Abstract
Objective
To describe the impact of comorbidities on in-hospital mortality and overall survival in patients with acute mesenteric ischemia (AMI) due to superior mesentery artery (SMA) thromboembolism.
Methods
A retrospective study was conducted for 40 patients with AMI due to SMA thromboembolism who were treated in our hospital between February 2013 and December 2019. The presence of comorbidities was described and their severities were classified into 1–4 levels by Geriatric Index of Comorbidity (GIC), the comorbidities were defined as any distinct additional clinical entity that has existed. Univariate and cox proportional-hazards analyses were performed to determine the effect of comorbidities on in-hospital mortality and overall survival.
Results
During a mean follow-up of 15.05 ± 18.02 months (range from 0.3 to 58 months) for the 40 patients with AMI due to SMA thromboembolism, In-hospital mortality rate was 52.5% (21/40). One-year, 2-year, and 3-year overall estimated survival rates by the Kaplan–Meier method were 45%, 34%, and 26.5%, respectively, the average overall survival time was 20.84 ± 3.95 (95% CI: 13.10–28.58) months. In-hospital mortality was significantly related to the GIC classification (χ2 = 7.86, p = 0.049). The average overall survival was significantly related to the malignant tumor in pre-existing comorbidities and GIC classification (log-rank, p = 0.001). Cox proportional-hazards regressions analysis showed that the class 4 of comorbidities was an independent prognostic factor of mortality (p = 0.031, HR = 10.45 [95% CI: 1.24–87.70]).
Conclusion
Comorbidity is common and an important factor associated with all-cause mortality in AMI patients due to SMA thromboembolism. In managing AMI patients, we recommend a timely diagnosis of both AMI condition and its associated comorbidities.
Keywords
Introduction
Acute mesenteric ischemia (AMI) caused by superior mesentery artery (SMA) thromboembolism is a life-threatening condition with a dismal prognosis; its mortality rate varies in reported studies with a range of 60–90%, despite advances in radiological and surgical techniques, anesthesia, and intensive care.1–3 It is well known that AMI due to SMA thromboembolism often affects elderly patients. 4 The elderly AMI patients often have hypertension, atherosclerosis, cardiovascular and cerebrovascular diseases, and many other comorbid conditions.5,6 Comorbidities are known to pose a challenge in the assessment and effective management of AMI patients due to SMA thromboembolism. 7 For example, AMI patients with cardiovascular diseases are at increased risk of surgical treatment and mortality.8,9 Moreover, multiple comorbidities may have cumulative effects on the prognosis of AMI.10,11
AMI patients caused by SMA thromboembolism can have one or more comorbid diseases. 12 Current management approaches provide clinicians with little guidance in terms of managing comorbidities for AMI patients. Considering the presence and seriousness of comorbidities would be very useful for clinicians who wish to undertake personalized disease management approaches, resulting in improved outcomes in individual patients. 13 In most studies, more attentions are paid to the presence of comorbidities in AMI patients.6,12,14 There are scant researches regarding the severity of comorbidities in relation to the overall clinical outcome of AMI management.
In this study, we described comorbidities and quantified their severities associated with all-cause mortality for AMI patients due to SMA thromboembolism to further explore the underlying mechanisms linking AMI with its comorbidities.
Materials and methods
Patients sampling
This research was designed as a retrospective, observational, non-interventional, monocentric study. The Institutional Ethical Committee approved the study protocol. A retrospective survey was performed for patients with AMI who underwent treatment in our institution between February 2013 and December 2019. Data were collected in reviewing the medical records of these patients. Inclusion criteria included patients with AMI due to SMA thromboembolism, exclusion criteria included patients with venous mesenteric ischemia, nonocclusive mesenteric ischemia, vasculitis induced mesenteric ischemia, inflammatory or other conditions affecting mesenteric vessels (e.g. pancreatitis, perforated ulcer, dissecting aneurysm, tumor), mechanically induced mesenteric ischemia (e.g. adhesion, bezoar, volvulus, incarcerated hernia), and chronic mesenteric ischemia. The diagnosis of AMI was confirmed by radiological (computed tomography (CT) scanning and/or arteriography), intraoperative, or histologic findings.
Finally, 40 patients who met the rigid inclusion criteria were enrolled in the study. There were 23 men and 17 females, with a mean age of 69.58 ± 11.23 years (ranged from 43 to 88 years).
