Abstract
Objectives
To demonstrate the ease with which steerable sheaths, designed for cardiac electrophysiological applications, can be used to aid endovascular treatment of a wide range of non-cardiac vascular disease and to assist with target vessel cannulation in branched and fenestrated aortic grafts.
Methods
A retrospective medical chart review was carried out to identify cases from a single vascular surgery unit (2019–2022) where the HeartSpan Steerable Sheath (HSS) (Merit Medical, South Jordan, UT, USA) was utilised to enable endovascular management of complex vascular pathology. A case presentation of branch graft insertion performed entirely via distal access is described and used to help identify pertinent sheath characteristics and technical considerations, and to illustrate the advantages and disadvantages of the design for modified use in target vessel cannulation.
Results
The HSS was used in the endovascular treatment of different vascular pathologies in 15 patients (23 target vessels) where access to the vessels using standard catheters and approaches was not possible. Cannulation and subsequent stenting were successful for 21 of the 23 target vessels in total. Of these cases, the HSS was used as an adjunct for deployment of fenestrated endovascular graft systems when conventional techniques for canulation of target vessels had failed on five occasions. On another four occasions, the HSS enabled full deployment of the entire Zenith® t-Branch™ Thoracoabdominal Endovascular Graft system from an exclusively femoral approach. An additional three cases involved use of the HSS for superior mesenteric artery cannulation in patients with mesenteric ischaemia. The device was also used once in each of the following cases: contralateral common iliac cannulation, cannulation of contralateral internal iliac artery for coil embolisation, and access of a contralateral iliac branched device. There were no stent dislocations and all aortic branches that were patent at the completion of each case remained so 1-year post procedure.
Conclusion
Steerable sheaths designed for cardiac electrophysical applications, like the HSS Introducer, can be successfully utilised for cannulation of challenging target vessels in a wide range of aortic endovascular procedures. This modified approach may salvage cases that would otherwise be considered inoperable in regions of the world where steerable sheaths designed for aortic use are not readily available.
Introduction
The use of endovascular grafts with fenestrations and branches for management of aneurysmal disease has become commonplace.1,2 However, even in experienced hands the deployment of such grafts can be technically demanding. 3 In particular, the cannulation of target vessels and subsequent passage of bridging stents can be challenging if the orientation of vessels is counter to the direction of approach.4,5
The concept of a steerable sheath to facilitate cannulation of target vessels of the aorta and peripheral vessels is attractive. 6 Sheaths designed and marketed for this intended use are only available in select regions of the world. Conversely, steerable sheaths designed for use in cardiac radiofrequency ablation procedures are commonplace in most regions of the world. 7
The HeartSpan Steerable Sheath (HSS) Introducer (Merit Medical, South Jordan, UT, USA) is an 8.5 French (F) sheath designed for cardiac electrophysiological (EP) applications. The HSS Introducer is a relatively inexpensive item (less than 3% of total consumable costs) compared to the total consumable cost of a four branch Thoracoabdominal endovascular graft at approximately $40,000 AUD.
In our experience, the HSS Introducer can be effectively utilised in many complex endovascular aortic pathologies. Contrary to the conventional clinical paradigm which advocates the use of larger sheath sizes (e.g. 12 F and larger) for deployment of bridging stents, 8 our experience presented herein has shown that the 8.5 F sheaths are adequate for most applications, including the placement of branched grafts in aortic repair.
In the three years of using the HSS, we have found that the device substantially improves the technical success of aortic procedures by reducing operating time, radiation exposure, and has even been used to salvage cases that were otherwise considered inoperable. The HSS is of particular benefit for use with aortic branch grafts. However, the surgical benefits associated with the HSS (e.g. availability to vascular surgeons and applicability for aortic procedures) are not widely recognised in Australasia, even among units that participate in high volumes of complex endovascular work.
To raise awareness regarding the utility of the HSS we summarise our experience and provide a detailed description of one case involving deployment of a Zenith® t-Branch™ Thoracoabdominal Endovascular Graft (Cook Medical, Bloomington, IN, USA) exclusively from groin access. We then highlight some of the characteristics of the HSS that, while ideal for cardiac applications, need to be considered when using the sheath for vascular cases.
