Abstract
Purpose
Surgical revascularization is the standard treatment for chronic limb-threatening ischemia (CLTI). However, some patients may require reintervention. The Global Anatomic Staging System (GLASS), which evaluates the complexity of infrainguinal lesions, was proposed. This study aimed to identify predictors for graft revision and evaluate whether GLASS impacts vein graft revision.
Methods
Between 2011 and 2018, CLTI patients who underwent de novo infrapopliteal bypass using autogenous veins were retrospectively analyzed. To assess anatomic complexity with GLASS, femoropopliteal, infrapopliteal, and inframalleolar/pedal (IM) disease grades were determined. The outcomes of patients with or without graft revision were compared. Cox regression analysis was performed.
Results
Thirty-six of the 80 patients underwent reintervention for graft revision. Compared to the non–graft revision group, the graft revision group exhibited significantly higher rates of GLASS stage III (66% vs 81%, p = 0.046) and grade P2 IM disease (25% vs 58%, p = 0.009). Multivariate analysis revealed that IM grade P2 (hazard ratio [HR], 3.35; 95% confidence interval [CI], 1.66–6.75; p = 0.001) and spliced vein grafts (HR, 3.18; 95% CI, 1.43–7.06; p = 0.005) were significantly associated with graft revision.
Conclusions
This study demonstrated that IM grade P2 and spliced vein grafts were predictors of graft revision. The GLASS stratification of IM disease grade may be useful in optimizing treatment for CLTI.
Introduction
Chronic limb-threatening ischemia (CLTI) is associated with amputation, mortality, and impaired quality of life, and is a condition that requires revascularization. 1 Surgical bypass with an autogenous vein has been the standard treatment for CLTI, while endovascular therapy (EVT) has been frequently performed because it is minimally invasive. Some studies have provided recommendations on selecting surgical bypass versus EVT, taking into consideration patient comorbidities, limb condition severity, anatomical features of the arterial disease, and vein availability.1–3
To achieve evidence-based revascularization, the Global Vascular Guidelines (GVGs) proposed evaluating patients with CLTI in terms of clinically important factors such as patient risk, limb condition severity, and arterial anatomic complexity. 1 Additionally, the GVG proposed a Global Anatomic Staging System (GLASS) that involves defining a preferred target artery path (TAP) and then estimating limb-based patency, leading to three stages of arterial anatomic complexity for endovascular intervention. 1
Surgical bypass using an autogenous vein for infrainguinal arterial reconstruction is preferred because of its durability regarding revascularization, especially in patients with a good prognosis and ambulatory status. However, some patients undergo graft revision procedures due to failed or failing grafts after initial bypass surgery. Lower extremity arterial bypass occlusion remains a challenging problem. Since failed grafts, specifically graft occlusion, may lead to major amputation,4,5 it is important to maintain long-term patency of vein grafts. Graft revision techniques include excision with primary anastomosis, vein patch angioplasty, interposition grafting, sequential or jump grafting, and percutaneous transluminal angioplasty. It is important for vascular surgeons to recognize predictors of graft revision and to determine risk stratification prior to surgical bypass. However, little information is available on the relationship between vein graft revision and GLASS. The purpose of this single-center, retrospective study was to evaluate predictors of graft revision and to determine whether GLASS impacts vein graft revision in CLTI patients undergoing infrapopliteal bypass surgery.
Methods
Study population
This clinical investigation analyzed preoperative medical risk factors, surgical characteristics, and outcomes of 89 CLTI patients with ischemic ulcers or gangrene due to atherosclerotic disease who underwent de novo infrapopliteal bypass using autogenous veins at our institution from 2011 to 2018. One limb per patient was included in this study. In cases of bilateral CLTI, which was present in 15% (12/80) of patients, the first treated limb was included in the study. Patients whose surgeries were attributable to acute arterial occlusion (n = 1) or non-atherosclerotic diseases (n = 8) were excluded, and 80 patients were finally enrolled.
Data were collected using a prospectively registered database. Patient records were carefully reviewed retrospectively. This study was conducted in accordance with the Declaration of Helsinki. The Nagoya University School of Medicine institutional review board approved the study (approval number 2021-0151). All patients provided written informed consent prior to intervention and data collection.
Revascularization Procedures and Follow-up
Arterial revascularization was considered for all CLTI patients at our institution. Routine evaluation included laboratory tests, cardiovascular function (electrocardiography, coronary angiography, and echocardiography), respiratory function, and other parameters. The treatment of each patient was discussed among vascular surgeons and the best procedure was determined considering the patient’s comorbidities, ambulatory status, vein availability, foot wound status, and the anatomical features of arterial disease. Surgical bypass was preferred as a first-line treatment, particularly in patients with extensive tissue loss and infrapopliteal lesions. At our institution, we generally perform EVT for TransAtlantic Inter-Society Consensus (TASC) II A and B femoropopliteal lesions, and surgical bypass for TASCII C and D lesions.6,7 We do not perform bypass in patients with extensive infection above the ankle, those with severe dementia, or those who have been bedridden for a long period of time due to neurogenic deficits.