Comorbidity assessment
The term comorbidity was defined as any distinct additional clinical entity that has existed. 15 Comorbidity was identified via patient history, laboratory values, and review of patients’ clinical data. We chose the Geriatric Index of Comorbidity (GIC) most widely used to assess the comorbidities. 16 In computing the GIC based on the presence and severity of clinical conditions, each clinical condition is graded on a 0–4 disease severity scale on the basis of the following general framework: 0 = absence of disease, 1 = asymptomatic disease, 2 = symptomatic disease requiring medication but under satisfactory control, 3 = symptomatic disease uncontrolled by therapy, and 4 = life-threatening or the most severe form of the disease. The GIC classifies patients into four classes of increasing comorbidity. Class 1 includes patients who have one or more conditions with a disease severity grade equal to or lower than 1. Class 2 includes patients who have one or more conditions with a disease severity grade of 2. Class 3 includes patients who have one condition with a disease severity of 3, other conditions having a disease severity equal to or lower than 2. Class 4 includes patients who have two or more conditions with a disease severity of 3 or one or more conditions with a disease severity of 4.
Follow-up
Follow-up started on the first day of admission. In-hospital mortality and overall survival were also recorded. In-hospital mortality was defined as any death occurring within 30 days that was considered to be the consequences of AMI or comorbidities complications. The overall survival was defined as the time between data of admission and the data of death from any cause or last follow-up visit. Survivors were followed in the outpatient clinic, checking their actual conditions (alive, dead, or missing) by reviewing the medical records or the general hospital database or by contacting patients’ families by telephone. The follow-up cut-off date was 30 June 2020. The mean follow-up time was 15.05 ± 18.02 months with a minimal observation of 0.3 months and a maximum of 58 months.
Statistical analysis
