Abstract
Introduction
The femoropopliteal sector endovascular treatment is particularly challenging due to its high tortuosity and torsional forces. Better results are still needed to ensure the long-term patency of stenting in this area. The Supera stent appears to change this paradigm.
Methods
This single-center retrospective cohort study aims to evaluate the efficacy and safety of femoropopliteal stenting with Supera in a real-world population. Seventy-nine patients were treated between January 2015 and December 2020, and the results are reported with a median follow-up of 28 months.
Results
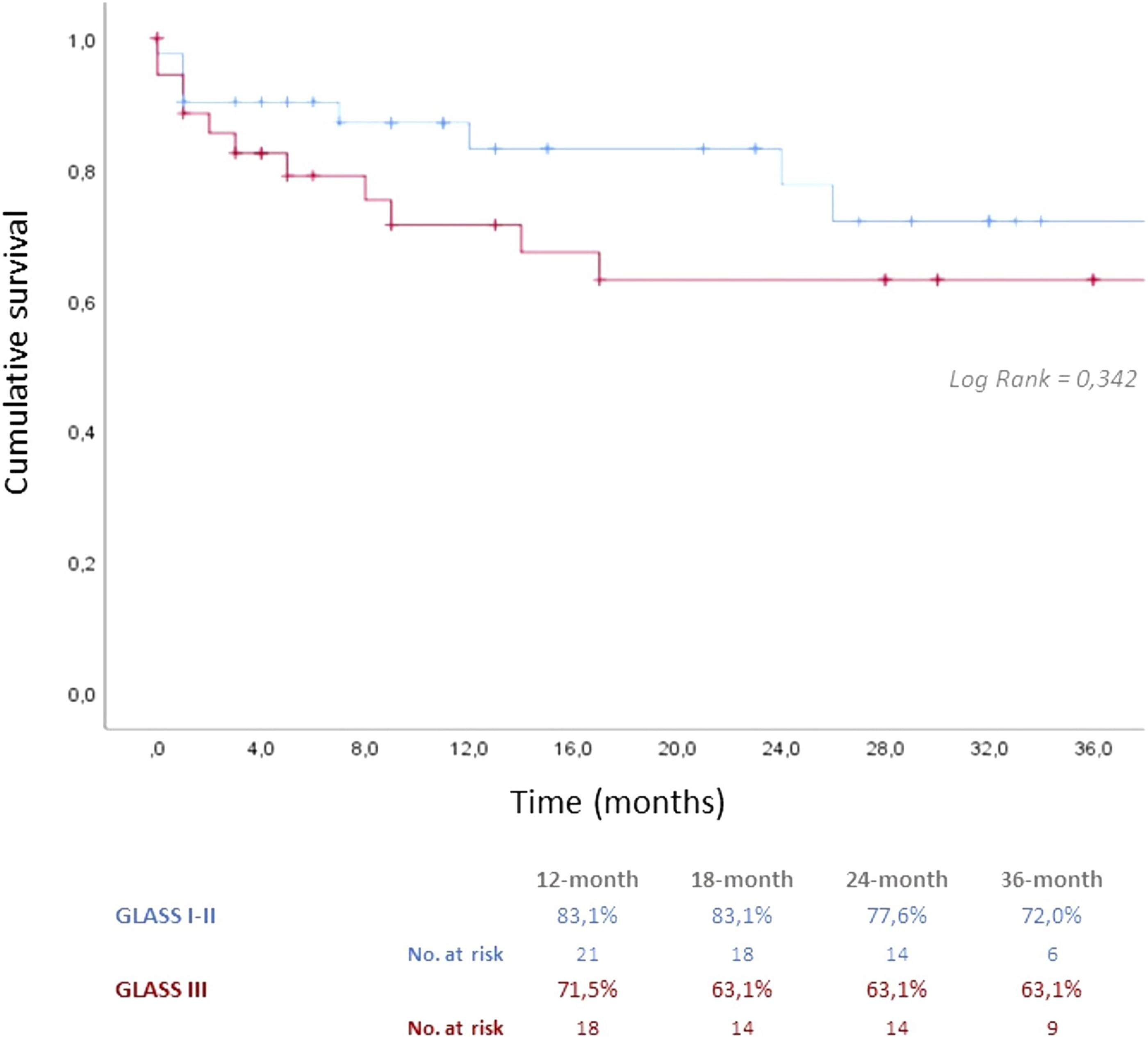
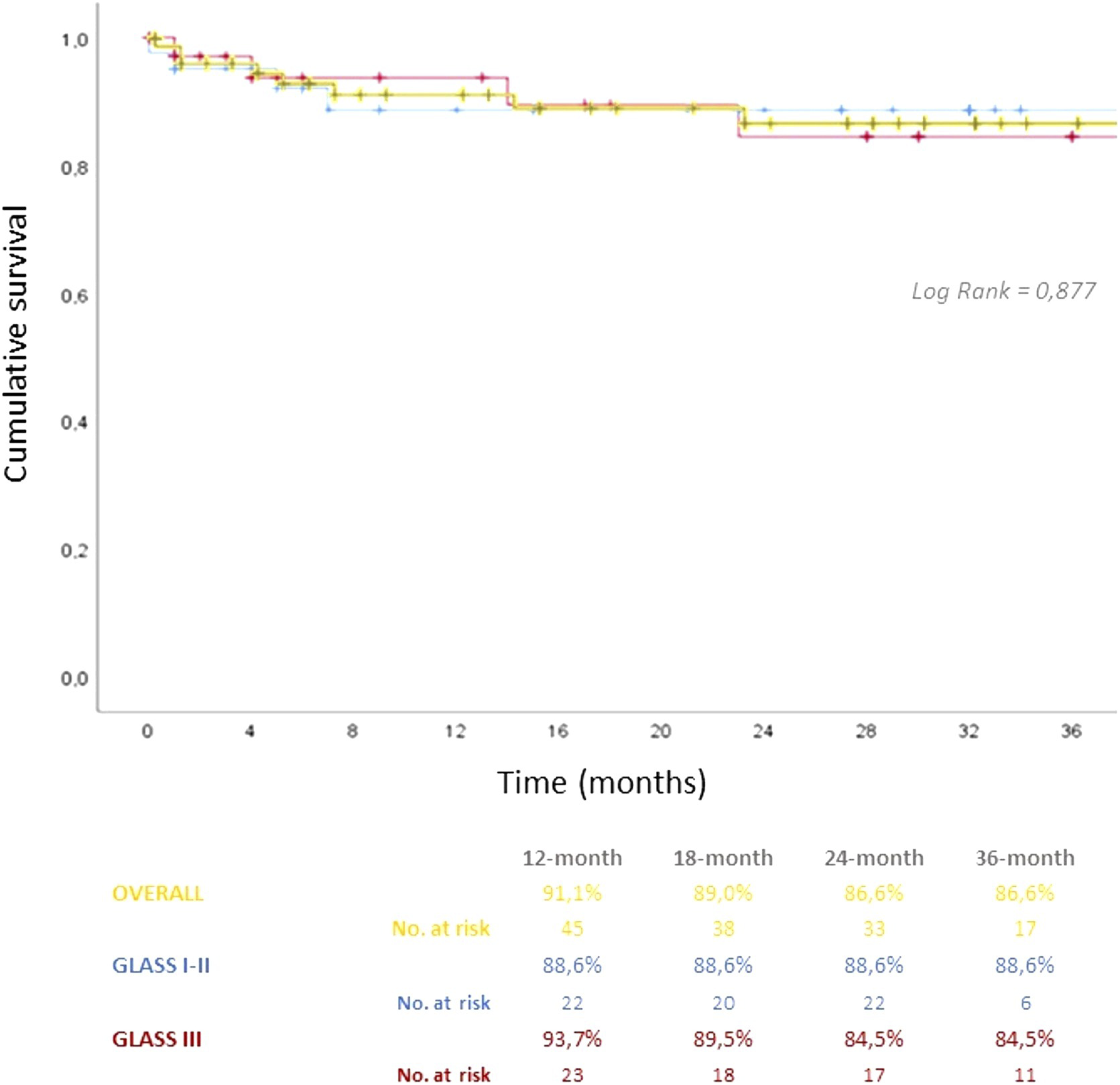
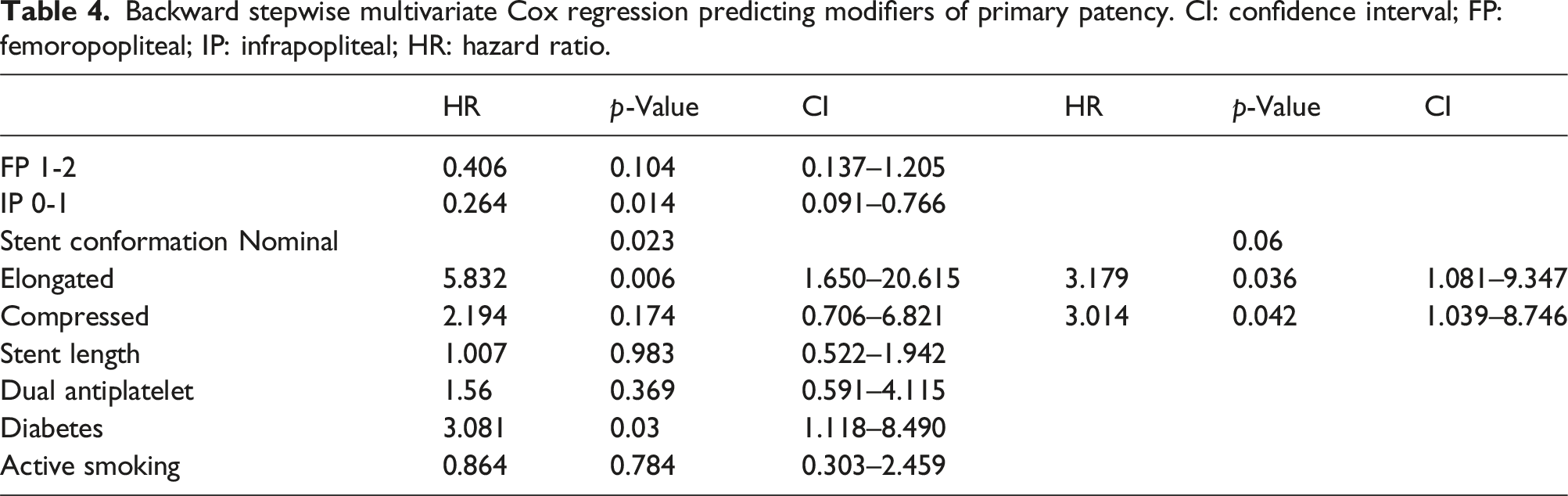