The technical details of the vein graft bypass procedure have been published previously.8,9 Generally, the distal anastomotic site was chosen based on the optimal runoff vessel to the ischemic wound according to the angiosome concept. However, if the vessel to the ischemic wound was diseased, a less diseased artery was selected regardless of the angiosome concept. Proximal anastomoses were performed with 6-0 polypropylene sutures, and distal anastomoses were performed with 7-0 or 8-0 polypropylene sutures. For distal anastomoses, we used the non-dissection method with pneumatic tourniquets to control blood flow. The great saphenous vein (GSV) was used as a first-choice conduit if its diameter was >2 mm, as determined by preoperative duplex scan imaging. If the GSV was not available, the small saphenous vein or an arm vein was used. If a single saphenous vein was too short, a spliced vein graft was created with 7-0 polypropylene sutures. To check for graft stenosis and anastomotic site problems, angiography was performed after completion of the anastomoses. Moreover, we assessed graft flow by ultrasonography using a linear transducer. The measurements were obtained with a transit time flowmeter (300-Series Flowmeter; Transonic System, Ithaca, NY, USA) after the completion of bypass grafting. Heparin was intravenously administered for 24 hours and prostaglandin E1 was also infused for several days postoperatively. All patients received antiplatelet drugs.
Routine follow-up consisted of postoperative visits every month for 3 months, followed by visits at 3-month intervals for 2 years and at 6-month intervals thereafter. Routine surveillance included ankle–brachial index, skin perfusion pressure, and duplex ultrasound measurements. Duplex scanning of the vein graft was the primary method for detecting occult graft lesions. A focal increase in graft velocity (peak systolic velocity >300 cm/s or peak systolic velocity ratio ≥4) was considered significant and was followed by arteriography and revision when appropriate.
Definitions
In this study, CLTI was defined by the presence of peripheral artery disease in combination with gangrene or tissue loss lasting >2 weeks. 1 Assessment of wound severity was performed using the Rutherford classification and the Wound, Ischemia, and Foot Infection (WIfI) classification system. Skin perfusion pressure (SPP) was routinely measured because ankle and toe pressures could not be evaluated due to intractable rest pain or a non-compressible artery secondary to severe calcification. 10 WIfI ischemia grades of 0, 1, 2, and 3 corresponded to SPPs of ≥60 mmHg, 41–59 mmHg, 31–40 mmHg, and ≤30 mmHg, respectively. 2 Patients were included in this study if they had limb-threatening atherosclerotic disease with mild-to-severe ulcers (W grade 1–3) and mild-to-severe ischemia (I grade 1–3) according to the WIfI classification system.
Coronary artery disease was defined as an abnormal result on coronary angiogram and a history of myocardial infarction or open or percutaneous coronary artery revascularization. Cerebrovascular disease was defined as a history of stroke and/or cerebral hemorrhage. Hypertension, dyslipidemia, and diabetes mellitus were diagnosed in patients receiving relevant active medical treatments or diet modification. A positive smoking history was defined as any prior smoking. Ambulatory status was defined by the patient’s functional status immediately prior to initial surgery. Patients were considered non-ambulatory if they were wheelchair-bound or bedridden, or if they were able to walk with a cane or by themselves preoperatively but who later could not walk owing to ischemic limb symptoms. Major amputation was defined as amputation above the ankle. Amputation data were obtained from outpatient clinic follow-up contact. Wound healing was defined as complete epithelialization of wounds without death or major amputation.