Methods
We conducted a review of medical records from our vascular surgery unit (1 June 2019 to 1 June 2022) to identify cases where the primary surgeon elected to use the HSS. Cases were excluded if the sheath was selected but not utilised throughout the case. Each case was reviewed by the authors and primary surgeon to identify pertinent aspects (e.g. manoeuvrability) that demonstrated the nuances of utilising the HSS in endovascular surgery. Human ethics approval requirements were completed through the local health authority human research ethics committee (HREC). Of the records reviewed, one case was selected to detail the benefits associated with the system for which consent to feature in this work was obtained from the patient.
Following medical record review of HSS cases, a list of technical considerations was established to identify criteria associated with the likelihood of clinical success for aortic procedures with the device.
Results
Frequency and indications for HeartSpan steerable sheath use.
The most common indication for use of the HSS in these cases was as an adjunct for deployment of fenestrated endovascular graft systems (Figure 1). This included cannulation (left renal artery cannulation Figures 1(a) and (b), right renal artery cannulation Figure 1(c)), angiographic imaging (right renal artery angiogram Figure 1(d)), and subsequent stenting of target vessels through graft fenestrations (superior mesenteric artery bridging stent deployment Figure 1(e), left renal artery stent deployment Figure 1(f)) when conventional cannulation techniques had failed. Stages of deployment of bridging stents into visceral target vessels during branched-graft deployment. (a) and (b) left renal artery cannulation. (c) right renal artery cannulation. (d) right renal angiogram. (e) superior mesenteric artery stent deployment. (f) left renal stent deployment. In all images the HeartSpan has been flexed to 180°.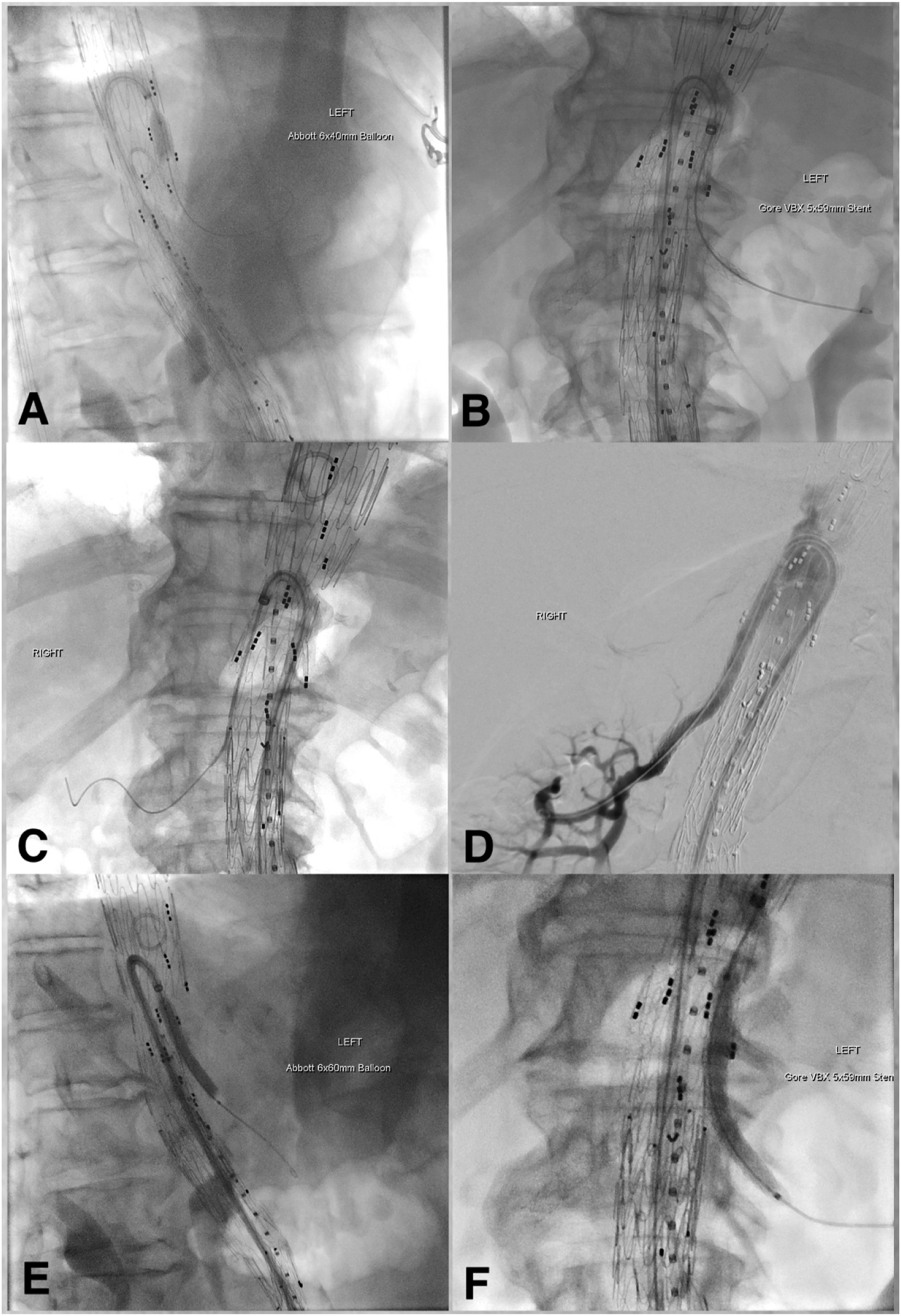