Statistical analyses were performed using SPSS 22.0 software (SPSS Inc., Chicago, USA), p ≤ 0.05 was considered significantly different. Categorical variables were expressed as percentages. Continuous variables were expressed as mean with standard deviation (SD). The percentages of 30-day mortality were calculated. One-year, 2-year, and 3-year survival rates, and average overall survival time were estimated by the Kaplan–Meier method. The relation of 30-day mortality to GIC was calculated by using Pearson χ2 test. Univariate analysis was performed for the relations of age, duration of symptoms before admission, comorbidities, and GIC to the average overall survival using a log-rank test, risk factors of p ≤ 0.05 were selected for cox proportional-hazards regressions analysis. The results of cox proportional-hazards regressions analysis were expressed as hazard ratio (HR) with 95% CI and corresponding p value.
Results
Patients’ characteristics
Clinical characteristics and treatment options of 40 patients with AMI due to SMA thromboembolism.

Note. *2 patients were admitted to hospital for acute cerebral infarction, concurrent acute mesenteric ischemia was presented during hospitalization.
Among 19 patients (47.5%) who underwent mesenteric recanalization by endovascular therapy (Figure 1), 5 patients underwent bowel resection because of worsening symptoms, suspicion of intestinal necrosis after mesenteric arterial recanalization treatment. Other patients’ treatment options are listed in Table 1. The treatment options were selected by attending physicians based on clinical manifestations, laboratory tests, and imaging findings. After mesenteric recanalization procedures, all patients were transferred into the surgical intensive care unit (ICU) receiving resuscitation, antibiotic therapy, multiple-organ function support, and nutrition therapy. A 72-year-old male patient with hypertension, arterial fibrillation, old cerebral infarction, and chronic obstructive pulmonary disease of more than 10 years, presented with acute onset of abdominal pain, nausea and vomiting, and hematochezia. Multidetector computed tomography coronal image of the abdomen (a) shows small bowel wall thickening (arrows), curved planar reformation (b) and volume rendering (c) images show superior mesenteric artery (SMA) embolism (arrows). Digital subtraction angiography (DSA) (d) after stent placement shows that a bare-metal stent (5 mm × 30 mm) (arrows) is placed in the previously occluded SMA segment, and the SMA and its branches are now patent.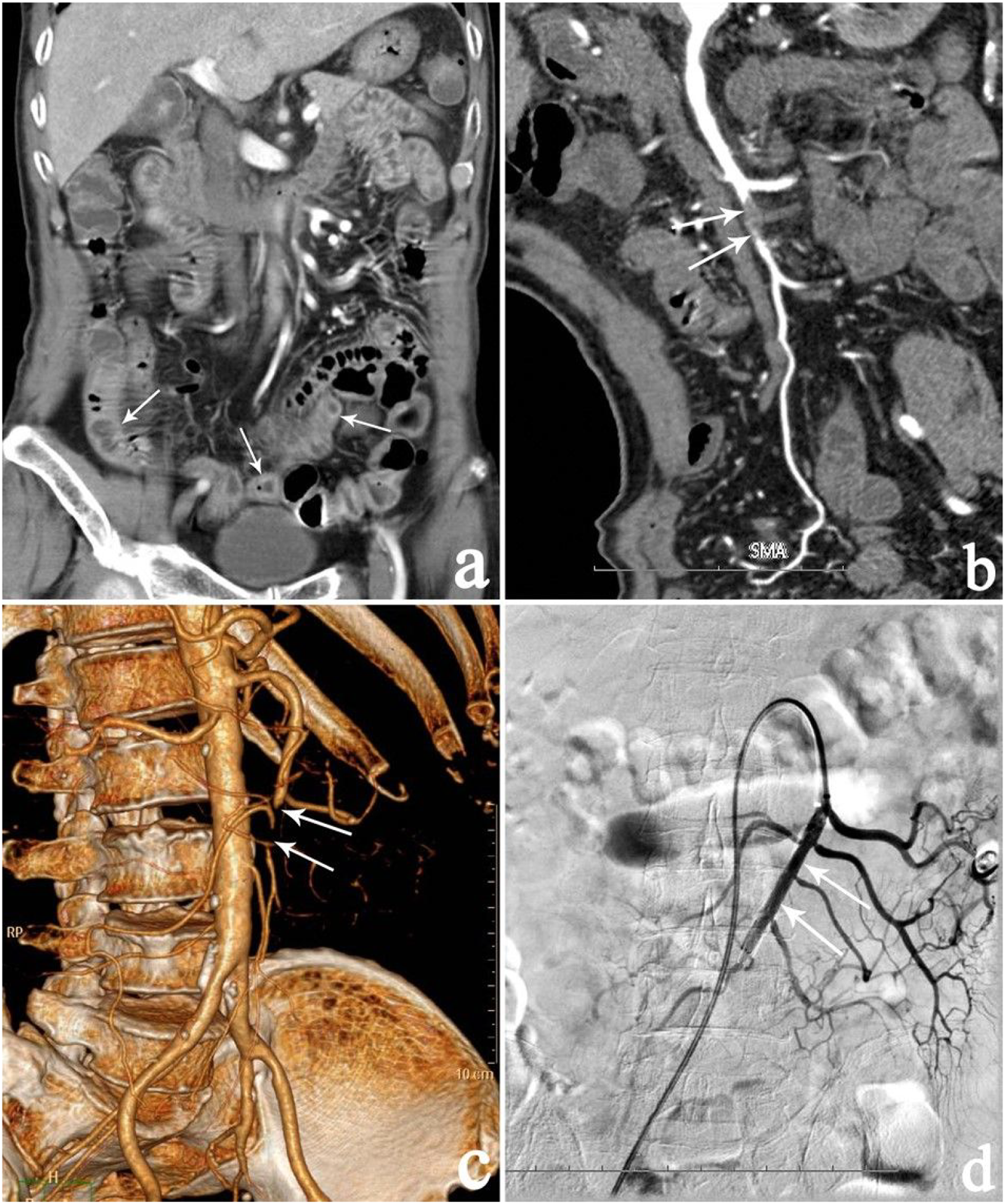
Comorbidities and GIC classification
The impact of age, duration of symptoms before admission, comorbidities on average overall survival of the 40 patients with AMI due to SMA thromboembolism.
