Abstract
Novel coronavirus 2019 (COVID-19) represents a significant risk factor for the development of venous thromboembolism (VTE) in hospitalized with both moderate and severe/critical COVID-19. Herein, we present a brief updated review on emerging robust data on diverse thromboprophylaxis strategies used to mitigate VTE complications, as well as a personal point of view of current controversies in regards the use of therapeutic and prophylactic anticoagulation strategies, particularly in the moderately-ill subgroup of patients with COVID-19.
Background
Novel coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) is clearly associated with a significant risk factor for the development of venous thromboembolism (VTE) and/or arterial thromboembolic events (ATE). Pooled rates of VTE were higher among severe/critically ill COVID-19 hospitalized in the intensive care unit (ICU) in comparison to patients with moderate COVID-19 hospitalized in the medical wards (prevalence rate of 27.9% vs 7.1%).1,2 COVID-19 associated coagulopathy (CAC) (also known as thromboinflammation or immunothrombosis) is a consequence of a very complex pathobiological process, in which there is an important interplay between endothelial cell activation, dysfunction, and injury, hyperinflammation due to a dysregulated cytokine-storm, and a prothrombotic state at both, micro and macro-vascular environments.2–4
It is well known from a plethora of studies that pharmacological thromboprophylaxis reduces the risk of in-hospital VTE in acutely-ill medical patients, despite no net benefit on mortality. 5 Thromboprophylaxis strategies with diverse regimens of heparins in hospitalized patients with COVID-19 has been an important arena for ongoing clinical research in the last 2 years, with the aim of understanding the potential role of systemic anticoagulation (AC) in a complex and challenging clinical scenario. An important unclear issue is which is the best thromboprophylaxis regimen for a given hospitalized patient with COVID-19. In this article we provide a critical review of the available literature on this topic, considering the recommendations recently published by the American College of Chest Physicians (ACCP) and the National Institutes of Health (NIH) in the U.S.6,7
List of selected, randomized trials tdat evaluated tdromboprophylaxis strategies in hospitalized patients witd COVID-19.
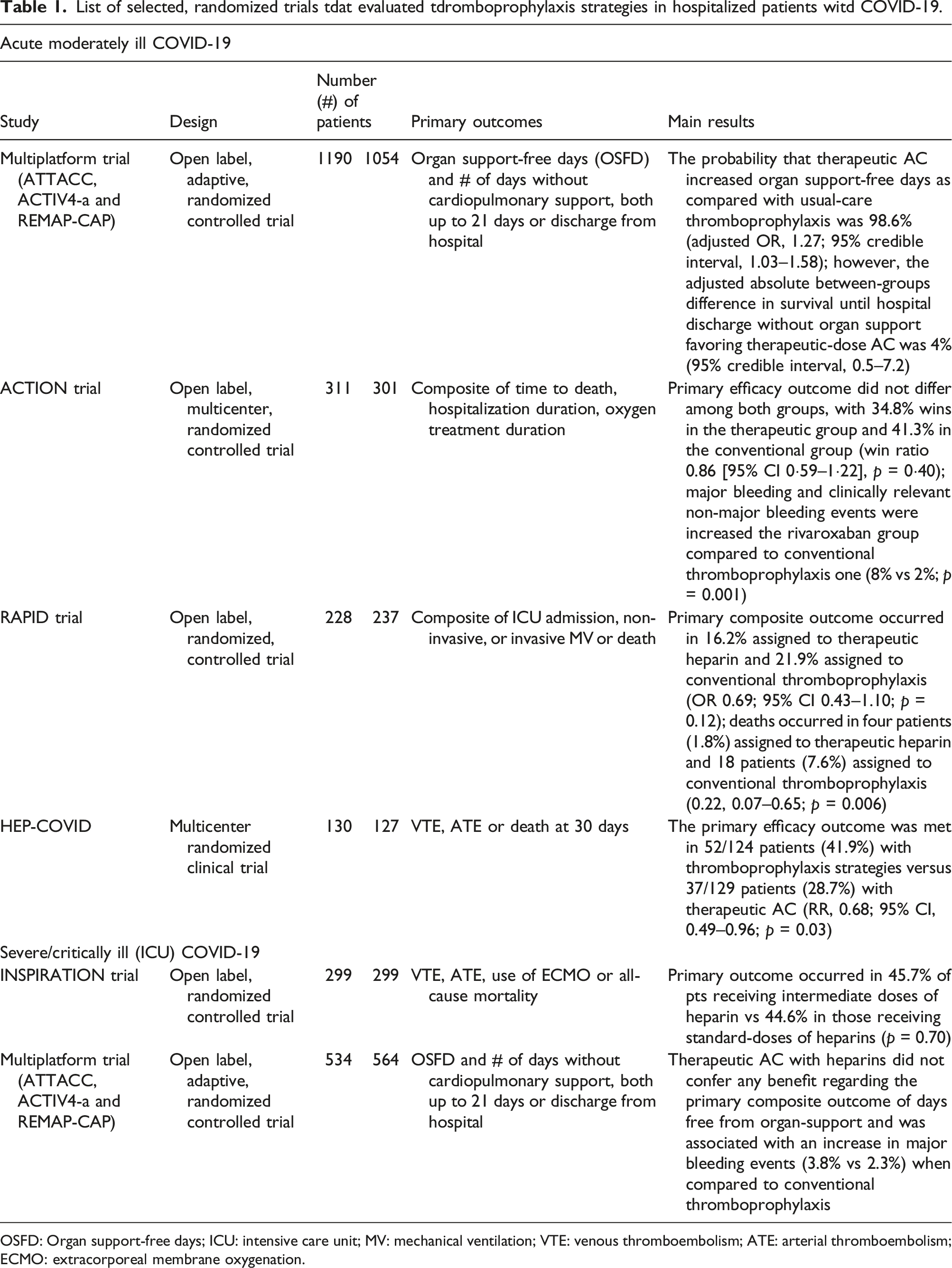
OSFD: Organ support-free days; ICU: intensive care unit; MV: mechanical ventilation; VTE: venous thromboembolism; ATE: arterial thromboembolism; ECMO: extracorporeal membrane oxygenation.
Strategies for moderate (non-critical) COVID-19
The multiplatform, open-label, prospective randomized trial performed by the ATTACC, ACTIV-4a, and REMAP-CAP investigators included 2,219 patients with moderate COVID-19 (defined by as an absence of requiring ICU-level of care, by means of any level organ support, like high-flow oxygenation, non-invasive or invasive mechanical ventilation (MV), new need for hemodialysis or vasopressors/inotropes. The primary composite outcome was the probability of the intervention resulting in less organ support-free days, combining in-hospital death and number of days free of cardiopulmonary support up to day 21 among patients who survived until hospital