At the 1-year follow-up, all aortic branches successfully cannulated with the HSS Introducer remained patent, there were no instances of stent dislocation.
Case presentation
An 83-year-old male presented with a symptomatic but unruptured 11 cm juxta-renal aortic aneurysm. A Zenith t-Branch Thoracoabdominal Endovascular Graft was planned but an occluded left subclavian artery and high arch thrombus load made the normal technique of canulating branches from a proximal direction unacceptable. Although case reports exist that describe the use of steerable sheaths to canulate branches in similar situations, these have always described the use of larger sheath systems (12–16 F), which were not available.3,9,10 It was therefore decided to utilise an available 8.5 F HSS to assist in the deployment of all four branch stents from a femoral approach. The procedure was undertaken using general anaesthesia in a hybrid laboratory. Access was gained via bilateral common femoral percutaneous approach only.
The main t-branch stent body was positioned and fully deployed via the right side and followed by the bifurcated section, contralateral iliac limb, and ipsilateral limb. An HSS was inserted via the existing 20 F sheath in the groin. The tip of the catheter was advanced beyond the ‘waist’ section of the graft into the more capacious proximal graft to allow a 180° curve to be created in the catheter. The device was then pulled back to engage into each branch. This process was noted to be remarkably easy, and every target branch was selected for the first time with a single pass.