Global Anatomic Staging System
Digital subtraction angiography of each arterial segment was routinely performed before infrainguinal bypass surgery. In accordance with a recent report,
1
after the TAP was identified, femoropopliteal and infrapopliteal arterial segments were graded on a scale of 0 to 4, and the combination of these segmental grades was assigned to three overall GLASS stages (I to III) for each limb using a consensus-based matrix. Additionally, pedal grades were determined (P0 to P2) to define the status of the inframalleolar arteries. GLASS considers limbs to be patent if there is flow through the TAP, which is defined as the physician-preferred route from the superficial femoral artery to the foot.
1
Examples with angiography of how inframalleolar/pedal disease grades P0, P1, and P2 look like are shown in Figure 1. Inframalleolar/pedal pedal grade. (a) P0: Complete pedal arch. Patent plantar artery and patent arteria dorsalis pedis. (b) P1: Incomplete pedal arch. Either of plantar artery or patent arteria dorsalis pedis was patent. (c) P2: No pedal arch. Occluded plantar artery and occluded arteria dorsalis pedis.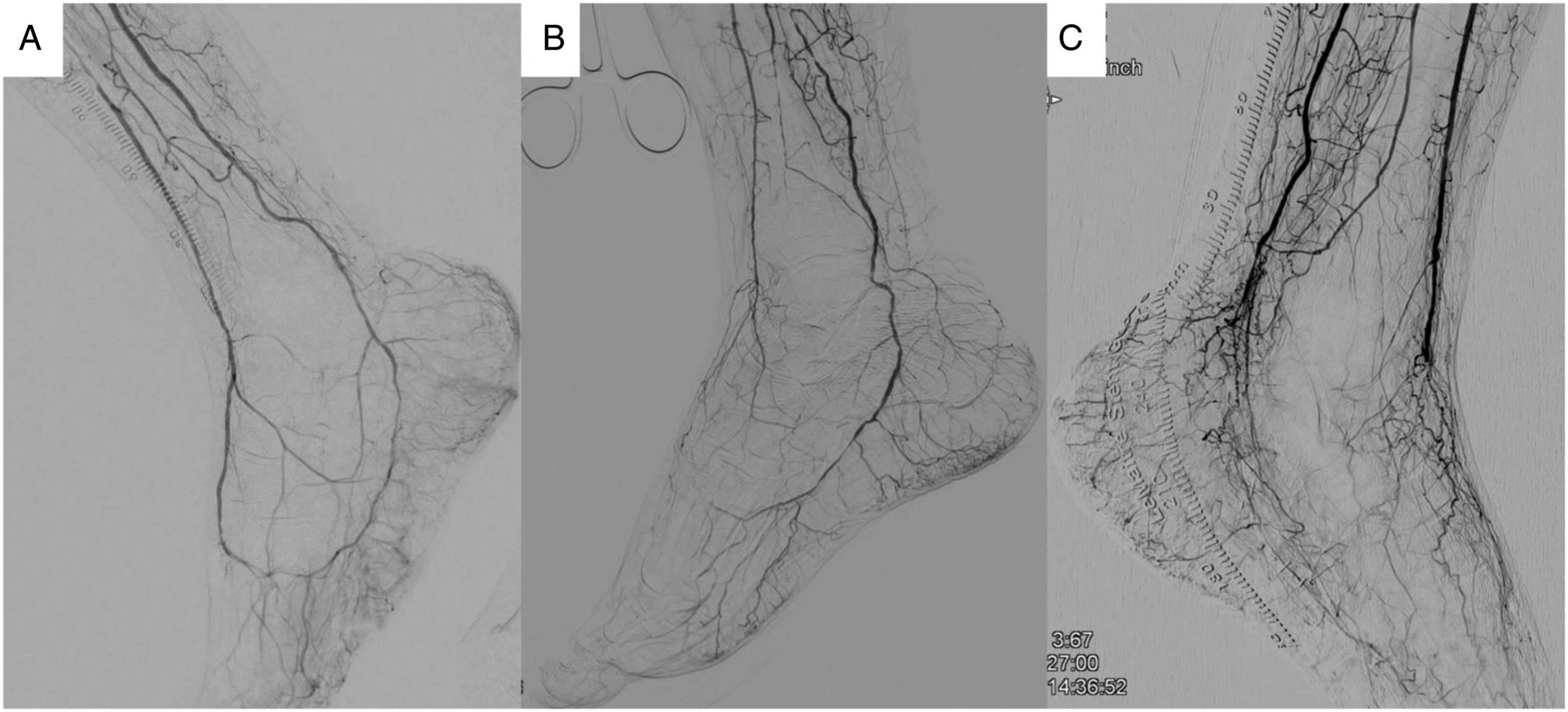
Graft revision
A failing graft was defined as any graft with duplex ultrasound evidence of significant stenosis that threatened patency at any follow-up time point. A failed graft was defined as any occluded graft with or without symptomatic lower limb ischemia. 11 Patients with failing or failed grafts were discussed in our team meetings before a final determination was made as to the best strategy for graft salvage.
Surgical interventions (vein patch angioplasty, interposition graft, and jump graft) or EVT (percutaneous transluminal angioplasty) were performed to repair stenotic or occluded lesions. In failed grafts, thrombectomy was performed before graft revision. The locations of graft revision were categorized as proximal anastomosis, graft body, distal anastomosis, or multiple. The graft body was defined as any location other than the anastomotic sites. Lesions treated in the native artery, either proximal (inflow) or distal (outflow) to the graft, were excluded. In cases of repeated graft revision, the first revision data were analyzed.
Clinical endpoints
The endpoints of this study were twofold: to compare patients who underwent de novo infrapopliteal bypass using autogenous veins with or without subsequent graft revision, and to identify predictors of graft revision.
Statistical analysis
Normally distributed continuous variables are expressed as means ± standard deviation (SD). Medians and interquartile ranges (IQRs) are presented for other continuous variables. Categorical variables are presented as percentages. Statistical significance was calculated and compared between the two groups using the χ2 test or unpaired t-test as appropriate. Patency rate, amputation-free survival rate, and overall survival rate were assessed using Kaplan–Meier life-table analysis, and the log-rank test was used to compare both groups. Cox multivariate regression analysis was used to assess the association between each variable and subsequent graft revision following infrapopliteal bypass. p values <0.05 were considered statistically significant. All statistical analyses were performed using IBM Statistics Statistical Package for Social Science (SPSS) version 26 (IBM Corp., Armonk, NY, USA).
Results
During the study period, 80 CLTI patients with ischemic ulcer or gangrene underwent de novo infrapopliteal bypass using autogenous veins at our institution. Of the 80 patients included in this study, 36 underwent graft revision (45%; graft revision group) and 44 did not (55%; non–graft revision group).
Patient characteristics
Patient characteristics.
