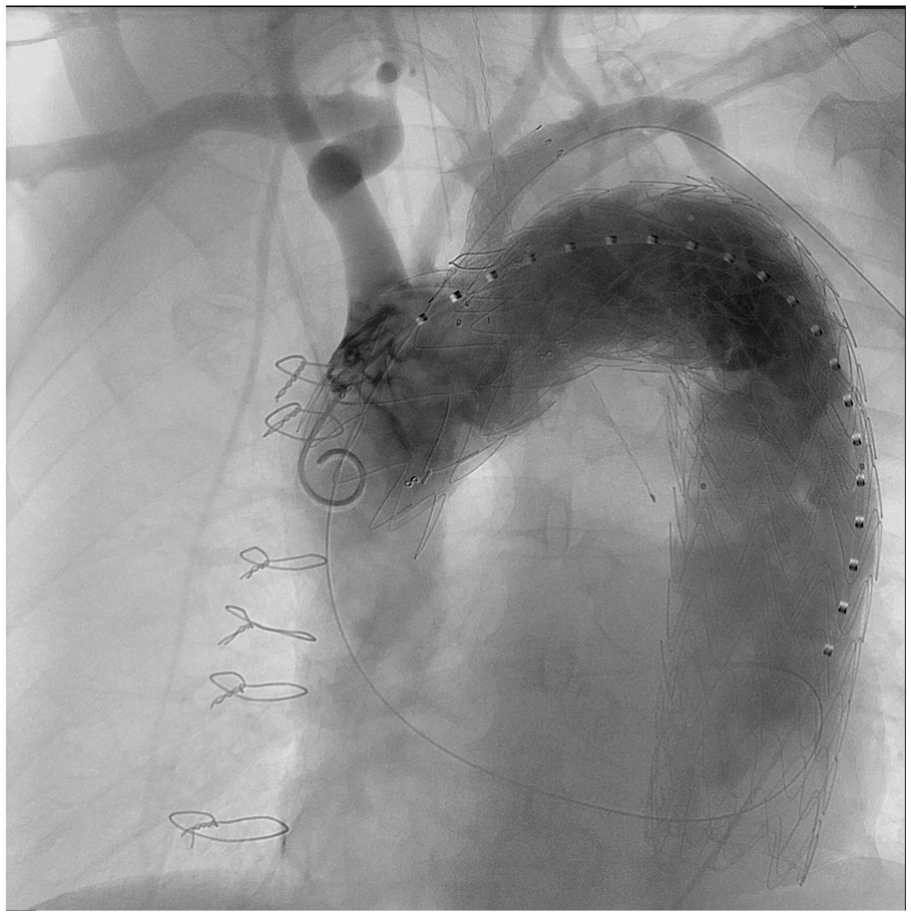
Completion angiography performed to verify the correct position of the physician-modified endovascular graft with patency of all supra-aortic vessel trunks and exclude other potential complications.
Follow-up
Follow-up surveillance was performed with serial computed tomographic scans at 1 week, then at 3, 6, and 12 months, and annually thereafter (Figure 3).
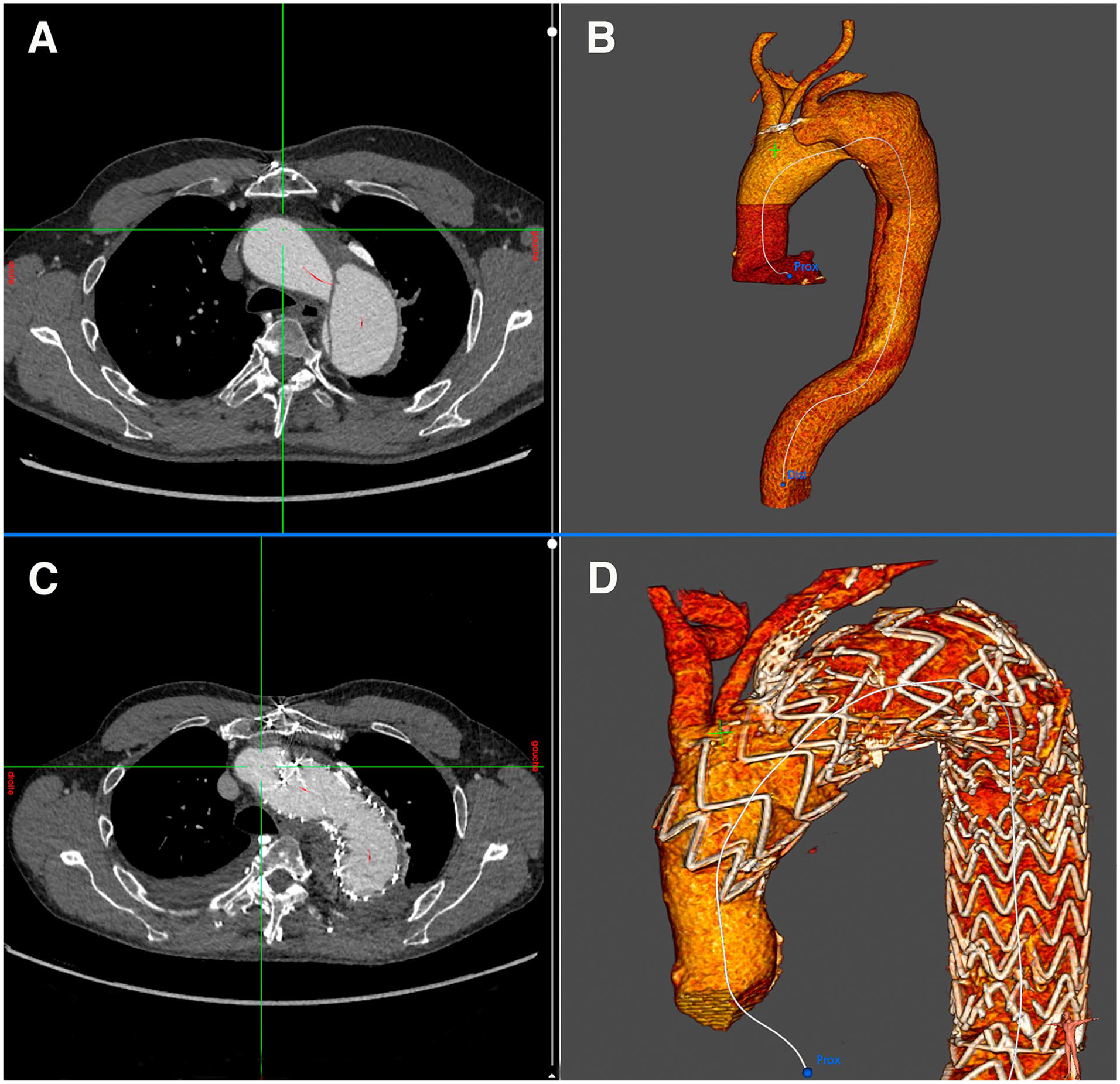
Axial computed tomographic (CT) scan demonstrating a symptomatic dissecting aortic arch aneurysm (A). Three-dimensional volume rendered reconstruction (B). Axial CT scan after double fenestrated physician-modified endovascular graft (PMEG) in zone 0 treatment in the same patient. Note the left subclavian artery covered stent (C). Three-dimensional volume rendered reconstruction demonstrating the double fenestrated PMEG and patency of the supra-aortic trunks (D).
Statistical Analysis
Categorical data are presented as frequencies; continuous variables are expressed as median and range or standard deviation. All statistical analyses were performed using IBM SPSS Statistics 24 software (version 24.0.0.0).