Once in the branch, a hydrophilic wire and 120 cm 5 F vertebral catheter were used to select the desired vessel. We chose to start distally and treat in the order of right renal, left renal, superior mesenteric, and coeliac artery (Figure 2). Once the target vessel was cannulated, an angiogram was performed via the vertebral catheter in the vessel and via the steerable sheath to ensure correct anatomical location. Each target vessel was infused with 100 μg of glyceryl trinitrate in 1 mL of saline. Cannulation procedure. (a) cannulation and stenting of critical stenosis of coeliac artery. (b) cannulation of renal fenestration. (c) accessing internal iliac artery in patient with complex arteriovenous abnormality. (d) example of poor filling of aorta during angiography via the steerable sheath.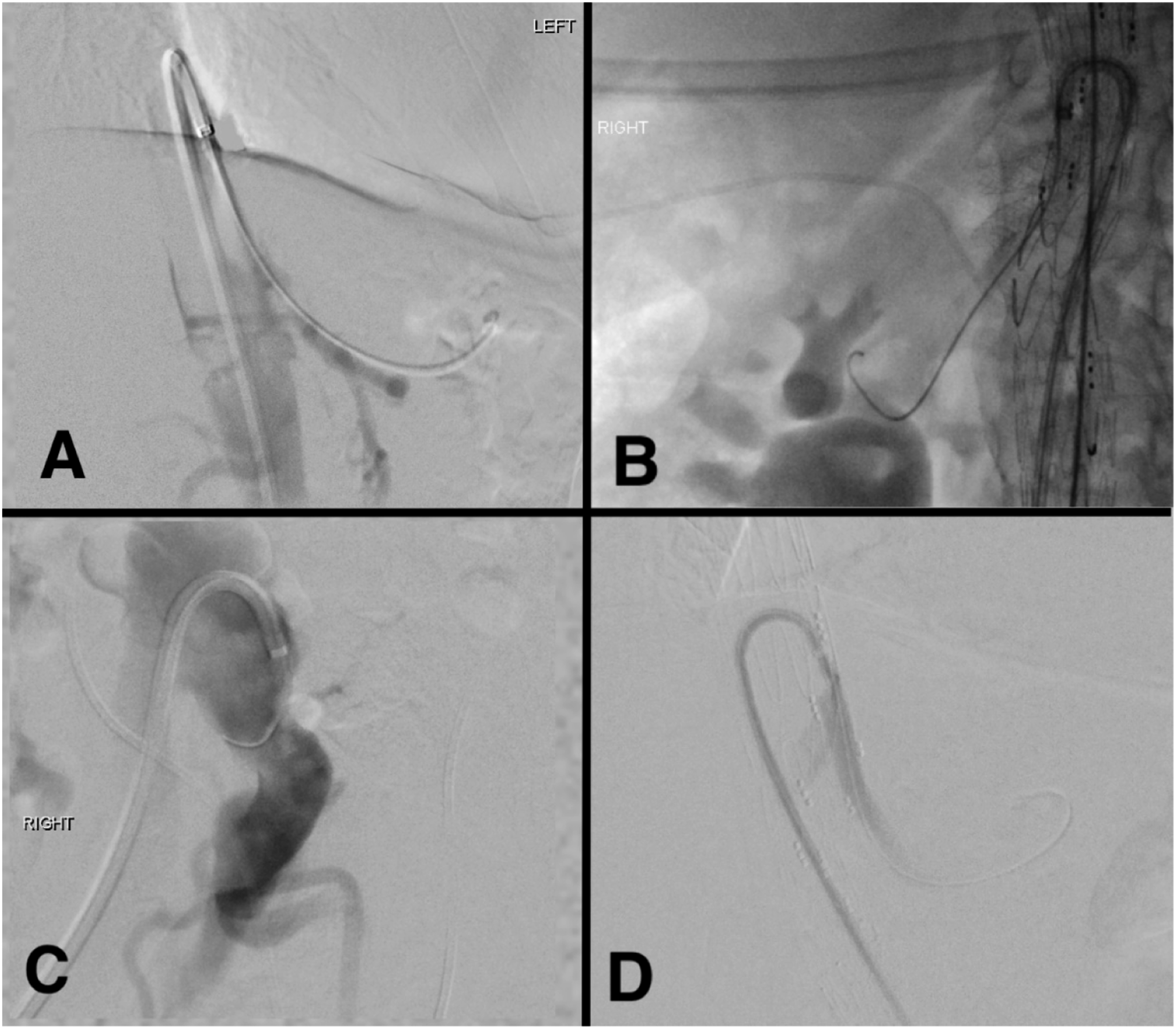
A 280 mm Rosen wire was positioned within the distal target vessel and an angioplasty balloon was used to gauge the desired length of the bridging stent. This system was used in preference to a marking pigtail to avoid the sawing action that marking catheters can have on the vessel orifice and the anticipated problems of tracking the catheter. Furthermore, the balloon was inflated to ensure that a track was present to ease the subsequent passage of stents.