Indications for revascularization were chronic limb-threatening ischemia with tissue loss (73.6%) or ischemic rest pain (17.7%) and claudication (7.6%). Thirty-six patients (45.6%) were classified as GLASS stage III according to the Global Limb Anatomic Staging System, with 65.8% and 30.4% in grades 3 and 4 of femoropopliteal and infrapopliteal sectors, respectively. The 36-month primary, primary-assisted, and secondary patency rates were 68.6%, 72.0%, and 79.0%, respectively, with an amputation-free survival rate of 86.6%. There was no significant difference between primary patency rates in GLASS stages I–II compared with GLASS stage III (36-month primary patency rates of 72% vs 63% respectively, p = 0.342) nor in amputation-free survival (88% vs 84%, p = 0.877). After adjusting for potential confounders, only the stent conformation significantly affected the primary patency rates, with a higher hazard of reintervention for the elongated (HR = 3.179; p = 0.36; CI 1.081–9.347) and the compressed (HR = 3.014; p = 0.42; CI 1.039–8.746) forms.
Conclusions
The 36-month patency of the Supera stents in our real-world cohort was similar to other reported series. The GLASS stage did not interfere with the stent patency, proving it is a good choice even in the most adverse anatomy patients. Only the non-nominal stent conformation affected the primary patency rates in our patients.