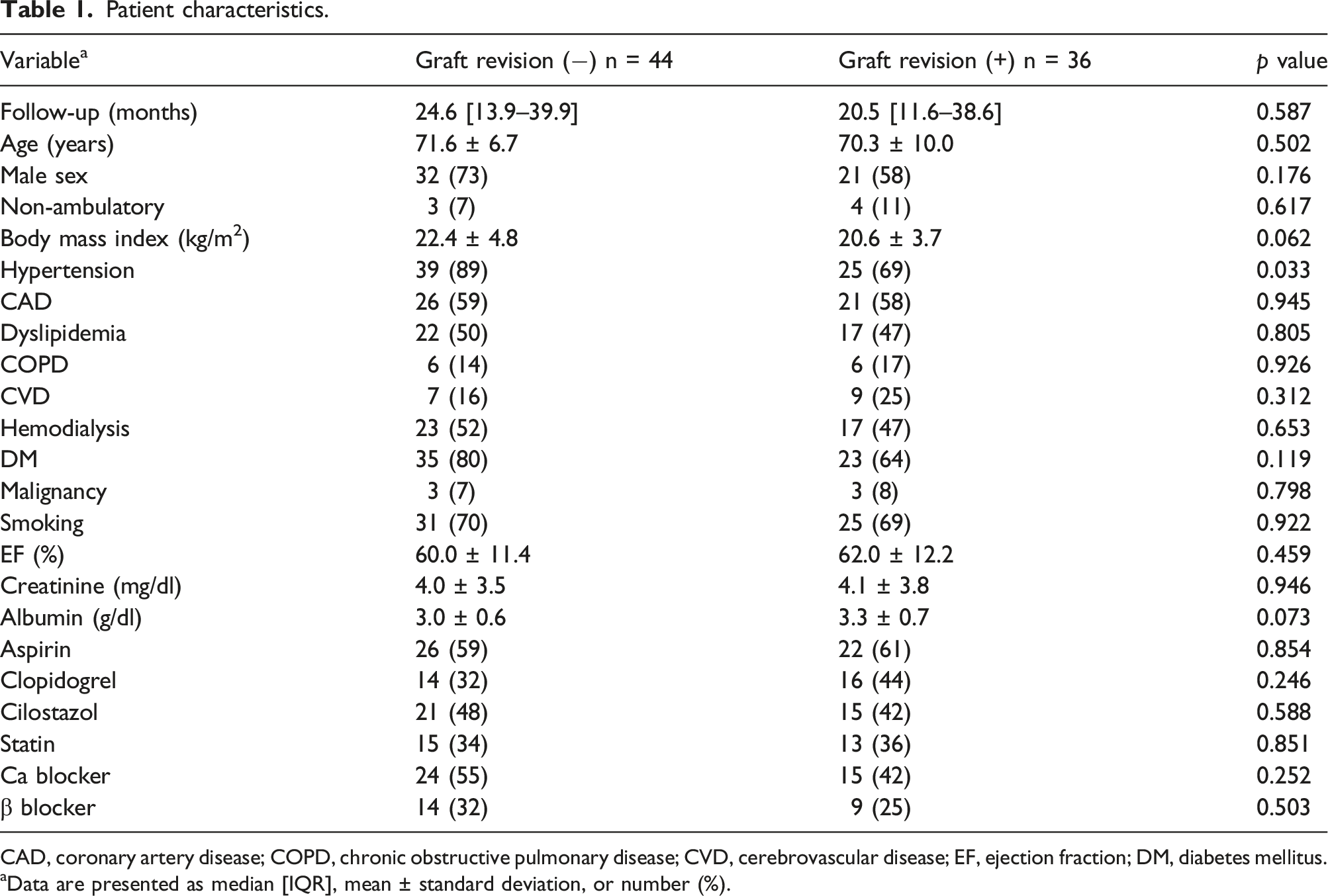
CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CVD, cerebrovascular disease; EF, ejection fraction; DM, diabetes mellitus.
Data are presented as median [IQR], mean ± standard deviation, or number (%).
Preoperative limb characteristics
Clinical characteristics of limbs.

WIfI, Wound, Ischemia, and Foot Infection; GLASS, Global Limb Anatomic Staging System.
Data are presented as mean ± standard deviation or number (%).
Surgical procedures
Surgical characteristics.
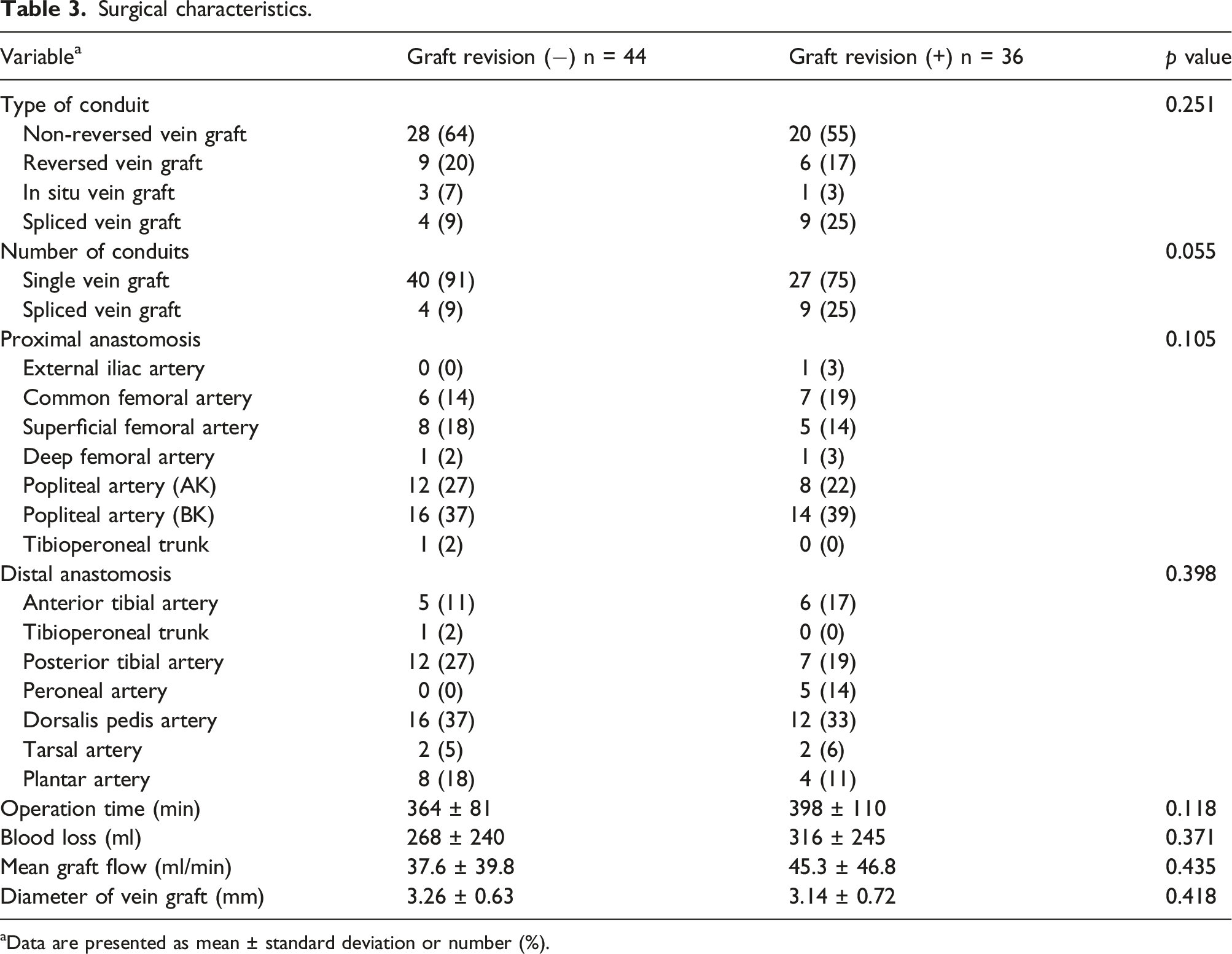
aData are presented as mean ± standard deviation or number (%).