Two 6 mm, one 7 mm, and one 9 mm Viabahn VBX Balloon Expandable Endoprosthesis (W. L. Gore and Associates, Flagstaff, AZ, USA) were used for the two renal superior mesenteric arteries and coeliac branches, respectively. They were deployed via the sheath and “barebacked” into the branch and target vessel. Each was flared within the branch as appropriate.
Most stents pass through the 180° curve of the steerable sheath easily; however, moderate pressure is required when using 9 mm diameter stents. It was found that the most significant resistance was encountered at the point where the proximal section of the stent exits the tip of the sheath. This is possibly due to the soft ‘atraumatic’ tip of the HSS becoming kinked where it was pressed against the wall of the main body. This resistance was overcome by applying slight upward pressure on the steerable sheath whilst simultaneously applying a rocking forward pressure on the bridging stent. Once the proximal end of the stent had passed out of the sheath, forward movement continued with relative ease. Gently reducing the degree of angulation of the HSS Introducer also facilitated easier passage of the stents from the sheath.
All four vessels were cannulated using the same technique. The total time for vessel canulation was 65 min, and the total procedure time was 230 min. A contrast volume of 200 mL was used for the procedure. Completion runs confirmed successful exclusion of the aneurysm sac and patency of all vessels. No further intervention was required, and the patient was discharged three days after surgery.
Technical considerations
In our experience, using the HSS in many different clinical applications, we have identified several technical aspects and procedural techniques that are likely to improve the clinical success of vascular procedures that are summarised below. • The HSS is successfully used both ‘bareback’ into groin arteries or via a 10 F or larger sheath. If used ‘bareback’ it can be replaced by an 8 F short sheath when removed. The device can also fit simultaneously along with two 6 F sheaths through a single 20 F flexor (Cook Medical) sheath, if multiply punctured. • In all cases, we used the small curve size (16.4 mm). It is likely that the medium and large curve sizes will be more difficult to reform within the confines of the aorta and may limit their manoeuvrability to the desired position. • The sheath, with introducer, will only travel on a 0.032″ wire, which is an uncommon platform for non-cardiac interventionalists. However, once the catheter is placed in the main graft body, the introducer and 0.032″ wire can be discarded and are not required again. • The sheath has a 3 mm atraumatic flexible tip which is designed to reduce the risk of intra-septal injury in the heart for cardiac EP cases. However, this tip is less desirable for use in aortic utilisations and can act like a ‘flap’ if pressed against the inner wall of the main stent. This may subsequently limit the passing of balloons and stents out of the sheath. We have overcome this by gently pushing the sheath upwards, thereby relieving the pressure at the tip and straightening the flexible end to open up the sheath lumen at the tip. • The HSS has a highly visible distal radiopaque ring marker which is an excellent guide during placement. However, the flexible tip, which is less visible, extends a few millimetres beyond the marker band which may be confusing if operators are unaware. • Contrast runs performed through the sheath do not opacify a capacious aorta due to the narrow spray of contrast produced by the tapered tip. This is worsened when the tip is pushed against an aortic wall (Figure 2(d)). • The precise clock face orientation can be hard to calculate as there are no facilitating radioopaque markers on the tip. • The tip of the HSS can be angled in two opposing directions away from neutral (i.e. straight). Although this may seem advantageous, it can be difficult to remember which direction to rotate the handle to tighten or reduce the angulation of the catheter tip without looking directly at the handle. A single direction of deflection would be easier to use. • Adequate space is required to reform the catheter tip which may be a concern in small aortas. Undertaking the movement in a graft is protective but caution should be exercised if undertaking in small aortas or those with a particularly high burden of mural thrombus.
Discussion
To our knowledge, steerable sheaths designed and licenced for general vascular applications are not broadly available worldwide. However, based on our experience, the HSS for cardiac applications is widely available in Australasia. The HSS is a 74 cm long, 8.5 F, reinforced, steerable sheath designed to support transseptal cardiac EP procedures. It has a controllable tip that can bend through 180° in either direction in a single plane. It is available with three tip curve sizes, small (16.4 mm), medium (22.4 mm), and large (50 mm). The system is highly radiopaque and robust and includes a marker band at the tip. Its length allows it to reach all target visceral vessels including the great vessels of the aortic arch from the common femoral artery.