Introduction
The femoropopliteal sector is particularly affected by peripheral arterial disease. Its biomechanical characteristics with high tortuosity and susceptibility to extrinsic compressive and torsional forces associated with low flow and high resistance hemodynamic conditions make the endovascular treatment particularly challenging.1–5 Despite several studies reporting longer patency of femoropopliteal stenting than that of balloon angioplasty,6–12 long-term outcomes are still marginal due to the high rate of restenosis, stent fracture, and thrombosis.2,13–15 However, the Supera stent is changing this paradigm. Its unique interwoven nitinol conformation provides greater radial strength, flexibility, and fracture resistance than conventional self-expanding stents,4,16–19 with promising long-term results. This study evaluates the efficacy and safety of femoropopliteal stenting with Supera in a real-world population and the extrinsic factors that may influence the results.
Materials and methods
This single-center retrospective cohort study includes all patients with peripheral arterial disease who underwent femoropopliteal intervention with a Supera stent between January 2015 and December 2020. Patients were recruited by searching for the word “Supera” in the center’s database, and procedure-related information and follow-ups were analyzed by consulting the electronic health records. Angiographic findings for anatomical staging (Global Limb Anatomical Staging System—GLASS) 20 and stent conformation details were analyzed by a single independent reviewer (LL). Our institutional review board approved the retrospective case series study. Written informed consent was not required because of the retrospective nature of this study. The indications for stenting were (1) restenosis, (2) residual stenosis, or (3) dissection after plain balloon angioplasty. The vessel was previously dilated with a balloon 0.5–1.0 mm larger than the target vessel diameter. All vessels were prepared with balloon angioplasty only. None of the patients were submitted for adjunctive atherectomy or drug-coated technology before implantation of the Supera stent. Our center did not have intravascular ultrasound at the time of this study. The patients were kept under surveillance in the outpatient clinic with clinical and ultrasound monitoring. An invasive study with angiography was only performed if clinical worsening would justify additional intervention. Stent patency rates were the main outcome considered, defined as follows: primary patency includes the period from stent deployment until the first reintervention; assisted primary patency includes the previous, ending with the first stent thrombosis/occlusion; secondary patency includes the whole period from stent deployment until definite occlusion; in the absence of occlusion, the stents were considered patent until the last clinical observation. In addition, independent variables such as diabetes, active smoking, intra-operative anatomic staging (femoropopliteal and infrapopliteal grading), total stent length coverage, deployment conformation (nominal, compressed, and elongated), and antiplatelet regimen (single or dual) were considered potential modifiers of patency and were further analyzed.
Statistical analysis
Statistical analysis was performed in IBM’s SPSS statistics v.25, and the 0.05 significance level was adopted. Categorical variables are reported as frequencies and percentages; non-categorical variables with normal distribution are presented as means and standard deviations. The median follow-up time was calculated with the reverse Kaplan–Meier method. The primary, assisted primary, and secondary patency rates and amputation free-survival were calculated using the Kaplan–Meier statistical method. GLASS I–II and III primary patency rates were calculated using the Kaplan–Meier statistical method and compared through the Mantel–Cox log-rank test. The potential modifiers of primary patency were analyzed through a backward stepwise multivariate Cox regression.