Outcomes of graft revision
Graft revision details.
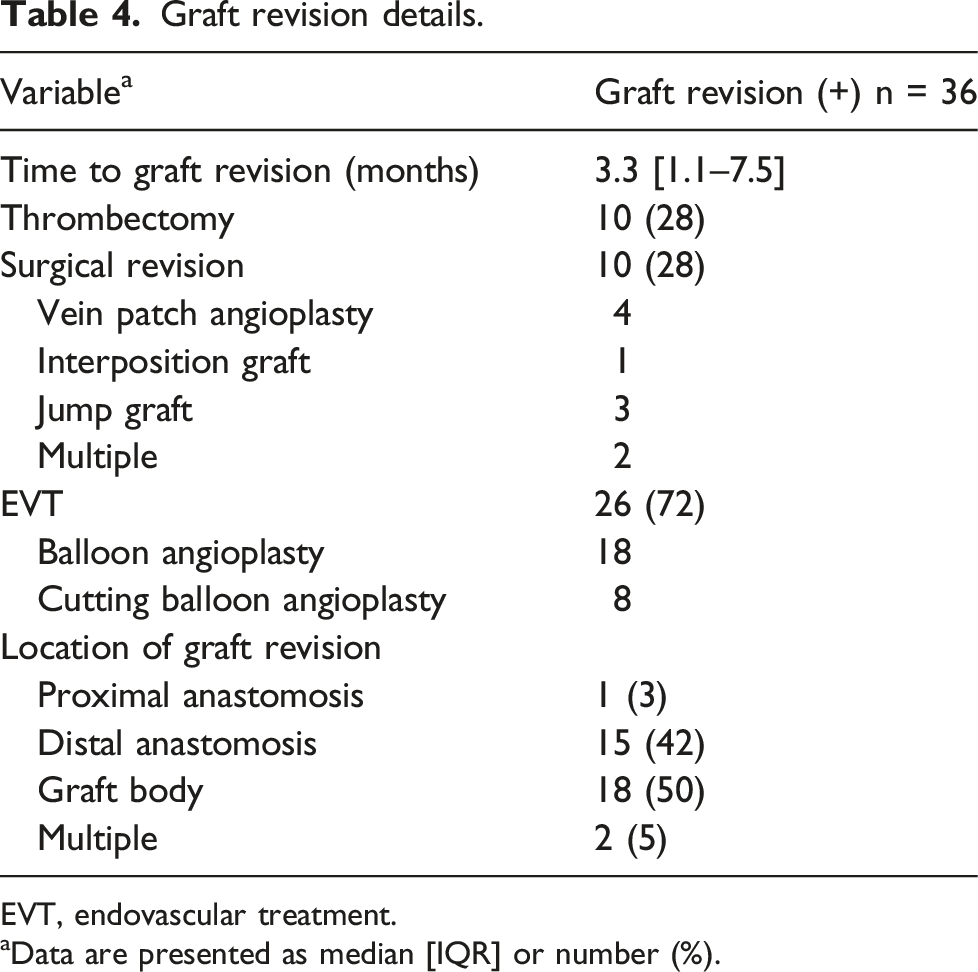
EVT, endovascular treatment.
Data are presented as median [IQR] or number (%).