Results
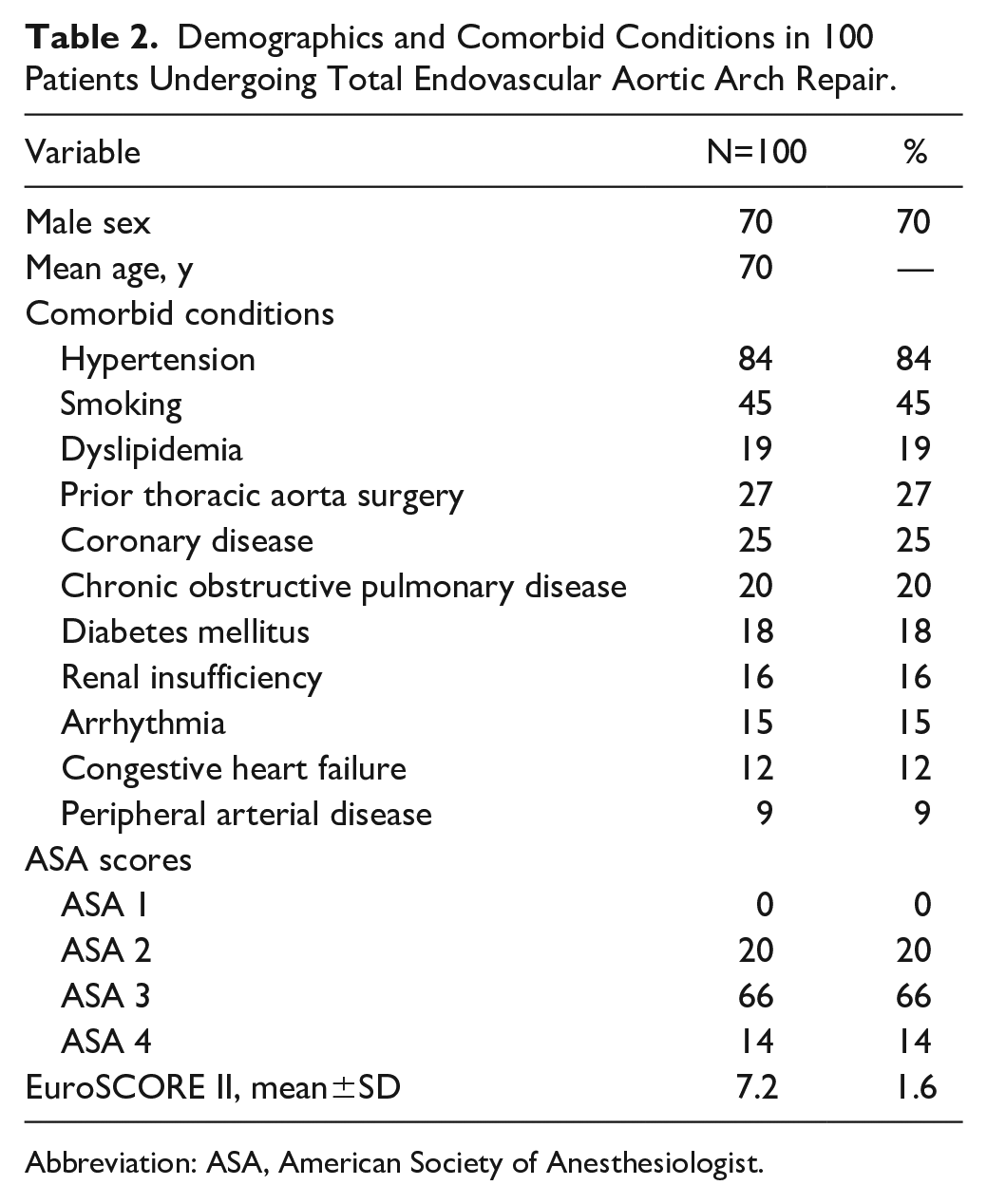
From January 2017 to December 2021, 100 patients (74 men; mean age 70±10.5 years; range, 24–90 years) had undergone total endovascular aortic arch repair. Of the 100 procedures, 74 (74%) were elective. All the elective patients were judged to be unsuitable for open surgical repair because of high surgical risk and associated probability of major complication and mortality. This cohort included patients who have not been able to tolerate aortic cross-clamping or thoracotomy. Medical comorbidities included significant chronic obstructive pulmonary disease, coronary artery disease, congestive heart failure, and prior thoracic aorta surgery. Detailed demographics and comorbid conditions are described in Table 2. Indications for total endovascular arch repair were varied and are summarized in Table 3.
Demographics and Comorbid Conditions in 100 Patients Undergoing Total Endovascular Aortic Arch Repair.
Abbreviation: ASA, American Society of Anesthesiologist.
Indications and Operative Details of 100 Patients Undergoing Total Endovascular Aortic Arch Repair.