discharge. Patients were randomized to either therapeutic doses of heparin (94% received LMWH, mostly enoxaparin) or conventional prophylaxis doses of heparin (71.6% received conventional doses of and 26.5% intermediate-dose) and stratified by baseline D-dimer levels. The study was stopped when the pre-specified criteria for superiority of therapeutic AC were met. The probability that therapeutic AC increased organ support-free days as compared with usual-care thromboprophylaxis was 98.6% (adjusted OR, 1.27; 95% credible interval, 1.03–1.58). However, the adjusted absolute between-groups difference in survival until hospital discharge without organ support favoring therapeutic-dose AC was 4.0 percentage points (95% credible interval, 0.5–7.2). There were no differences in the different strata according to D-dimer levels. Overall rates of major bleeding events were 1.9% in the therapeutic AC arm versus 0.9% in the conventional thromboprophylaxis one. Investigators concluded that in moderate COVID-19 patients, the initial strategy of therapeutic AC regimen with heparins increased the probability of surviving to hospital discharge with reduced utilization of cardiorespiratory organ-support when compared to conventional usual-care thromboprophylaxis. 8
Lopes et al. reported the results of the ACTION trial; 9 615 patients from Brazil with confirmed COVID-19 and elevated D-dimer, primarily with non-critical moderate disease, were randomized to receive therapeutic doses of rivaroxaban (20 mg daily or 15 mg daily if renally adjusted) or conventional thromboprophylaxis with either LMWH or UFH. The primary composite end-pointed consisted of time of death, duration of hospitalization, and duration of supplemental oxygen using the win ratio method. The primary efficacy outcome did not differ among both groups, with 34.8% wins in the therapeutic group and 41.3% in the conventional group (win ratio 0.86 [95% CI 0.59–1.22], p = 0.40); consistent results were observed across both groups, moderate COVID-19 (94%) and in critically ill COVID-19 patients (6%). However, major bleeding and clinically relevant non-major bleeding events were increased the rivaroxaban group compared to conventional thromboprophylaxis one (8% vs 2%; p = 0.001); investigators concluded that in patients hospitalized with COVID-19 and elevated D-dimer concentration, therapeutic doses of rivaroxaban did not improve clinical outcomes and increased bleeding events when compared with conventional thromboprophylaxis using heparins; therefore, the use of rivaroxaban, and other direct oral anticoagulants (DOACs), should be avoided in these patients. 9
The RAPID trial included 465 patients with moderate COVID-19 with elevated D-dimers >2 times the ULN, randomly assigned to heparin at therapeutic- (n = 228) or prophylactic (n = 237) doses; 10 the primary composite endpoints were death, invasive and non-invasive MV, or admission to an ICU up to 28 days or until hospital discharge; secondary outcomes included all-cause death, the composite of all-cause death or any MV, and VTE. The primary composite outcome occurred in 16.2% assigned to therapeutic heparin and 21.9% assigned to prophylaxis (OR 0.69; 95% CI 0.43–1.10; p = 0.12); deaths occurred in four patients (1.8%) assigned to therapeutic heparin and 18 patients (7.6%) assigned to prophylactic heparin (0.22, 0.07–0.65; p = 0.006); Major bleeding occurred in 0.9% assigned to therapeutic heparins and 1.7% in thromboprophylaxis heparins (0.52, 0.09–2.85; p = 0.69); concluding that therapeutic AC was not significantly associated with a reduction in the primary outcome but the odds of death at 28 days were lower, with an acceptably low risk of major bleeding events. 10