Follow-up outcomes
The secondary patency rates at 1 and 2 years were 63% and 63% in the non–graft revision group, and 68% and 56% in the graft revision group, respectively (p = 0.923). The amputation-free survival rates at 1 and 2 years were 73% and 62% in the non–graft revision group and 83% and 67% in the graft revision group, respectively (p = 0.367). The wound healing rates at 2, 6, and 10 months were 27%, 90%, and 93% in the non–graft revision group and 36%, 73%, and 92% in the graft revision group, respectively (p = 0.331) (Figure 2). Kaplan–Meier curves comparing secondary patency rate (a), amputation-free survival (b), and wound healing rate (c). The log-rank test showed no significant differences between patients with and without graft revision.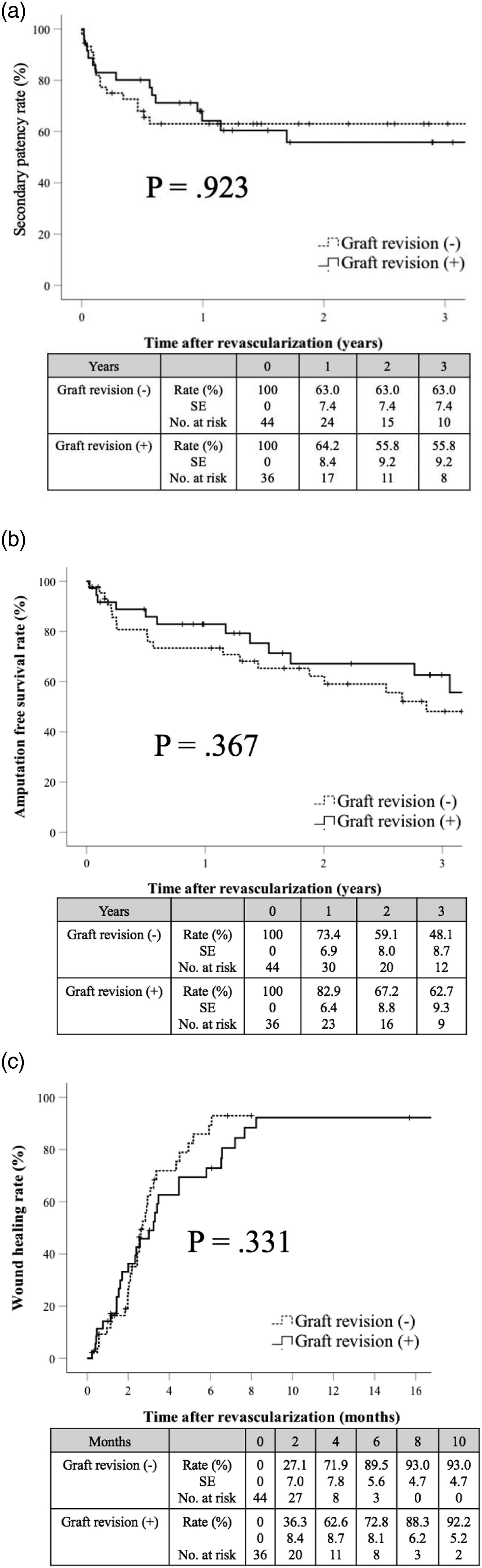
Predictors of graft revision after infrainguinal bypass surgery
Multivariate analysis of predictors of graft revision.
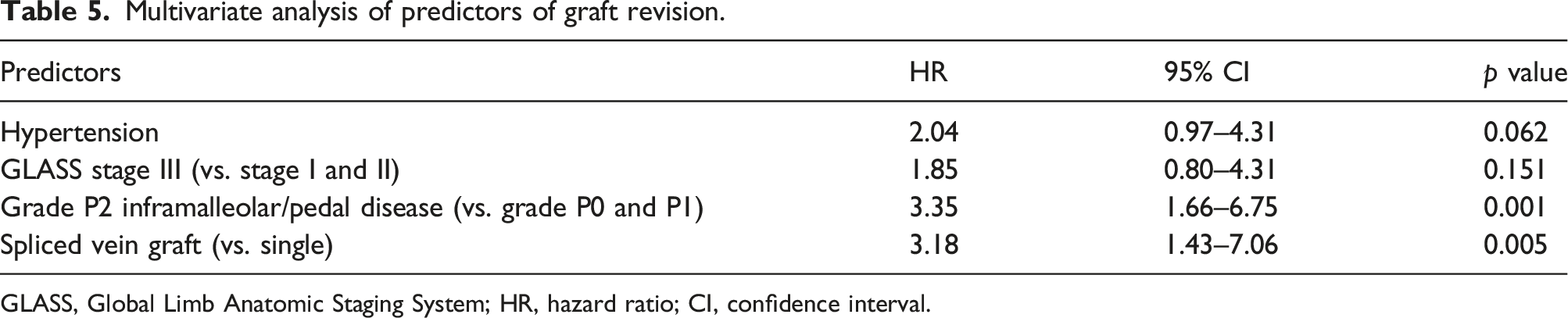
GLASS, Global Limb Anatomic Staging System; HR, hazard ratio; CI, confidence interval.
Graft revision sites
Graft revision sites.
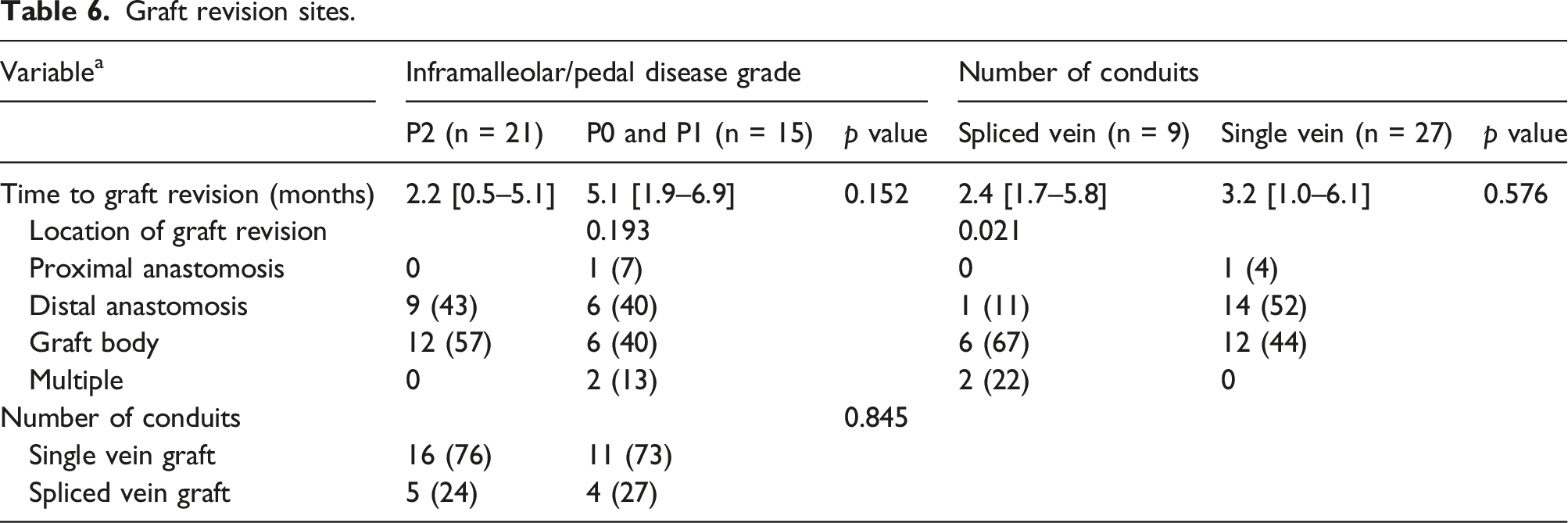
In 8 of 9 patients who underwent distal bypass with a spliced vein graft, the graft revision site was the graft body, which matched the veno–veno anastomosis with reference to surgical record.