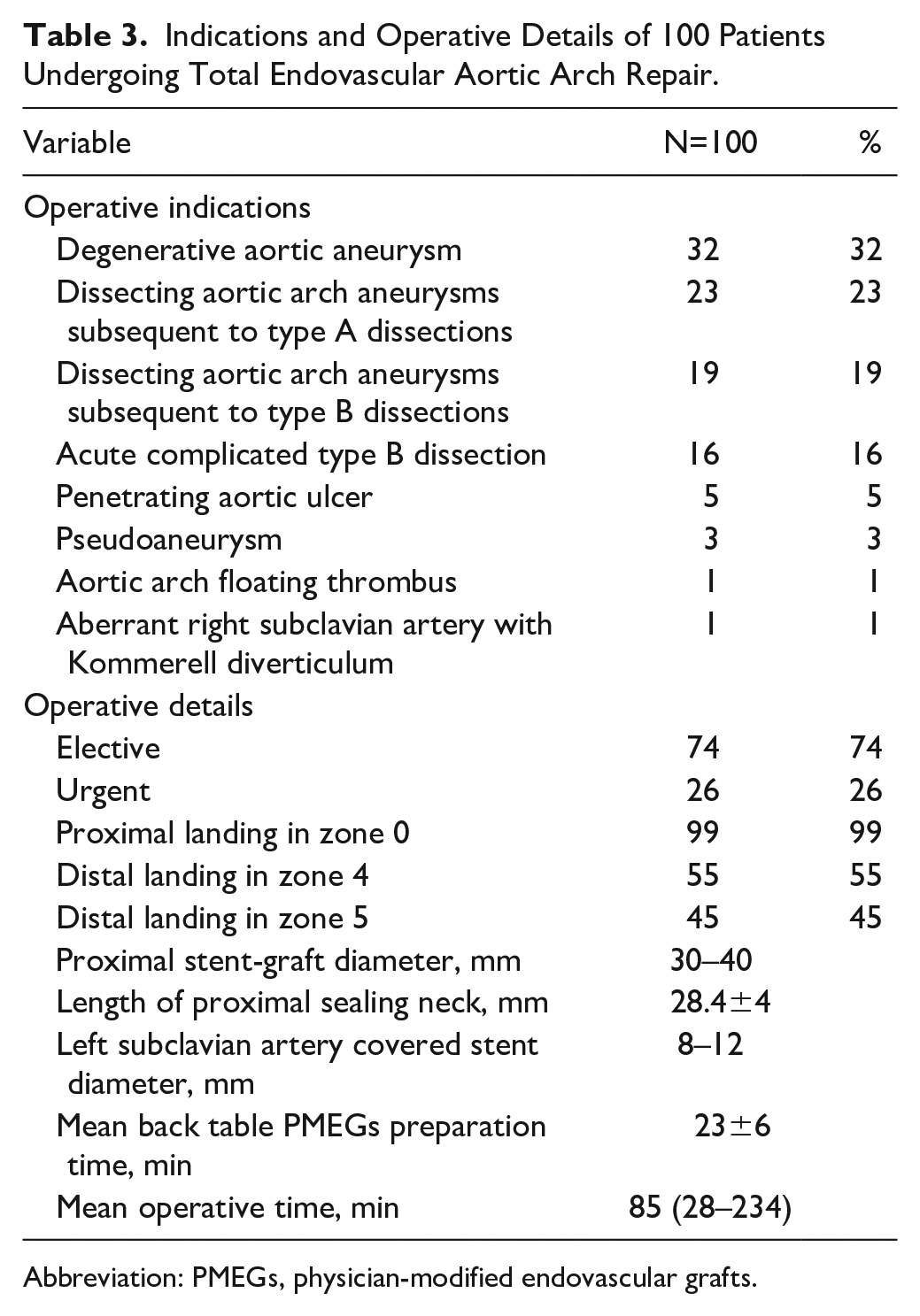
Abbreviation: PMEGs, physician-modified endovascular grafts.
An average of 2.1 stent-grafts (range, 1–3) was deployed. The mean proximal sealing length was 28.4±4 mm. The mean length of the proximal fenestration was 28.8 mm (18–30 mm). Mean length between the fenestrations was 5.4 mm (5–8 mm). Associated procedures included supra-aortic trunk reentry tear closure by covered stent-graft placement in 6 patients, left carotid-axillary bypass in 2 patients (unplanned), transcatheter aortic valve replacement in 1 patient, LSA chimney in 1 patient (unplanned), femoral endarterectomy in 1 patient, femoral-femoral bypass in 2 patients, and 3 unplanned iliac conduits were necessary to introduce the stent-graft delivery system. The operative details are also provided in Table 3.
Thirty Day Outcomes
The median duration for stent-graft modification was 23±6 minutes, range 16 to 41 minutes. Endovascular exclusion of the aortic arch was achieved in all cases. In 2 patients, catheterization of the stent-graft fenestration from the LSA was not achieved and revascularization was performed by left carotid-axillary bypass. Coverage of the LSA fenestration by additional distal stent-graft placement was used to prevent an endoleak. In 1 patient, the LSA stent-graft fenestration was misaligned. Having confirmed patency of BT and LCCA with aortography, a chimney was used to manage the LSA. This was the third patient in the series.