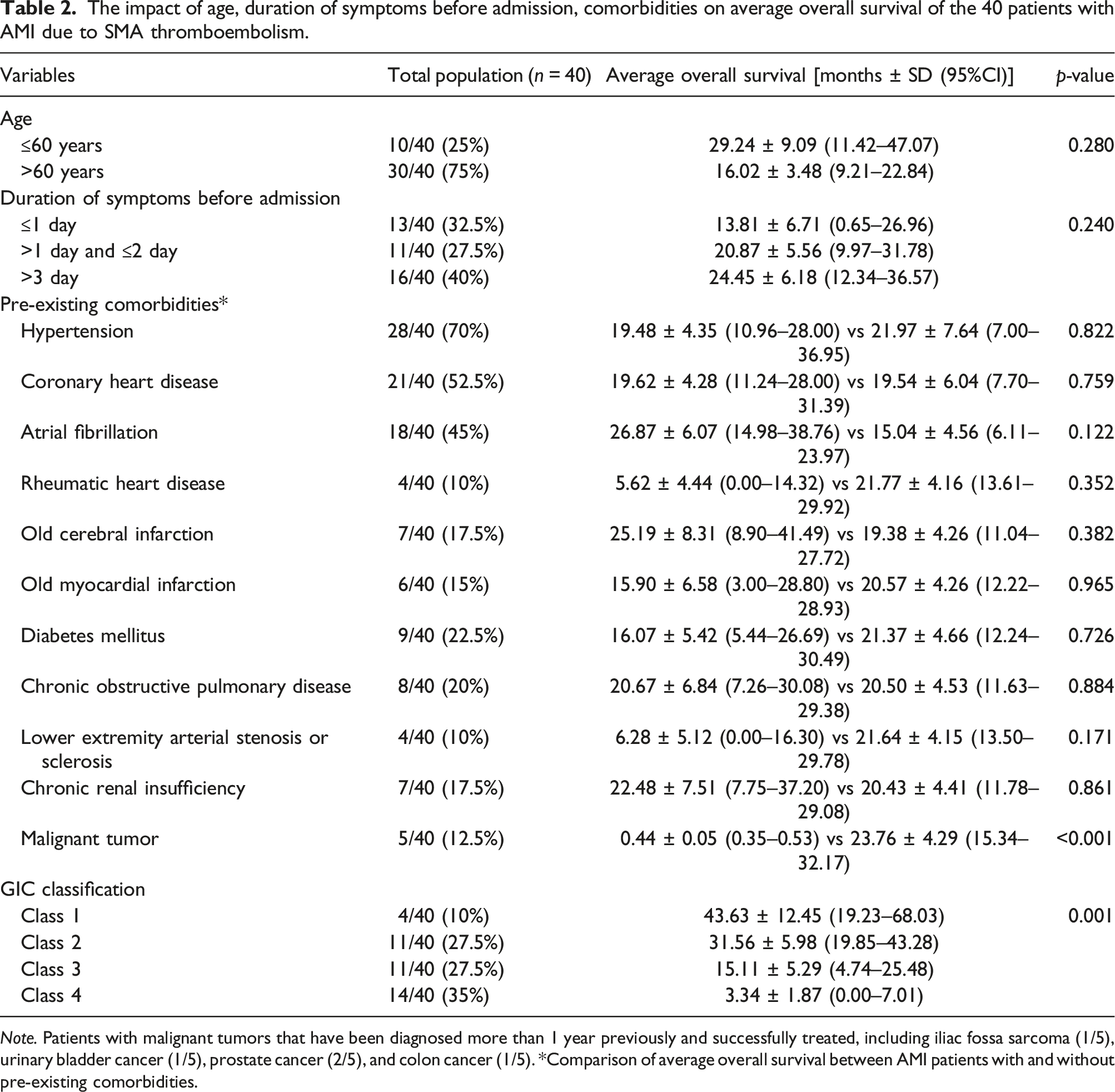
Note. Patients with malignant tumors that have been diagnosed more than 1 year previously and successfully treated, including iliac fossa sarcoma (1/5), urinary bladder cancer (1/5), prostate cancer (2/5), and colon cancer (1/5). *Comparison of average overall survival between AMI patients with and without pre-existing comorbidities.