Discussion
This study analyzed factors associated with graft revision after infrapopliteal vein bypass in CLTI patients with ischemic ulcer or gangrene. We demonstrated that grade P2 inframalleolar/pedal disease and spliced vein grafts were predictors of graft revision. This study is the first demonstration of the relationship between vein bypass graft revision and GLASS severity.
Wound healing is the goal of treatment for ischemic ulcer or gangrene secondary to atherosclerotic disease. Although EVT has been widely performed for CLTI because it is the least invasive option and there have been advances in the devices used, infrainguinal bypass using an autogenous vein graft is still the standard treatment, especially for infrapopliteal disease. 12 Maintaining graft patency is important to achieve wound healing, and graft occlusion can lead to extremely poor limb outcome. 13 Previous studies have reported that several factors, such as poor runoff, poor conduit quality, graft length, inadequate inflow, female sex, and hemodialysis, were associated with poor graft patency outcome.8, 14–20
Graft failure sometimes occurs soon after surgery, mainly due to hypercoagulability, technical errors, graft-related problems, and compromised runoff. Midterm graft occlusion is usually caused by the development of focal stenosis within a conduit or anastomotic area due to intimal hyperplasia. 21 To avoid early and midterm bypass failure, it is important to identify predictors of failing or failed grafts. Therefore, we compared the results of patients with and without graft revision and sought to identify predictors of graft failure.
GLASS, a new anatomical staging system proposed by the GVG for CLTI, separately grades (from 1 to 4) the severity and extent of disease in the femoropopliteal and infrapopliteal arterial segments to give an overall limb GLASS stage (from I to III). The GVG states that GLASS should be used to correlate the angiographic pattern of disease with immediate technical failure and clinical outcomes following infrainguinal endovascular intervention to facilitate shared decision-making and evidence-based revascularization. Additionally, to evaluate the status of the inframalleolar arteries, pedal grades (from P0 to P2) are determined. 1 Several studies showed that GLASS was associated with the clinical results of CLTI patients who underwent EVT.22,23 Recently, Kodama et al. reported a relationship between GLASS grades and outcome following bypass surgery or EVT in the Basil-1 trial cohort. 24 However, little information is available about the relationship between GLASS and graft revision. In this study, although univariate analysis showed that significant differences were observed between the graft revision and non–graft revision groups in terms of GLASS stage and inframalleolar/pedal disease grade, multivariate analysis showed that grade P2 inframalleolar/pedal disease was a significant predictor of graft revision. Our result suggests that GLASS IM might be a useful predictor of vein graft revision after bypass surgery.
Runoff is one of the predictors of graft performance, and it can be assessed in several ways, including the Bollinger scoring system and the Rutherford runoff score.25,26 Recently, Matsukura et al. and Miyake et al. reported new ways to evaluate runoff status.27,28 However, these scoring systems are too complicated for daily practice, and the Bollinger scoring system does not include the foot region. The GLASS stage classification is a simple means of evaluating femoropopliteal, infrapopliteal, and inframalleolar anatomical extension, as well as the complexity of infrainguinal diseases. For this reason, we opted to use GLASS to assess both runoff status and obstructive limb disease as a whole. Grade P2 inframalleolar/pedal disease was a predictor of graft revision, indicating that the presence of at least one patent inframalleolar artery entering the foot might be the minimum condition for infrapopliteal bypass surgery. In this study, a total of 16 patients with GLASS stage I of femoropopliteal classification underwent bypass surgery. There are two reasons for the use of bypass in these cases. First, 12 of 16 patients with GLASS I had an inframalleolar/pedal disease of grade P1 or P2. We prefer surgical bypass as a first-line treatment for patients with CLTI. Second, 10 of the 16 patients with GLASS I had a WIfI wound grade of 2 or 3. We think that bypass surgery is also appropriate for patients with extensive tissue loss. Thus we performed distal bypass for the 16 patients with GLASS I, taking into consideration poor run-off and large tissue defects.
It is controversial whether the quality of the pedal arch is associated with clinical outcomes such as patency rate and limb salvage rate. Some studies identified no significant relationship between pedal arch and graft patency.29,30 This may be because the blood flow feeding the foot is redundant as a result of multiple arterial–arterial connections. On the other hand, several studies demonstrated that the status of runoff vessels could affect graft patency, possibly by influencing outflow resistance.31,32 Progression of atherosclerotic disease or intimal hyperplasia may contribute to graft failure by increasing the resistance to flow.33–35 We agree with this conclusion and believe that the quality of the pedal arch is associated with graft patency. However, outflow resistance was not measured in this study. Further research on this topic is needed.