Four patients (4%) had a stroke (visual loss and confusion, both with cerebral computed tomographic changes), 2 of them with full recovery by discharge and no long-term deficit.
Thirty day mortality was 2% (2 patients). One patient died on the first postoperative day of multiorgan failure after repair of an iliac rupture during stent-graft insertion. The other patient died on the fourth postoperative day after a reintervention because of bleeding from the proximal anastomosis of the remnant iliac conduit.
Six patients (6%) required reintervention. One was for a type IB endoleak, successfully treated with the deployment of a distal thoracic stent-graft on the third postoperative day. The remaining 5 patients had required reintervention for access-related complications.
All supra-aortic trunks were patent; no spinal cord ischemia, aortic rupture, and conversion to surgical repair were recorded in the immediate perioperative period.
Follow-up
One patient had a retrograde type A aortic dissection on the 32nd postoperative day. The patient underwent emergent ascending aortic replacement with a distal anastomosis between the prosthetic graft and the endograft. Postoperative course was uneventful.
One patient had a type IA endoleak discovered on the 189th postoperative day. He was successfully treated by performing a left carotid-axillary bypass, occlusion of the left subclavian stent using an occlude, and proximal deployment of a double fenestrated physician-modified stent-graft with the distal fenestration centered on the left carotid artery. Postoperative course was uneventful.
Two patients (2%) had a type II endoleak from the LSA, both of them without sac expansion; both of them were observed and spontaneously disappeared.
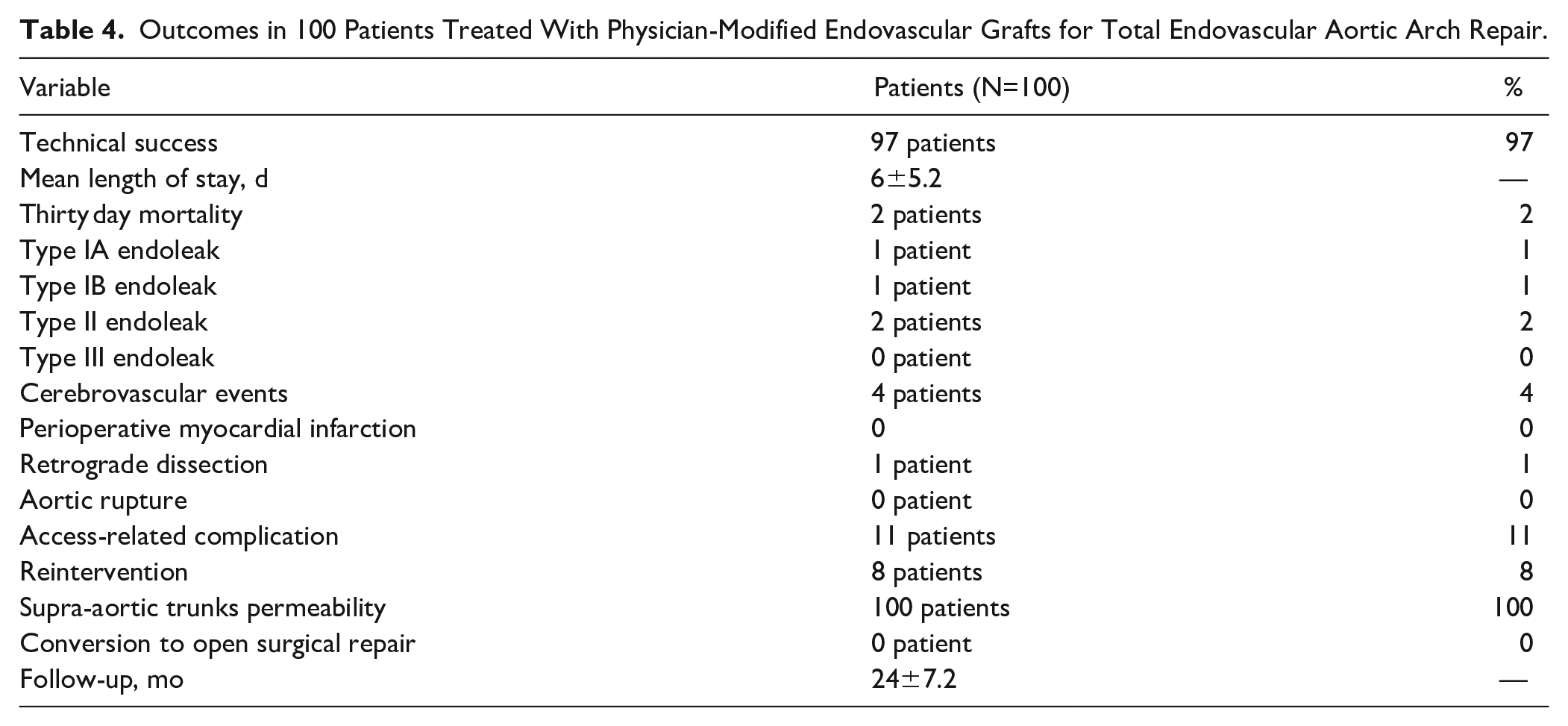
During a mean follow-up of 24±7.2 months, 7 patients died of non–aortic related conditions, an overall mortality of 9% with aortic-related mortality of 2%. There was no aortic rupture, paraplegia, graft migration, graft collapse, or aneurysmal diameter evolution. All supra-aortic trunks were patent. Outcomes in 100 patients treated with PMEGs for total endovascular aortic arch repair are summarized in Table 4.