The relations of comorbidities and GIC classification to mortality and average overall survival time
The follow-up results for the 40 patients with AMI due to SMA thromboembolism between February 2013 and December 2019 were listed as follows: 21 patients died within 30-day, including 4 patients in 2013, 4 patients in 2014, 5 patients in 2015, 1 patient in 2016, 1 patient in 2017, 3 patients in 2018, and 3 patients in 2019; 1 patient in 2018 died within 1 year; 3 patients in 2013 and 1 patient in 2017 died within 2 years; 1 patient in 2013 and 1 patient in 2015 died within 3 years. Two patients in 2015 and 2 patients in 2016 were followed up to 2019 year, and 1 patient in 2016 was followed up to 2018 year, and then the follow-up was lost. The rest of patients survived during follow-up to Jun 2022. Overall, in-hospital mortality rate was 52.5% (21/40), the mortality within 1-year, 2-year, and 3-year were 2.5% (1/40), 10% (4/40), and 5% (2/40), respectively. The causes of mortality included septic shock, multiple-organ failure, stroke, cardiac arrest, or/and cardiac failure. One-year, 2-year, and 3-year overall estimated survival rates by the Kaplan–Meier method were 45%, 34%, and 26.5%, respectively, and the average overall survival time was 20.84 ± 3.95 (95% CI: 13.10–28.58) months (Figure 2). Estimated overall cumulative survival in patients with AMI due to SMA thromboembolism (Kaplan–Meier method).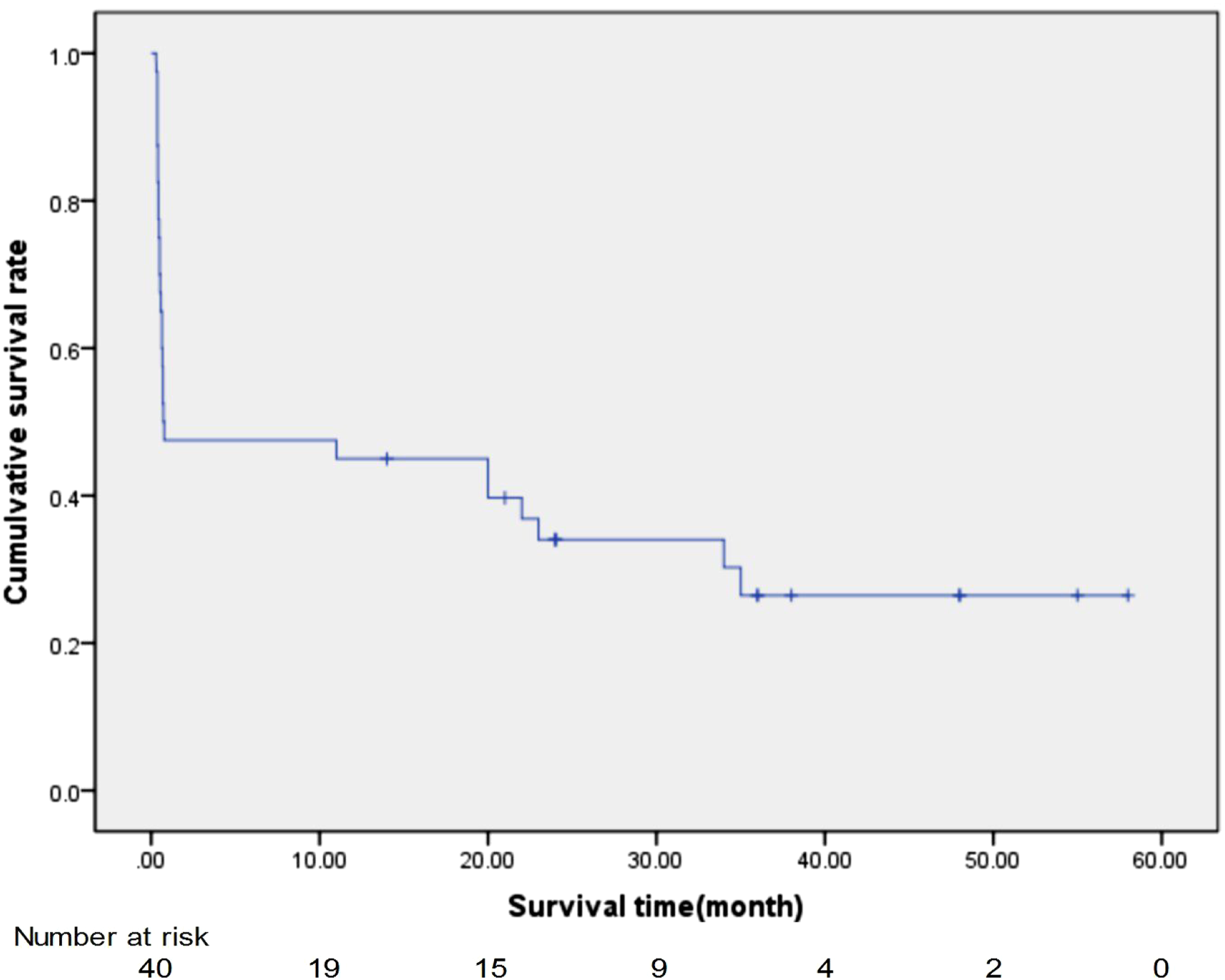
Among these pre-existing comorbidities, only malignant tumor was significantly related to the average overall survival time (log-rank test, p < 0.001), AMI patients with the medical history of malignant tumors had a shorter average overall survival time (0.44 ± 0.05 months (95% CI: 0.35–0.53 months)) than that of AMI patients without the medical history of malignant tumors (23.76 ± 4.29 months [95% CI: 15.34–32.17 months]) (Table 2). The age and duration of symptoms before admission were not significantly related to the average overall survival time (Table 2).