Several previous series reported that graft type had an impact on the outcome of graft patency.15,36–40 Chew et al. analyzed 226 patients who underwent autologous infrainguinal reconstruction without an adequate ipsilateral GSV. 37 They demonstrated a primary patency rate of 61% for contralateral GSV grafts compared with 39% for spliced vein bypass, while the patency rate did not differ significantly between groups, at 73% versus 63%, respectively. They concluded that the contralateral GSV is the preferred alternative conduit. Van Mierlo et al. evaluated 308 CLTI patients with an infrapopliteal autologous bypass. 38 They found that the use of a spliced vein resulted in similar outcomes compared with a single-segment GSV. As shown in Table 6, there was no significant difference in the number of conduits between patients with grade P2 and non-P2 IM disease. On the basis of the above results, grade P2 IM disease and spliced vein grafts are independent predictors of graft revision.
Previous studies showed that intraoperative graft flow and vein diameter were predictors of graft prognosis,15,16,41,42 but it is difficult to determine the absolute cutoff values of these measurements, and low graft flow and small vein diameter do not necessarily lead to poor outcome. 43 This is because low graft flow depends on several unfavorable conditions, such as poor runoff. In this study, we observed no significant difference in mean graft flow or vein graft diameter between patients with and without graft revision.
The WIfI classification, which was introduced by the Society for Vascular Surgery, stratifies risk relating to limb prognosis and has been widely used for decision-making when treating CLTI patients. 44 The WIfI classification can identify ischemic wounds more precisely than the Rutherford classification, and a higher WIfI stage indicates greater difficulty in achieving wound healing. In this study, however, patients with and without graft revision showed no significant difference in WIfI stage or in wound healing rate. This may be because strict surveillance can detect failing grafts in the early phase, and early intervention can contribute to comparable wound healing rates in the groups with and without graft revision.
In this study, we analyzed the data from the first graft revision. Thirty-six limbs underwent graft revision; of these, 10 (28%) underwent surgical revision and 26 limbs (72%) received EVT. Avino et al. demonstrated that the revision of vein graft stenosis with surgical or endovascular techniques resulted in an excellent patency rate, and EVT for focal vein graft stenosis was effective, durable, and comparable with surgical intervention of more extensive lesions. 45 Berceli et al. showed that surgical revision of vein grafts was associated with a reduced incidence of reintervention or major amputation, but the early success rate of EVT for vein grafts, particularly for failing grafts, was similar to that of the surgical procedure. 46 In our study, patients with failed grafts tended to undergo surgical revision, whereas those with failing grafts usually underwent endovascular revision. Graft revision was achieved with cutting balloon angioplasty in eight limbs. Schneider et al. reported that cutting balloon angioplasty was a reasonable initial treatment for vein graft stenosis and was a safe procedure with patency rates comparable to those of surgical procedures and superior to those of plain balloon angioplasty. 47
Causes of failing grafts include stenosis or occlusion in a number of locations, such as in the inflow or outflow arteries, in the anastomosis, or within the vein graft itself. Intrinsic vein graft lesions are known to be the most common cause of failing grafts. 48 In this study, graft revision was performed on the graft body in half of all patients. In most patients receiving distal bypass with a spliced vein graft, the graft revision site was the graft body, which matched with veno–veno anastomosis with surgical record. In patients who underwent bypass surgery with a single vein graft, the causes of vein graft lesions were unclear, but may have included valve-related stenosis or intimal hyperplasia. In patients receiving graft revision, there were no significant differences in graft revision sites between patients with grade P2 versus non-P2 inframalleolar/pedal disease. A poor runoff artery generally affects distal atherosclerotic disease or intimal hyperplasia, 35 but in this study, the revision site did not defer based on pedal disease grade. On this point, further research with a larger cohort is needed. Furthermore, 13 patients underwent multiple graft revisions during this study. Among them, 4 received graft revisions at the same location. Most had grade P2 inframalleolar/pedal disease.
The limitations of the current study should be mentioned. First, this study was performed at a single vascular surgery institution; therefore, our results might not be free from bias. Second, the identification of TAPs might have been inaccurate due to the retrospective analysis of angiographic images. The concept of the TAP was not conceived early in this study. Third, the number of patients was small. A larger sample size might be necessary to achieve sufficient statistical power to define the relationship between GLASS and graft revision. Fourth, the median follow-up was approximately 2 years, which might have been too short to accurately determine clinical outcome. Fifth, we believe that the quality of the pedal arch is associated with graft patency, because progression of atherosclerotic disease or intimal hyperplasia may contribute to graft failure by increasing the resistance to flow. However, this is just one hypothesis. Further study on this topic is needed. Finally, patients with failed grafts and no revision intervention were included in the non–graft revision group, because the goal of this study was to identify predictors of graft revision as opposed to graft patency. It will be important in the future to identify factors affecting graft revision, such as comorbidities, clinical status of limbs, anatomical characteristics of artery, and surgical details.
Conclusions
Our study demonstrated that grade P2 inframalleolar/pedal disease and spliced vein grafts were predictors of graft revision. The GLASS stratification of IM disease grade might be useful in identifying and optimizing treatment for CLTI.
ORCID iDs
Yohei Kawai https://orcid.org/0000-0002-7030-1541
Masayuki Sugimoto https://orcid.org/0000-0002-1712-4398
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