Outcomes in 100 Patients Treated With Physician-Modified Endovascular Grafts for Total Endovascular Aortic Arch Repair.
Discussion
This study evaluates a large series of aortic arch lesions treated with a double fenestrated PMEGs for zone 0 TEVAR. Studies on total endovascular repair of arch lesions are typically limited by small case numbers and early follow-up. With 100 patients and 2 year follow-up, this represents the largest study currently reported. Published series of patients treated with total endovascular aortic arch repair are summarized in Table 5.
Published Series of Patients Treated With Custom-Made Branched Stent-Grafts for Zone 0 Thoracic Endovascular Aortic Repair.
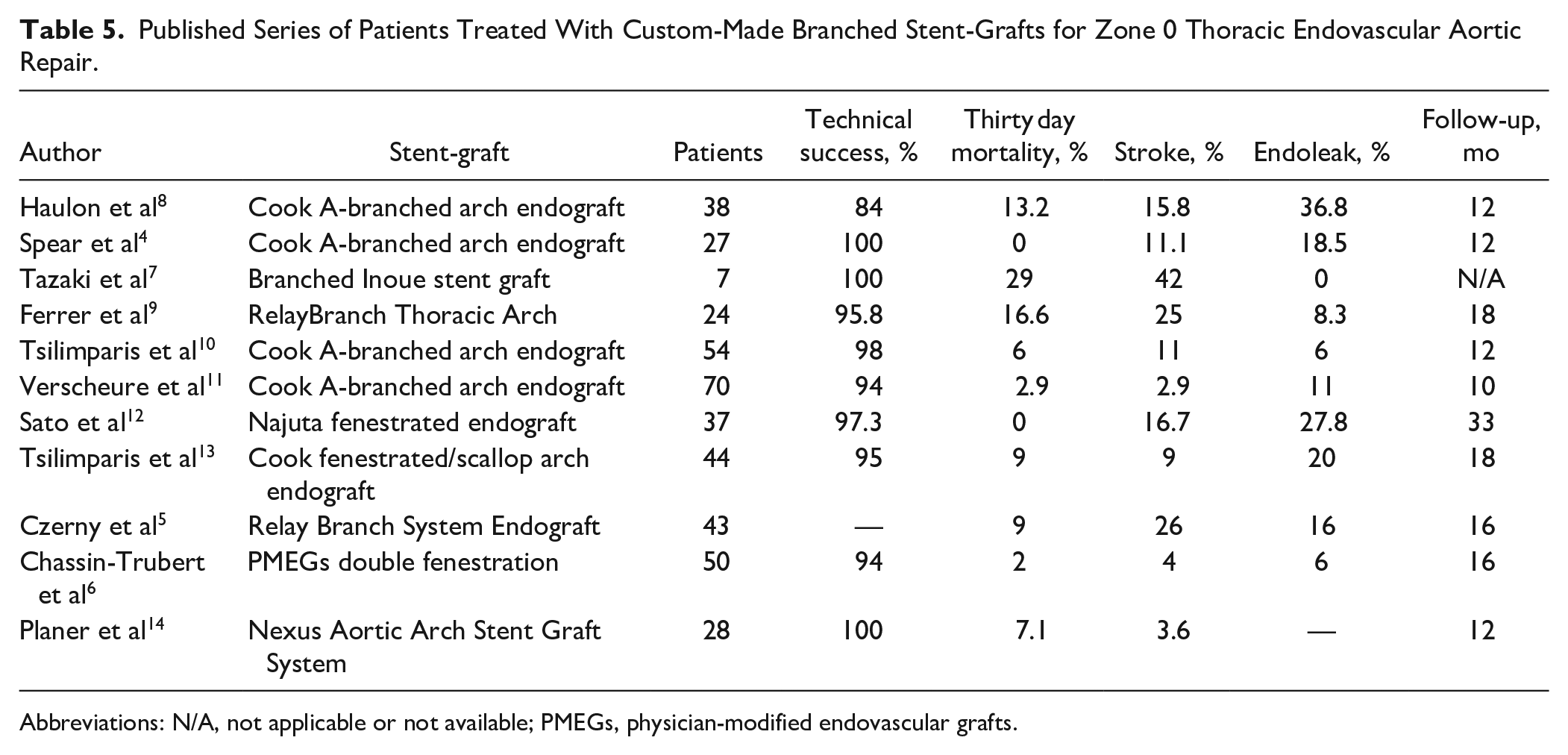
Abbreviations: N/A, not applicable or not available; PMEGs, physician-modified endovascular grafts.
The standout feature of this double fenestrated device is its simple handling during operation with the proximal fenestrations being directed to the orifices of the BT and LCCA automatically when the LSA fenestration is catheterized and secured by covered stent placement. The large proximal fenestration accommodates the trunks with a low risk of occlusion. Bare metal or covered stents are not required for these branches. The deployment algorithm actively steers the operator away from superfluous manipulations of the device within the arch and avoids guidewire manipulation in the BT and the LCCA.
Pre-cannulation of the fenestration with a preloaded guidewire reduces operating time, intra-aortic manipulations, and therefore the neurological risk, and maximizes the technical success rate 15 and reproducibility. 16 The preloaded guidewire reduces the technical difficulty of what could be the most challenging step for this procedure, especially in challenging anatomical scenarios. Most importantly, since its introduction in our experience we have observed 100% technical success whatever the pathology treated.