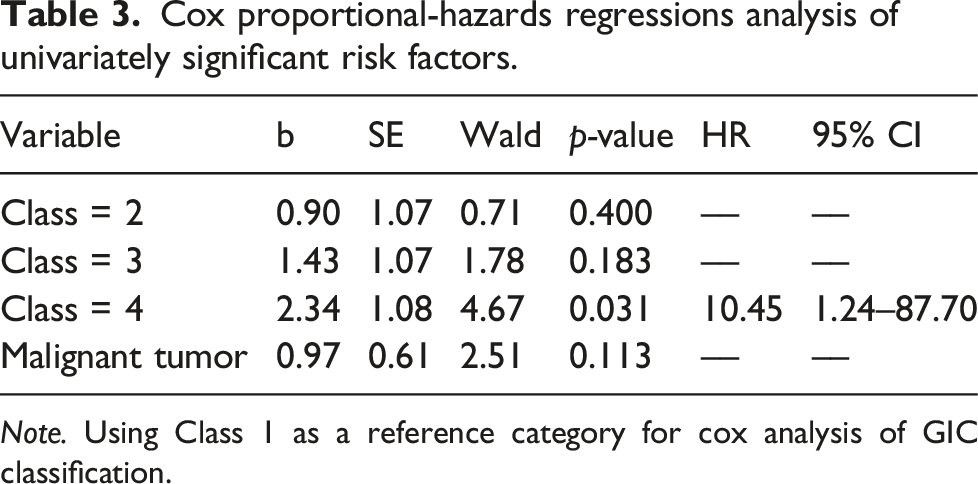
According to the GIC classification, in-hospital mortality rates of AMI patients with class 1, class 2, class 3, and class 4 were 25%, 27.3%, 54.5%, and 78.6%, respectively. The average overall survival time of AMI patients with class 1, class 2, class 3, and class 4 were 43.63 ± 12.45 months (95% CI: 19.23–68.03 months), 31.56 ± 5.98 months (95% CI: 19.85–43.28 months), 15.11 ± 5.29 months (95% CI: 4.74–25.48 months), and 3.34 ± 1.87 months (95% CI: 0.00–7.01 months) respectively (Table 2, Figure 3).GIC classification was significantly related to the in-hospital mortality (Pearson χ2 = 7.86, p = 0.049) and average overall survival time (log-rank, p = 0.001). AMI patients with higher GIC classification had higher in-hospital mortality rates and lower average overall survival time. Cox proportional-hazards regressions analysis showed that class 4 of comorbidities was an independent prognostic factor of mortality (p = 0.031, HR = 10.45 [95% CI: 1.24–87.70]) (Table 3). Kaplan–Meier survival analysis for patients with AMI due to SMA thromboembolism using the Geriatric Index of Comorbidity (p = 0.001) (log-rank test). Cox proportional-hazards regressions analysis of univariately significant risk factors. Note. Using Class 1 as a reference category for cox analysis of GIC classification.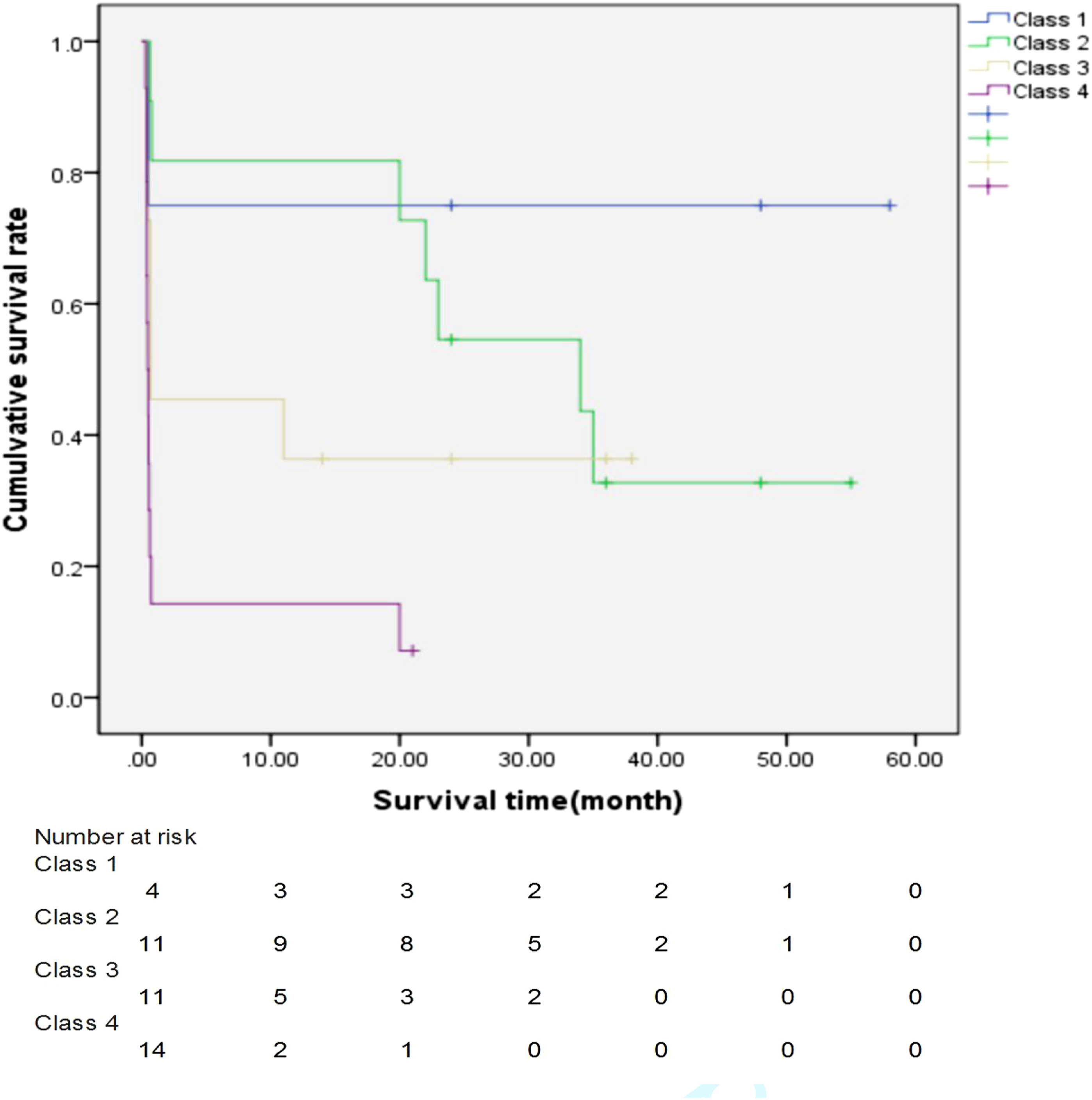
Discussion
AMI due to SMA thromboembolism is an uncommon condition of high mortality and morbidity, early diagnosis and timely modern management are key steps to improve clinical outcomes but still made slight progress in reducing mortality in the past decade. 17 The reasons are as follows: higher prevalence of AMI due to SMA thromboembolism in elderly people, most of elderly AMI patients with comorbidities, and unspecific complaints may lead to delayed diagnosis and treatment.4–6 Any delay in diagnosis and treatment remains the greatest challenge to reducing morbidity and mortality of AMI patients. Comprehensive knowledge and individual management of comorbidities in AMI patients may be one of the clinical pathways to improve the clinical outcome. 18