Results
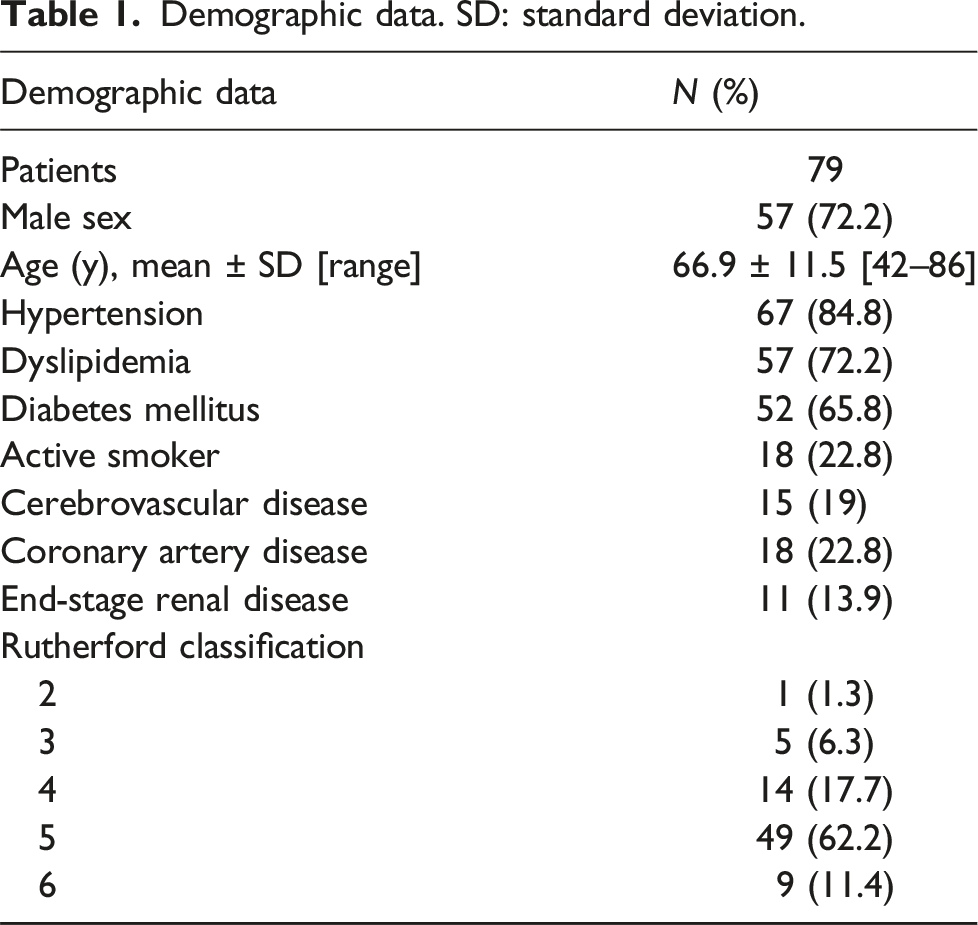
Demographic data. SD: standard deviation.
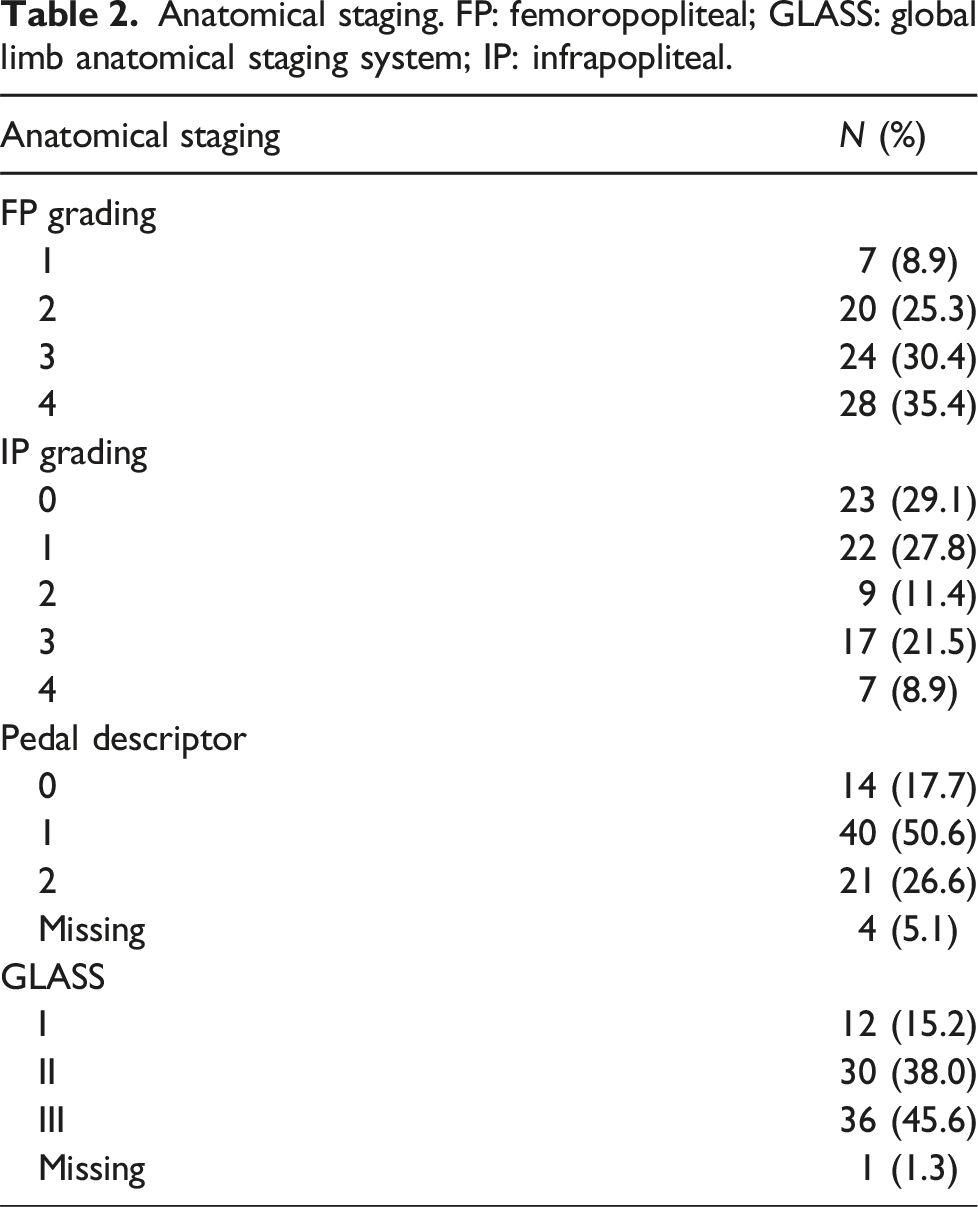
Anatomical staging. FP: femoropopliteal; GLASS: global limb anatomical staging system; IP: infrapopliteal.
Procedural features.

The 36-month primary, primary-assisted, and secondary patency rates were 68.6%, 72.0%, and 79.0%, respectively (Figure 1), with an amputation-free survival rate of 86.6%. There was no significant difference between primary patency rates in GLASS stages I–II compared with