The HEP-COVID trial randomized 257 patients with elevated D-dimer levels >4 times ULN or objective evidence of CAC to either conventional thromboprophylaxis or intermediate-doses and therapeutic doses of heparins; 11 efficacy outcomes were VTE, ATE, or death from any cause; safety outcome was major bleeding events. 83 (32.8%) patients were categorized as critically ill COVID-19. The primary efficacy outcome was met in 52/124 patients (41.9%) (28.2% VTE, 3.2% ATE, 25.0% death) with thromboprophylaxis strategies versus 37/129 patients (28.7%) (11.7% VTE, 3.2% ATE, 19.4% death) with therapeutic AC with heparins (RR, 0.68; 95% CI, 0.49–0.96; p = 0.03); additionally, the primary efficacy outcome was reduced in non-ICU patients (36.1% vs 16.7%; RR, 0.46; 95% CI, 0.27–0.81; p = 0.004) but not in ICU patients (55.3% vs 51.1%; RR, 0.92; 95% CI, 0.62–1.39; p = 0.71); concluding that therapeutic AC with heparins reduced major thromboembolic events and death; however, such treatment effects were not seen in critically-ill COVID-19. 11
Strategies for severe/critical (ICU) COVID-19
The INSPIRATION trial compared intermediate doses of LMWH with conventional thromboprophylaxis in 562 patients being admitted to the ICU, with follow-ups up to 90 days, irrespective of hospital discharge or not. The study did not show any difference in the primary composite outcome of adjudicated acute VTE, ATE, need for ECMO, or death (45.7% vs 44.1%; OR 1.06; p = 0.70). Major bleeding occurred in 2.5% in the intermediate-dose group and 1.4% in the conventional thromboprophylaxis group, with significant thrombocytopenia noted in the intermediate-dose group as well (p = 0.01).12,13
The multiplatform trials ATTACC, ACTIV-4a and REMAP-CAP investigators found that therapeutic AC with heparins did not confer any benefit in regards to the primary composite outcome of days free from organ-support, and also was associated with an increase in major bleeding events (3.8% vs 2.3%) when compared to conventional usual-care thromboprophylaxis in 1098 patients. 14 Recently the American Society of Hematology (ASH) issued a conditional recommendation in favor of conventional thromboprophylaxis over intermediate-dose AC in patients with severe/critical COVID-19 patients who do not have confirmed or suspected VTE. 15
Discussion and future directions
Based on the previous studies, the most recent guidelines from the ACCP recommend that in patients with acutely, moderately ill COVID-19, and acceptable/low bleeding risk, the use of therapeutic AC with heparins (UFH or LMWH) over conventional usual thromboprophylaxis (conditional recommendation, ungraded consensus-based statement) Critically ill COVID-19 patients should receive conventional thromboprophylaxis; they saw no role for intermediate-doses of AC in both clinical scenarios. 6
Risk factors and clinical predictors for bleeding events in hospitalized patients with COVID-19.