The term of comorbidities has historically been defined as any distinct additional clinical entity that has existed. 15 Starfield defines comorbidity as the simultaneous presence of multiple conditions when there is an index disease and other unrelated conditions. 19 Also, a broader view has been proposed that recognizes that a patient may suffer from multiple diseases, termed multimorbidity. 20 Much of the discussion about the accuracy of the term revolves around which of the multiple conditions ought to be the primary or index disease and the manner in which these multiple conditions interact. 21 Lin et al. had previously reported that SMA calcification has a strong association with major cardiovascular risk factors, cardiovascular mortality, and all-cause mortality. 22 Also, hypertension and diabetes mellitus are the risk factors of atherosclerosis; the process of atherosclerosis can affect all vascular beds, such as renal artery, coronary artery, peripheral artery et al. On the basis of atherosclerosis, the development of SMA thrombosis and stenosis can induce acute mesenteric ischemia. Hence, Hypertension, cardiovascular and cerebrovascular diseases, diabetes mellitus, and other comorbidities correlate with SMA thromboembolism to some extent and affect the prognosis of AMI.1,8–10
In pre-existing comorbidities, our study results revealed that AMI patients with malignant tumors have a significantly shorter average overall survival time than that of AMI patients without malignant tumors, other pre-existing comorbidities impacted little on mortality. Although these malignant tumors had been diagnosed more than 1 year previously and successfully treated, malignant tumors related to systemic inflammation and local immune response may contribute to the development and progression of AMI, and the shorter average overall survival time and higher mortality rates. 23 Elderly AMI patients with malignant tumors can make diagnosis and therapy more difficult, and prone to occur complications during the clinical course. The complex interplay between AMI and malignant tumors is also unknown and needs to be further study.