GLASS stage III (36-month PPR of 72% vs 63%, respectively, p = 0.342) (Figure 2) nor in amputation-free survival (88% vs 84%, p = 0.877) (Figure 3). After adjusting for potential confounders, only the stent conformation significantly affected the PPR, with a higher hazard of reintervention for the elongated (HR = 3179; p = 0.36; CI 1081–9347) and the compressed (HR = 3014; p = 0.42; CI 1039–8746) forms. In contrast, the variables related to the disease severity, represented as GLASS femoropopliteal and infrapopliteal grading, diabetes, active smoking, stent length, or antiplatelet regimen, did not reach statistical significance (Table 4). Overall patency rates. Patency rates and GLASS stage. Amputation-free survival and GLASS stage. Backward stepwise multivariate Cox regression predicting modifiers of primary patency. CI: confidence interval; FP: femoropopliteal; IP: infrapopliteal; HR: hazard ratio.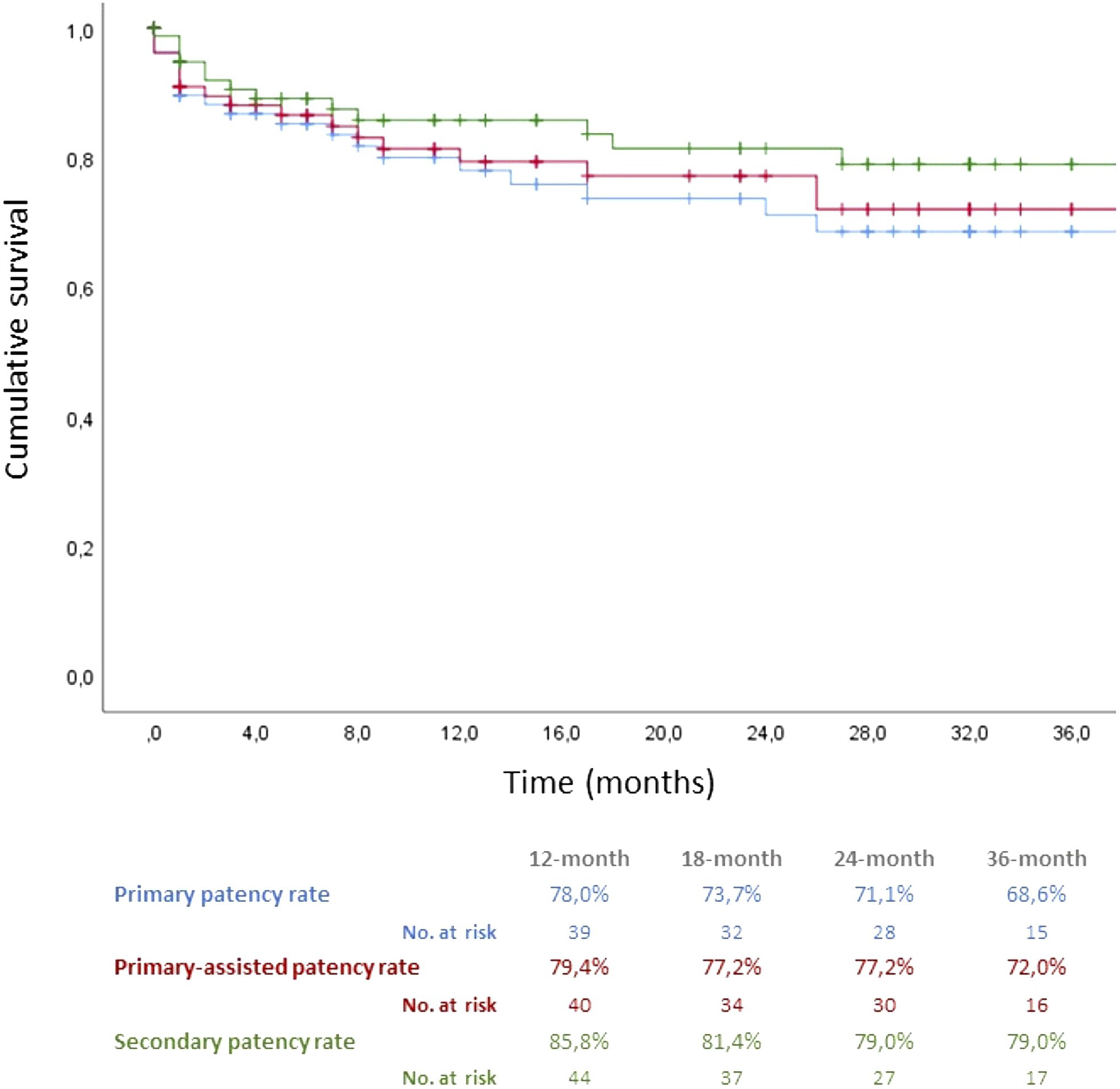
There were no stent fractures or procedure-related complications other than stent occlusion by the end of follow-up.