ICU: Intensive care unit; INR: international normalized ratio; ULN: upper limit of normal; aPTT: activated partial thromboplastin time; TA: therapeutic anticoagulation; GFR: glomerular filtration rate; sCr: serum creatinine.
Additionally, DOACs are not currently indicated for their use in hospitalized medical patients, with or without COVID-19. In this regard, results from the ongoing FREEDOM trial (NCT04512079), which is evaluating the effectiveness and safety of enoxaparin and apixaban in hospitalized patients with COVID-19 that are not requiring MV yet may be of great value in addition to current available data. 17
Recently, the American Society of Hematology (ASH) in their most recent updated clinical living guidelines, issued a conditional recommendation, favoring therapeutic AC over conventional thromboprophylaxis in moderately-ill COVID-19 patients who do not have confirmed VTE; however, such recommendation was based on very-low certainty of clinical evidence, emphasizing the need for further high-quality, prospective studies. 18 Moreover, the AC Forum, in their 2022 updated guidelines, have suggested to consider therapeutic AC in moderately-ill COVID-19 patients at increased risk for VTE, and who are not at high risk for AC-related bleeding events. 19
The International Society for Thrombosis and Haemostasis (ISTH) recently published its guidelines of antithrombotic therapies in hospitalized patients with COVID-19; the panel issued a Class-I, level of evidence A recommendation that in select moderately-ill COVID-19 patients, therapeutic AC with either LMWH or UFH is beneficial in preference to conventional thromboprophylaxis or intermediate-doses of LMWH or UFH to reduce the risk of VTE and end-organ failure. 20
Another unresolved issue is the potential impact of anti-inflammatory and antiviral strategies currently used like systemic corticosteroids, tocilizumab, remdesivir, and more recently nirmatrelvir/ritonavir, and sotrovimab. It is very possible that these therapies may reduce thromboembolic complications, by modifying the thromboinflammatory response in COVID-19 patients. This might be an additional confounding factor in interpreting the available information given that therapeutic AC regimens were not utilized consistently by the time robust data was collected.
An issue complicating clinical decisions is the fact that extensively validated and easily applicable risk assessment models for VTE in medical patients are scarce, were developed many years ago, and could not be applicable to COVID-19 patients. In the case of bleeding risk assessment models, these are even less explored.21,22 To date, the VTE risk scores published for COVID-19 patients are quite diverse, population dependent, not well validated, and primarily focused on patients with severe/critical disease, thus limiting the recommendations for their use of a specific one in general and particularly for moderately ill patients. 21
Based on the previous considerations, it is the personal point of view of the authors that for now, and despite emerging data, the use of intermediate or therapeutic doses of AC in non-critically ill patients should still be done within the context of rigorous prospective clinical trials. In some instances, if consideration is given to this strategy, it should be based on individualizing case by case scenarios, comprehensively assessing thromboembolic and bleeding risks, discussing complex cases in a multidisciplinary fashion, and obtaining and documenting informed consent from patients or their families since from our perspective it should not be a standard recommendation yet.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