The desire to stent branches in the aortic arch is driven by concerns regarding achieving seal and durability. These decisions are extrapolations derived from experience managing visceral branches in the setting of extensive abdominal or thoracoabdominal aneurysm, where the need for stenting of fenestrations is intuitive and clear. The results presented here suggest that near total stenting of fenestrations is not necessarily generalizable to fenestrated/branch devices at all anatomical locations. The requirements for seal are determined by the interaction of the anatomy, physiology, and device in the treated zone. In an abdominal aortic aneurysm, the stent-graft is positioned in the middle of the sac leaving a significant gap between the fenestration and the ostium of the target, but in the aortic arch, once the stent-graft is fully deployed, the hemodynamic forces and angulation align the deployed stent-graft against the greater curvature or aortic dome, eliminating the gap between the fenestration and the vessel ostia. The authors believe that this phenomenon explains the sound results of this strategy, the absence of endoleaks, despite the use of a bridging covered stent exclusively for the LSA. The LSA bridging stent is only to secure the alignment of the fenestrations and prevent migration over time. Using an unstented fenestration for the BT and LCCA increases the theoretical risk of stent-graft migration and shuttering. However, the stent-graft is anchored by the LSA stent and migration has not been observed. It is also reassuring that in proximal scalloped TEVAR no cases of stent-graft migration have been reported. There are, however, special anatomic considerations when utilizing this approach. Zone 0 aortic arch lesions, except saccular aneurysms at the lesser curvature of the arch, are unsuitable. In this context, the size of the proximal fenestration, proximally and laterally larger than that of the BT and LCCA orifice, would be associated with an unacceptably high rate of endoleak.
The shaggy score has been described 17 as a tool to prevent overt neurological events (stroke, paraplegia), by improving patient selection. Aortic shagginess is a significant predictor of periprocedural ischemic strokes. In this series, transient ischaemic attack (TIA)/stroke rate was associated with the presence of thrombus in the proximal landing zone and all of cases had atheroma protruding into the aortic lumen. The authors would recommend consideration of alternative strategies or even conservative management with surgical turn down.