There are several published examples of steerable sheaths being used to deploy branch grafts.3,9,10 In 2018, Watkins et al. 9 described the use of the 16 F Aptus Heli-Fx steerable sheath to deploy a t-Branch graft in a patient with a ruptured aneurysm. Also 2018, Makaloski et al. 3 described the use of a 12 F Destino steerable sheath (Oscor, Palm Harbor, FL, USA) through 18 F access sheaths for retrograde cannulation of antegrade branches in branched graft repair of aortic aneurysms in four patients. While in 2021, Vaccarino et al. 10 reported on the utilisation of a HSS through a 12 F sheath to deploy an iliac branched device for the management of a 1b endoleak in an existing fenestrated endovascular aortic repair. These reports reinforce the widely held belief that sheaths less than 12 F are inadequate to allow the passage of large bridging stents. However, in our experience, the 8.5 F steerable sheath, although considerably smaller, was adequate in all cases and results in a far smaller access cannulation. The small size also had the advantage that the HSS can fit alongside two other 6 F guide sheaths, within a multiply punctured 20 F sheath. Moreover, we found that the most notable use of the steerable sheath is its ability to allow the deployment of the Zenith t-Branch Thoracoabdominal Endovascular Graft system using a retrograde groin only approach.
Limitations
This article is intended to increase awareness of the availability and utility of the HSS to vascular surgeons practicing in areas where steerable sheaths designed for aortic work are not readily available. The need for an article of this nature was acknowledged after reflecting on the increasing success observed in our unit with the unique utilisation of the HSS in complex aortic cases. While considering the benefits associated with this work, it is important to acknowledge its limitations. First, as this is a preliminary retrospective review of cases, it is subject to all the usual limitations of a retrospective analysis including limited data availability and risk of bias. Second, this work was based on a small sample of patients analysed which limits broad generalizability of our observations. Finally, this work is based on observations from a single surgical unit; additional research is needed to confirm the generalisability of our observations across cases from other institutions.
Initially, the HSS was used only as a salvage device for complex and challenging cases. However, as the unique benefits and relative ease of use became more apparent to surgeons within our unit, utilisation of the HSS shifted from that of a salvage device to that of an intentional primary strategy for cannulation of aortic branch vessels. It was difficult to determine from the available data for each case what the pre-operative intention of the primary surgeon was with respect to utilisation of the HSS, and therefore could not be presented in this article.
This article highlights the need for additional research to identify characteristics of aortic cases that would indicate utilisation of the HSS or whether a similar device would be reasonable or even indicated. Furthermore, it is our hope that by increasing the awareness of the availability of this approach to practitioners carrying out complex aortic work, we may increase its uptake. This in turn will lead to increased clinical experience and feedback that would be invaluable for research focused on the design of a steerable device specifically intended for intra-aortic use in the future.
Conclusion
Our experience with the HSS has shown that it can be successfully utilised for cannulating challenging target vessels in a wide range of aortic endovascular situations. In particular, we found it advantageous during deployment of aortic branch grafts, allowing these devices to be deployed entirely from a femoral access point rather than requiring an additional superior access point from the upper limb. Despite previous reports to the contrary, we find the 8.5 F system suitable for branched bridging stents of up to 9 mm and it does not require an external stabilising sheath. As this sheath has been designed for cardiac EP procedures, there are minor design characteristics that need to be considered when using the device in vascular procedures to enhance clinical success. This novel approach may offer practitioners, particularly those from regions where steerable sheath technology designed for aortic use is not widely available, another course of action for negotiating challenging branch vessel cannulation.
Footnotes
Authors note
Administrative and editorial assistance in the preparation of this article was provided by Gloria DeWalt, PhD, an employee of Merit Medical.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