Discussion
Peripheral artery disease is a new epidemic affecting more patients worldwide, older and more fragile with high surgical risk. Advances in endovascular techniques allow for a safe and effective revascularization alternative. However, the results for complex lesions with extensive calcified obstructive disease or chronic total occlusions are far from ideal. According to the Global Vascular Guidelines on the Management of Chronic Limb-Threatening Ischemia in high-complexity lesions, classified by the Global Limb Anatomic Staging System (GLASS) as stage III, the endovascular treatment has an estimated immediate technical failure >20% and 1-year limb-based patency <50%. 20 Although, in the absence of an adequate autologous conduit or if high surgical risk, the endovascular treatment may be the only revascularization approach in the setting of advanced lesion complexity (GLASS FP grades 2–4), adjuncts to balloon angioplasty (stents or drug-eluting devices) may be considered (recommendation 6, grade 2, level of evidence B). 20 Despite several studies reporting longer patency of femoropopliteal stenting than balloon angioplasty,6–12 long-term outcomes are still marginal due to the high rate of restenosis, stent fracture, and thrombosis.2,13–15 The literature reports 12-month PPR ranging from 72.6% to 94.1%,1,3,17,21–29 with sustained results by the third year of follow-up (PPR 69.5%–76.7%).1,3,21,26 Despite some studies reporting the worst outcomes for high-complexity anatomies,1,25,27 the 12-month PPR is still >64% for this subgroup. Those findings were not supported by other centers,3,21,24,26 and the “Supersub Study” 23 including TASC C and D lesions with a mean stented lesion length of 279 ± 12 mm exclusively reported a 12-month PPR of 94.1%; also, the recently published “STELLA-SUPERA Trial” 29 (exclusively TASC C and D lesions with a mean stented lesion length of 273 ± 127 mm) reported a 24-month PPR of 77.9%. In our study, the Supera stent achieved very satisfactory outcomes, with a primary patency rate of 68.6% and an amputation-free survival rate of 86.6% by the third year of follow-up; we also corroborate the stent’s effectiveness in complex anatomies with no significant difference between GLASS I–II and GLASS III lesions (36-month PPR of 72% vs 63%, respectively, p = 0.342). Nonetheless, a successful recanalization remains a significant obstacle, requiring high technical skills; even in this scenario, the need for subintimal recanalization does not seem to impair the effectiveness of this stent. 23 Under constant exposure to biodynamic forces (compression, torsion, rotation, flexion, and extension), the stent extension to the popliteal sector would presumably hold a high risk of stent stenosis, fracture, and thrombosis. Supera’s unique interwoven nitinol conformation provides greater radial strength, flexibility, and fracture resistance,4,16–19 and the stent extension to the popliteal sector appears safe without compromising efficacy.1,24,26 In this study, most stents were deployed or extended to the popliteal sector, not allowing a direct comparison between stent locations; however, the sound overall results led us to validate the stent’s good performance in the popliteal sector. The fact that all vessels were prepared with only balloon angioplasty under the same sizing protocol (+0.5–1 mm of the intended vessel diameter) provided an excellent uniformization of the results in our retrospective study. The stent conformation has been proposed as a modifier of patency with inferior results for the elongated form and best outcomes achieved with stent compression after deployment; 30 our results showed that both elongated and compressed conformations impair the primary patency rate. On the other hand, the stent length and complexity of femoropopliteal and infrapopliteal disease did not reach statistical significance, supporting the previous results with non-inferior patency rates for GLASS III compared to GLASS I–II lesions. Notably, 70% of the patients had a GLASS Infrapopliteal Grade >1. This means most patients had at least a focal chronic total occlusion or stenosis involving a third of the best run-off vessel at the beginning of the revascularization procedure. Surprisingly, active smoking and diabetes did not significantly affect the outcomes; however, this may be justified by the small sample size. Most published studies on Supera performance1,3–5,16,17,21–30 had all patients under a dual antiplatelet regimen for at least 4 weeks after the implantation. Ours is one of the few studies that compare the impact of the antiplatelet regimen on Supera’s patency rates, as only 39.2% of the patients were on dual antiplatelets after the implantation. We underline that the antiplatelet regimen did not significantly affect the outcomes in our cohort.
Conclusions
The Supera stent is changing the endovascular treatment of femoropopliteal lesions, highlighting its performance in high-complexity anatomic patterns. The only characteristic that affected the results was the conformation of the stent after deployment. Further investigations, including randomized studies comparing the Supera stent with other stents, drug-eluting angioplasty, or atherectomy devices, are advisable.
Footnotes
Author contributions
Luis Loureiro, MD, FEBVS, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António: Study design, data gathering, data analysis, and manuscript production; Andreia Pinelo, MD, MSc, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António: data statistical analysis, data gathering, and manuscript production; Carlos Veterano, MD, MSc, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António: data gathering; Henrique Rocha, MD, MSc, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António: data gathering; João Castro, MD, MSc, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António: data gathering; Rui Machado, MD, PhD, Angiology and Vascular Surgery Department, Centro Hospitalar Universitário de Santo António, Instituto de Ciências Biomédicas Abel Salazar - Universidade do Porto: study design and manuscript revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