This seems to be crucial to the excellent perioperative outcomes reported here and contrasts with reports for inner branched stent-grafts in the arch. Although there are many technical challenges specific to the aortic arch for total endovascular arch repair, over the last decade manufactured branched custom stent-grafts have gained acceptance.3–5 Two custom-made branched devices are currently available: the Cook A-branched arch endograft (third-generation arch design; Cook Medical; Bloomington, Indiana) and the RelayBranch Thoracic Arch System (Terumo Aortic; Sunrise, Florida). Both devices are intended for zone 0 TEVAR with 2 inner or 3 side branches for BT, LCCA, and LSA. The world experience with 27 Cook A-branched arch endografts was reported as a multicenter experience in 2016 by Spear et al 4 with 0% 30 day mortality, 11% stroke, 18.5% endoleak, 100% technical success, and a secondary procedure rate of 22.2% during 12 month follow-up. 8 A more recent report with 54 patients treated with this same custom-made branched stent-graft was published by Tsilimparis et al 10 in 2019 with 6% 30 day mortality rate, 11% stroke rate, 6% endoleak, 98% technical success, and a secondary procedure rate of 24% during 12 month follow-up. The experience with the RelayBranch Thoracic Arch System was reported in 2019 by Ferrer et al 9 in 24 patients with 16.6% 30 day mortality rate, 25% stroke rate, 8.3% endoleak, 95.8% technical success rate, and a secondary procedure rate of 16.7% during 18 month follow-up. Despite the experience and skill of the operators, the technical difficulty of side branch catheterization, compounded by the need for surgical revascularization for the LSA, resulted in an inherently high risk of cerebral embolism and other adverse outcomes. In the present series of 100 patients, aligning the graft is based on only the LSA fenestration. This is one of the most important intraoperative advantages of this approach. The deployment algorithm actively steers the operator away from superfluous manipulations of the device within the arch, guidewire manipulation, instrumentation, and clamping of the BT and LCCA. In addition, no surgical revascularization of the LSA is required. A significantly lower procedural stroke risk is observed. The outcomes with double fenestrated PMEGs for total endovascular aortic repair with 2% 30 day mortality rate, 4% stroke rate, during 24 month follow-up seem to be superior to the published outcomes available for the 2 manufactured branched custom stent-grafts.
This approach is an off-label use of the Valiant Captivia thoracic stent-graft. Concerns about the long-term fabric, stent-graft durability, and impact on the general graft ring stability are understandable. In our series, no stent fractures were detected during routine radiologic follow-up. The Montpellier group has had already published its series of 15 patients with single fenestration PMEGs for zone 2 TEVAR with more than 3 years of follow-up with no stent fractures, endoleaks, and with all supra-aortic trunks’ vessels permeable. 18 Naturally, further data are needed to establish durability. Careful long-term monitoring and strict surveillance of patients are required to identify complications resulting from potential device failure.
The availability of industry-made devices for zone 0 TEVAR would alleviate concerns regarding physician modification and limitations of PMEGs as a not established treatment option in American or European vascular guidelines. Although variations of the aortic arch anatomy are numerous and common, the Montpellier group has recently published an anatomical study that defines and establishes 2 areas of the aortic arch from within which all the supra-aortic branches originate in 97% of patients. 19 These morphological data associated with the encouraging outcomes of this series of 100 patients suggest that an off-the-shelf double fenestrated thoracic stent-graft is at least conceptually possible. Industry could therefore use this “universal double fenestration pattern” to create a commercially-available stent-graft for zone 0 TEVAR, decreasing costs and increasing availability.
Although the technique described is reproducible, it should be noted that the Montpellier group has extensive experience in physician modification of thoracic stent-grafts. Prior to the total endovascular aortic arch repair series described, the unit gained experience by performing experimental and clinical studies of proximal scalloped PMEGs, followed by single fenestrated PMEGs for zone 2 TEVAR and finally single fenestrated PMEGs for TEVAR of zone 0 and zone 1 aortic lesions combined with cervical debranching procedures. 20
Conclusion
Endovascular aortic arch repair using this model of double fenestrated PMEGs for lesions of the aortic arch is a feasible, safe, and reproducible technique with a low perioperative neurological complication rate. This technique is not meant to replace other approaches such as conventional surgery but should be considered complementary allowing to treat urgent patients that cannot wait for a custom endovascular device or high-risk surgical patients. Long-term follow-up is needed to confirm or improve these results.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