While multiple comorbidity indices are available, each with unique advantages and disadvantages, no single index has emerged as clearly superior to the others. 24 Dina Zekry et al. reported that the GIC classifies patients based on increasing somatic comorbidities and takes into account disease severity, the index remained statistically significant and the best predictor for death for elderly patients with acute disease.16,24 Hence, we selected GIC as classification system for elderly AMI patients’ comorbidities. Our results revealed that AMI patients with higher GIC classification had higher in-hospital mortality rates and lower average overall survival time. And class 4 of comorbidities was an independent prognostic factor of mortality (p = 0.031, HR = 10.45 [95% CI: 1.24–87.70]). For AMI patients with more serious comorbidities, it is difficult to manage for attending physicians because of the high risk of complications and mortality in the emergency settings. In the future, personalized management strategies may improve the prognosis of AMI patients with comorbidities according to the GIC classification.
In addition, it is important to note that concurrent cerebrovascular and SMA thromboembolism occurred in 2 patients in our study samples. Autopsy findings in 213 patients with acute thromboembolic occlusion of the SMA verified that concurrent acute embolism was present in 7.9% of AMI patients, including acute ischemic stroke, acute myocardial infarction, or lower extremities embolism. 25 Concurrent acute embolism affecting other arterial segments than the SMA can occur in arterial occluded AMI patients with coexisting heart disease, for example, atrial fibrillation, rheumatic heart. Usually, concurrent the branches of SMA and celiac trunk embolism are common in arterial occluded AMI patients, leading to splenic, renal, and hepatic focal infarction. Concurrent cerebrovascular and cardiovascular embolism in patients with AMI due to SMA thromboembolism is rare in clinical practice. However, attending physicians must be aware of the occurrence of concurrent acute embolism because of the high in-hospital mortality rates.
Our study has some limitations. First, a major limitation of this study is the retrospective nature of the data. Second, the patient sample size is small, and the enrolled AMI patients in our study sampling might behave different clinical characteristics from other AMI patient groups. It is likely that it is difficult to generalize its conclusion to all AMI patient groups. Third, our study included a highly heterogeneous cohort treated over a comparably long period of time. This may cause differences regarding mortality and survival between ours and other studies. However, because of the emergency setting in which most AMI patients are seen with individually tailored multidisciplinary approaches, it is difficult to perform evidence-based treatment of the disease.
In conclusion, Comorbidity is common and an important factor associated with all-cause mortality in AMI patients due to SMA thromboembolism. The GIC is an available clinical tool to quantify the severity of comorbidities. 16 In managing AMI patients, we recommend a timely diagnosis of both AMI condition and its associated comorbidities. The effective management of comorbidities in AMI patients is the selection of reliable measurement tools that can assist in improving clinical outcomes.
Footnotes
Acknowledgments
The authors thank computed tomography technician Qisheng Ran and Xinhui Li for their help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Clinical Research Centre of Imaging and Nuclear Medicine (CSTC2015YFPT-gcjsyjzx0175), Scientific research development project of Affiliated Hospital of North Sichuan Medical College (2021-210832), and the opening project of Medical Imaging Key Laboratory of Sichuan Province (MIKLSP202006).
Ethical statement
This study was approved by the ethics board of our institution and the requirement to obtain informed consent was waived because of the retrospective design.
